Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (6): 971-977.doi: 10.3969/j.issn.2095-4344.1548
Previous Articles Next Articles
Maxillary sinus floor augmentation: with or without bone grafting
Li Fang, Cheng Yuting, Huang Xiaolin, Zhou Qian, Wu Chao, Shi Qianhui, Wang Yong, Liao Jian
- School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2018-10-26Online:2019-02-28Published:2019-02-28 -
Contact:Liao Jian, MD, Associate professor, Associate chief physician, Master’s supervisor, School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Li Fang, Master candidate, School/Hospital of Stomatology of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:the National Natural Science Foundation of China, No. 81660179 (to LJ); the Science and Technology Plan Project of Guizhou Province, No. LH[2016]7257 (to LJ)
CLC Number:
Cite this article
Li Fang, Cheng Yuting, Huang Xiaolin, Zhou Qian, Wu Chao, Shi Qianhui, Wang Yong, Liao Jian. Maxillary sinus floor augmentation: with or without bone grafting[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(6): 971-977.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 上颌窦底提升术 上颌窦底提升术又称上颌窦底骨增量术,是解决口腔种植中上颌后牙区牙槽骨骨量不足最有效而可靠的手段。目前最常用且较为有效的方法主要有2种:上颌窦底外提升术(又称外侧壁开窗法)和上颌窦底内提升术(又称穿牙槽嵴顶法)。当上颌窦底骨高度严重不足,在行骨增量同期需要植入种植体时,上颌窦底内、外提升术可能需要同时用到。 2.1.1 上颌窦底外提升术 上颌窦底外提升术是在20世纪70年代中期由Tatum发展而来的,包括翻入式、揭盖式及磨除式3种方法。前者技术包括轻敲侧方骨壁将其作为移植材料上方的顶部,后两者技术可彻底移开骨壁,可使移植材料更好地进入窦底[2]。对于这3种技术来说,最重要的是从周围骨壁掀起窦膜,确保到达牙槽嵴顶骨壁,以获得移植材料的空间。窦膜提升高度可达10-12 mm[3],在形成足够的空间之后,移植材料应该充满准备好的窦腔,用生物膜覆盖在骨窗内移植材料上,以促进重要骨的形成,最后是关闭黏骨膜瓣,以减少口腔微生物对移植材料的污染。目前,这种外科手术仍然是种植体植入前或同期植入时提升上颌后部牙槽骨高度的常用方法,且效果已得到认 可[4-6]。其优点有:①外提升入路时可提升的高度较高;②可在直视下进行手术,有效保护窦黏膜;③若术中发生黏膜,可及时进行处理。缺点为:①术中涉及范围广、创伤大;②术后反应明显、费用高;③愈合期及修复时间较长。 2.1.2 上颌窦底内提升术 Summer在1994年首次描述了上颌窦底内提升术,其创伤较小,通过从正常预备的种植窝入路,使用钻针预备到距离窦底1.0-2.0 mm,最后一级扩孔钻预备后,将骨挤压器放入种植窝,进行敲击,使窦底出现青枝骨折,压向窦腔并抬高窦膜来提升上颌窦底。后来他又改善了此项技术,在预备好的种植窝内加入骨移植材料后进行敲击,使包含血液的移植材料从断裂骨壁周围进入窦底黏膜下方,由于机械冲击力和液压原理的协同作用,可安全、快速地剥离周围黏膜,有效地增加窦底高度。经研究发现,窦膜提升的高度可达2.5-5.7 mm[3]。此术的优点有:简化手术操作,缩短手术和愈合时间,减小术后反应;缺点为:提升的高度有限,不能在直视下操作,黏膜穿孔时不能及时发现和修补。 2.1.3 术式选择 以往主要是根据剩余牙槽骨高度决定术式:当剩余骨高度>5 mm时,上颌窦内提升术是较为理想的选择;当剩余骨高度≤5 mm时,认为应该使用上颌窦外提升术[7-9]。也有学者认为,为了获取种植体的初期稳定性,要求剩余牙槽骨高度大于4.0-5.0 mm[10]。随着种植技术逐渐的成熟和器械的逐渐改进,上颌后牙区行种植的适应证也逐渐扩大。一般情况下,当窦底提升高度≤3 mm时,仅使用上颌窦底内提升术而不需要骨移植材料即可;当窦底提升高度>3 mm时,术中需要骨移植材料来获得充足的骨量;当窦底提升高度>5 mm时,往往需要上颌窦外提升术+骨移植材料才能获得良好的临床效果。在近期研究中,有学者将患者的上颌后牙区剩余骨高度分为2 mm≤剩余骨高度<4 mm组、4 mm≤剩余骨高度<6 mm组、6 mm≤剩余骨高度<8 mm组进行比较,结果发现3组中种植体初期稳定性差异无统计学意义,为种植体初期稳定性与剩余牙槽骨高度无关,而与周围皮质骨厚度密切相关[11]。相同的结果也出现在了Gonzalez等[12]的研究中,其将102例患者分为剩余骨高度≤4 mm组和剩余骨高度>4 mm组2组,2组的种植体的成功率分别为100%,98.51%,再一次证明了决定种植体初期稳定性的关键因素不是剩余骨高度,而是其周围骨密度[13-14]。 2.2 骨移植材料的应用 将骨移植材料放入上颌窦底中,现已被广泛应用于临床,其效果也得到了高度认可。目前临床上的主要骨移植材料包括:自体骨、同种异体骨、异种骨、人工骨材料。 2.2.1 自体骨 自体骨是一种较为理想的骨移植材料,拥有其他材料不能达到的优良性能[15-17]。自体骨内包含有活细胞、可吸收的引导骨支架及多种细胞因子,其常取自于下颌颏部、升支和外斜线,上颌结节,髂骨等处。自体骨移植提高了种植体存活率[4],且骨生成量最多[4,18-19],在上颌窦底骨增量中被认为是“金标准”[20-24],因为它表现出理想的骨增量特征:骨生成、骨诱导和骨传导特性。另外,Pisoni等[25]比较了微粒和块状自体骨移植的上颌窦底垂直骨增量和骨吸收情况,结果发现两者差异有统计学意义,并建议口腔医生应在任何可行的情况下选择块状自体骨移植。但取自体骨的限制也非常多:创伤大、取骨量有限、吸收过快[26]、较高的供区发病率、成本高昂等。因此,有学者开始使用自体牙作为一种新的骨替代材料。 自体牙来源于新鲜拔除的第三磨牙、乳牙、前磨牙,或是患有牙周病的牙齿经脱矿后制成,由55%的无机材料和45%的有机材料组成,还包括4种类型的磷酸钙:羟基磷灰石、磷酸三钙、磷酸八钙和无定形磷酸钙,是具有骨诱导性和骨传导性的生物相容性材料。Kim等[27]将自体牙作为上颌窦底骨增量的骨替代材料,行同期植入种植体,结果发现种植体周围获得了充足的骨量,取得了良好的临床效果。又因其可减少费用,因此被认为可作为良好的骨替代材料[28]。但其冠部自体牙结晶含量高,存在导致缓慢吸收和骨传导性较差的趋势;根部自体牙结晶含量低,有致骨质疏松的倾向。 2.2.2 同种异体骨 同种异体骨是目前骨科最常见的骨替代材料,具有骨传导特性,其通常是从人类尸体中获得,与自体骨相比具有可控性。由于处理过程中成骨细胞等成分已被去除,一般同种异体骨不具有成骨性。同种异体骨包括同种异体新鲜冷冻骨、同种异体冻干骨、同种异体脱水矿化骨。由于其存在免疫排斥风险,因此不是上颌窦底骨增量的首选。相比于窦底提升术中使用自体骨,新鲜冷冻骨表现出相似的临床结果[29]。组织学结果表明,同种异体新鲜冷冻骨中的成骨细胞与类骨质基质紧密接触,通过移植骨颗粒与新骨之间形成桥连接,具有与自体骨相当的骨传导性。Reikeras等[30]在同种异体移植物潜在免疫原性研究中发现,-80 ℃储存条件具有消除免疫原性的潜力。因此临床上使用的同种异体新鲜冷冻骨,已通过组织处理和消毒作用阻断了疾病的传播。同种异体冻干骨是采用冻干法处理得到的骨材料,其保留了有机和无机成分(钙和磷酸盐),当用作移植材料时,矿物质含量被破骨细胞分解,成为诱导新骨形成的诱导蛋白,能在较短时间内形成较多的骨组织[21,31]。虽然脱矿的同种异体冻干骨具有骨诱导潜力,但也存在更高的吸收率和更低的体积稳定性;同种异体脱水矿化骨是从尸体松质骨或皮质骨经矿化溶剂脱水得到,保留了骨结构的生物力学特性,最大限度地降低了抗原性和感染潜力[21]。从目前的组织学研究来看,同种异体脱水矿化骨样本显示出较高的生物相容性,无急性炎症反应,新骨与移植颗粒间可形成桥接[21,32]。与自体骨相比,同种异体脱水矿化骨形成的新骨量略少,但差异并不显著[21]。 2.2.3 异种骨 随着临床工作中对骨替代材料的需求不断增长,自体骨与同种异体骨的来源无法达到其需求量,因此对于新材料的研究便成为了重中之重。高等哺乳动物骨组织在成分、结构上与人体骨组织相似,力学性能接近同种骨。异种骨的材料来源较广,可大量获取,但相比于同种异种骨而言,其更易引起生物体免疫反应。因此,降低异种骨的免疫原性,减少机体对移植物的排异反应是目前研究的重点。 常见的处理异种骨方法有超低温冷冻、反复冻融、煅烧、照射、脱蛋白或联合多种方法运用[33],不同处理方法对异种骨的力学特性与生物学特性影响不同。 Bio-oss骨粉:是脱蛋白的小牛骨,与人类松质骨具有相似的多孔性,兼备良好的骨传导性和骨诱导性[31,34-37],是应用最广的骨替代品,其成骨效果已得到认可[38]。有专家比较Bio-Oss骨粉、自体骨与种植体骨结合的效果,3个月时发现,Bio-Oss、自体骨接触种植体的面积分别为52.16%和11.46%;6个月时,Bio-Oss的接触率为63.43%,自体骨为42.22%,因此认为Bio-Oss可作为永久性移植材料[39]。Starch-Jensen等[40]用循证医学和Meta分析法检验上颌窦底骨增量中使用自体骨移植相比于自体骨与Bio-Oss骨粉混合或单独使用Bio-Oss骨粉5年的治疗效果,自体骨移植的种植体存活率为97%,单独Bio-Oss骨粉的种植体存活率95%。因此其结论为Bio-Oss骨粉可作为理想的骨移植材料。 马衍生骨:是一种马衍生的骨组织,通过蛋白水解低温过程脱毒,保留了Ⅰ型骨胶原并使其成为无机物,保持了羟基磷灰石的矿物结构不变[41]。当移植6个月后,在马衍生骨颗粒间可看到骨小梁和血管形成,表面有成骨细胞附着[21]。马衍生骨快速形成血管及新骨的能力,可促进种植体的早期骨结合。与脱矿牛骨相比,马衍生骨能生成更多的新骨及残留更少的移植材料,在骨替代材料中也是不错的选择,但在临床上尚未得到普及。 2.2.4 人工骨材料 目前应用的人工骨材料主要有:生物陶瓷(羟基磷灰石,磷酸三钙,磷酸钙水泥)、高分子聚合物。此类材料主要用作骨传导支架。与异体骨和异种骨相比,人工骨材料的制备方法、降解和力学特性可控,可塑性强且无抗原性和疾病传播的潜在风险,具有巨大的临床应用和开发潜力[42]。 生物陶瓷:主要包括羟基磷灰石、磷酸三钙、磷酸钙水泥、双相磷酸钙陶瓷等。羟基磷灰石是一种生物活性陶瓷,主要为人体硬组织(骨骼和牙齿)的无机成分,因其具有良好的生物相容性,成为骨组织工程中广泛研究和应用的生物材料[43-46]。羟基磷灰石植入体内后,可与周围骨组织形成直接的化学键结合,具有骨传导性、无毒、非炎性、无免疫性等特征。在理化性能上主要分为致密羟基磷灰石和多孔羟基磷灰石,其强度与组成有关,结晶度和孔隙度有关,见表1。"

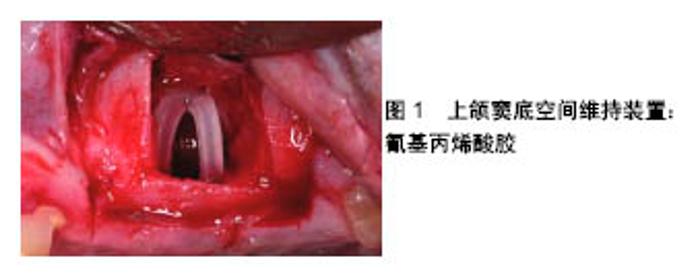
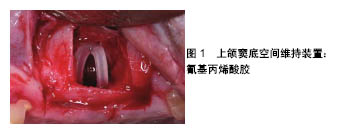
致密羟基磷灰石的压缩强度和抗弯强度较高,多孔羟基磷灰石的强度随着孔隙率的增加而下降。人工合成羟基磷灰石与天然羟基磷灰石在化学组成上非常接近,其组成成分Ca/P为1.67,具有一定的生物活性,植入骨内后通过释放磷酸盐及钙离子在骨组织周围,形成促进骨形成的机械界面。Proussaefs等[47]在上颌窦底提升时使用羟基磷灰石颗粒移植并植入羟基磷灰石涂层种植体,多年后经过病理检验等途径发现种植体与周围骨结合良好,新生骨内可见很多骨小梁排列紧密,承担长期咬合力作用相当明显,而没有明显吸收。 磷酸三钙具有近似于人体骨组织的无机成分,生物相容性、降解性和骨传导性较好,在人体内溶解度较大,化学稳定性不佳,容易被水化,是典型的生物降解类陶瓷。磷酸钙水泥一般由2种或3种组分构成,包括1份或2份粉剂结构及1份液状结构。当将粉剂及液状组分混合后,将形成可塑外形的糊剂。双相磷酸钙陶瓷是含有羟基磷灰石和磷酸三钙2种成分的生物陶瓷,根据两者比例不同,钙磷比主要在1.5-1.67之间。磷酸三钙的溶解度较高,溶解较快,而羟基磷灰石的溶解度相对较低,因此可通过控制二者的比例来制备溶解性能合适的双相磷酸钙陶瓷。有学者曾为1例62岁男性患者行上颌窦提升同期种植,使用的骨增量材料为双相磷酸钙陶瓷,6年后患者因膀胱癌去世,尸体被捐赠,其尸检报告显示种植体周围存在充足的骨量,并与种植体形成了稳定的骨结合[48]。使用环钻活检技术在植入双相磷酸钙陶瓷的种植体周围取骨,发现其愈合时间较长[49],具有良好的骨传导性和上颌窦底空间维持性,残留的双相磷酸钙陶瓷和成熟骨组织可在长的愈合期内维持。 高分子聚合物:分为天然高分子聚合物和人工高分子聚合物。天然高分子聚合物包括胶原、纤维蛋白、甲壳素及衍生物等,这些材料有较好的生物相容性,携带细胞识别信号,有助于细胞的黏附、增殖和分化,但其缺点也很明显,如难以进行大规模生产、不同批号的产品存在较大质量差异、材料机械强度和降解速度不易调控等。人工合成高分子聚合物包括聚乳酸、聚乙醇酸及聚乳酸和聚乙醇酸的共聚体,是研究最多的人工高分子聚合物,其应用也十分广泛。 2.3 不使用骨移植材料 种植方案中在保证修复效果的前提下,为患者选择更简单的方案、缩短治疗周期、减少创伤和费用一直是口腔医生努力的方向。Silva等[50]在对16篇文献(共计436例患者868颗植体)的系统评价后发现,用或不用骨移植材料的上颌窦提升外科,是一种安全的、并发症较低和可预期结果的外科手术。Si等[51]通过对45例种植患者3年的随机对照临床研究发现:在上颌窦底提升同期植入种植体,用或不用骨移植材料都可获得预期的效果,使用骨移植材料的种植临床成功率方面较不使用骨移植材料无明显优势。Pinchasov等[52]通过研究对上颌窦底提升术中植骨、不植骨的效果做出了比较,结果得出不论植骨与否,都是有着较少的并发症及可预测的结果。Lai等[53]在202例上颌后牙区牙槽骨萎缩患者行上颌窦底提升同期植入280颗植体的初步研究中发现,使用或不使用骨移植材料都可获得可预期的骨整合,不使用骨移植材料的上颌窦底提升并同期植入种植体,可获得预期的自主性新骨形成。Sohn等[54]在对用或不用骨移植材料上颌窦骨增量的动物实验组织形态学中发现,不使用骨移植材料组可获得更快更致密的新骨形成,相对于覆盖屏障膜的骨移植材料,可替换的骨窗在骨愈合早期可加快新骨的形成,其结果揭示可替换的骨窗和提升的上颌窦黏膜具有骨诱导潜能。还有学者认为对比使用了植骨材料,不使用植骨材料似乎可增加新骨形成,提高种植体存活率[55-57]。其原理一方面是血凝块的骨再生作用,另一方面可通过帐篷技术或空间维持网使上颌窦膜抬高,以获得骨形成的空间[58]。 2.3.1 血凝块的骨再生作用 Lundgren等[59]在2003年首次报道了摘除囊肿后上颌窦内自发垂直骨形成,证明了窦内血凝块的成骨潜能,并鼓励了进一步的研究。有人发现未使用移植材料上颌窦底提升后的新骨生成和种植体骨结合是可能的[60],可见仅使用血凝块而不需要放置骨移植材料就可实现良好的骨再生[61-62]。因为上颌窦本身具有形成新骨、自我愈合的能力,且粗糙种植体表面会促进细胞的黏附、增殖,提高种植体的初期稳定性[63]。上颌窦黏膜提升后,来源于上颌窦骨膜和松质骨的骨细胞在此吸收和沉积,形成了新骨。所以窦膜和窦壁在血凝块成骨过程中起着非常重要的作用。然而,也有人认为膜塌陷进入所产生的空间后会限制新骨的生成[64]。另外,Moon等[61]将静脉血放到上颌窦腔内,6个月后发现相比于使用植骨材料组,未使用组有更多明显的新骨形成。然而这个结果在6个月以上的时间中则变得不显著。Falah等[56]在对30例(植入72颗植体)无骨移植的上颌窦底增量同期植入种植体临床病例中发现,种植体存留率为94%,其认为血凝块是能够被称为自体骨生成的骨移植材料,是一种能够迁移、分化、再生骨的骨源细胞。无骨移植的上颌窦提升具有高的成功率,可避免进行复杂的骨移植外科流程。 2.3.2 空间维持装置 空间维持装置由可吸收生物支架材料或不可吸收钛网构成。Cossellu等[65]将聚合的可逆热塑性凝胶放在抬高的上颌窦黏膜下方维持空间,6个月后结果显示,新骨形成百分比在54%-60%范围内。这种空间维持凝胶似乎刺激了可预测的骨形成,因此促进了上颌窦中的新骨形成。然而,该研究仅报告了在种植体周围有少量新骨,在新骨形成过程中不足以稳定。另外,Cricchio等[66]用氰基丙烯酸胶作为空间维持装置,评估其是否利于新骨形成及是否利于同期和后期植入种植体的稳定,见图1,组织学结果显示在大多数移位情况下,可看到与上颌窦膜和此装置接触的骨形成。但是,同期植入的部位显示沿种植体表面有骨形成,而延期植入的部位显示很少或没有骨形成和/或形成致密的纤维组织。近期也有专家将钛网放在窦嵴距均低于5 mm的病例中维持上颌窦膜高度,结果获得了理想的新骨形成[67]。"
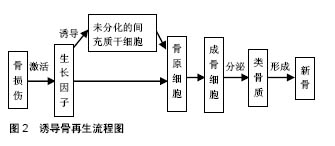
2.3.3 短种植体 对于短种植体的标准定义,目前尚缺乏统一的标准,宿玉成教授认为长度低于6 mm的种植体称为短种植体,也有人定义短种植体的长度≤8 mm[68]。有学者通过随机对照试验比较上颌窦内行侧壁开窗同期植入长种植体和经牙槽嵴顶植入短种植体的效果,随访5年发现,2组种植体存留率和牙周状况间差异无统计学意义[69]。Teng等[70]对32例牙槽骨严重萎缩、上颌窦底骨高度低于5 mm (平均3.34 mm)的上颌后牙区患者,采用上颌窦内嵌骨提升术同期植入50颗短种植体,术后1年随访发现,种植体的存留率为100%,上颌窦底平均提升骨高度为5.38 mm,认为这种不使用骨移植材料的上颌窦内嵌骨提升术可避免复杂的手术操作,简化手术流程。另外,短种植体不但能为患者避免植骨、减少疼痛和节约费用,而且术中和术后并发症也很少[70]。由此可见,对于那些不能承受更多创伤和手术的患者来说,短种植体是一个更好的选择。但是,如果种植体较短,修复后的义齿牙冠与种植体长度比例不符合正常生理状态下的冠根比,行牙冠修复后的并发症可能会增加。因此,关于短种植体的长期成功率,目前仍需更多可靠及更长时间的医学证据来证实。 2.3.4 倾斜种植体 由于倾斜种植体拥有更长的植体,除了具有短种植体的优点外,同时还能增加与骨组织接触的面积,以增强植体的稳定性。近年来,广受大众喜爱的All-on-Four方案不但能充分利用垂直骨量不足的上颌窦底周围骨质,还能获得良好的种植体成功率和理想的修复效果,其前景可观。有研究得出上颌All-on-Four修复的5年随访病例成功率和种植体成功率分别为93%和98%,其中锥形种植体设计更有利于获得初期稳定性,因此作者推荐用于All-on-Four即刻负重病例中[71]。也有学者在无牙颌患者上颌将种植体穿通窦底固位在其前侧壁和鼻骨的骨质间,以增加植体的稳定性,连同前牙区的2颗植体进行上颌固定义齿修复,随访3年结果发现,种植体存留率为96.4%,修复体成功率为100%[72]。另外,有学者通过系统评价和Meta分析得出倾斜种植体存留率为98.37%,垂直种植体存留率为98.19%,二者结果无统计学差异[73]。由此可见,使用倾斜种植体是上颌窦区种植修复的一个良好选择,但目前尚缺乏长期的随访证据。 2.4 成骨机制 修复性骨再生、种植体骨结合的3种生物学机制为:骨生成、骨诱导和骨引导。上颌窦底区的骨原细胞和成骨细胞,通过直接成骨机制在窦壁断面进行骨塑性和骨改建。上颌窦黏膜和骨髓组织中含有大量间充质干细胞,在信号分子作用下可分化为骨原细胞和成骨细胞,分泌类骨质形成新骨,见图2。当使用骨移植材料作为支架时,其可引导血管原细胞和骨原细胞长入并形成新骨。另外,血液黏附到钛种植体表面时可产生凝血酶,有助于成骨细胞活化,增强骨生长效果。有研究证实,骨生成潜能是上颌骨窦底或更确切地说是上颌骨结节和上颌骨骨膜所固有的[74],因为这些位点是骨祖细胞的来源,并且具有早期表达成骨标记物的细胞,其可在异位移植后形成骨结构,因此提出上颌窦黏膜具有成骨能力的结论[13,60],认为上颌窦黏膜细胞被诱导后可产生具有成骨的能力细胞因子,包括碱性磷酸酶、骨钙蛋白、骨桥蛋白、骨形成蛋白等。另外,机械刺激还可激起骨膜潜在的成骨潜能。但这一观点却被质疑[75-76],因为Jungner等[75]通过组织学和免疫组织化学研究发现,灵长动物上颌窦膜似乎不呈现骨诱导能力。?Scala?等[77-78]在灵长类动物上颌后牙区行上颌窦黏膜提升同期行种植手术,术后组织学结果显示:上颌窦黏膜周围下方没有明显的新骨形成,且新骨主要出现在窦底的血凝块处,没有直接证据指出上颌窦黏膜直接参与了早期成骨过程。因此,进一步的相关研究仍需进行。"
| [1] 邵磊,张波,江鹭鹭,等.不同术式上颌窦提升术应用于上颌后牙种植修复临床疗效评价[J].中国实用口腔科杂志, 2015,8(4):214-218.[2] Danesh-Sani SA,Loomer PM,Wallace SS.A comprehensive clinical review of maxillary sinus floor elevation: anatomy, techniques, biomaterials and complications. Br J Oral Maxillofac Surg.2016;54(7): 724-730.[3] Jang JW,Chang HY,Pi SH,et al.Alveolar Crestal Approach for Maxillary Sinus Membrane Elevation with <4 mm of Residual Bone Height: A Case Report.Int J Dent.2018;2018:1063459.[4] Corbella S,Taschieri S,Del Fabbro M. Long-term outcomes for the treatment of atrophic posterior maxilla: a systematic review of literature.Clin Implant Dent Relat Res. 2015;17(1): 120-132.[5] Aoki N,Baba J,Iwai T,et al.Lateral Bone Window Approach with Micross Mini Bone Scraper for Sinus Floor Elevation.J Maxillofac Oral Surg.2018;17(3):291-295.[6] Kao SY,Lui MT,Cheng DH,et al.Lateral trap-door window approach with maxillary sinus membrane lifting for dental implant placement in atrophied edentulous alveolar ridge.J Chin Med Assoc. 2015;78(2):85-88.[7] Fermergard R,Astrand P.Osteotome sinus floor elevation without bone grafts--a 3-year retrospective study with Astra Tech implants.Clin Implant Dent Relat Res. 2012;14(2):198-205.[8] Al-Dajani M.Recent Trends in Sinus Lift Surgery and Their Clinical Implications. Clin Implant Dent Relat Res. 2016;18(1): 204-212.[9] Si MS,Shou YW,Shi YT,et al.Long-term outcomes of osteotome sinus floor elevation without bone grafts: a clinical retrospective study of 4-9 years.Clin Oral Implants Res. 2016; 27(11):1392-1400.[10] Pjetursson BE,Lang NP.Sinus floor elevation utilizing the transalveolar approah. Periodontology 2000. 2014; 66(1): 59-71.[11] Qian SJ,Gu YX,Mo JJ,et al.Resonance frequency analysis of implants placed with osteotome sinus floor elevation in posterior maxillae.Clin Oral Implants Res.2016;27(1):113-119.[12] Gonzalez S,Tuan MC,Ahn KM,et al.Crestal approach for maxillary sinus augmentation in patients with </= 4 mm of residual alveolar bone.Clin Implant Dent Relat Res. 2014; 16(6):827-835.[13] Nedir R, Nurdin N,Khoury P,et al. Short Implants Placed with or without Grafting in Atrophic Sinuses: The 3-Year Results of a Prospective Randomized Controlled Study.Clin Implant Dent Relat Res. 2016; 18(1):10-18.[14] Pommer B,Hof M,Fadler A,et al.Primary implant stability in the atrophic sinus floor of human cadaver maxillae: impact of residual ridge height, bone density, and implant diameter. Clin Oral Implants Res. 2014; 25(2):e109-113.[15] Smeets R,Kolk A,Gerressen M,et al.A new biphasic osteoinductive calcium composite material with a negative Zeta potential for bone augmentation. Head Face Med. 2009;5:13.[16] Aebi M, Zuber K, Marchesi D.Treatment of cervical spine injuries with anterior plating. Spine.1991;16:S38-S45.[17] Horch HH, Sader R,Pautke C,et al.Synthetic, pure-phase beta-tricalcium phosphate ceramic granules (Cerasorb) for bone regeneration in the reconstructive surgery of the jaws.Int J Oral Maxillofac Surg.2006;35(8):708-713.[18] Corbella S,Taschieri S,Weinstein R,et al.Histomorphometric outcomes after lateral sinus floor elevation procedure: a systematic review of the literature and meta-analysis.Clin Oral Implants Res. 2016;27(9): 1106-1122.[19] Danesh-Sani SA, Engebretson SP,Janal MN. Histomorphometric results of different grafting materials and effect of healing time on bone maturation after sinus floor augmentation: a systematic review and meta-analysis.J Periodontal Res.2017;52(3):301-312.[20] Cardoso CL,Curra C,Santos PL,et al.Current considerations on bone substitutes in maxillary sinus lifting. Revista Clínica de Periodoncia, Implantología Y Rehabilitación Oral. 2016; 9(2):102-107.[21] La Monaca G,Iezzi G,Cristalli MP,et al.Comparative Histological and Histomorphometric Results of Six Biomaterials Used in Two-Stage Maxillary Sinus Augmentation Model after 6-Month Healing. Biomed Res Int. 2018;2018:9430989.[22] de Molon RS,de Paula WN,Spin-Neto R,et al.Correlation of fractal dimension with histomorphometry in maxillary sinus lifting using autogenous bone graft.Braz Dent J. 2015; 26(1): 11-18.[23] Santagata M, Tozzi U,Tartaro G,et al.Maxillary Sinus Augmentation with Autologous and Heterologous Bone Graft: A Clinical and Radiographic Report of Immediate and Delayed Implant Placement. J Maxillofac Oral Surg. 2014; 13(4):401-408.[24] Maddalone M,Mirabelli L,Venino PM,et al.Long-term stability of autologous bone graft of intraoral origin after lateral sinus floor elevation with simultaneous implant placement. Clin Implant Dent Relat Res.2018:1-9.[25] Pisoni L,Lucchi A,Persia M,et al.Sinus lift: 3 years follow up comparing autogenous bone block versus autogenous particulated grafts.J Dent Sci.2016;11(3):231-237.[26] Shanbhag S,Shanbhag V,Stavropoulos A.Volume Changes of Maxillary Sinus Augmentations over Time: A Systematic Review.Int J Oral Maxillofac Implants.2014;29(4):881-892.[27] Kim YK,Lee J,Yun JY,et al.Comparison of autogenous tooth bone graft and synthetic bone graft materials used for bone resorption around implants after crestal approach sinus lifting: a retrospective study.J Periodontal Implant Sci. 2014;44(5): 216-221.[28] Kim ES,Kang JY,Kim JJ,et al.Space maintenance in autogenous fresh demineralized tooth blocks with platelet-rich plasma for maxillary sinus bone formation: a prospective study.Springerplus.2016;5:274.[29] Sehn FP,Dias RR,de Santana Santos T,et al.Fresh-frozen allografts combined with bovine bone mineral enhance bone formation in sinus augmentation.J Biomater Appl. 2015; 29(7): 1003-1013.[30] Reikeras O,Sigurdsen UW,Shegarfi H.Impact of freezing on immunology and incorporation of bone allograft. J Orthop Res. 2010; 28(9):1215-1219.[31] Zizzari VL,Zara S,Tete G,et al.Biologic and clinical aspects of integration of different bone substitutes in oral surgery: a literature review.Oral Surg Oral Med Oral Pathol Oral Radiol. 2016;122(4):392-402.[32] Annibali S,Iezzi G,Sfasciotti GL,et al.Histological and Histomorphometric Human Results of HA-Beta-TCP 30/70 Compared to Three Different Biomaterials in Maxillary Sinus Augmentation at 6 Months: A Preliminary Report.Biomed Res Int.2015;2015:156850.[33] 刘宏波,李梦云,田清业.同种异体骨制备贮存的研究进展[J].医药前沿, 2013,35(3):336-338.[34] Smith MM,Duncan WJ,Coates DE. Attributes of Bio-Oss((R)) and Moa-Bone((R)) graft materials in a pilot study using the sheep maxillary sinus model.J Periodontal Res. 2018;53(1):80-90.[35] Aludden H,Dahlin A,Starch-Jensen T,et al. Histomorphometric analyses of area fraction of different ratios of Bio-Oss((R)) and bone prior to grafting procedures -An in vitro study to demonstrate a baseline. Clin Oral Implants Res 2018;29(2): 185-191.[36] Shirmohammadi A, Roshangar L, Chitsazi MT, et al. Corrigendum to "Comparative Study on the Efficacy of Anorganic Bovine Bone (Bio-Oss) and Nanocrystalline Hydroxyapatite (Ostim) in Maxillary Sinus Floor Augmentation".Int Sch Res Notices.2017;2017:7258513.[37] De Santis E,Lang NP,Ferreira S,et al. Healing at implants installed concurrently to maxillary sinus floor elevation with Bio-Oss((R)) or autologous bone grafts. A histo-morphometric study in rabbits.Clin Oral Implants Res. 2017;28(5):503-511.[38] Kurkcu M,Benlidayi ME,Cam B,et al.Anorganic bovine-derived hydroxyapatite vs beta-tricalcium phosphate in sinus augmentation: a comparative histomorphometric study. J Oral Implantol. 2012;38 Spec No: 519-526.[39] Schlegel KA,Fichtner G,Schultze-Mosgau S,et al. Histologic Findings in sinus Augmentation with Autogenous Bone chips versus a Bovine Bone Substitute.Int J Oral Maxillofac Implants. 2003;18(1):53-58.[40] Starch-Jensen T,Aludden H,Hallman M,et al.A systematic review and meta-analysis of long-term studies (five or more years) assessing maxillary sinus floor augmentation. Int J Oral Maxillofac Surg. 2018; 47(1):103-116.[41] Di Stefano DA,Gastaldi G,Vinci R,et al.Bone Formation Following Sinus Augmentation with an Equine-Derived Bone Graft: A Retrospective Histologic and Histomorphometric Study with 36-Month Follow-up.Int J Oral Maxillofac Implants. 2016;31(2):406-412.[42] 宋会平,王志强.骨移植的过去、现在和未来[J].中国修复重建外科杂志, 2009,23(5):513-516.[43] Pighinelli L,Kucharska M.Chitosan-hydroxyapatite composites. Carbohydr Polym.2013;93(1): 256-262.[44] Holmes R,Bucholz R,Mooney V.Porous hydroxyapatite as a bone-graft substitute in metaphyseal defects.J Bone Joint Surg.1986;68A(6):904-911.[45] Herath H,Di Silvio L,Evans J.Porous hydroxyapatite ceramics for tissue engineering. J Appl Biomater Biomech. 2005;3(3): 192-198.[46] Ishihara K,Arai J,Nakabayashi N,et al. Adhesive bone cement containing hydroxyapatite particle as bone compatible filler.J Biomed Mater Res. 1992;26:937-945.[47] Proussaefs P,Olivier HS,Lozada J.Histologic evaluation of a 12-year-old threaded hydroxyapatite-coated implant placed in conjunction with subantral augmentation procedure: a clinical report. J Prosthetic Dent. 2004;92(1):17-22.[48] Lee JS,Cha JK,Thoma DS,et al.Report of a human autopsy case in maxillary sinuses augmented using a synthetic bone substitute: Micro-computed tomographic and histologic observations. Clin Oral Implants Res.2018;29(3):339-345.[49] Bouwman WF,Bravenboer N,Frenken J,et al.The use of a biphasic calcium phosphate in a maxillary sinus floor elevation procedure: a clinical, radiological, histological, and histomorphometric evaluation with 9- and 12-month healing times. Int J Implant Dent.2017;3(1):34.[50] Silva LD,de Lima VN,Faverani LP,et al. Maxillary sinus lift surgery-with or without graft material? A systematic review.Int J Oral Maxillofac Surg. 2016;45(12):1570-1576.[51] Si MS,Zhuang LF,Gu YX,et al.Osteotome sinus floor elevation with or without grafting: a 3-year randomized controlled clinical trial.J Clin Periodontol.2013;40(4):396-403.[52] Pinchasov G,Juodzbalys G. Graft-free sinus augmentation procedure: a literature review. J Oral Maxillofac Res. 2014; 5(1):e1.[53] Lai HC,Zhuang LF,Lv XF,et al.Osteotome sinus floor elevation with or without grafting: a preliminary clinical trial. Clin Oral Implants Res. 2010; 21(5):520-526.[54] Sohn DS, Kim WS, An KM,et al.Comparative histomorphometric analysis of maxillary sinus augmentation with and without bone grafting in rabbit. Implant Dent. 2010; 19(3):259-270.[55] Nasr S,Slot DE,Bahaa S,et al.Dental implants combined with sinus augmentation: What is the merit of bone grafting? A systematic review. J Craniomaxillofac Surg. 2016;44(10): 1607-1617.[56] Falah M, Sohn DS, Srouji S. Graftless sinus augmentation with simultaneous dental implant placement: clinical results and biological perspectives.Int J Oral Maxillofac Surg. 2016; 45(9):1147-1153.[57] Srinivasan M,Meyer S,Mombelli A,et al.Dental implants in the elderly population: a systematic review and meta-analysis.Clin Oral Implants Res. 2017;28(8):920-930.[58] Ahmed M,Abu Shama A,Hamdy RM,et al.Bioresorbable versus titanium space-maintaining mesh in maxillary sinus floor elevation: a split-mouth study.Int J Oral Maxillofac Surg. 2017;46(9): 1178-1187.[59] Lundgren S,Anderson S,Sennerby L.Spontaneous bone formation in the maxillary sinus after removal of a cyst. Clin Implant Dent Relat Res. 2003; 5(2):78-81.[60] Bassi AP,Pioto R,Faverani LP,et al.Maxillary sinus lift without grafting, and simultaneous implant placement: a prospective clinical study with a 51-month follow-up.Int J Oral Maxillofac Surg. 2015;44(7):902-907.[61] Moon JW,Sohn DS,Heo JU,et al. New bone formation in the maxillary sinus using peripheral venous blood alone.J Oral Maxillofac Surg. 2011; 69(9):2357-2367.[62] Parra M,Atala-Acevedo C,Farina R,et al.Graftless Maxillary Sinus Lift Using Lateral Window Approach: A Systematic Review.Implant Dent. 2018;27(1):111-118.[63] Boyan BD,Cheng A,Olivares-Navarrete R,et al.Implant Surface Design Regulates Mesenchymal Stem Cell Differentiation and Maturation.Adv Dent Res. 2016; 28(1):10-17.[64] Kaneko T,Masuda I,Horie N,et al.New bone formation in nongrafted sinus lifting with space-maintaining management: a novel technique using a titanium bone fixation device.J Oral Maxillofac Surg. 2012;70(3): e217-224.[65] Cossellu G, Farronato G, Farronato D,et al. Space-maintaining management in maxillary sinus lifting: a novel technique using a resorbable polymeric thermo-reversible gel.Int J Oral Maxillofac Surg. 2017;46(5): 648-654.[66] Cricchio G, Palma VC, Faria PE, et al. Histological outcomes on the development of new space-making devices for maxillary sinus floor augmentation.Clin Implant Dent Relat Res.2011;13(3):224-230.[67] Atef M, Hakam MM, ElFaramawey MI, et al. Nongrafted sinus floor elevation with a space-maintaining titanium mesh: case-series study on four patients. Clin Implant Dent Relat Res.2014;16(6):893-903.[68] Renouard F, Nisand D.Impact of implant length and diameter on survival rates. Clin Oral Implants Res.2006;17(2):35-51.[69] Cannizzaro G, Felice P, Leone M, et al. Early loading of implants in the atrophic posterior maxilla: lateral sinus lift with autogenous bone and Bio-Oss versus crestal mini sinus lift and 8-mm hydroxyapatite-coated implants. A randomised controlled clinical trial.Eur J Oral Implantol. 2009; 2(1):25-38.[70] Teng M, Liang X,Yuan Q, et al.The inlay osteotome sinus augmentation technique for placing short implants simultaneously with reduced crestal bone height. A short-term follow-up.Clin Implant Dent Relat Res.2013; 15(6):918-926.[71] Malo P,de Araujo Nobre M,Lopes A,et al. "All-on-4" immediate-function concept for completely edentulous maxillae: a clinical report on the medium (3 years) and long-term (5 years) outcomes. Clin Implant Dent Relat Res. 2012;14 Suppl 1:e139-150.[72] Malo P, Nobre MdA, Lopes A. Immediate loading of’ All-on-4’. Eur J Oral Implantol.2013;6(3):273-283.[73] Chrcanovic BR,Albrektsson T,Wennerberg A.Tilted versus axially placed dental implants: a meta-analysis.J Dent. 2015;43(2):149-170.[74] Cicconetti A, Sacchetti B, Bartoli A,et al. Human maxillary tuberosity and jaw periosteum as sources of osteoprogenitor cells for tissue engineering. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2007;104(5): 618.e1-12.[75] Jungner M, Cricchio G, Salata LA, et al. On the Early Mechanisms of Bone Formation after Maxillary Sinus Membrane Elevation: An Experimental Histological and Immunohistochemical Study. Clin Implant Dent Relat Res. 2015;17(6):1092-1102.[76] de Oliveira GR, Olate S, Cavalieri-Pereira L, et al. Maxillary sinus floor augmentation using blood without graft material. Preliminary results in 10 patients.J Oral Maxillofac Surg. 2013;71(10): 1670-1675.[77] Scala A, Botticelli D, Faeda RS, et al. Lack of influence of the Schneiderian membrane in forming new bone apical to implants simultaneously installed with sinus floor elevation: an experimental study in monkeys.Clin Oral Implants Res. 2012; 23(2):175-181.[78] Scala A,Botticelli D,Rangel IG Jr, et al.Early healing after elevation of the maxillary sinus floor applying a lateral access: a histological study in monkeys.Clin Oral Implants Res, 2010;21(12):1320-1326. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Zeng Xianghong, Liang Bowei. A new strategy for the treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 431-437. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||