Chinese Journal of Tissue Engineering Research ›› 2013, Vol. 17 ›› Issue (8): 1505-1512.doi: 10.3969/j.issn.2095-4344.2013.08.028
Previous Articles Next Articles
Hemocompatibility of anticoagulant biomaterials
Zhang Ming-hua1, Zheng Hai-yan2
- 1 Affiliated Hospital of the Armed Police Logistics College, Tianjin 300162, China
2 Department of Outpatient, Military Transportation University, Tianjin 300162, China
-
Received:2012-10-20Revised:2012-12-27Online:2013-02-19Published:2013-02-19 -
Contact:Zheng Hai-yan, Attending physician, Department of Outpatient, Military Transportation University, Tianjin 300162, China -
About author:Zhang Ming-hua, Associate chief technician, Affiliated Hospital of the Armed Police Logistics College, Tianjin 300162, China zhangminghua1234@163.com
CLC Number:
Cite this article
Zhang Ming-hua, Zheng Hai-yan. Hemocompatibility of anticoagulant biomaterials[J]. Chinese Journal of Tissue Engineering Research, 2013, 17(8): 1505-1512.
share this article
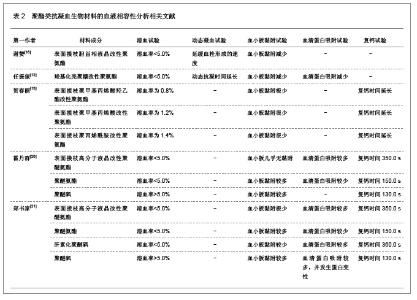
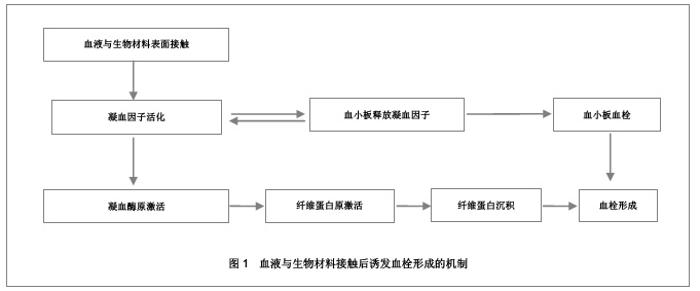
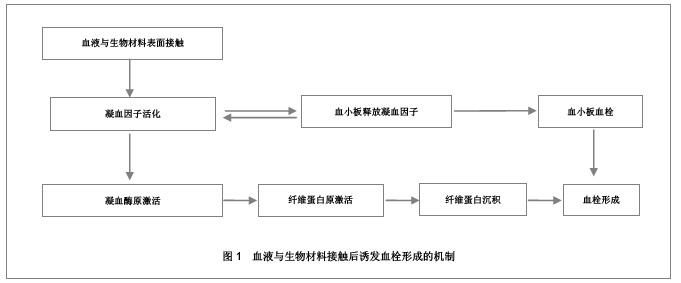
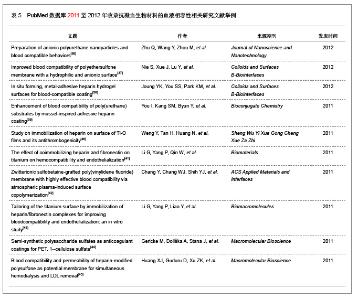
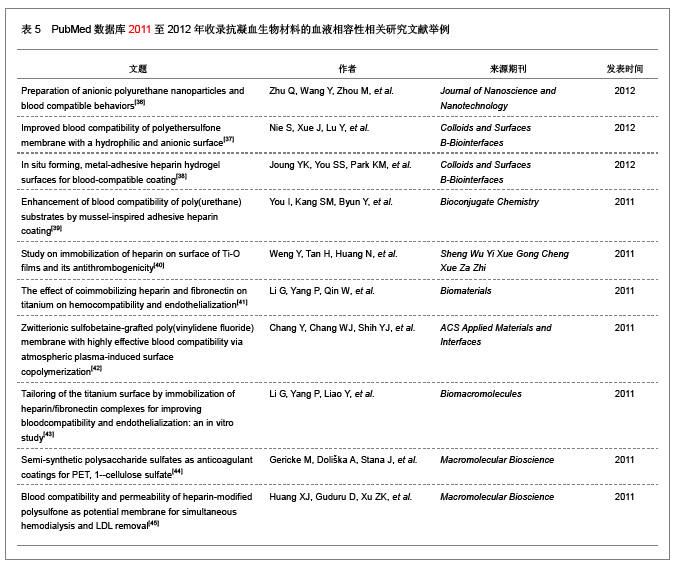
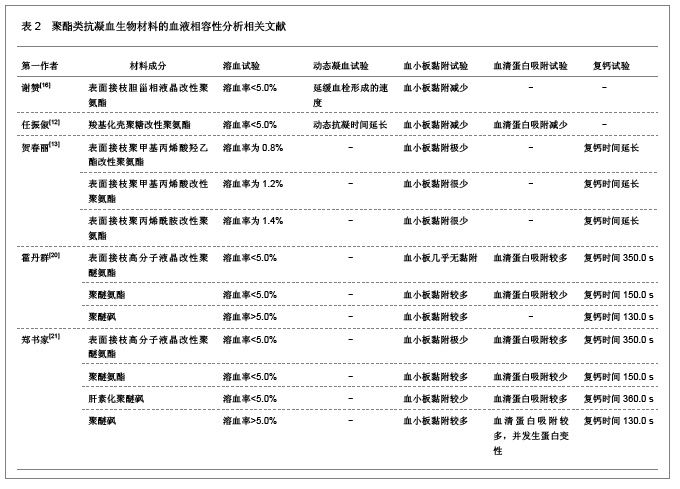
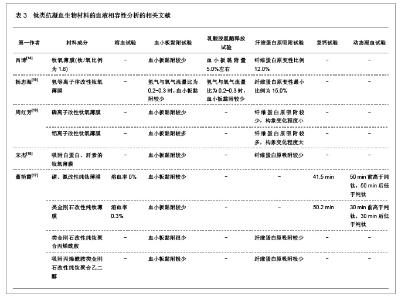
2.2 抗凝血生物材料的血液相容性 2.2.1 聚酯类抗凝血生物材料的血液相容性 抗凝血生物材料分为可降解生物材料和非降解生物材料,可降解生物材料在生物环境的作用下能够发生结构和性能的变化,降解产物能够被机体完全吸收或者排除体外。非降解生物材料能够在机体环境中长期保持结构和性能的稳定,不发生降解和磨损等,具有良好的理化性能。通过人工合成的方法将可降解类生物材料和非降解类生物材料进行聚合改性处理,提高二者的理化性能以及与机体的生物相容性。聚氨酯、聚乙烯、聚丙烯、聚丙烯酸酯等属于非降解生物材料,壳聚糖、纤维素、聚乙烯醇等属于可降解生物材料。谢赞[16]、任振俶[12]、贺春丽[13]、霍丹群[20]和郑书家[21]分别对聚酯类抗凝血生物材料的抗凝血性能进行了分析,发现聚酯类抗凝血生物材料中除了聚醚砜以外,其余大多数聚酯类抗凝血生物材料的溶血率均小于5.0%,不会发生溶血现象,并且血小板的黏附较少,动态凝血时间和复钙时间明显延长,表现出良好的血液相容性。具体分析结果见表2。"
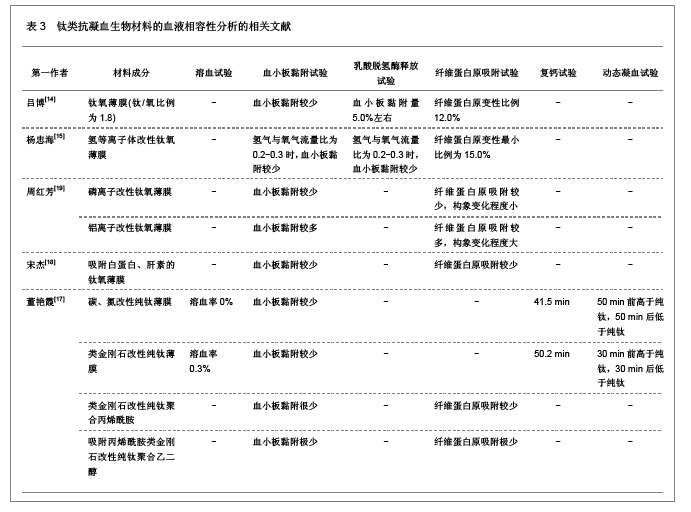
2.2.2 钛类抗凝血生物材料的血液相容性 钛金属具有良好的延展性,密度小,机械强度大,可朔性强,并且具有良好的耐腐蚀性能,不易被酸性和碱性溶液腐蚀变性。此外,钛金属还具有良好的细胞相容性和组织相容性。纯钛及钛金属复合物均有良好的机械性能、理化性能以及良好的组织相容性和血液相容性,是近年来生物材料研究的热点之一。吕博[14]、杨忠海[15]、周红芳[19]、宋杰等[18]和董艳霞[17]分别以纯钛和氧化钛为基体,通过氢离子、磷离子、铝离子等离子体注入以及碳、氮化合物、肝素等多种物质的聚合对钛金属进行改性,并对改性后的钛类抗凝血生物材料的抗凝性能进行了研究分析,发现以钛氧薄膜和纯钛为基体的钛类抗凝血生物材料同样表现出较高的抗凝血性能,纯钛的溶血率为0,而改性后的钛类抗凝血生物材料的溶血率仍小于1.0%,并且血小板的吸附量较少,纤维蛋白原的吸附也较少,动态凝血时间及复钙时间均明显延长,表现出较高的抗凝血性能,具有良好的血液相容性,具体分析结果见表3。"
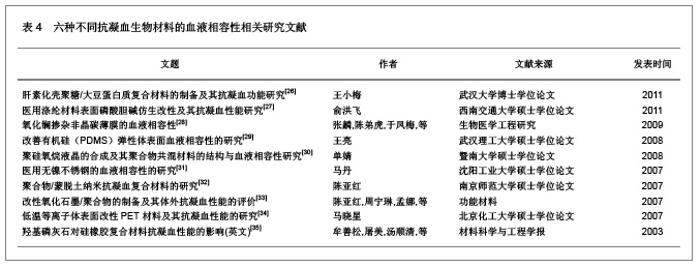
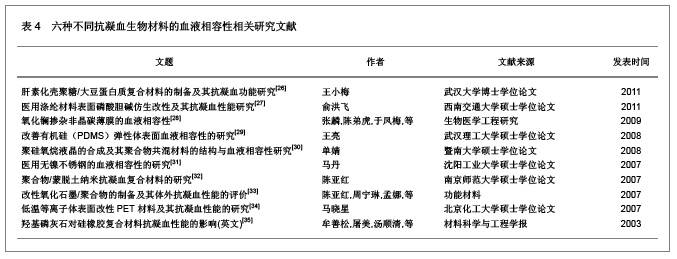
| [1] Macocinschi D, Filip D, Vlad S, et al. Segmented biopolyurethanes for medical applications. J Mater Sci Mater Med. 2009;20(8):1659-1668. [2] Kütting M, Roggenkamp J, Urban U, et al. Polyurethane heart valves: past, present and future. Expert Rev Med Devices. 2011;8(2):227-233. [3] Gastaldello K, Melot C, Kahn RJ, et al. Comparison of cellulose diacetate and polysulfone membranes in the outcome of acute renal failure. A prospective randomized study. Nephrol Dial Transplant. 2000;15(2):224-230. [4] Suzuki Y, Daitoku K, Minakawa M, et al. Poly-2-methoxyethylacrylate-coated bypass circuits reduce activation of coagulation system and inflammatory response in congenital cardiac surgery. J Artif Organs. 2008;11(3): 111-116. [5] Kwok SC, Yang P, Wang J, et al. Hemocompatibility of nitrogen-doped, hydrogen-free diamond-like carbon prepared by nitrogen plasma immersion ion implantation-deposition. J Biomed Mater Res A. 2004;70(1):107-114. [6] Santerre JP, Woodhouse K, Laroche G, et al. Understanding the biodegradation of polyurethanes: from classical implants to tissue engineering materials. Biomaterials. 2005;26(35): 7457-7470. [7] Losi P, Lombardi S, Briganti E, et al. Luminal surface microgeometry affects platelet adhesion in small-diameter synthetic grafts. Biomaterials. 2004;25(18):4447-4455. [8] 沈健.生物医用高分子材料的研制及其基础研究[D].江苏:南京理工大学,2004:1-396.[9] 马银陈,周宁琳,陈亚红,等.新型抗凝血纳米复合材料的制备及其性能研究[J].功能材料,2007,38(10):1713-1716.[10] 程莉萍,孙树东,乐以伦,等.血液净化膜材和抗凝剂对凝血过程中接触活化的影响[J].四川大学学报:医学版,2005,36(3):411-414.[11] 中国知网.中国学术期刊总库[DB/OL].2012-11-18. https://www.cnki.net.[12] 任振俶.羧基化壳聚糖改性聚氨酯的制备和表征[D].辽宁:大连理工大学,2012:1-67.[13] 贺春丽.不同类型亲水结构表面修饰聚氨酯的合成及其生物相容性研究[D].江苏:南京师范大学,2011:1-84.[14] 吕博.抗凝血Ti-O薄膜的电化学行为研究[D].四川:西南交通大学, 2010:1-52.[15] 杨忠海.氢等离子体改性Ti-O薄膜的血液相容性研究[D].四川:西南交通大学,2009:1-87.[16] 谢赞.表面接枝胆甾相液晶改善PU的抗凝血性能研究[D].广东:暨南大学,2007:1-59.[17] 董艳霞.钛表面改性及其血液相容性研究[D].陕西:陕西师范大学, 2007:1-68.[18] 宋杰,吴熹,黄楠,等.纤维蛋白原与吸附白蛋白、肝素的新型血管支架材料氧化钛的血液相容性[J].生物医学工程学杂志,2007, 24(5):1097-1101.[19] 周红芳.离子注入钛氧薄膜的血液相容性研究[D].四川:西南交通大学,2006:1-66.[20] 霍丹群.肝素化高分子液晶/聚醚氨酯生物材料的研究[D].重庆:重庆大学,2004:1-157.[21] 郑书家.抗凝血生物材料的血液相容性研究[D].重庆:重庆大学,2004:1-62.[22] Lelah M, Pierce JA, Lambrecht LK, et al. Polyether-urethane ionomers:surface property/ex vivo blood compatibility relationships. J Colloid Interface Sci. 1985;104(2):422-439. [23] Ito Y, Sisido M, Imanishi Y. Synthesis and antithrombogenicity of anionic polyurethanes and heparin-bound polyurethanes. J Biomed Mater Res. 1986;20(8):1157-1177. [24] Goosen MF, Sefon MV. Properties of a heparin-poly(vinyl alcohol) hydrogelcoating. J Biomed Mater Res. 1993;17: 359-361. [25] Bélanger MC, Marois Y, Roy R, et al. Selection of a polyurethane membrane for the manufacture of ventricles for a totally implantable artificial heart: blood compatibility and biocompatibility studies. Artif Organs. 2000;24(11):879-888. [26] 王小梅.肝素化壳聚糖/大豆蛋白质复合材料的制备及其抗凝血功能研究[D].湖北:武汉大学,2011:1-149.[27] 俞洪飞.医用涤纶材料表面磷酸胆碱仿生改性及其抗凝血性能研究[D].四川:西南交通大学,2011:1-63.[28] 张麟,陈弟虎,于凤梅,等.氧化镧掺杂非晶碳薄膜的血液相容性[J].生物医学工程研究,2009,28(2):96-99. [29] 王亮.改善有机硅(PDMS)弹性体表面血液相容性的研究[D].湖北:武汉理工大学,2008:1-60.[30] 单婧.聚硅氧烷液晶的合成及其聚合物共混材料的结构与血液相容性研究[D].广东:暨南大学,2008:1-90.[31] 马丹.医用无镍不锈钢的血液相容性的研究[D].辽宁:沈阳工业大学,2007:1-66.[32] 陈亚红.聚合物/蒙脱土纳米抗凝血复合材料的研究[D].江苏:南京师范大学,2007:1-75.[33] 陈亚红,周宁琳,孟娜,等.改性氧化石墨/聚合物的制备及其体外抗凝血性能的评价[J].功能材料,2007,38(3):438-440.[34] 马晓星.低温等离子体表面改性PET材料及其抗凝血性能的研究[D].北京:北京化工大学,2007:1-77.[35] 牟善松,屠美,汤顺清,等.羟基磷灰石对硅橡胶复合材料抗凝血性能的影响(英文)[J].材料科学与工程学报,2003,21(2):162-164.[36] Zhu Q, Wang Y, Zhou M, et al. Preparation of anionic polyurethane nanoparticles and blood compatible behaviors. J Nanosci Nanotechnol. 2012;12(5):4051-4056.[37] Nie S, Xue J, Lu Y, et al. Improved blood compatibility of polyethersulfone membrane with a hydrophilic and anionic surface. Colloids Surf B Biointerfaces. 2012;100:116-125. [38] Joung YK, You SS, Park KM, et al. In situ forming, metal-adhesive heparin hydrogel surfaces for blood-compatible coating. Colloids Surf B Biointerfaces. 2012;99:102-107. [39] You I, Kang SM, Byun Y, et al. Enhancement of blood compatibility of poly(urethane) substrates by mussel-inspired adhesive heparin coating. Bioconjug Chem. 2011;22(7):1264-1269.[40] Weng Y, Tan H, Huang N, et al. Study on immobilization of heparin on surface of Ti-O films and its antithrombogenicity. Sheng Wu Yi Xue Gong Cheng Xue Za Zhi. 2011;28(1):86-89.[41] Li G, Yang P, Qin W, et al. The effect of coimmobilizing heparin and fibronectin on titanium on hemocompatibility and endothelialization. Biomaterials. 2011;32(21):4691-4703. [42] Chang Y, Chang WJ, Shih YJ, et al. Zwitterionic sulfobetaine-grafted poly(vinylidene fluoride) membrane with highly effective blood compatibility via atmospheric plasma-induced surface copolymerization. ACS Appl Mater Interfaces. 2011;3(4):1228-1237. [43] Li G, Yang P, Liao Y, et al. Tailoring of the titanium surface by immobilization of heparin/fibronectin complexes for improving bloodcompatibility and endothelialization: an in vitro study. Biomacromolecules. 2011;12(4):1155-1168. [44] Gericke M, Doliška A, Stana J, et al. Semi-synthetic polysaccharide sulfates as anticoagulant coatings for PET, 1--cellulose sulfate. Macromol Biosci. 2011;11(4):549-556.[45] Huang XJ, Guduru D, Xu ZK, et al. Blood compatibility and permeability of heparin-modified polysulfone as potential membrane for simultaneous hemodialysis and LDL removal. Macromol Biosci. 2011;11(1):131-140. [46] Wagner WR, Johnson PC, Thompson KA, et al. Heparin-coated cardiopulmonary bypass circuits: hemostatic alterations and postoperative blood loss. Ann Thorac Surg. 1994;58(3):734-740; discussion 741.[47] Bozdayi M, Borowiec J, Nilsson L, et al. Effects of heparin coating of cardiopulmonary bypass circuits on in vitro oxygen free radical production during coronary bypass surgery. Artif Organs. 1996;20(9):1008-1016. [48] Høgevold HE, Moen O, Fosse E, et al. Effects of heparin coating on the expression of CD11b, CD11c and CD62L by leucocytes in extracorporeal circulation in vitro. Perfusion. 1997;12(1):9-20. [49] Moen O, Fosse E, Brockmeier V, et al. Disparity in blood activation by two different heparin-coated cardiopulmonary bypass systems. Ann Thorac Surg. 1995;60(5):1317-1323. |
| [1] | Li Xingping, Xiao Dongqin, Zhao Qiao, Chen Shuo, Bai Yiguang, Liu Kang, Feng Gang, Duan Ke. Preparation and properties of copper-loaded antibacterial functional film on titanium surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 553-557. |
| [2] | Cheng Jun, Tan Jun, Zhao Yun, Cheng Fangdong, Shi Guojia. Effect of thrombin concentration on the prevention of postoperative cerebrospinal leakage by fibrin glue [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 570-575. |
| [3] | Li Li, Ma Li. Immobilization of lactase on magnetic chitosan microspheres and its effect on enzymatic properties [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 576-581. |
| [4] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [5] | Bi Qingwei, Liu Chengpu, Li Yan, Zhao Wenwen, Han Mei. Structure analysis of platelet-rich fibrin derived from two centrifugation procedures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3534-3539. |
| [6] | Zhou Anqi, Tang Yufei, Wu Bingfeng, Xiang Lin. Designing of periosteum tissue engineering: combination of generality and individuality [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3551-3557. |
| [7] | Lang Limin, He Sheng, Jiang Zengyu, Hu Yiyi, Zhang Zhixing, Liang Minqian. Application progress of conductive composite materials in the field of tissue engineering treatment of myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3584-3590. |
| [8] | Chen Deng, Zhang Yaxin, Dai Jihang, Chen Duoyun, Sun Yu. Analysis on relative factors affecting pyrexia following total hip replacement [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(18): 2846-2850. |
| [9] | Zhang Xianjun, Zhao Xijiang. In vivo osteogenic properties of silicon-incorporated titanium dioxide nanotubes on titanium screw surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2461-2465. |
| [10] | Xie Jian, Su Jiansheng. Advantages and characteristics of electrospun aligned nanofibers as scaffolds for tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2575-2581. |
| [11] | Ji Qi, Yu Zhengwen, Zhang Jian. Problems and trends of technique and clinical application of metallic biomaterials prepared by three-dimensional printing technology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(16): 2597-2604. |
| [12] | Liu Zige, Liu Xinrui, Li Yan, Song Guorui, Zhang Chen, Chen Desheng. In vitro experiment of tetrandrine on the model of osteolysis induced by wear particles around the prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(15): 2358-2363. |
| [13] | Qian Nannan, Zhang Qian, Yang Rui, Ao Jun, Zhang Tao. Mesenchymal stem cells in the treatment of spinal cord injury: cell therapy and combination of new drugs and biomaterials [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2114-2120. |
| [14] | Wu Shengxiang, Liu Yuan, Lu Shuai. Mini-locking titanium plate system fixation in the treatment of carpal scaphoid fracture [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(12): 1874-1878. |
| [15] | Jia Wei, Zhang Mandong, Chen Weiyi, Wang Chenyan, Guo Yuan. Effects of femoral prosthetic materials on artificial knee arthroplasty performance [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1477-1481. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||