Chinese Journal of Tissue Engineering Research
Previous Articles Next Articles
Nano drug delivery system in targeted therapy for tumor
Zhang Peng1, Chen Yong-zhong1, Zhou Qing-bi1, Bai Jing-ping2, Xilin Baoleri2
- 1 Institute of Orthopedics, Fuzhou General Hospital of Nanjing Military Command of Chinese PLA (the 476 Hospital), Fuzhou 350001, Fujian Province, China
2 Department of Orthopaedic Oncology, Tumor Hospital of Xinjiang Medical University, Urumqi 830011, Xinjiang Uygur Autonomous Region, China
-
Received:2013-01-18Revised:2013-03-25Online:2013-05-21Published:2013-05-21 -
Contact:Xilin Baoleri, M.D., Chief physician, Associate professor, Doctoral supervisor, Department of Orthopaedic Oncology, Tumor Hospital of Xinjiang Medical University, Urumqi 830011, Xinjiang Uygur Autonomous Region, China xjbonetumor@126.com -
About author:Zhang Peng★, Master, Physician, Institute of Orthopedics, Fuzhou General Hospital of Nanjing Military Command of Chinese PLA (the 476 Hospital), Fuzhou 350001, Fujian Province, China zpzippo@hotmail.com
CLC Number:
Cite this article
Zhang Peng, Chen Yong-zhong, Zhou Qing-bi, Bai Jing-ping, Xilin Baoleri. Nano drug delivery system in targeted therapy for tumor[J]. Chinese Journal of Tissue Engineering Research, doi: 10.3969/j.issn.2095-4344.2013.21.024.
share this article
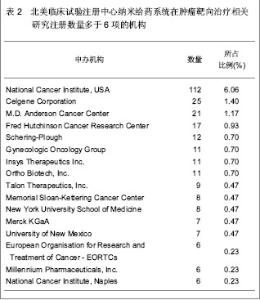
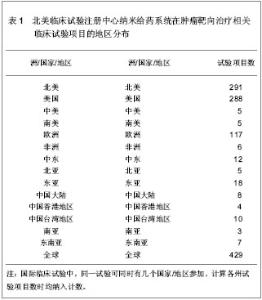
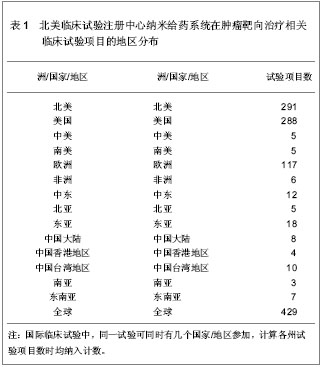
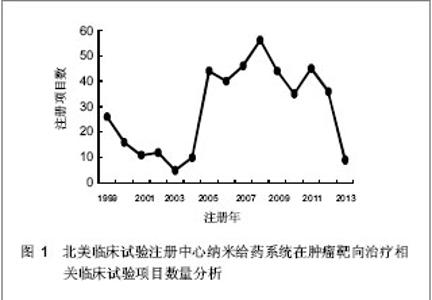
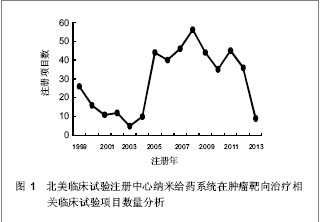
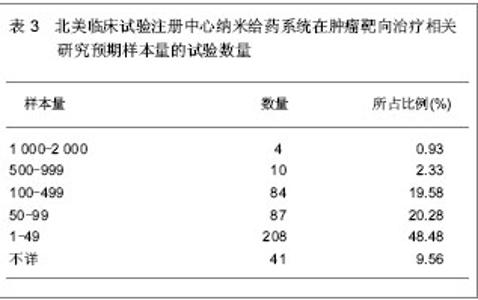
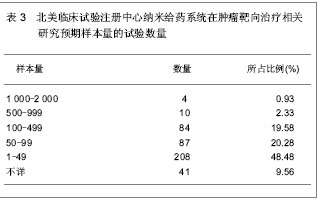
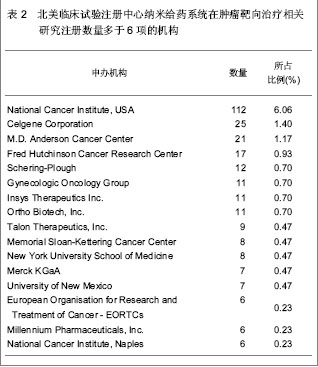
北美临床试验注册中心关于纳米给药系统在肿瘤靶向治疗的相关临床试验项目注册开始于1999年,此后,注册数量逐年下降,2003年达到谷底,只有5项研究项目,之后数量大幅度升高,在2008年注册项目数量达到顶峰,为56项,占总项目数量的12.87%。2008年后,注册的项目数量处于波动状态。 2.3 北美临床试验注册中心注册的临床试验项目申办机构分布 以注册项目数量排名,美国国立癌症研究所(National Cancer Institute, USA)在纳米给药系统在肿瘤靶向治疗的临床试验研究方面较为重视,已注册同类研究112项,紧随其后的是来自美国的Celgene 公司。北美临床试验注册纳米给药系统在肿瘤靶向治疗相关研究数量多于5项的机构,见表2。"

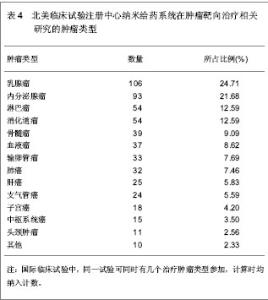
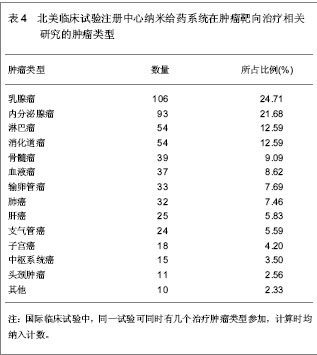
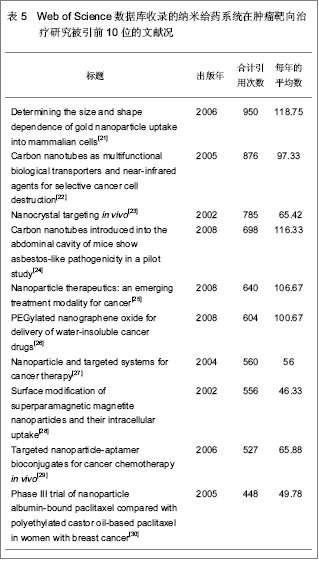
2.6 中国在北美临床试验注册中心注册的已完成的纳米给药系统在肿瘤靶向治疗相关研究的项目分析 中国在北美临床试验注册中心共注册22项,其中,已完成的项目3项,中国大陆,中国香港地区,及中国台湾地区各完成1项。 中国香港地区已完成的注册项目,临床注册号NCT00093444,中国和美国联合注册,以研究阿霉素脂质体联合射频消融治疗原发性及转移性肝肿瘤疗效为目的Ⅰ期临床试验,研究开始于2004年10月,结束于2010年1月,共纳入30名试验对象,主要观察注射15,30,45,60,75, 90 min,2,3,4,6,24,46 h,及 4,8 d的药代动力学及药效学水平。阿霉素脂质体治疗的最大耐受剂量;其次MRI观测 28 d后消融治疗对肿瘤血流量及肿瘤血管密度的影响。纳入的疾病特征:组织学确诊原发性或转移性肝肿瘤:≤4个病灶;单一肿瘤最大直径<7 cm。病理学或先前手术表明不适合进行手术切除。 患者的特点:年龄≥18岁, 体能状态:ECOG评分0-2分;预期寿命≥3个月, 造血系统:血小板计数≥75 000/ mm3;白细胞计数≥1 500/mm3;血红蛋白≥100 g/L的(允许输血达到水平≥100 g/L);肝:胆红素≤34.2 μmol/L;凝血酶原时间或部分凝血致活酶时间≤1.5倍对照值(患者接受抗凝治疗除外)。 肾:肌酐≤221 μmol/L。 心血管:心脏放射性核素扫描射血分数≥50%;无充血性心脏衰竭;过去6个月内无心肌梗死,无脑血管意外;没有危及生命的心律失常。 其他:体质量<136 kg;血糖≤16.65 mmol/L;无不受控制的糖尿病;对使用的造影剂没有不良反应史;无蛋或蛋制品过敏史;无其他严重内科疾病;非怀孕期或哺乳期;妊娠试验阴性;育龄妇女必须有效的避孕。 同时疗法: 生物治疗:无治疗干扰;免疫抑制患者在试验时及试验后30 d无活疫苗接种。 其它:肝肿瘤治疗≥3周;全身治疗非危及生命的肝外疾病并恢复超过>3周;无其他并发全身性治疗;试验期间及试验后30 d无任何下列药物给药:环孢素、苯巴比妥、苯妥英钠、链脲佐菌素。未同时给予给下药物:注射两性霉素B、抗甲状腺剂用于甲状腺亢进、硫唑嘌呤、氯霉素、秋水仙碱、氟胞嘧啶、更昔洛韦、普卡、齐多夫定、丙磺舒、磺吡酮。 临床注册号NCT00606515,中国大陆地区注册的以研究紫杉醇脂质体治疗中国实体瘤患者的Ⅳ期药代动力学研究,开始于2008年1月,结束于2008年10月。纳入标准:病理学确诊实体瘤晚期患者;仅适用醇脂质体治疗;体能状态ECOG评分0-2分;预期寿命≥3个月;血液指标,肝、肾功能正常;其他器官功能正常;过去4周内无全身化疗;无激素过敏史。排除标准:有对试验研究药物过敏史;有影响患者完成试验的严重并发症;毒性评价标准NCI CTCAE3.0规定的≥1级神经病变;怀孕期或哺乳期。共16例患者纳入分析。随机分成A,B两组,A组患者接受静脉注射紫杉醇脂质体,3 h内注射175 mg/(m2•d), 21 d 一个疗程,治疗3个疗程;B组患者接受静脉注射紫杉醇注射液,3 h内注射175 mg/(m2•d),21 d 一个疗程,治疗3个疗程。主要观察治疗72 h 的药代动力学参数。 临床注册号NCT00813072,中国台湾地区联合波黑共和国、克罗地亚、韩国、西班牙、英国的国际多中心研究PEP02, 依立替康及多西他奇治疗胃或胃食管肿瘤的Ⅱ期有效性分析。研究开始于2008年12月,结束于2010年12月。纳入标准:病理学或细胞学确诊的局部晚期(不能手术切除)或转移性胃或胃食管肿瘤患者;局部晚期或转移性疾病仅一次全身化疗失败,包括辅助化疗后6个月内疾病复发的患者,同步放化疗给药不算入全身化疗;根据RECIST标准,至少有一个可测量的病灶;有能力签署患者知情同意书;ECOG体力状态评分0-2分;预期寿命≥3个月;器官和血液指标正常。 排除标准:试验开始前3周接受全身化疗;试验开始前4周接受放疗;脑转移瘤;试验前5年内接受活动性多重肿瘤或其他肿瘤的治疗(治愈的非黑色素瘤和原位宫颈癌患者除外);曾接受伊立替康或紫杉烷类(紫杉醇,多西紫杉醇)治疗;曾接受辐射导致骨髓活性>30%;试验开始前4周内接受重大手术(剖腹手术,导管置入除外);周围神经病变> 2级;对脂质体产品或其他含聚山梨酯成分药物有过敏史;有不受控制的并发症;试验开始前3周内接受任何药物试验;妊娠期或哺乳期女性(所有女性受试者进入研究之前都必须进行妊娠试验,结果必须是阴性);肠梗阻患者;试验开始前2周内接受St. John's Wort, CYP3A4诱导抗痉挛药(苯妥英钠,苯巴比妥,卡马西平)、利福平和利福布汀,或试验开始前1周内接受酮康唑,伊曲康唑,醋竹桃霉素,红霉素,地尔硫卓,维拉帕米药物。135例患者随机分为2组,PEP02组:静脉注射PEP02, 90 min 内注射120 mg/(m2•d),21 d 一个疗程,直到肿瘤恶化或出现无法耐受的毒副作用。依立替康组:静脉注射依立替康, 300 mg/(m2•d),21 d一个疗程,直到肿瘤恶化或出现无法耐受的毒副作用。 多西他奇组,静脉注射依立替康, 75 mg/(m2•d), 60 min注完,21d 一个疗程,直到肿瘤恶化或出现无法耐受的毒副作用。 主要观察指标:治疗的客观有效率。 次要观察指标:无进展生存期,肿瘤反应时间,肿瘤进展时间,治疗失败时间,疾病控制率,1年存活率,总存活率,以及PEP02和依立替康的药代动力学及药物遗传学检测。 2.7 Web of Science数据库纳米给药系统在肿瘤靶向治疗相关文章分析 见表5。 Web of Science数据库2000-01-01/2012-12- 31共收录纳米给药系统在肿瘤靶向治疗相关文章 4 497篇,发文量最多的国家为美国,数量为2 987篇,中国发表文章134篇。被引较高的前10篇文章中有3篇发表在Proceedings of the National Academy of Sciences of the United States of America (《美国科学院院刊》)上。被引次数最高的文章为2006年在Nano Letters (《纳米快报》)由Chithrani BD, Ghazani AA, Chan WC发表的Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells[21],截止检索日止,在Web of Science数据库中共计被引用950次,年平均被引118.75次。"
| [1] Kodaira H, Tsutsumi Y, Yoshioka Y, et al. The targeting of anionized polyvinylpyrrolidone to the renal system. Biomaterials. 2004;25(18):4309-4315.[2] Langer R. Drug delivery and targeting. Nature. 1998;392 (6679 Suppl):5-10.[3] Pierce WM, Leonard WC. Bone targeted inhibitors of carbonic anhydraseP. Eur Patent: 201057, 1986-11-12.[4] Hwang HY, Kim IS, Kwon IC, et al. Tumor targetability and antitumor effect of docetaxel-loaded hydrophobically modified glycol chitosan nanoparticles. J Control Release. 2008;128(1): 23-31.[5] Omelyanenko V, Kopecková P, Gentry C, et al. Targetable HPMA copolymer-adriamycin conjugates. recognition, internalization, and subcellular fate. J Control Release. 1998;53(1-3):25-37.[6] Goldberg M, Langer R, Jia X. Nanostructured materials for applications in drug delivery and tissue engineering. J Biomater Sci Polym Ed. 2007;18(3):241-268.[7] Donbrow M, Friedman M .Timed release from polymeril films containing drugs and kinetics of drug release. J Pharm Sci. 1975;64(1):76-80.[8] Saltzman WM, Fung LK. Polymeric implants for cancer chemotherapy. Adv Drug Deliv Rev. 1997;26(2-3):209-230.[9] Yoshioka Y, Tsutsumi Y, Mukai Y, et al.Effective accumulation of poly(vinylpyrrolidone-co-vinyl laurate) into the spleen.J Biomed Mater Res A. 2004;70(2):219-223.[10] Nishiyama N, Kataoka K. Current state, achievements, and future prospects of polymeric micelles as nanocarriers for drug and gene delivery. Pharmacol Ther. 2006;112(3): 630-648.[11] Archakov AI, Ivanov YD. Analytical nanobiotechnology for medicine diagnostics. Mol Biosyst. 2007;3(5):336-342.[12] Han M, He CX, Fang QL, et al. A novel camptothecin derivative incorporated in nano-carrier induced distinguished improvement in solubility, stability and anti-tumor activity both in vitro and in vivo. Pharm Res. 2009;26(4):926-935.[13] 易承学,余江南,徐希明.纳米药物载体在中药制剂研发中的应用[J].中国中药杂志,2008;33(16):1936-1940.[14] Thompson WW, Anderson DB, Heiman ML. Biodegradable microspheres as a delivery system for rismorelin porcine, a porcine-growth-hormone-releasing-hormone. J Control Release. 1997;43(1):9-22.[15] Davis SS. Biomedical applications of nanotechnology implications for drug targeting and gene therapy. Trends Biotechnol. 1997;15(6):217-224.[16] Foster TP, Moseley WM, Caputo JF, et al. Sustained elevated serum somatotropin concentrations in Holstein steers following subcutaneous delivery of a growth hormone releasing factor analog dispersed in water, oil or microspheres. J Control Release.1997;47(1):91-99.[17] Janes KA, Calvo P, Alonso MJ. Polysaccharide colloidal particles as delivery systems for macromolecules. Adv Drug Deliv Rev. 2001;47(1):83-97.[18] 王斌.靶向抗肿瘤药物载体系统研究近况[J].广东药学学报, 1998,14(4):311-314.[19] Takeuchi H, Yamamoto H, Kawashima Y. Mucoadhesive nanoparticulate systems for peptide drug delivery. Adv Drug Deliv Rev. 2001;47(1):39-54.[20] Jain KK. Nanomedicine: application of nanobiotechnology in medical practice. Med Prine Pract.2008;17(2):89-101.[21] Chithrani BD, Ghazani AA, Chan WC. Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett. 2006;6(4):662-668.[22] Kam NW, O'Connell M, Wisdom JA, et al. Carbon nanotubes as multifunctional biological transporters and near-infrared agents for selective cancer cell destruction. Proc Natl Acad Sci U S A. 2005;102(33):11600-11605. [23] Akerman ME, Chan WC, Laakkonen P, et al. Nanocrystal targeting in vivo. Proc Natl Acad Sci U S A. 2002;99(20): 12617-12621.[24] Poland CA, Duffin R, Kinloch I, et al. Carbon nanotubes introduced into the abdominal cavity of mice show asbestos-like pathogenicity in a pilot study. Nat Nanotechnol. 2008;3(7):423-428. [25] Davis ME, Chen ZG, Shin DM. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nat Rev Drug Discov. 2008;7(9):771-782. [26] Liu Z, Robinson JT, Sun X, et al. PEGylated nanographene oxide for delivery of water-insoluble cancer drugs. J Am Chem Soc. 2008;130(33):10876-10877. [27] Brannon-Peppas L, Blanchette JO.Nanoparticle and targeted systems for cancer therapy. Adv Drug Deliv Rev. 2004;56(11): 1649-1659.[28] Zhang Y, Kohler N, Zhang M. Surface modification of superparamagnetic magnetite nanoparticles and their intracellular uptake. Biomaterials. 2002;23(7):1553-1561.[29] Farokhzad OC, Cheng J, Teply BA, et al. Targeted nanoparticle-aptamer bioconjugates for cancer chemotherapy in vivo. Proc Natl Acad Sci U S A. 2006;103(16):6315-6320. [30] Gradishar WJ, Tjulandin S, Davidson N, et al. Phase III trial of nanoparticle albumin-bound paclitaxel compared with polyethylated castor oil-based paclitaxel in women with breast cancer. J Clin Oncol. 2005;23(31):7794-7803. [31] Cancer centers. http://cancercenters.cancer.gov/cancer_centers/map-cancer-centers.html. 2011-12-03.[32] Thangapazham RL, Puri A, Tele S, et al. Evaluation of a nanotechnology-based carrier for delivery of curcumin in prostate cancer cells. Int J Oncol. 2008;32(5):1119-1123.[33] Sarin H, Kanevsky AS, Wu H, et al. Effective transvascular delivery of nanoparticles across the blood-brain tumor barrier into malignant glioma cells. J Transl Med. 2008;6:80.[34] Nellis DF, Ekstrom DL, Kirpotin DB, et al. Preclinical manufacture of an anti-HER2 scFv-PEG-DSPE, liposome-inserting conjugate. 1. Gram-scale production and purification. Biotechnol Prog. 2005;21(1):205-220.[35] Nellis DF, Giardina SL, Janini GM, et al. Preclinical manufacture of anti-HER2 liposome-inserting, scFv-PEG-lipid conjugate. 2. Conjugate micelle identity, purity, tability, and potency analysis. Biotechnol Prog. 2005;21(1):221-232.[36] Jia L, Wong H, Wang Y, et al. Carbendazim: disposition, cellular permeability, metabolite identification, and pharmacokinetic comparison with its nanoparticle. J Pharm Sci. 2003;92(1):161-172.[37] Dinh B, Dove K, Jappar D, et al. Effect of hydrophobic structure on the catalysis of nitric oxide release from zwitterionic diazeniumdiolates in surfactant and liposome media. Nitric Oxide. 2005;13(3):204-209.[38] Cachau RE, Gonzalez-Nilo FD, Ventura ON, et al. In-silico nanobio-design. A new frontier in computational biology. Curr Top Med Chem. 2007;7(15):1537-1540.[39] Kosaka N, Bernardo M, Mitsunaga M, et al. MR and optical imaging of early micrometastases in lymph nodes: triple labeling with nano-sized agents yielding distinct signals. Contrast Media Mol Imaging. 2012;7(2):247-253. [40] Clogston JD, Zheng JW, Ramalinga U, et al. ANYL 198-Nanoparticle metrology: Reporting the hydrodynamic size by DLS. Front Nanoscale Mater Anal. The 236th ACS National Meeting, Philadelphia, PA, August 17-21, 2008. |
| [1] | Jiao Hui, Zhang Yining, Song Yuqing, Lin Yu, Wang Xiuli. Advances in research and application of breast cancer organoids [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1122-1128. |
| [2] | Liu Yang, Gong Yi, Fan Wei. Anti-hepatoma activity of targeted Pluronic F127/formononetin nanocomposite system in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 526-531. |
| [3] | Zhou Jihui, Yao Meng, Wang Yansong, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Influence of novel nanoscaffolds on biological behaviors of neural stem cells and the related gene expression [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 532-536. |
| [4] | Liu Jiangfeng. Nano-hydroxyapatite/polyamide 66 composite filling combined with locking plate in the treatment of fibrous dysplasia of femoral bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 542-547. |
| [5] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [6] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| [7] | Chen Song, He Yuanli, Xie Wenjia, Zhong Linna, Wang Jian. Advantages of calcium phosphate nanoparticles for drug delivery in bone tissue engineering research and application [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3565-3570. |
| [8] | Lang Limin, He Sheng, Jiang Zengyu, Hu Yiyi, Zhang Zhixing, Liang Minqian. Application progress of conductive composite materials in the field of tissue engineering treatment of myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3584-3590. |
| [9] | Qin Yanchun, Rong Zhen, Jiang Ruiyuan, Fu Bin, Hong Xiaohua, Mo Chunmei. Chinese medicine compound preparation inhibits proliferation of CD133+ liver cancer stem cells and the expression of stemness transcription factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3016-3023. |
| [10] | Chen Jie, Liao Chengcheng, Chen Zhiwei, Wang Yan. Bladder cancer stem cell markers and related signaling pathways: antibody targeted therapy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3090-3096. |
| [11] | Zhao Shuangdan, Zheng Jiahua, Qi Wenbo, Huang Xianghua. Role and mechanism of exosomes derived from mesenchymal stem cells in reproductive system diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(19): 3097-3102. |
| [12] | Zhang Xianjun, Zhao Xijiang. In vivo osteogenic properties of silicon-incorporated titanium dioxide nanotubes on titanium screw surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2461-2465. |
| [13] | Wang Renxian, Cao Jingjing, Wang Honggang, Wan Ben, Liu Weifeng. Effects of dispersants on aggregation, intracellular distribution and cell proliferation of nano-hydroxyapatite [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2500-2505. |
| [14] | Gan Zhoujie, Pei Xibo. Enzyme-responsive nanoparticles in tumor therapy: superiority of nanoparticles in accumulation and drug release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2562-2568. |
| [15] | Xie Jian, Su Jiansheng. Advantages and characteristics of electrospun aligned nanofibers as scaffolds for tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2575-2581. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||