Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (6): 963-970.doi: 10.3969/j.issn.2095-4344.1553
Previous Articles Next Articles
Application and progress of bioactive scaffolds in bone tissue engineering
Leng Yi, Li Zuhao, Ren Guangkai, Wang Zhonghan, Gao Chaohua, Shi Chenyu, Liu He, Wu Dankai
- Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China
-
Received:2018-11-06Online:2019-02-28Published:2019-02-28 -
Contact:Wu Dankai, Chief physician, Professor, Master’s supervisor, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
About author:Leng Yi, Master candidate, Department of Orthopedics, Second Hospital of Jilin University, Changchun 130041, Jilin Province, China -
Supported by:the National Natural Science Foundation of China, No. 81671804, 81171681 and 81772456
CLC Number:
Cite this article
Leng Yi, Li Zuhao, Ren Guangkai, Wang Zhonghan, Gao Chaohua, Shi Chenyu, Liu He, Wu Dankai. Application and progress of bioactive scaffolds in bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(6): 963-970.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
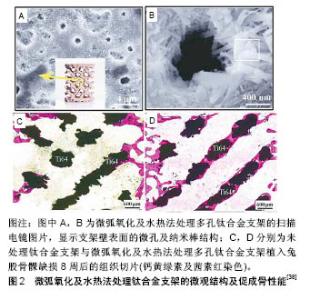
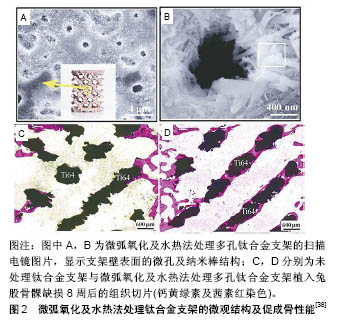
所以在3D生物活性支架的效果选择上,应在有利于细胞功能、细胞活力及负载下的机械完整性之间做出更加平衡的调整。 2.2 支架制作方法 现如今多种技术已被用于3D支架制作中,有时一种支架的制作会应用多种技术。通常使用常规技术制作的支架,难以精确控制形成复杂的微观结构。然而随着计算机辅助设计建模将3D打印技术应用于骨组织工程中,显著提高了支架制造的制造精度和可重复性,并可控制支架的宏观和微观结构。因此,此节介绍传统支架制造与3D打印技术的方法及优缺点。 2.2.1 传统支架制造 传统支架制造方法包括溶剂浇铸/颗粒浸出、气体发泡、乳化冷冻干燥、静电纺丝等制造方法,每种方法都有其不同的优缺点。 溶剂浇铸/颗粒浸出技术的具体方法是将致孔剂与目标材料溶液混合,随后形成聚合物-致孔剂网络,复合物中溶剂蒸发,使支架硬化,随后用水溶解致孔剂,从而形成了具有孔隙的支架[20]。致孔剂可分散到模塑结构中,并且一旦结构凝固随后就溶解,从而产生孔隙。这种制作方法有着相对简单的技术,可形成具有规则孔隙率、孔径的支架。但由于难以控制支架产生的孔隙形状和孔隙互连并同时使用有机溶剂,就不能将细胞或生长因子直接添加于支架中,所以现在已很少使用此类方法。 气体发泡法消除了溶剂浇铸/颗粒浸出方法中有机溶剂的使用,首先通过压缩成型技术将目标材料压缩成圆形盘,然后在室温下暴露于5.5 MPa CO2压力下72 h,然后将CO2压力降低至大气压水平,快速降低气体在聚合物中的溶解度,使CO2气体聚集在一起,产生孔隙。使用该技术可达到93%的孔隙率和高达100 mm的孔径。然而,通过气体发泡仍难以控制孔隙连通性和孔径[21]。 冷冻干燥法首先将聚合物溶液冻结,于是便得到了被聚合物骨架包围的溶剂结晶,然后通过真空将周围压力降低至低于冷冻溶剂的平衡蒸气压水平,触发溶剂直接升华为气体;当溶剂完全升华时,保留具有互连多孔结构的干燥聚合物支架。乳化冷冻干燥也可用做主要支架制造方法。该方法首先将聚合物/陶瓷溶解在溶剂中,然后与水混合得到乳液,将混合物倒入模具中并冷冻,然后分离两相;将冷冻的乳液冷冻干燥,以除去溶剂和分散的水,在固化支架中产生孔隙结构。虽然这种方法避免了固体致孔剂的应用,但仍未减少有机溶剂的使用,并且处理时间长且孔隙率难以控制。 静电纺丝是另一种流行的支架制造技术,能产生纳米纤维互连的多孔支架。该方法使用外部施加的电场,将聚合物溶液或聚合物熔体的带电线从毛细管朝向集电板拉出,可依次创建和沉积由复合材料和生物分子构成的微米和纳米级别纤维,以形成支架[22]。静电纺丝所制造的支架具有较大的表面积,以促进细胞附着,而且技术简单且廉价,研究人员也在对这种技术进行不断的开发。但此法难以精确掌控支架的机械性能及微观结构,仍需要研究及攻克。 2.2.2 3D打印技术 传统支架制造常面临着造孔剂残留、重复性低、孔隙率难以控制等缺点。较常规支架制造技术而言,3D打印技术在空间上控制支架微架构和空间内容的能力已得到改善。通常,3D打印技术通过粉末、液体或固体材料的逐层处理来制造物体,从底部开始构建,每个新形成的层黏附到前一层,产生逐渐增大的构造,而且通过计算机辅助设计的帮助更能实现个体化定制。常见的3D打印技术包括立体激光光刻、熔融沉积成型及选择性激光烧结等[23]。 立体激光光刻是最早得到开发的3D打印技术之一,它使用定向激光固化液体光敏聚合物(如树脂等物质)形成3D模型,其中紫外激光器可以固化模型横截面,留下液体形式的剩余区域,然后去除过量的未固化树脂,得到完整的支架。虽然立体激光光刻可快速生产具有受控结构和微米水平的支架,但范围较小的材料选择和昂贵的技术仍制约着立体激光光刻的发展[24]。 熔融沉积成型使用温控打印头以逐层方式将热塑性材料沉积到平台上,以构建3D构造。将热塑性长丝驱入加热的印刷头中,使长丝熔化,使得半熔融聚合物(如聚己内酯)顺序精确沉积。熔融长丝在打印环境中的空气中冷却,使长丝快速熔合在一起,形成支架。尽管较高的制造温度限制了水凝胶或生物分子的应用,但熔融沉积成型作为生产合成支架的方法,也已成功应用于骨组织工程中[25]。 选择性激光烧结使用计算机控制的激光束来熔化粉末层,将粉末材料烧结在一起,以形成实心3D结构。选择性激光烧结能生产具有与骨小梁相似机械特性的生物活性复合支架,并初步应用于骨组织工程中[26]。然而该过程中涉及的升高温度,限制了选择性激光烧结生产包含细胞和生物材料的支架。 由此可看出,上述方法都有着温度过高而不能加入生物活性物质的缺点,所以3D生物打印由此产生。生物打印可从上而下制造包含活细胞、细胞外基质及其他生物材料的复杂结构支架,也能够改善血管向支架内长入的能力。相比常规3D打印技术,3D生物打印实现了细胞或生物活性成分的分层放置[27]。常用的生物打印技术包括喷墨[28]、激光辅助[29]、微型阀和挤出生物打印 等[30-31]。打印成功后,可直接植入患者体内或在体外培养,构成被称为生物反应器的生物活性培养环境,促使细胞向特定方向成长,所以3D生物打印技术正不断被研究者们开发利用。 2.3 生物活性支架应用的材料 2.3.1 金属材料复合支架 钴铬合金、锆合金、钛合金和不锈钢等金属合金具有优异的生物相容性和强度,因此通常被用于关节置换和骨折固定植入物,以支持骨愈合[32]。然而其缺乏生物降解性,不太适合骨组织工程,作为骨折内固定物还需要额外手术来移除金属植入物,特别是当它们用于未达到骨骼成熟度的儿童患者时[33]。虽然有这些限制,但通过制造复合金属支架已取得了一些成功。Chen等[34]发现给予3D打印钛支架孔隙率在达到50%的情况下,抗压强度可达到109 MPa,并且具有粗糙的微观结构,在一定程度上促进了骨髓间充质干细胞在支架上的附着与增殖。而在支架中加入锶、镁的等一系列金属微量元素,可促进骨生成和血管生成,在增强骨重建和修复过程中起重要作用[35]。其中锶得到了广泛研究,因为它具有与钙相似的大小及电荷,现已证明锶可在成骨细胞介导的骨再生过程中取代钙,增强骨生成,同时防止破骨细胞再吸收。此外,锶已被证明可增强成骨细胞向骨细胞的分化[36]。Lei等[37]通过冷冻干燥法将锶与羟基磷灰石和壳聚糖混合,用以制作复合纳米杂化支架,发现支架中存在的锶-羟基磷灰石纳米晶体可显著增强人骨髓间充质干细胞的增殖和成骨分化能力,显著改善骨再生。通过微弧氧化及水热法处理钛合金支架后,不但增强了羟基磷灰石的形成能力,而且在兔股骨髁缺损中促进了骨整合及骨再生,见图2。"
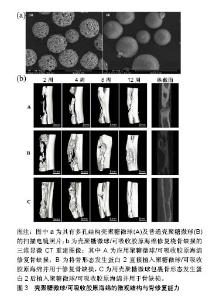
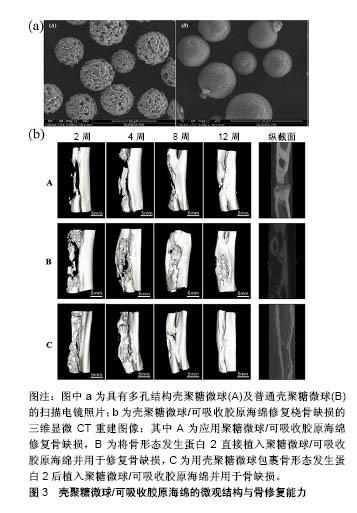
近些年铁作为支架原材料正得到不断的应用,铁基支架在满足可生物降解的条件下,还能拥有优秀的机械性能,而且有研究显示其植入18个月不会产生明显的炎症反应或全身毒性[39]。Chou等[40]在此基础上利用3D喷墨印刷制作铁镁复合支架,发现3D打印后铁镁构建体具有开放的多孔结构,具有与松质骨相似的拉伸机械性质。体外分析发现暴露于支架后细胞活力良好,细胞也渗透到孔隙中。金属作为应用历史最久的支架材料,正在不断得到新的使用方式。 2.3.2 生物陶瓷材料复合支架 生物陶瓷包括陶瓷复合材料、无定形玻璃和生物玻璃等,具有良好的生物活性、耐腐蚀性和抗压缩性[41]。其中骨组织工程中最常用的材料为羟基磷灰石、磷酸三钙和他们不同比例的混合物双相磷酸钙,这些材料作为骨组织工程支架在体内经由细胞降解为钙和磷酸根离子,从而通过骨诱导促进骨再生[42]。生物陶瓷在结构和化学组成方面也与天然骨的矿物质含量具有很大程度的相似性。这使得生物陶瓷构建体能够提供生物相容的骨传导性界面,能够促进与宿主组织的整合,而不会形成瘢痕组织[43]。 通过Tang等[44]进行的4种生物陶瓷支架的对比实验,可以清楚地了解到生物陶瓷支架拥有着良好的骨传导性及骨诱导性,而且随着组成成分的不同,产生效果的侧重点也不同,但也证明了生物陶瓷支架具有优秀的应用前景。然而,在骨组织工程的背景下,陶瓷支架相对易于脆化。与金属基支架一样,它们也难以精细控制的降解速率。因此,越来越多的注意力被用于开发陶瓷复合支架。而且生物陶瓷相比其他骨组织工程材料更易于表面添加涂层,增强其功能。Mohseni等[45]将纳米复合磷酸三钙/胶原支架植入兔股骨节段性骨缺损中,45 d后在组织学上发现了良好的骨形成,且比单纯的羟基磷灰石支架表现更为优异。而添加聚合物涂层的另一优势在于可以搭载药物,并能达到药物缓释的目的,从而产生更优秀的骨再生效果。Tarafder等[46]在3D打印了磷酸三钙支架后,对其进行了聚己内酯/阿仑膦酸涂层,发现与裸露的磷酸三钙或聚己内酯包被的磷酸三钙支架相比,具有聚己内酯与阿仑膦酸复合涂层的支架可局部缓慢释放阿仑膦酸,促进早期骨形成增加。而含有二氧化硅的生物陶瓷复合材料在骨组织工程中也受到了极大的关注,因为含Si支架的离子分解产物具有优秀的骨传导性[22]。Dai等[47]烧结碳酸钙和二氧化硅以产生新型多孔β-硅酸二钙支架,机械分析发现这种复合支架最大抗压强度达到28.1 MPa,孔隙率高达71%,表现出了良好的生物相容性,体外细胞增殖实验发现MG-63细胞和间充质干细胞增殖良好。同时将支架放置在模拟体液培养后发现了表面磷灰石的形成。他们还发现在小鼠体内植入支架9周后,支架中有骨组织生成,提示支架的骨诱导特性。 诸如耐腐蚀性、生物相容性和刚度的性质导致生物陶瓷在临床骨科广泛使用。成千上万的患者已经从陶瓷关节置换组件,以及用于填充骨缺损的陶瓷粉末和颗粒中受益[48]。 生物活性玻璃代表了陶瓷材料的另一个子组,已在骨组织工程领域进行了广泛研究。生物活性玻璃可被视为具有无定形结构的表面活性玻璃-陶瓷生物材料。植入体内后,生物活性玻璃溶解有助于在生物玻璃表面上形成羟基磷灰石层,其反过来与宿主骨中的胶原原纤维相互作用,以产生强烈的结合。事实上,与骨骼形成的黏合是如此强烈,以至于通常不能通过破坏周围骨骼来去除生物活性玻璃。生物活性玻璃相互连接的多孔结构类似于骨小梁,为细胞提供了一个临时模板,促进骨再生[49]。目前生物活性玻璃已开始被用于临床,包括用于整形外科和颌面外科领域的假体;颗粒和微粒也被用来代替传统的骨移植物,来帮助治疗慢性骨髓炎、软组织缺损和伤口[50-51]。虽然生物活性玻璃在生物活性和骨传导性等具有吸引力,但固有的脆性使其临床应用具有局限性。所以可通过掺入可生物降解的聚合物,来改善生物活性玻璃的孔隙率、降解速率和弹性模量等性能[52]。 Wessuser等[53]研究了多种聚合物涂层3D打印生物活性玻璃支架的骨诱导特性,包括明胶及交联明胶,并在接种间充质干细胞后植入免疫缺陷小鼠中,8周后组织形态测定和微计算机断层扫描发现,明胶包被的支架整体表现最好,同时还发现生物活性玻璃支架具有优秀的药物传递能力。Jiang等[54]用酰胺涂覆中孔生物活性玻璃支架,随后将庆大霉素加载到支架中孔表面上,体外与未经过胺化的生物活性玻璃支架相比实现了更高的庆大霉素负载和更长的药物释放;体外生物活性也得到改善,在模拟体液中浸泡3 d后,酰胺涂覆中孔生物活性玻璃支架表面羟基磷灰石形成增加。生物活性玻璃支架在一些实验中也被用于可注射黏合剂搭载抗生素或聚合物,被发现具有良好的细胞相容性并能够治疗体内骨缺损。Zhang等[55]制备了掺杂锶的硼酸盐生物活性玻璃-壳聚糖复合骨水泥并注入兔股骨髁缺损中,有明显的新骨形成。因此,这种可注射生物支架为通过微创手术治疗不规则形状的骨缺损提供了新的思路。 2.3.3 聚合物材料复合支架 用于骨组织工程支架制备的聚合物材料,包括天然聚合物和人工合成聚合物。由于天然聚合物具有延展性、生物相容性和生物降解性等有利性质,使其在骨组织工程中得到了很大的关注,而且通常天然聚合物表面含有生物功能分子,可帮助支架上的细胞附着、整合和分化。已在骨组织工程中研究的天然聚合物包括胶原、蚕丝、藻酸盐、壳聚糖和透明质酸。然而,使用天然聚合物也可能受到限制,包括存在致病性杂质(如内毒素[56])、缺乏降解速率的可调性和降解相关的局部细胞抑制。尽管交联可在一定程度上提高天然聚合物的结构性能[57],但其机械性能也未能达到最佳标准。已在骨组织工程中应用的合成聚合物包括聚乳酸、聚乙醇酸、聚己内酯、聚乙二醇及共聚物聚乳酸-乙醇酸等。将聚合物与3D打印技术相结合,可根据需要制造具有定制孔径、孔隙率、降解速率和机械强度的聚合物支架[58]。由壳聚糖及丙烯酰胺共聚形成的温敏水凝胶,不但具有理想的多孔结构,而且具有良好的互连性、弹性和显著的骨传导性,体外可显著增强骨髓间充质干细胞的增殖活力及碱性磷酸酶活性,且细胞/水凝胶构建体在成骨分化后也显示出良好的机械强度和弹性,体内实验也验证了水凝胶聚合物的生物相容及促进骨形成的特性[59]。然而无论合成和天然聚合物,都面临着单独使用时相对较差的承载能力,与金属和陶瓷化合物相比具有较低的弹性模量[60]。在骨组织工程中,对支架的抗压缩与拉伸、抗疲劳性能机械要求是复杂的[61]。为了解决这一问题,可将生物陶瓷作为涂层或填料添加到聚合物基质中,提高聚合物的生物活性及机械性能[62]。而通过表面改性也可增加聚合物支架的性能与生物活性,包括生长因子和表面配体,以帮助细胞黏附和增殖[63]。 Villa等[64]通过共沉淀和冷冻铸造工艺开发了胶原-羟基磷灰石支架,所形成的支架具有高度的渗透性,适合细胞浸润、附着和成骨,具有99%的孔隙互连性。将小鼠骨髓间充质干细胞种到此支架上,在体外培养12 h后观察到良好附着。将该支架植入小鼠颅骨缺损中,3周后在射线照相和矿化分析中发现接近完全填充颅骨缺损的再生骨组织;数周后胶原蛋白的基质金属蛋白酶分解,必然导致支架降解。相比之下Marcacci等[65]将纯羟基磷灰石支架植入4例长骨缺损患者,发现6年支架仍未能降解。Hou等[66]将壳聚糖微球与可吸收胶原海绵混合并进行冷冻干燥,并将载有骨形态发生蛋白的壳聚糖微球/可吸收胶原海绵复合支架植入兔长15 mm桡骨缺损中,早在4周时发现缺损就会被新骨桥接,12周X射线和组织学分析可见骨髓腔完全愈合和再通,见图3。这些结果表明,复合壳聚糖-胶原支架是骨形态发生蛋白2用于治疗节段性骨缺损的有希望载体。在支架中添加不同的成分,对骨再生也会产生不同的效果。有研究证明,在聚合物支架中加入骨骼组成成分(如羟基磷灰石),可显著增强其骨诱导能力。Chang等[67]将透明质酸作为羟基磷灰石-β-磷酸三钙颗粒的水性黏合剂,观察是否可减少所需的骨移植量并增加临床情况下移植物处理的便捷性。将羟基磷灰石-β-磷酸三钙加载到交联的透明质酸中,形成新的透明质酸/羟基磷灰石-β-磷酸三钙复合物,然后将其注入体内兔颅骨缺损,组织学和微CT分析发现,即使羟基磷灰石-β-磷酸三钙颗粒数量减少,透明质酸也能维持骨再生。事实上,与对照支架相比,透明质酸/羟基磷灰石-β-磷酸三钙样品在2周后的骨形成量增加了1.7倍。总之,向骨移植物中添加透明质酸,不仅促进了骨传导,而且在临床情况下也改善了处理特性。 在合成聚合物方面,Yang等[68]利用聚乳酸、聚己内酯和疏水改性的纳米二氧化硅粒子制造了生物支架,不但具有分层结构,更能支持骨髓间充质干细胞的黏附和增殖,表现了良好的生物相容性。Shaui等[69]发现纳米羟基磷灰石可提高共聚物聚乳酸-乙醇酸的成骨能力和弹性模量,而共聚物聚乳酸-乙醇酸可以平衡羟基磷灰石的脆性和降解速率,并通过实验证明纳米羟基磷灰石含量最多为20%。Haider等[70]证明,与球形共聚物聚乳酸-乙醇酸-羟基磷灰石和纯共聚物聚乳酸-乙醇酸支架相比,共聚物聚乳酸-乙醇酸-羟基磷灰石纳米纤维支架显示出更高的细胞黏附、增殖,增强的成骨效应和增加的钙离子释放。但与合成聚合物相比,天然聚合物(如胶原和壳聚糖)具有改善的矿化特性。天然聚合物中存在许多离子分子基团,允许发生诸如钙螯合的过程,并且可通过负表面电荷改善矿化速率。合成聚合物表面很大程度上是疏水的,不利于细胞黏附。因此已尝试改变合成聚合物的表面,以增加其细胞亲和力。 "
| [1] Ashman O,Phillips AM.Treatment of non-unions with bone defects: which option and why? Injury. 2013;44 Suppl 1:S43-45.[2] Thaller PH,Furmetz J,Wolf F,et al.Limb lengthening with fully implantable magnetically actuated mechanical nails (PHENIX((R)))-preliminary results.Injury.2014;45 Suppl 1:S60-65.[3] Hollister SJ.Porous scaffold design for tissue engineering.Nat Mater.2005;4(7):518-524.[4] Garcia-Gareta E,Coathup MJ,Blunn GW.Osteoinduction of bone grafting materials for bone repair and regeneration.Bone. 2015; 81:112-121.[5] Brydone AS,Meek D,Maclaine S.Bone grafting, orthopaedic biomaterials, and the clinical need for bone engineering.Proc Inst Mech Eng H.2010;224(12):1329-1343.[6] Higgins A,Glover M,Yang Y,et al.EXOGEN ultrasound bone healing system for long bone fractures with non-union or delayed healing: a NICE medical technology guidance. Appl Health Econ Health Policy. 2014;12(5):477-484.[7] Faour O,Dimitriou R,Cousins CA, Giannoudis PV. The use of bone graft substitutes in large cancellous voids: any specific needs? Injury.2011;42 Suppl 2:S87-90.[8] O'Keefe RJ, Mao J. Bone tissue engineering and regeneration: from discovery to the clinic--an overview. Tissue Eng Part B Rev. 2011;17(6):389-392.[9] Bose S,Roy M,Bandyopadhyay A.Recent advances in bone tissue engineering scaffolds.Trends Biotechnol.2012;30(10):546-554.[10] Wu S, Liu X,Yeung KWK,et al. Biomimetic porous scaffolds for bone tissue engineering.Mater Sci Eng R. 2014;80:1-36.[11] Webber MJ,Khan OF,Sydlik SA, et al. R. A perspective on the clinical translation of scaffolds for tissue engineering.Ann Biomed Eng.2015;43(3):641-656.[12] Boskey AL,Roy R.Cell culture systems for studies of bone and tooth mineralization. Chem Rev. 2008;108(11):4716-4733.[13] Young MF.Bone matrix proteins: their function, regulation, and relationship to osteoporosis. Osteoporos Int.2003;14 Suppl 3: S35-42.[14] Kim SS,Sun Park M,Jeon O,et al. Poly(lactide-co-glycolide)/ hydroxyapatite composite scaffolds for bone tissue engineering. Biomaterials.2006;27(8):1399-1409.[15] Hench LL.Opening paper 2015- Some comments on Bioglass: Four Eras of Discovery and Development. Biomed Glasses. 2015;1(1):1-11.[16] Albrektsson T,Johansson C. Osteoinduction, osteoconduction and osseointegration. Eur Spine J. 2001;10 Suppl 2:S96-101.[17] Vaccaro AR.The role of the osteoconductive scaffold in synthetic bone graft. Orthopedics. 2002;25(5 Suppl):s571-578.[18] Gao C, Deng Y,Feng P, et al.Current progress in bioactive ceramic scaffolds for bone repair and regeneration. Int J Mol Sci. 2014; 15(3):4714-4732.[19] Loh QL, Choong C. Three-dimensional scaffolds for tissue engineering applications: role of porosity and pore size.Tissue Eng Part B Rev.2013;19(6):485-502.[20] Johnson T, Bahrampourian R,Patel A,et al.Fabrication of highly porous tissue-engineering scaffolds using selective spherical porogens.Biomed Mater Eng.2010;20(2):107-118.[21] Dehghani F, Annabi N. Engineering porous scaffolds using gas-based techniques. Curr Opin Biotechnol. 2011;22(5):661-666.[22] Huang Y, Han S, Pang X,et al. Electrodeposition of porous hydroxyapatite/calcium silicate composite coating on titanium for biomedical applications.Appl Surf Sci.2013;271:299-302.[23] Ligon SC,Liska R,Stampfl J,et al. Polymers for 3D Printing and Customized Additive Manufacturing. Chem Rev. 2017;117(15): 10212-10290.[24] Kim K,Yeatts A,Dean D,et al.Stereolithographic bone scaffold design parameters: osteogenic differentiation and signal expression.Tissue Eng Part B Rev.2010;16(5):523-539.[25] Do AV, Khorsand B, Geary SM, et al. 3D Printing of Scaffolds for Tissue Regeneration Applications. Adv Healthc Mater. 2015;4(12): 1742-1762.[26] Xia Y,Zhou P,Cheng X,et al.Selective laser sintering fabrication of nano-hydroxyapatite/poly-epsilon-caprolactone scaffolds for bone tissue engineering applications. Int J Nanomedicine. 2013;8: 4197-4213.[27] Murphy SV,Atala A.3D bioprinting of tissues and organs.Nat Biotechnol.2014;32(8):773-785.[28] Saunders RE, Derby B. Inkjet printing biomaterials for tissue engineering: bioprinting. Int Mater Rev.2014;59(8):430-448.[29] Koch L,Gruene M,Unger C,et al.Laser assisted cell printing.Curr Pharm Biotechnol. 2013;14(1):91-97.[30] Faulkner-Jones A,Fyfe C,Cornelissen DJ,et al.Bioprinting of human pluripotent stem cells and their directed differentiation into hepatocyte-like cells for the generation of mini-livers in 3D. Biofabrication.2015;7(4):044102.[31] Ozbolat IT, Hospodiuk M. Current advances and future perspectives in extrusion-based bioprinting. Biomaterials. 2016;76:321-343.[32] Staiger MP,Pietak AM,Huadmai J, et al.Magnesium and its alloys as orthopedic biomaterials: a review. Biomaterials. 2006; 27(9): 1728-1734.[33] Lopez-Heredia MA,Sohier J,Gaillard C,et al.Rapid prototyped porous titanium coated with calcium phosphate as a scaffold for bone tissue engineering. Biomaterials.2008;29(17):2608-2615.[34] Chen H,Wang C,Zhu X,et al.Fabrication of porous titanium scaffolds by stack sintering of microporous titanium spheres produced with centrifugal granulation technology. Mater Sci Eng C Mater Biol Appl.2014;43:182-188.[35] Bose S,Fielding G,Tarafder S,et al.Understanding of dopant-induced osteogenesis and angiogenesis in calcium phosphate ceramics.Trends Biotechnol.2013;31(10):594-605.[36] Atkins GJ,Welldon KJ,Halbout P,et al.Strontium ranelate treatment of human primary osteoblasts promotes an osteocyte-like phenotype while eliciting an osteoprotegerin response. Osteoporos Int.2009;20(4):653-664.[37] Lei Y,Xu Z,Ke Q,et al.Strontium hydroxyapatite/chitosan nanohybrid scaffolds with enhanced osteoinductivity for bone tissue engineering. Mater Sci Eng C Mater Biol Appl. 2017;72: 134-142.[38] Xiu P,Jia ZJ,Lv J, et al.Hierarchical Micropore/Nanorod Apatite Hybrids In-Situ Grown from 3-D Printed Macroporous Ti6Al4V Implants with Improved Bioactivity and Osseointegration.J Mater Sci Technol.2017;33(2):179-186.[39] Peuster M,Hesse C,Schloo T,et al.Long-term biocompatibility of a corrodible peripheral iron stent in the porcine descending aorta. Biomaterials.2006;27(28):4955-4962.[40] Chou DT,Wells D,Hong D,et al. Novel processing of iron-manganese alloy-based biomaterials by inkjet 3-D printing. Acta Biomater.2013;9(10):8593-8603.[41] Ho GW, Matinlinna JP.Insights on Ceramics as Dental Materials. Part I: Ceramic Material Types in Dentistry. Silicon. 2011;3(3): 109-115.[42] Bohner M, Galea L, Doebelin N. Calcium phosphate bone graft substitutes: Failures and hopes. J Eur Ceram Soc. 2012;32(11): 2663-2671.[43] Ko CL, Chen WC, Chen JC, et al. Properties of osteoconductive biomaterials: calcium phosphate cement with different ratios of platelet-rich plasma as identifiers. Mater Sci Eng C Mater Biol Appl.2013;33(6):3537-3544.[44] Tang Z,Tan Y,Ni Y,et al.Comparison of ectopic bone formation process induced by four calcium phosphate ceramics in mice. Mater Sci Eng C Mater Biol Appl.2017;70(Pt 2):1000-1010.[45] Mohseni M,Jahandideh A,Abedi G,et al.Assessment of tricalcium phosphate/collagen (TCP/collagene)nanocomposite scaffold compared with hydroxyapatite (HA) on healing of segmental femur bone defect in rabbits.Artif Cells Nanomed Biotechnol. 2018;46(2):242-249.[46] Tarafder S,Bose S.Polycaprolactone-coated 3D printed tricalcium phosphate scaffolds for bone tissue engineering: in vitro alendronate release behavior and local delivery effect on in vivo osteogenesis.ACS Appl Mater Interfaces.2014;6(13):9955-9965.[47] Dai Y,Liu H,Liu B,et al.Porous β-Ca2SiO4 ceramic scaffolds for bone tissue engineering: In vitro and in vivo characterization. Ceram Int.2015;41(4):5894-5902.[48] Pasold J,Markhoff J,Tillmann J,et al.Direct influence of titanium and zirconia particles on the morphology and functionality of mature human osteoclasts. J Biomed Mater Res A. 2017; 105(9): 2608-2615.[49] Jones JR.Review of bioactive glass: from Hench to hybrids. Acta Biomater.2013;9(1):4457-4486.[50] Rahaman MN,Day DE,Bal BS,et al.Bioactive glass in tissue engineering.Acta Biomater.2011;7(6):2355-2373.[51] Miguez-Pacheco V,Hench LL,Boccaccini AR.Bioactive glasses beyond bone and teeth: emerging applications in contact with soft tissues. Acta Biomater.2015;13:1-15.[52] Habraken WJ, Wolke JG, Jansen JA. Ceramic composites as matrices and scaffolds for drug delivery in tissue engineering.Adv Drug Deliv Rev.2007;59(4-5):234-248.[53] Westhauser F,Weis C,Prokscha M,et al. Three-dimensional polymer coated 45S5-type bioactive glass scaffolds seeded with human mesenchymal stem cells show bone formation in vivo. J Mater Sci Mater Med. 2016;27(7):119.[54] Jiang S, Zhang Y, Shu Y, et al.Amino-functionalized mesoporous bioactive glass for drug delivery. Biomed Mater. 2017; 12(2): 025017.[55] Zhang Y,Cui X,Zhao S, et al. Evaluation of injectable strontium-containing borate bioactive glass cement with enhanced osteogenic capacity in a critical-sized rabbit femoral condyle defect model.ACS Appl Mater Interfaces. 2015;7(4): 2393-2403.[56] Ravi S, Chaikof EL. Biomaterials for vascular tissue engineering. Regen Med. 2010;5(1):107-120.[57] Zhu J,Marchant RE. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev Med Devices. 2011;8(5): 607-626.[58] Sheikh FA, Ju HW, Moon BM, et al. Hybrid scaffolds based on PLGA and silk for bone tissue engineering. J Tissue Eng Regen Med.2016;10(3):209-221.[59] Liao HT, Chen CT, Chen JP. Osteogenic differentiation and ectopic bone formation of canine bone marrow-derived mesenchymal stem cells in injectable thermo-responsive polymer hydrogel. Tissue Eng Part C Methods.2011;17(11):1139-1149.[60] Woodruff MA, Hutmacher DW. The return of a forgotten polymer - Polycaprolactone in the 21st century. Prog Polym Sci. 2010; 35(10):1217-1256.[61] Hollister SJ, Murphy WL. Scaffold translation: barriers between concept and clinic.Tissue Eng Part B Rev.2011;17(6):459-474.[62] Gloria A,De Santis R,Ambrosio L. Polymer-based composite scaffolds for tissue engineering. J Appl Biomater Biomech. 2010; 8(2):57-67.[63] Bellis SL. Advantages of RGD peptides for directing cell association with biomaterials. Biomaterials. 2011;32(18): 4205-4210.[64] Villa MM,Wang L,Huang J,et al.Bone tissue engineering with a collagen-hydroxyapatite scaffold and culture expanded bone marrow stromal cells. J Biomed Mater Res B Appl Biomater. 2015; 103(2):243-253.[65] Marcacci M,Kon E,Moukhachev V,et al.Stem cells associated with macroporous bioceramics for long bone repair: 6- to 7-year outcome of a pilot clinical study.Tissue Eng.2007;13(5):947-955.[66] Hou J,Wang J,Cao LY,et al.Segmental bone regeneration using rhBMP-2-loaded collagen/chitosan microspheres composite scaffold in a rabbit model.Biomed Mater.2012;7(3):035002.[67] Chang YS,Huang HD,Yeh KT, et al.Genetic alterations in endometrial cancer by targeted next-generation sequencing.Exp Mol Pathol.2016;100(1):8-12.[68] Yang T,Hu Y,Wang CY,et al.Fabrication of Hierarchical Macroporous Biocompatible Scaffolds by Combining Pickering High Internal Phase Emulsion Templates with Three-Dimensional Printing. ACS Appl Mater Interfaces.2017;9(27):22950-22958.[69] Shuai CJ,Yang B,Peng SP,et al.Development of composite porous scaffolds based on poly(lactide-co-glycolide)/ nano-hydroxyapatite via selective laser sintering. Int J Adv Manuf Tech. 2013;69(1-4):51-7.[70] Haider A, Gupta KC, Kang IK. Morphological effects of HA on the cell compatibility of electrospun HA/PLGA composite nanofiber scaffolds. Biomed Res Int.2014;2014:308306. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [5] | Liu Jiangfeng. Nano-hydroxyapatite/polyamide 66 composite filling combined with locking plate in the treatment of fibrous dysplasia of femoral bone [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 542-547. |
| [6] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

