Chinese Journal of Tissue Engineering Research ›› 2020, Vol. 24 ›› Issue (2): 276-281.doi: 10.3969/j.issn.2095-4344.1922
Previous Articles Next Articles
Mechanism of action of energy metabolism molecule SIRT1 in improving bone metabolism of type 2 diabetes
Zhang Luyao
- Department of Physical Education, Huaiyin Institute of Technology, Huaian 223003, Jiangsu Province, China
-
Received:2019-05-30Revised:2019-06-04Accepted:2019-07-15Online:2020-01-18Published:2019-12-25 -
About author:Zhang Luyao, Master, Associate professor, Department of Physical Education, Huaiyin Institute of Technology, Huaian 223003, Jiangsu Province, China
CLC Number:
Cite this article
Zhang Luyao. Mechanism of action of energy metabolism molecule SIRT1 in improving bone metabolism of type 2 diabetes[J]. Chinese Journal of Tissue Engineering Research, 2020, 24(2): 276-281.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
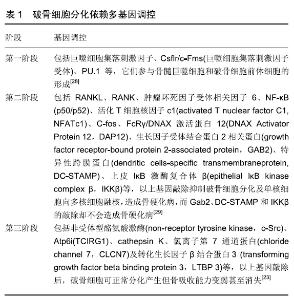
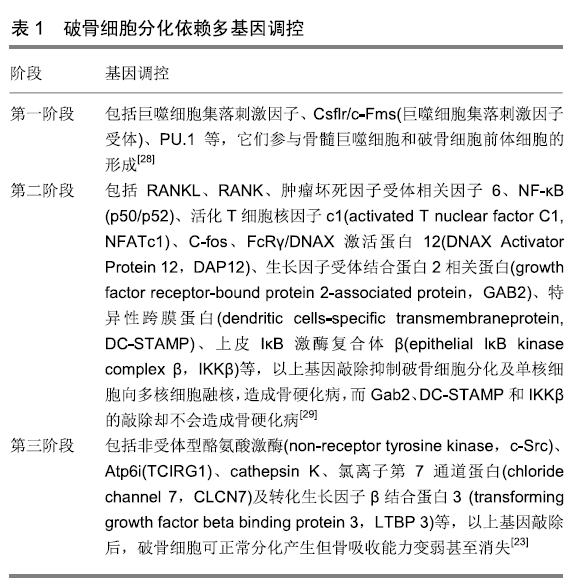
2.1 SIRT1概述 SIRT1作为Sirt家族成员,含有500个氨基酸残基,于1999年在人体中被首次发现,基因定位于染色体10q21.3,全长33 kb,含9个外显子和8个内含子[9]。其DNA双链上含有一长275氨基酸的烟酰胺腺嘌呤二核苷酸(nicotinamide adenine dinucleotide,NAD+)结合区域和催化结构域,其裂隙是底物与SIRT1发生反应的场所。当细胞内NAD+上调时,其介导脱乙酰化(或O-ADP-核糖基化),引起细胞结构和功能的相应改变[10]。 SIRT1作为一种NAD+依赖的Ⅲ型组蛋白去乙酰化酶,能够催化组蛋白底物和非组蛋白底物的乙酰赖氨酸残基的去乙酰化,其通过氧化还原状态的改变及内环境稳态来调控细胞能量代谢等生理过程[11]。蛋白质组学检测小鼠的133个线粒体蛋白发现了277个乙酰化位点,对1 750个蛋白检测发现了3 600个乙酰化位点,表明蛋白的乙酰化/去乙酰化是细胞内分子信号调控途径的重要影响因 素[12]。 SIRT1将蛋白底物中赖氨酸的乙酰基转移到NAD+的二磷酸腺苷核糖上,产生去乙酰化蛋白,烟酰胺(NAM)和2’-O-乙酰基-二磷酸腺苷核糖[13]。进而调控过氧化物酶体增殖物激活受体γ辅激活因子1α(PGC-1α)、叉头框转录因子O亚家族蛋白1(forkhead box O1, FOXO1)、PI3K、肝X受体等去乙酰化,参与炎症、2型糖尿病发生、骨代谢等过程[14]。SIRT1还可通过与各底物结合参与基因修复、细胞代谢、细胞能量平衡和细胞寿命调节等[15]。研究证实,SIRT1不仅是调控糖脂等能量代谢的关键因子,其功能缺失亦是2型糖尿病等能量代谢紊乱型疾病发生及病理改变的始动环节[16]。并且,SIRT1基因表达变化通过调控成骨细胞、破骨细胞分化、功能发挥来影响,2型糖尿病骨代谢。 2.2 2型糖尿病对骨代谢的影响 能量代谢紊乱导致的胰岛素抵抗是2型糖尿病发生的病理生理学基础,而胰岛素抵抗引起的高血糖亦是骨质疏松出现的生物机制[17]。更深一层次,2型糖尿病骨质疏松的发生起于机体内成骨细胞介导的骨形成与破骨细胞介导的骨吸收之间的代谢平衡紊乱。成骨细胞由骨髓间充质干细胞分化产生,而其分化、成熟过程分为4个阶段:增殖、细胞外基质成熟、矿化及凋亡[18]。阶段不同调控的细胞因子亦存在较大差异。2型糖尿病大鼠或小鼠骨形成和骨沉积速率下降导致的骨质疏松与骨髓间充质干细胞分化产生的成骨细胞数量和骨形成能力显著下降密切相关[19]。胰岛素抵抗造成2型糖尿病进而引起胰岛素分泌相对或绝对减少,抑制成骨细胞分化及骨形成[20]。这与胰岛素抵抗水平升高,羧化不全骨钙素去磷酸化抑制β细胞增殖及胰岛素分泌,使得胰岛素受体α亚基不能充分与胰岛素特异结合,抑制β亚基3个酪氨酸残基(Tyr1158、Tyr1162、Tyr1163)磷酸化,下调胰岛素受体表达[21]。胰岛素受体底物1不能正常募集刺激蛋白1和CCAAT增强子结合蛋白β与下游PI3K p85-p110亚基结合,抑制磷脂酰肌醇(3,4)二磷酸(PIP2)和磷脂酰肌醇(3,4,5)三磷酸(PIP3)表达及其与磷脂酰肌醇依赖性激酶1 (phosphoinositide dependent kinase-1,PDK-1)结合,下游Akt丝氨酸残基去磷酸化[22]。关键基因Runx2、Osx和Ⅰ型胶原蛋白表达下调抑制成骨细胞分化及骨形成,导致骨质疏松发生[23]。并且,近来研究发现的能量代谢调控关键因子——蔗糖非酵解型蛋白激酶(sucrose non-fermenting1-related protein kinase,SNRK)亦可通过以上信号途径调控成骨细胞分化[24]。高血糖是2型糖尿病的另一病理表征,当蛋白激酶C活化、糖基化终末产物形成及氧化应激等多途径被激活后会诱导成骨细胞凋亡,下调BGP、转录因子D1x5c-bfa1/Runx2及Ⅰ型胶原等表达和激活环磷酸腺苷/蛋白激酶A/细胞外调节激酶(ERK)/PPAR-γ途径,导致成骨细胞分化被抑制[25]。 破骨细胞由骨髓巨噬细胞分化产生,RANKL和巨噬细胞集落刺激因子在此过程中必不可少,RANK激活促进破骨细胞分化产生[26]。破骨细胞分化依赖多基因调控,而调控啮齿类和人破骨细胞分化的基因至少有24种[27]。根据以上基因在破骨细胞分化、成熟及功能发挥上的作用,将其分为3阶段,见表1。"

2型糖尿病机体破骨细胞分化异常,骨髓巨噬细胞体外诱导分化产生的破骨细胞和多核破骨细胞(≥ 5核)数量及骨吸收功能异常升高[30]。研究亦证实,2型糖尿病促进破骨细胞分化、融核及其骨吸收功能[31-32]。在此过程中,RANK、肿瘤坏死因子α、血管内皮生长因子A及巨噬细胞集落刺激因子等表达显著上调,激活PU.1、GAB2、DC-STAMP、Atp6i(TCIRG1)、CTSK等,促进破骨细胞分化、融核及骨吸收功能[31]。2型糖尿病小鼠胰岛素抵抗水平升高,胰岛素分泌减少,下调胰岛素受体及其靶基因白细胞介素6表达,促进破骨细胞分化[32]。并且,破骨细胞作为新发现的胰岛素作用位点,其胰岛素抵抗水平将会直接影响全身胰岛素抵抗及2型糖尿病并发症骨质疏松发生。有学者在探究SNRK对骨代谢影响时,发现其可通过白细胞介素6调节破骨细胞的胰岛素抵抗标志性羧化不全骨钙素磷酸化及PI3K/Akt途径来影响破骨细胞分化[33]。另一研究中,2型糖尿病小鼠颅骨愈合程度明显低于正常小鼠,仅为后者40%,而利用糖基化终末产物对正常小鼠预处理,其颅骨愈合程度相较于未处理者明显降低,提示糖基化终末产物参与了骨重建过程,具有增加骨吸收、减少骨形成的作用[29]。其机制与高血糖可通过磷酸化糖基化终末产物促进转化生长因子α、CTSK等表达,提高破骨细胞活性,促进骨吸收密切相关[32]。再者,2型糖尿病引起的高血糖可激活于丝裂原活化蛋白激酶(mitogen-activated protein kinase,MAPK)的p38亚型及下游途径CTSK、TRAP等表达上调,促进破骨细胞分化及骨吸收,导致骨密度下降[34]。 2.3 SIRT1介导2型糖尿病骨代谢 SIRT1是调控能量代谢的关键分子,NAD+/NADH比值增加上调其蛋白表达,当比值降低时又会显著抑制SIRT1表达及活性,胰岛素抵抗和血糖水平升高,导致2型糖尿病发生[35]。因此,SIRT1被认为是2型糖尿病发生的启动因子[36]。而有关2型糖尿病中SIRT1生物学作用的相关研究集中在睾丸内质网应激、肝脏脂质沉积、糖尿病肾病、心肌组织损伤等领域[37]。骨代谢中的相关研究较少,当敲除SIRT1后,SIRT1-/-小鼠骨组织病理特征与2型糖尿病小鼠一致,且其骨中Wnt/β-catenin途径被抑制后骨髓间充质干细胞分化产生的成骨细胞数量和骨形成能力降低,股骨和椎骨皮质骨厚度显著下降[38]。并且,SIRT1表达下调抑制2型糖尿病小鼠骨髓间充质干细胞向成骨细胞分化[39]。并且,当利用高糖和脂肪酸模拟体外2型糖尿病对MC3T3-E1细胞的影响时,发现SIRT1表达下调抑制MC3T3-E1向成骨细胞分化,且碱性磷酸酶和茜素红染色显示成骨细胞骨形成能力显著下降[40]。综上表明,2型糖尿病的成骨细胞分化及骨形成能力下降导致的骨质疏松与SIRT1表达下调密切相关。 SIRT1在成骨细胞中表达上调对改善2型糖尿病小鼠骨密度有重要意义,烟酰胺(SIRT1抑制剂)抑制成骨细胞分化,增加骨髓脂肪细胞形成和破骨细胞数量[4]。而其激活剂白藜芦醇在骨髓间充质干细胞向成骨细胞分化中促进SIRT1与PPAR-γ(主要成脂转录因子)结合,抑制PPAR-γ活性,阻碍脂肪细胞分化产生,促进成骨细胞分化[41]。FoxO3a的C末端结构域存在一个与SIRT1的结合位点,一旦结合形成SIRT1-FOXO3A复合物将增强FoxO3a转录活性[42]。而FoxO3a激活后抑制成骨细胞凋亡,改善2型糖尿病骨质疏松。β-catenin是启动成骨转录程序的早期主要转录因子,可诱导成骨细胞特异性基因表达(如骨桥蛋白、骨钙蛋白和碱性磷酸酶等)。SIRT1-FOXO3A的过表达或沉默都影响着β-catenin启动子活性,沉默FOXO1也可降低β-catenin表达[43],并损害骨形成。研究表明,MC3T3-E1细胞转染SIRT1过表达后,会抑制肿瘤坏死因子α诱导的细胞凋亡,提高碱性磷酸酶活性,增加β-catenin及下游Runx2和骨钙蛋白表达,此外还可明显抑制肿瘤坏死因子α诱导NF-κB激活,减少一氧化氮合酶、一氧化氮表达可被SIRT1的抑制剂烟酰胺所逆转[44]。由此可见,SIRT1蛋白通过与PPAR-γ、FoxO3a、FOXO1等蛋白作用来参与2型糖尿病的成骨细胞分化及骨形成过程,而β-catenin蛋白是最终调节目标。β-catenin作为Wnt/β-catenin等途径的下游靶基因,在成骨细胞分化及骨形成上具有重要调控作用[45]。SIRT1表达下调后作用于下游Wnt/β-catenin、TGF-β/骨形态发生蛋白等途径及Runx2、Osx等靶基因表达,抑制成骨细胞分化及骨形成,导致骨质疏松发生[46]。由此可见,SIRT1是研究2型糖尿病影响成骨细胞分化及其骨形成能力的重要靶点。 破骨细胞主导骨吸收,当分化产生的破骨细胞及融核后的多核破骨细胞数量显著增多、骨吸收功能紊乱时,2型糖尿病骨质疏松发生。能量代谢紊乱作为2型糖尿病的发病基础,其亦是调控破骨细胞分化及融核的关键因 素[47]。SIRT1作为能量传感器,将其敲除后小鼠糖脂代谢酶活性及蛋白表达紊乱,出现高血糖和高血脂症状,并且骨组织病理特征与2型糖尿病小鼠表型相一致[48]。SIRT1调控2型糖尿病的破骨细胞分化,基因缺失造成破骨细胞分化、融核及骨吸收功能紊乱[22]。并且,其作为RANKL负调节因子,通过磷酸化RANK及下游C-fos-NFATc1途径,上调关键因子Oscar、CTSK、破骨细胞质子泵亚单位(v-ATPase V0subunit d2,Atp6v0d2)、DC-STAMP等表达,使得破骨细胞分化、融核及骨吸收能力异常升 高[18,49]。因此,2型糖尿病骨中SIRT1表达下调是破骨细胞分化、骨吸收异常升高及骨质疏松发生的原因所在。体外研究中,模拟2型糖尿病体内环境对RAW264.7细胞进行高糖干预时,抑制SIRT1后活化T细胞核因子c1(Activated T nuclear factor C1,NFATc1)、CTSK等靶基因表达上调,破骨细胞分化及其蚀骨能力显著增强[50]。 2.4 SIRT1介导运动改善2型糖尿病骨代谢 2型糖尿病骨质疏松发生与成骨细胞分化被显著抑制密切相关。而SIRT1在2型糖尿病抑制成骨细胞分化上具有重要调控作用。JIA等[51]研究发现,SIRT1-/-小鼠糖脂代谢酶活性及蛋白表达紊乱,出现高血糖和高血脂症状,骨组织病理特征与2型糖尿病小鼠表型相一致。SIRT1作为能量传感器和Wnt/β-catenin途径的调控因子,通过磷酸化下游Runx2、Osx、骨钙蛋白等关键因子表达,促进成骨细胞化及骨形成能力[48]。因此,2型糖尿病骨中SIRT1表达下调是成骨细胞分化、骨形成下降及骨折疏松发生的原因所在。那么,运动是否通过激活SIRT1促进2型糖尿病的成骨细胞分化产生?NAD+/NADH比值增加能激活SIRT1,运动过程中细胞呼吸供给能量阶段,NAD+含量增加且NADH量减少,NAD+/NADH比值增加激活SIRT1[12]。国内外研究及作者前期成果发现,运动改善2型糖尿病的成骨细胞分化及骨质疏松,但相关研究较少,仅局限于BMPs/Smad、TGF-β/Smad途径和cAMP/CREB/ATF4途径[52-54],其分子调控网络尚待完善。基于以上研究,运动可通过激活SIRT1来促进2型糖尿病的成骨细胞分化及骨形成进而改善其骨组织形态微细结构。 骨是机体内运动器官,其腔隙-小管系统可将力学刺激转换为胞内水平的腔液流动,骨细胞、成骨细胞等上的初级纤毛在流动腔液产生的剪切力下发生弯曲,胞内耦联蛋白可将力学刺激信号转到胞内,调控细胞代谢及活性[55]。骨质疏松作为2型糖尿病重要并发症,它的发生与成骨细胞分化、成熟及骨形成能力下降密切相关。而运动可通过促进成骨细胞分化及骨形成来改善骨质疏松,且在运动过程中细胞呼吸供给能量阶段,NAD+含量增加且NADH减少,NAD+/NADH比值增加可激活SIRT1[12]。那么,SIRT1作为2型糖尿病始动因子且运动亦可将其激活,运动促2型糖尿病的成骨细胞分化是否通过SIRT1进行调控呢?其分子机制又是否是通过Wnt/β-catenin途径来实现呢?目前,有关运动通过SIRT1调控2型糖尿病破骨细胞分化的体内研究较少,BOYLE等[56]研究发现,8周跑台运动可显著上调2型糖尿病大鼠骨中SIRT表达,抑制RANK及下游C-fos-NFATc1途径,上调关键因子CTSK、Atp6v0d2等表达,抑制破骨细胞分化、融核。RANK、RANKL激活NF-κB的同时可上调P300表达,促进NF-κB乙酰化来调控破骨细胞分化[57-58]。运动干预后,SIRT1表达上调并与P300在破骨细胞前体细胞中结合,导致NF-κB去乙酰化,并抑制IκBα酶激活及其磷酸化、降解,导致破骨细胞分化减少且骨吸收功能下降[35]。而对模拟2型糖尿病的体外高糖环境下RAW264.7细胞施加力学刺激时,发现SIRT1蛋白表达上调,且分化产生的破骨细胞数量及骨吸收能力显著下降[40]。利用SIRT1siRNA对RAW264.7进行转染低表达后,破骨细胞分化下降,而力学刺激可显著改善细胞转染对其造成的作用影响[59]。2型糖尿病抑制破骨细胞分化的研究较多,对其机制研究较深入,除骨保护素(OPG)/ RANKL/RANK分子轴外,发现钙调磷酸酶/NFAT、PI3K/Akt、转化生长因子α等途径或关键分子具有重要调控作用,但有关SIRT1介导运动抑制2型糖尿病破骨细胞分化的相关研究尚待补充。 "

| [1] MACDONALD IA. A review of recent evidence relating to sugars, insulin resistance and diabetes. Eur J Nutr.2016; 55(2):17-23. [2] USALA RL, FERNANDEZ SJ, METE M, et al. Hyponatremia is associated with increased osteoporosis and bone fractures in patients with diabetes with matched glycemic control. J Endocr Soc.2019; 3(2):411-426. [3] ZHOU S, TANG XQ, CHEN HZ. Sirtuins and insulin resistance. Front Endocrinol.2018;9(3):748. [4] KAYVAN Z, CASSIE JL, ALISON L MC, et al. SIRT1 is a positive regulator of in vivo bone mass and a therapeutic target for osteoporosis. PLoS One.2017;12(9):e0185236. [5] LIU W, ZHANG LQ, XUAN K, et al. Alkaline phosphatase controls Lineage switching of mesenchymal stem cells by regulating the LRP6/GSK3β complex in hypophosphatasia. Theranostics. 2018; 8(20):5575-5592. [6] MELONI L,VERSTREPEN L,KREIKE M,et al.Mepazine inhibits RANK-induced osteoclastogenesis independent of its MALT1 inhibitory function. Molecules.2018;23(12):3144. [7] LOMBARDI G, ZIEMANN E, BANFI G. Physical activity and bone health: What is the role of immune system? A narrative review of the third way. Front Endocrinol.2019;10:60. [8] SPINDLER C, SEGABINAZI E, MEIRELES A, et al. Paternal physical exercise modulates global DNA methylation status in the hippocampus of male rat offspring. Neural Regen Res. 2019;14(3): 491-500. [9] WANG SS, LUO Q, FAN PH.Cannabisin F from hemp (Cannabis sativa) seed suppresses lipopolysaccharide-induced inflammatory responses in BV2 microglia as SIRT1 modulator. Int J Mol Sci. 2019;20(3):507. [10] RICKERT E, FERNANDEZ MO, CHOI I, et al. Neuronal SIRT1 regulates metabolic and reproductive function and the response to caloric restriction. J Endocr Soc.2019; 3(2):427-445. [11] JĘŚKO H, WENCEL P, STROSZNAJDER RP, et al. Sirtuins and their roles in brain aging and neurodegenerative disorders. Neurochem Res.2017;42(3):876-890. [12] LIU XF, CAI SY, ZHANG CF, et al. Deacetylation of NAT10 by Sirt1 promotes the transition from rRNA biogenesis to autophagy upon energy stress. Nucleic Acids Res. 2018;46(18):9601-9616. [13] ONOFRIO ND,SERVILLO L, BALESTRIERI ML. SIRT1 and SIRT6 signaling pathways in cardiovascular disease protection. Antioxid Redox Signal. 2018; 28(8):711-732. [14] ABDESSELEM H,MADANI A,HANI A, et al. SIRT1 limits adipocyte hyperplasia through c-myc inhibition. J Biol Chem. 2016; 291(5):2119-2135. [15] LI MZ, ZHENG LJ, SHEN J, et al. SIRT1 facilitates amyloid beta peptide degradation by upregulating lysosome number in primary astrocytes. Neural Regen Res.2018;13(11):2005-2013. [16] ALI S, NAFIS S, KALAIARASAN P, et al.Understanding genetic heterogeneity in type 2 diabetes by delineating physiological phenotypes: SIRT1 and its gene network in impaired insulin secretion. Rev Diabet Stud.2016;13(1):17-34. [17] KIM YA, PARK JB, WOO MS, et al. Persistent organic pollutant-mediated insulin resistance. Int J Environ Res Public Health.2019;16(3):448. [18] AGAS D, GUSMÃO SILVA G, LAUS F, et al. INF-γ encoding plasmid administration triggers bone loss and disrupts bone marrow microenvironment. J Endocrinol.2017; 232 (2):309-321. [19] ZHANG WL, MENG HZ, YANG RF, et al. Melatonin suppresses autophagy in type 2 diabetic osteoporosis. Oncotarget. 2016; 7(32):52179-52194. [20] MA XY, CHEN FQ, HONG H, et al. The relationship between serum osteocalcin concentration and glucose and lipid metabolism in patients with type 2 diabetes mellitus-the role of osteocalcin in energy metabolism. Ann Nutr Metab.2015;66(2):110-116. [21] HAN J, WEI M, WANG Q, et al. Association of genetic variants of SIRT1 with type 2 diabetes mellitus. Gene Expr. 2015;16(4): 177-185. [22] IYER S, HAN L, BARTELL SM, et al.Sirtuin1 (Sirt1) promotes cortical bone formation by preventing β-catenin sequestration by FoxO transcription factors in osteoblast progenitors. J Biol Chem. 2014;289(35):24069- 24078. [23] ALBIERO M, PONCINA N, TJWA M, et al. Diabetes causes bone marrow autonomic neuropathy and impairs stem cell mobilization via dysregulated p66Shc and Sirt1. Diabetes.2014; 63(4):1353-1365. [24] STEPHANIE MC, VIJESH JB, BAO XP, et al. Sucrose non-fermenting related kinase enzyme mediated Rho-associated kinase signaling is responsible for cardiac function. Circ Cardiovasc Genet.2016;9(6):474-486. [25] MCCARTY MF. AMPK activation-protean potential for boosting healthspan. Age (Dordr). 2014;36(2):641-663. [26] PEREIRA M, GOHIN S, LUND N, et al. Sclerostin does not play a major role in the pathogenesis of skeletal complications in type 2 diabetes mellitus. Osteoporos Int.2017; 28(1):309-320. [27] CARLOS VG, ALICE C, AUDREY P, et al. The nuclear factor-erythroid 2-related factor/heme oxygenase-1 axis is critical for the inflammatory features of type 2 diabetes-associated osteoarthritis. J Biol Chem.2017;292(35):14505-14515. [28] QIAO YQ, JIANG PF, GAO YZ.Lutein prevents osteoarthritis through nrf2 activation and downregulation of inflammation. Arch Med Sci. 2018;14(3):617-624. [29] LIU W, ZHOU L, ZHOU C, et al. GDF11 decreases bone mass by stimulating osteoclastogenesis and inhibiting osteoblast differentiation. Nat Commun.2016;22(7):12794. [30] LEE SH, PARK Y, SONG M, et al. Orai1 mediates osteogenic differentiation via BMP signaling pathway in bone marrow mesenchymal stem cells. Biochem Biophys Res Commun.2016; 473(4):1309-1314. [31] PATEL JJ, BUTTERS OR, ARNETT TR.PPAR agonists stimulate adipogenesis at the expense of osteoblast differentiation while inhibiting osteoclast formation and activity. Cell Biochem Funct. 2014; 32(4):368-377. [32] GALLAGHER EJ, SUN H, KORNHAUSER C, et al. The effect of dipeptidyl peptidase-IV inhibition on bone in a mouse model of type 2 diabetes. Diabetes Metab Res Rev.2014; 30(3):191-200. [33] FU C, ZHANG X, YE F, et al. High insulin levels in KK-Ay diabetic mice cause increased cortical bone mass and impaired trabecular micro-structure. Int J Mol Sci.2015; 16(4):8213-8226. [34] SHAKIBAEI M, BUHRMANN C, MOBASHERI A. Resveratrol- mediated SIRT-1 interactions with p300 modulate receptor activator of NF-κB ligand(RANKL) activation of NF-κB signaling and inhibit osteoclastogenesis in bone-derived cells. J Biol Chem. 2011; 286(13):11492-11505. [35] SHAKIBAEI M, SHAYAN P, BUSCH F, et al. Resveratrol mediated modulation of Sirt-1/ Runx2 promotes osteogenic differentiation of mesenchymal stem cells: potential role of Runx2 deacetylation. PLoS One.2012;7(4):e35712. [36] TSENG PC, HOU SM, CHEN RJ, et al. Resvertrol promotes osteogenesis of human mesenchymal stem cells by upregulating RUNX2 gene expression via the SIRT1 / FOXO3A axis. J Bone Miner Res.2011;26(10):2552-2563. [37] BAE SJ, SHIN MW, SON T, et al. Ninjurin1 positively regulates osteoclast development by enhancing the survival of prefusion osteoclasts. Exp Mol Med. 2019; 51(1):7. [38] HSIEH YM, LEE WJ, SHEU WH, et al. Inpatient screening for albuminuria and retinopathy to predict long-term mortality in type 2 diabetic patients: a retrospective cohort study. Diabetol Metab Syndr.2017;9(3):29-36. [39] LAURA JL, ANIL KC, AIDI N, et al. Increased trabecular bone and improved biomechanics in an osteocalcin-null rat model created by CRISPR/Cas9 technology. Dis Model Mech.2016; 9(10):1169-1179. [40] TANG YP, LIU JY, YAN YM, et al. 1,25-dihydroxyvitamin-D3 promotes neutrophil apoptosis in periodontitis with type 2 diabetes mellitus patients via the p38/MAPK pathway. Medicine (Baltimore). 2018;97(52):e13903. [41] KITADA M, KOYA D. SIRT1 in Type 2 Diabetes: mechanisms and therapeutic potential. Diabetes Metab J. 2013; 37(5):315-325. [42] GONG K, QU B, LIAO D, et al.MiR-132 regulates osteogenic differentiation via downregulating Sirtuin1 in a peroxisome proliferator-activated receptor β/δ-dependent manner. Biochem Biophys Res Commun.2016; 478(1):260-267. [43] DUCY P. Cbfa1: a molecular switch in osteoblast biology. Dev Dyn. 2000;219(4):461-471. [44] TEIXEIRA CC, LIU Y, THANT LM, et al. Foxo1, a nover regulator of osteoblast differentiation and skeletogenesis. J Biol Chem. 2010;285(40):31055-31065. [45] DAO DY, JONASON JH, ZHANG YC, et al. Cartilage-specific β-CATENIN signaling regulates chondrocyte maturation, generation of ossification centers, and perichondrial bone formation during skeletal development. J Bone Miner Res. 2012; 27(8):1680-1694. [46] FRANCESCA BL, WILKINSON FL, ALEX WW, et al. The Interplay of SIRT1 and Wnt Signaling in Vascular Calcification. Front Cardiovasc Med. 2018;5:183. [47] ZHANG L, MENG S, TU QS, et al. Adiponectin ameliorates experimental periodontitis in diet-Induced obesity mice. PLoS One. 2014;9(5):e97824. [48] CAO X, LI H, TAO H, et al. Metformin inhibits vascular calcification in female rat aortic smooth muscle cells via the AMPK-eNOS-NO pathway. Endocrinology.2013;154(10):3680-3689. [49] HSIAO EC, MILLARD SM, NISSENSON RA.Gs/Gi Regulation of bone cell differentiation: review and insights from engineered receptors. Horm Metab Res.2016;48(11):689-699. [50] JIA YH, ZHENG Z, WANG YC, et al. SIRT1 is a regulator in high glucose-induced inflammatory response in raw264.7 cells. PLoS One.2015;10(3):e0120849. [51] JIA S, HU Y, ZHANG W,et al.Hypoglycemic and hypolipidemic effects of neohesperidin derived from citrus aurantium L in diabetic KK-A(y) mice. Food Funct.2015;6(3):878-886. [52] 陈祥和,彭海霞,李世昌,等.不同运动方式对Ⅱ型糖尿病小鼠骨形成及BMPs/Smad途径相关分子表达的影响[J].北京体育大学学报, 2017,40(05):51-58. [53] 陈祥和,彭海霞,孙朋,等.不同力学刺激对T2DM小鼠骨中TGF-β/ Smad途径及骨形成的影响[J].上海体育学院学报,2018,42(3): 95-102. [54] 陈祥和,孙朋,陈爱国,等.不同运动对2型糖尿病小鼠骨中cAMP/ CREB/ Atf4途径及骨形成的影响[J].中国运动医学杂志,2017,36(11):977-983. [55] BONEWALD LF. The amazing osteocyte. J Bone Miner Res.2011; 26(2):229-238. [56] BOYLE WJ, SIMONET WS, LACEY DL. Osteoclast differentiation and activation. Nature.2003;423( 6937):337-342. [57] 刘波,谢珍,徐彭.Sirt1与骨质疏松症的研究进展[J].中国药理学学报, 2013,29(8):1054-1056. [58] 李俊.有氧运动对2型糖尿病大鼠血管炎症及SIRT1/NF-κB信号通路的影响[J].北京体育大学学报,2018,41(5):57-63. [59] ZHANG RK, LI GW, ZENG C, et al. Mechanical stress contributes to osteoarthritis development through the activation of transforming growth factor beta 1 (TGF-β1). Bone Joint Res. 2018;7(11):587-594. |
| [1] | Tan Xinfang, Guo Yanxing, Qin Xiaofei, Zhang Binqing, Zhao Dongliang, Pan Kunkun, Li Yuzhuo, Chen Haoyu. Effect of uniaxial fatigue exercise on patellofemoral cartilage injury in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(在线): 1-6. |
| [2] | Wu Min, Zhang Yeting, Wang Lu, Wang Junwei, Jin Yu, Shan Jixin, Bai Bingyi, Yuan Qiongjia. Effect of concurrent training sequences on body composition and hormone response: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1305-1312. |
| [3] | Gu Zhengqiu, Xu Fei, Wei Jia, Zou Yongdi, Wang Xiaolu, Li Yongming. Exploratory study on talk test as a measure of intensity in blood flow restriction training [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1154-1159. |
| [4] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [5] | Shui Xiaoping, Li Chunying, Li Shunchang, Sun Junzhi, Su Quansheng . Effects of aerobic and resistance exercises on brain-derived neurotrophic factor, nuclear factor-kappa B and inflammatory cytokines in skeletal muscle of type II diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 669-675. |
| [6] | Liu Yiyi, Qiu Junqiang, Yi Longyan, Zhou Cailiang. Effect of resistance training on interleukin-6 and C-reactive protein in middle-age and elderly people: a Meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 804-812. |
| [7] | Deng Shuang, Pu Rui, Chen Ziyang, Zhang Jianchao, Yuan Lingyan . Effects of exercise preconditioning on myocardial protection and apoptosis in a mouse model of myocardial remodeling due to early stress overload [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 717-723. |
| [8] | Mo Weibin, Huang Tianchang, Zeng Zhiwei, Yan Linbo. Effects of Pueraria lobata flavonoids on expressions of beta-catenin and glycogen synthase kinase 3beta in the brain of rats undergoing exhaustive exercise after long endurance exercise [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 736-741. |
| [9] | Wei Xing, Liu Shufang, Mao Ning. Roles and values of blood flow restriction training in the rehabilitation of knee joint diseases [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 774-779. |
| [10] | Tang Jiping, Zhang Yeting. Exercise regulates adult hippocampal neurogenesis in Alzheimer’s disease: mechanism and role [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 798-803. |
| [11] | Wei Zhoudan, Li Wenjin, Zhu Li, Wang Yu, Zhao Jiaoyang, Chen Yanan, Guo Dong, Hao Min. Platelet-rich fibrin as a material for alveolar ridge preservation significantly reduces the resorption of alveolar bone height and width after tooth extraction: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 643-648. |
| [12] | Jia Ruizhen, Jiang Chao, Jin Qiguan, Lou Xujia, Hu Yulong. Effects and mechanism of the combined use of L-carnitine, pantothenic acid and coenzyme Q10 on aerobic exercise-induced fatigue in mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 165-170. |
| [13] | Sun Youqiang, Ma Chao, Liang Mengmeng, Xin Pengfei, Zhang Hua, Xiang Xiaobing. The pivotal role of autophagy in bone cells: bone-related cell activity and bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 276-282. |
| [14] | Zeng Xinyu, Chen Xianghe, Liu Bo, Lu Pengcheng, Jin Shengjie, Li Wenxiu, Tian Zhikai, Sun Changliang. Mechanism of exercise improving bone metabolism in type 2 diabetics mellitus based on "Muscle-Bone" Crosstalk [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 289-295. |
| [15] | Kan Jie. Guidelines for exercise during pregnancy in developed countries and revelation for the Healthy China Action [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(2): 308-314. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||