Chinese Journal of Tissue Engineering Research
Previous Articles Next Articles
In vitro culture of human embryonic striatum-derived neural stem cells
Fan Ming-chao1, Wang Qiao-ling2, Liu Ke1, Zhang Xin1, Guan Yun-qian3, Sun Peng4
- 1 Department of Neurosurgical Intensive Care Unit, the Affiliated Hospital of Medical College, Qingdao University, Qingdao 266003, Shandong Province, China
2 Community Medical Service Center of Zhenjiang Road, Qingdao 266033, Shandong Province, China
3 Cell Therapy Center, Xuanwu Hospital Capital Medical University, Beijing 100053, China
4 Department of Neurosurgery, the Affiliated Hospital of Medical College, Qingdao University, Qingdao 266003, Shandong Province, China
-
Received:2012-09-19Revised:2012-11-07Online:2013-07-02Published:2013-07-02 -
Contact:Sun Peng, M.D., Professor, Doctoral supervisor, Department of Neurosurgery, the Affiliated Hospital of Medical College, Qingdao University, Qingdao 266003, Shandong Province, China, 266003 sunpengqd@163.com -
About author:Fan Ming-chao★, Master, Attending physician, Department of Neurosurgical Intensive Care Unit, the Affiliated Hospital of Medical College, Qingdao University, Qingdao 266003, Shandong Province, China fanmcchina@126.com Wang Qiao-ling, Physician, Community Medical Service Center of Zhenjiang Road, Qingdao 266033, Shandong Province, China -
Supported by:Shandong Natural Science Foundation, No. z2008c06*; National Natural Science Foundation of China, No. 81171208*
CLC Number:
Cite this article
Fan Ming-chao, Wang Qiao-ling, Liu Ke, Zhang Xin, Guan Yun-qian, Sun Peng. In vitro culture of human embryonic striatum-derived neural stem cells[J]. Chinese Journal of Tissue Engineering Research, doi: 10.3969/j.issn.2095-4344.2013.27.016.
share this article
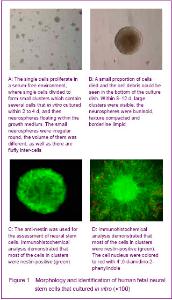
Culture and identification of human fetal neural stem cells Single cells were isolated from the dissociated striatum of human 8-16 weeks old fetuses described above. The primary-generation cells were globular, and the percentage of living cells was more than 95% with tryphan blue staining. The cells were continued to proliferate in a serum-free environment, where single cells divided to form small clusters which contained several cells that in vitro cultured within 2 to 4 days, and then neurospheres floating within the growth medium were formed. The small neurospheres were irregular round; the volume of them was different as well as its fluffy inter-cells (Figure 1A). At the same time, small proportion of cells was died and the cell debris could be seen in the bottom of the culture dish. Within 8-12 days, large clusters were visible; the neurospheres were buninoid, texture compacted and borderline limpid (Figure 1B)."
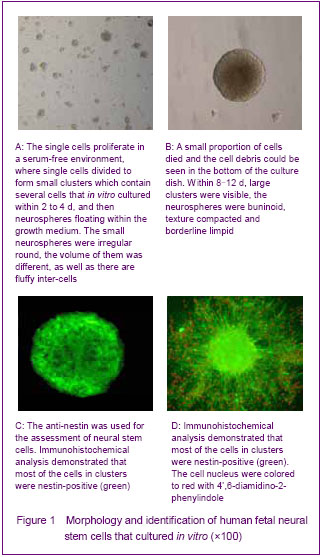
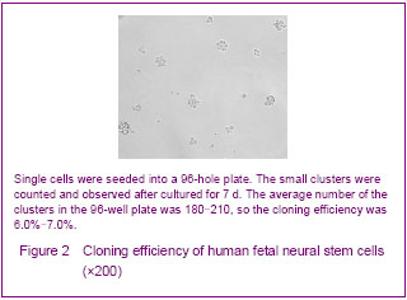
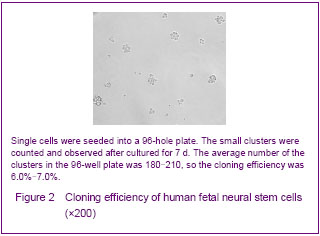
The operation of passage ought to be done when the diameter of clusters about 0.6-0.8 mm. New neurospheres will form after passage. The proliferation was rapid and the passage must to be done in a short time at the beginning culture. This kind of condition was changed after 2 months, the proliferation was slow and the generation time became longer. The identification of neural progenitor cells was impeded by the shortage of specific markers. So far, the expression of nestin was the only acceptable marker to identify neural stem cells in an early stage of development. So anti-nestin was used for the assessment of the neural stem cells. Immunohistochemical analysis demonstrated that most of the cells in clusters were nestin-positive (Figures 1C, D). Proliferation of human fetal neural stem cells Cloning efficiency was used for analyzing the growth rates of human fetal neural stem cells, and the single cells were seeded into a 96-hole plate described above. The small clusters were counted and observed after cultured for 7 days (Figure 2). The average number of the clusters in the 96-hole plate was 180-210, so the cloning efficiency was 6.0%-7.0%."

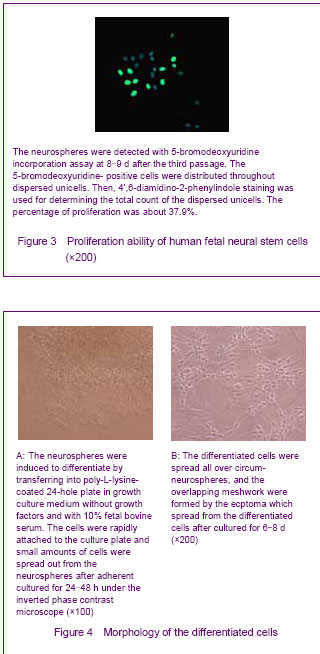
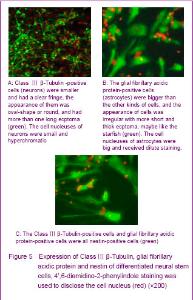
To further detect the proliferation ability of the human fetal neural stem cells, the neurospheres were used for 5-bromodeoxyuridine incorporation assay at 8-9 days after the third passage. The 5-bromodeoxyuridine- positive cells were distributed throughout dispersed unicells. Then, 4’,6-diamidino-2-phenylindole staining was used for determining the total count of the dispersed unicells. The percentage of proliferation was about 37.9% (Figure 3). Differentiation and Iimmunofluorescence labeling of human fetal neural stem cells The neurospheres were induced to differentiate by transferring into poly-L-lysine-coated 24-hole plate in growth culture medium without growth factors and with 10% fetal bovine serum. The cells rapidly attached to the culture plate and small amounts of cells spread out from the neurospheres after adherent culture for 24-48 hours under the inverted phase contrast microscope. The cells were irregular and have short ecphymas (Figure 4A). The quantity of cells spread out from the neurospheres was increased with culturing, and the cells with different morphologies were apparent. The differentiated cells spread all over the circum-neurospheres, overlapping meshwork were formed by the ecptoma which spread from the differentiated cells after cultured for 6-8 days (Figure 4B)."
To further detect the differentiation ability of the human fetal neural stem cells, the anti-nestin, anti-myelin basic protein, anti-Class Ⅲ β-Tubulin and anti-glial fibrillary acidic protein were used for immunofluorescence labeling. Differentiated neurospheres were found containing different cell types. Class Ⅲ β-Tubulin-positive cells were smaller and had a clear fringe, the appearance of them was oval-shape or round, and had more than one long ecptoma (Figure 5A). The proportion of Class Ⅲ β-Tubulin-positive in differentiated neural stem cells separated from striatum was about 56.8%. The glial fibrillary acidic protein-positive cells were bigger than the other kind cells, and the appearance of them was irregular with short and thick ecptoma, maybe like the starfish (Figure 5B). The proportion of glial fibrillary acidic protein-positive in differentiated neural stem cells separated from striatum was about 39.8%. The myelin basic protein-positive cells were never seen in the differentiated neural stem cells that separated from striatum in our study. The nestin-positive cells were located on the interior neurospheres, and the number of circum-neurosphere was decreased (Figure 5C). The Class Ⅲ β-Tubulin-positive cells and glial fibrillary acidic protein-positive cells were all nestin-positive cells, but the fluorescence of differentiated cells was dimmish when compared with the cells before differentiation."
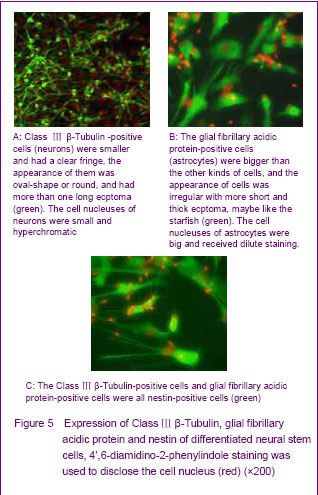

The Class Ⅲ β-Tubulin-positive cells had a tendency that they like aggregation when compared with the glial fibrillary acidic protein-positive cells. The glial fibrillary acidic protein-positive cells were hypodispersion in the corona radiata of differentiated neurospheres, but the Class Ⅲ β-Tubulin-positive cells could form cancellous cells cluster usually. The myelin basic protein-positive cells were unseen."

| [1] Cameron HA, Hazel TG, Mckay RD. Regulation of neurogenesis by growth factors and neurotransmitters. J Neurobiol. 1998;36(2):287-306.[2] Reynolds BA, Weiss S. Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. Science. 1992;255(5052): 1707-1710.[3] Temple S. The development of neural stem cells. Nature. 2001;414(6859):112-117.[4] Arsenijevic Y, Villemure JG, Brunet JF, et al. Isolation of multipotent neural precursors residing in the cortex of the adult human brain. Exp Neurol. 2001;170(1):48-62.[5] Park TI, Monzo H, Mee EW, et al. Adult human brain neural progenitor cells (NPCs) and fibroblast-like cells have similar properties in vitro but only NPCs differentiate into neurons. PLoS One. 2012;7(6):e37742.[6] Johansson CB, Momma S, Clarke DL, et al. Identification of a neural stem cell in the adult mammalian central nervous system. Cell. 1999;96(1):25-34.[7] Xu R, Wu C, Tao Y, et al. Nestin-positive cells in the spinal cord: a potential source of neural stem cells. Int J Dev Neurosci. 2008;26(7):813-820.[8] Walton RM, Wolfe JH. In vitro growth and differentiation of canine olfactory bulb-derived neural progenitor cells under variable culture conditions. J Neurosci Methods. 2008; 169(1): 158-167.[9] Ryu KJ, Cho SJ, Hatori K, et al. Proactive transplantation of human neural stem cells prevents degeneration of striatal neurons in a rat model of Huntington disease. Neurobiol Dis. 2004;16(1):68-77.[10] Emborg ME, Ebert AD, Moirano J, et al. GDNF-secreting human neural progenitor cells increase tyrosine hydroxylase and VMAT2 expression in MPTP-treated cynomolgus monkeys. Cell Transplant. 2008;17(4):383-395.[11] Cusimano M, Biziato D, Brambilla E, et al. Transplanted neural stem/precursor cells instruct phagocytes and reduce secondary tissue damage in the injured spinal cord. Brain. 2012;135(Pt 2):447-460.[12] Xuan AG, Long DH, Yang DD, et al. BDNF improves the effects of neural stem cells on the rat model of Alzheimer's disease with unilateral lesion of fimbria-fornix. Neurosci Lett. 2008;440(3):331-335.[13] Xuan AG, Luo M, Ji WD, et al. Effects of engrafted neural stem cells in Alzheimer's disease rats. Neurosci Lett. 2009; 450(2):167-171.[14] Fauza DO, Jennings RW, Teng YD, et al. Neural stem cell delivery to the spinal cord in an ovine model of fetal surgery for spina bifida. Surgery. 2008;144(3):367-373.[15] Tsai RY, Mckay RD. Cell contact regulates fate choice by cortical stem cells. J Neurosci. 2000;20(10):3725-3735.[16] Schumm MA, Castellanos DA, Frydel BR, et al. Direct cell-cell contact required for neurotrophic effect of chromaffin cells on neural progenitor cells. Brain Res Dev Brain Res. 2003;146(1-2):1-13.[17] Wang JH, Hung CH, Young TH. Proliferation and differentiation of neural stem cells on lysine-alanine sequential polymer substrates. Biomaterials. 2006;27(18):3441-3450.[18] Tsai RY, Mckay RD. Cell contact regulates fate choice by cortical stem cells. J Neurosci. 2000;20(10):3725-3735.[19] Johansson CB, Svensson M, Wallstedt L, et al. Neural stem cells in the adult human brain. Exp Cell Res. 1999;253(2):733-736.[20] Castiglione M, Calafiore M, Costa L, et al. Group I metabotropic glutamate receptors control proliferation, survival and differentiation of cultured neural progenitor cells isolated from the subventricular zone of adult mice. Neuropharmacology. 2008;55(4):560-567.[21] Lin HJ, O’Shaughnessy TJ, Kelly J, et al. Neural stem cell differentiation in a cell-collagen-bioreactor culture system. Brain Res Dev Brain Res. 2004;153(2):163-173.[22] El Seady R, Huisman MA, Löwik CW, et al. Uncomplicated differentiation of stem cells into bipolar neurons and myelinating glia. Biochem Biophys Res Commun. 2008; 376(2):358-362.[23] Su L, Lü X, Xu J, et al. Neural stem cell differentiation is mediated by integrin beta 4 in vitro. Int J Biochem Cell Biol. 2009;41(1):916-924.[24] Dictus C, Tronnier V, Unterberg A, et al. Comparative analysis of in vitro conditions for rat adult neural progenitor cells. J Neurosci Methods. 2007;161(2):250-258.[25] Hulspas R, Tiarks C, Reilly J, et al. In vitro cell density-dependent clonal growth of EGF-responsive murine neural progenitor cells under serum-free conditions. Exp Neurol. 1997;148(1):147-156.[26] Storch A, Sabolek M, Milosevic J, et al. Midbrain-derived neural stem cells; from basic science to therapeutic approaches. Cell Tissue Res. 2004;318(1):15-22.[27] Suzuki M, Wright LS, Marwah P, et al. Mitotic and neurogenic effects of dehydroepiandrosterone (DHEA) on human neural stem cell cultures derived from the fetal cortex. Proc Natl Acad Sci U S A. 2004;101(3):3203-3207.[28] Liu YH, Zhao LH, Zhao H, et al. The experimental research of culture of nueral stem cells isolated from rat fetal brain cortex in vitro. Zhongguo Shiyong Shenjing Jibing Zazhi. 2006;9(6):3-4.[29] Li X, Xu J, Bai Y, et al. Isolation and characterization of neural stem cells from human fetal striatum. Biochem Biophys Res Commun. 2005;326(2):425-434.[30] Bez A, Corsini E, Curti D, et al. Neurosphere and neurosphere-forming cells: morphological and ultrastructural characterization. Brain Res. 2003;993:18-19.[31] Itahana K, Campisi J, Dimri GP. Methods to detect biomarkers of cellular senescence: the senescence associated beta-galactosidase assay. Methods Mol Biol. 2007;371:21-31.[32] Ostenfeld T, Caldwell MA, Prowse KR, et al. Human neural precursor cells express low levels of telomerase in vitro and show diminishing cell proliferation with extensive axonal outgrowth following transplantation. Exp Neurol. 2000;164(1):215-226.[33] Wu P, Ye Y, Svendsen CN. Transduction of human neural progenitor cells using recombinant adeno-associated viral vectors. Gene Ther. 2002;9(4):245-255.[34] Sun Y, Pollard S, Conti L, et al. Long-term tripotent differentiation capacity of human neural stem (NS) cells in adherent culture. Mol Cell Neurosci. 2008;38(2):245-258.[35] Westerlund U, Moe MC, Varqhese M, et al. Stem cells from the adult human brain develop into functional neurons in culture. Exp Cell Res. 2003;289(2):378-383.[36] Xu R, Wu C, Tao Y, et al. Nestin-positive cells in the spinal cord: a potential source of neural stem cells. Int J Dev Neurosci. 2008;26(7):813-820.[37] Wang WZ, Qi JP, Wang DS, et al. Expression of glial fibrillar acidic protein and cyclinD1 in the perihematomal tissues in human hypertensive intracerebral hemorrhage. Zhongfeng yu Shenjing Jibing Zazhi. 2008;25(2):158-160.[38] Roskams AJ, Cai X, Ronnett GV. Expression of neuron-specific beta-III tubulin during olfactory neurogenesis in the embryonic and adult rat. Neuroscience. 1998;83(1):191-200.[39] Yin XJ, Feng ZC, Du J, et al. Effects of neonatal calf serum on differentiation of human fetal neural stem cells in the hippocampus. Di Yi Jun Yi Da Xue Xue Bao. 2004;24(2):1164-1166.[40] Windrem MS, Nunes MC, Rashbaum WK, et al. Fetal and adult human oligodendrocyte progenitor cell isolates myelinate the congenitally dysmelinated brain. Nat med. 2004;10(1):93-97.[41] Murray K, Dubois-Daicq M. Emergence of oligodendrocytes from human neural spheres. J Neurosci Res. 1997;50(3):146-156.[42] Ostenfeld T, Joly E, Tai YT, et al. Regional specification of rodent and human neurospheres. Brain Res Dev Brain Res. 2002;134(2):43-55.[43] Chandran S, Compston A. Neural stem cells as a potential solll'ce ofoligodendrocytes for myelin repair. J Neurol Sci. 2005;233(1-2):178-181. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [3] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| [4] | Wang Xianyao, Guan Yalin, Liu Zhongshan. Strategies for improving the therapeutic efficacy of mesenchymal stem cells in the treatment of nonhealing wounds [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1081-1087. |
| [5] | Liao Chengcheng, An Jiaxing, Tan Zhangxue, Wang Qian, Liu Jianguo. Therapeutic target and application prospects of oral squamous cell carcinoma stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1096-1103. |
| [6] | Xie Wenjia, Xia Tianjiao, Zhou Qingyun, Liu Yujia, Gu Xiaoping. Role of microglia-mediated neuronal injury in neurodegenerative diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1109-1115. |
| [7] | Li Shanshan, Guo Xiaoxiao, You Ran, Yang Xiufen, Zhao Lu, Chen Xi, Wang Yanling. Photoreceptor cell replacement therapy for retinal degeneration diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1116-1121. |
| [8] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [9] | Wang Shiqi, Zhang Jinsheng. Effects of Chinese medicine on proliferation, differentiation and aging of bone marrow mesenchymal stem cells regulating ischemia-hypoxia microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1129-1134. |
| [10] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [11] | Kong Desheng, He Jingjing, Feng Baofeng, Guo Ruiyun, Asiamah Ernest Amponsah, Lü Fei, Zhang Shuhan, Zhang Xiaolin, Ma Jun, Cui Huixian. Efficacy of mesenchymal stem cells in the spinal cord injury of large animal models: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1142-1148. |
| [12] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [13] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [14] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [15] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||