Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (29): 4593-4600.doi: 10.3969/j.issn.2095-4344.0994
Safety of freeze-dried powder of human adipose-derived stem cells and its exosomes
Li Hong-chao, Jin Yin-peng, Wang Xi, Li Li, Fu Qing-chun
- Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, China
-
Revised:2018-06-12Online:2018-10-18Published:2018-10-18 -
Contact:Fu Qing-chun, Master, Chief physician, Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, China -
About author:Li Hong-chao, Master, Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, China. Jin Yin-peng, Master, Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, China. Li Hong-chao and Jin Yin-peng contributed equally to this work. -
Supported by:the Major Medical Innovation Project of the Nanjing Military Region, No. 14ZX01; the Chinese Foundation for Hepatitis Prevention and Control-the Tianqing Foundation for Liver Diseases, No. TQGB20150104
CLC Number:
Cite this article
Li Hong-chao, Jin Yin-peng, Wang Xi, Li Li, Fu Qing-chun. Safety of freeze-dried powder of human adipose-derived stem cells and its exosomes[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(29): 4593-4600.
share this article
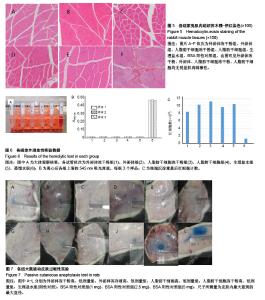
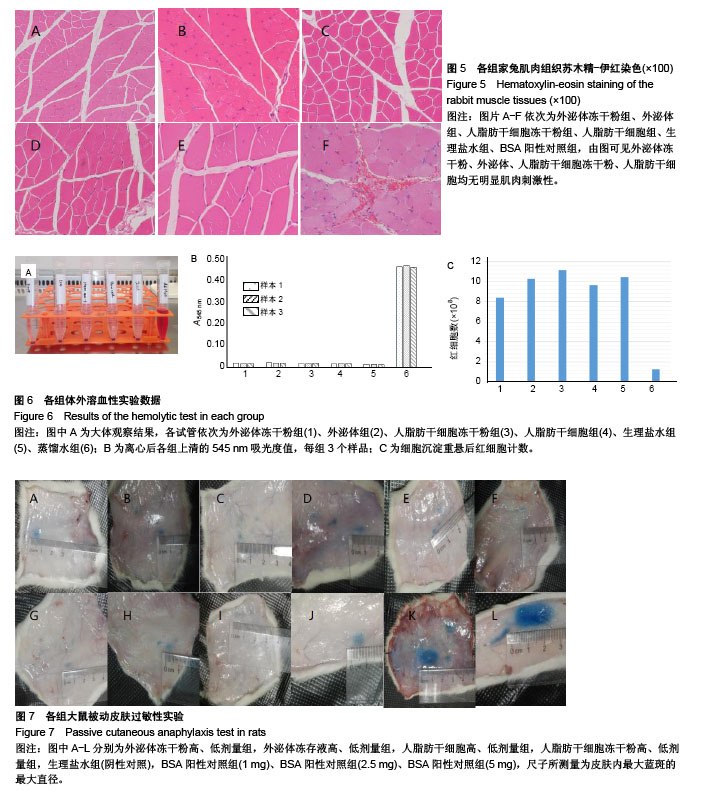
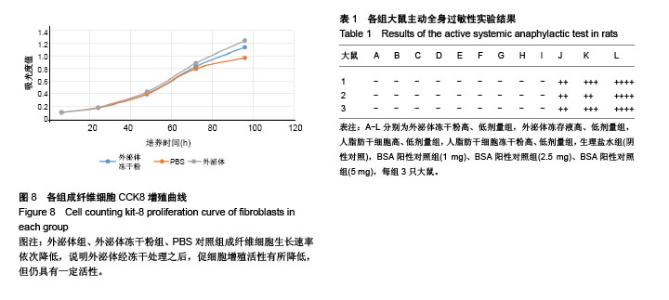
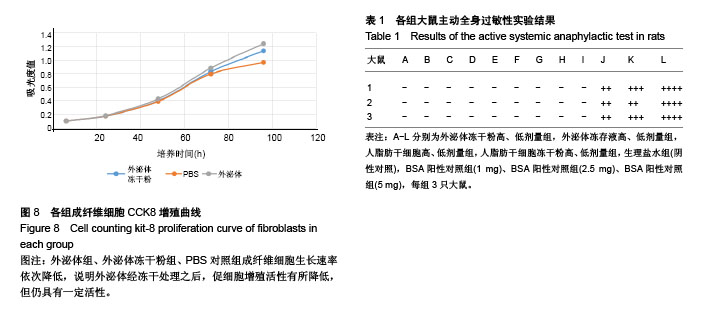
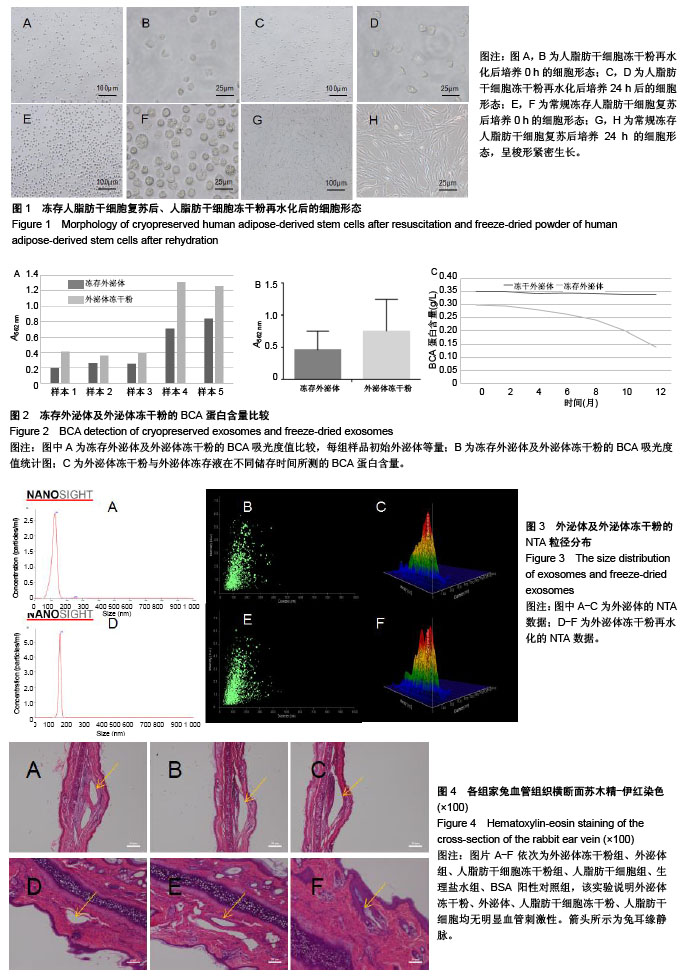
2.1 冻存人脂肪干细胞及人脂肪干细胞冻干粉的形态 见图1。普通冻存的人脂肪干细胞复苏时,细胞饱满、呈圆球形,37 ℃培养24 h后呈梭形贴壁生长。与普通冻存相比,冻干再水化人脂肪干细胞形态较不规则,数量较少,部分细胞成为碎片。培养24 h,冻干再水化人脂肪干细胞均不贴壁,丧失细胞活性。 2.2 冻存外泌体及外泌体冻干粉的BCA蛋白含量比较 图2A所示,外泌体冻干粉的562 nm吸光度值均略高于冻存外泌体,即蛋白测得量较高,考虑为外泌体在冻干过程中有部分外泌体双分子膜破碎,导致其中蛋白溢出,蛋白测得值升高。图2B为外泌体与外泌体冻干粉的BCA蛋白含量统计图,两组562 nm吸光度值差异无显著性意义。图2C为外泌体的蛋白含量变化,可以看出外泌体冻干粉的蛋白含量随时间延长减少不显著,而外泌体冻存液蛋白含量随时间变化大幅度下降,考虑为外泌体蛋白裂解所致。 2.3 人脂肪干细胞外泌体及冻干粉大小分布 由图3可见,外泌体的粒径多集中在130 nm左右,外泌体冻干粉的粒径多集中在150 nm左右。外泌体冻干粉的粒径较外泌体稍大,考虑为外泌体双分子膜在冻干的过程中可能丧失部分弹性,导致膜松弛,粒径变大。 2.4 人脂肪干细胞外泌体及冻干粉血管刺激实验结果 见图4。外泌体冻干粉组、外泌体组、人脂肪干细胞冻干粉组、人脂肪干细胞组与阴性对照生理盐水组给药部位均无明显反应,肉眼评价均为0级。从病理结果可以看出,外泌体冻干粉组、外泌体组、人脂肪干细胞冻干粉组、人脂肪干细胞组、生理盐水组以及BSA阳性对照组内皮完整,均无明显充血、变性、坏死等。BSA阳性对照组无明显充血、变性等改变可能因为该浓度的BSA血管刺激性不强。该实验可说明外泌体冻干粉、外泌体、人脂肪干细胞冻干粉、人脂肪干细胞均无明显血管刺激性。 2.5 人脂肪干细胞外泌体及冻干粉肌肉刺激实验结果 见图5。外泌体冻干粉组、外泌体组、人脂肪干细胞冻干粉组、人脂肪干细胞组与阴性对照生理盐水组内皮均完整,均无充血、变性、坏死等,评为0级,BSA阳性对照组有中度充血,评为2级。该实验说明外泌体冻干粉、外泌体、人脂肪干细胞冻干粉、人脂肪干细胞均无肌肉刺激性。 2.6 人脂肪干细胞外泌体及冻干粉体外溶血实验结果 由图6A可见,前5管上清清亮,细胞沉淀较多,第6管上清红染,沉淀较少,说明蒸馏水组有明显溶血现象,其他各组溶血均不明显。各组离心后取上清测545 nm吸光度值,蒸馏水阳性组吸光度值较高,有明显溶血,其他组吸光度值均较低,溶血不明显(图6B)。细胞沉淀重悬后显微镜下进行红细胞计数,蒸馏水阳性组红细胞最少,其他组红细胞均明显高于蒸馏水组(图6C)。以上结果均提示,外泌体、外泌体冻干粉、人脂肪干细胞、人脂肪干细胞冻干粉均不会造成大规模溶血。 2.7 人脂肪干细胞外泌体及冻干粉主动全身过敏实验结果 如表1所示,外泌体冻干粉高、低剂量组,外泌体冻存液高、低剂量组,人脂肪干细胞高、低剂量组,人脂肪干细胞冻干粉高、低剂量组,生理盐水组(阴性对照)均呈阴性,BSA(1.0,2.5,5.0 mg)阳性对照组呈阳性。外泌体、外泌体冻干粉、人脂肪干细胞、人脂肪干细胞冻干粉主动全身过敏实验均呈阴性,未引起Ⅰ型超敏反应,安全性可靠。 2.8 人脂肪干细胞外泌体及冻干粉被动皮肤过敏实验结果 如图7所示,外泌体冻干粉高、低剂量组,外泌体高、低剂量组,人脂肪干细胞高、低剂量组,人脂肪干细胞冻干粉高、低剂量组,生理盐水组的皮肤蓝斑最大直径分别为4,3,2,4,3,2,3,4,2,10,15,25 mm,均小于BSA(1.0,2.5,5.0 mg)阳性对照组,说明外泌体、外泌体冻干粉、人脂肪干细胞、人脂肪干细胞冻干粉主动全身过敏实验均呈阴性,不会引起明显皮肤反应。 2.9 人成纤维细胞的增殖曲线 图8为成纤维细胞的CCK8生长曲线,外泌体组、外泌体冻干粉组、PBS对照组成纤维细胞生长速率依次降低,说明外泌体经冻干处理之后,促细胞增殖活性有所降低,但仍具有一定活性。"
| [1] Chen G, Jin Y, Shi X, et al. Adipose-derived stem cell-based treatment for acute liver failure. Stem Cell Res Ther. 2015;6:40.[2] 罗永慧,刘漪沦.脂肪间充质干细胞研究应用进展[J].成都医学院学报, 2015,10(3):360-362.[3] Shi R, Jin Y, Cao C, et al. Localization of human adipose-derived stem cells and their effect in repair of diabetic foot ulcers in rats. Stem Cell Res Ther. 2016;7(1):155.[4] Zhu F, Chong Lee Shin OLS, Pei G, et al. Adipose-derived mesenchymal stem cells employed exosomes to attenuate AKI-CKD transition through tubular epithelial cell dependent Sox9 activation. Oncotarget. 2017;8(41):70707-70726.[5] Zhu CT, Li T, Hu YH, et al. Exosomes secreted by mice adipose-derived stem cells after low-level laser irradiation treatment reduce apoptosis of osteocyte induced by hypoxia. Eur Rev Med Pharmacol Sci 2017;21(24):5562-5570.[6] Patel RS, Carter G, El Bassit G, et al. Adipose-derived stem cells from lean and obese humans show depot specific differences in their stem cell markers, exosome contents and senescence: role of protein kinase C delta (PKCδ) in adipose stem cell niche. Stem Cell Investig. 2016;3:2.[7] García-Contreras M, Vera-Donoso CD, Hernández-Andreu JM, et al. Therapeutic potential of human adipose-derived stem cells (ADSCs) from cancer patients: a pilot study. PLoS One. 2014;9(11):e113288.[8] Camussi G, Deregibus MC, Bruno S, et al. Exosomes/ microvesicles as a mechanism of cell-to-cell communication. Kidney Int. 2010;78(9):838-848.[9] Gauthier SA, Pérez-González R, Sharma A, et al. Enhanced exosome secretion in Down syndrome brain - a protective mechanism to alleviate neuronal endosomal abnormalities. Acta Neuropathol Commun. 2017;5(1):65.[10] Zhou Y, Zhou G, Tian C, et al. Exosome-mediated small RNA delivery for gene therapy. Wiley Interdiscip Rev RNA. 2016; 7(6): 758-771.[11] Xu AT, Lu JT, Ran ZH, et al. Exosome in intestinal mucosal immunity. J Gastroenterol Hepatol. 2016;31(10):1694-1699.[12] Jia Y, Chen Y, Wang Q, et al. Exosome: emerging biomarker in breast cancer. Oncotarget. 2017;8(25):41717-41733.[13] Whiteside TL. Exosomes in Cancer: Another Mechanism of Tumor-Induced Immune Suppression. Adv Exp Med Biol. 2017; 1036:81-89.[14] Tatsumi K, Okano T. Hepatocyte Transplantation: Cell Sheet Technology for Liver Cell Transplantation. Curr Transplant Rep. 2017;4(3):184-192.[15] Wang J, Zheng Y, Zhao M. Exosome-Based Cancer Therapy: Implication for Targeting Cancer Stem Cells. Front Pharmacol. 2017;7:533.[16] Oishi Y, Manabe I. Adipose stem cell system. Clin Calcium. 2017; 27(6):795-801.[17] Kim SC, Im W. Adipose stem cell neurospheres for Huntington's disease. Cytotherapy. 2017;19(12):1546-1547.[18] Ben Nasr M, Frumento D, Fiorina P. Adipose Stem Cell Therapy for Chronic Pancreatitis. Mol Ther. 2017;25(11):2438-2439.[19] Sun Z, Gou W, Kim DS, et al. Adipose Stem Cell Therapy Mitigates Chronic Pancreatitis via Differentiation into Acinar-like Cells in Mice. Mol Ther. 2017;25(11):2490-2501.[20] Anticoli S, Aricò E, Arenaccio C, et al. Engineered exosomes emerging from muscle cells break immune tolerance to HER2 in transgenic mice and induce antigen-specific CTLs upon challenge by human dendritic cells. J Mol Med (Berl). 2018;96(2):211-221.[21] Figueroa J, Phillips LM, Shahar T, et al. Exosomes from Glioma-Associated Mesenchymal Stem Cells Increase the Tumorigenicity of Glioma Stem-like Cells via Transfer of miR-1587. Cancer Res. 2017;77(21):5808-5819.[22] Liu L, Jin X, Hu CF, et al. Exosomes Derived from Mesenchymal Stem Cells Rescue Myocardial Ischaemia/Reperfusion Injury by Inducing Cardiomyocyte Autophagy Via AMPK and Akt Pathways. Cell Physiol Biochem. 2017;43(1):52-68.[23] Xu QQ, Ge RL, Sun HY, et al. Research Advance on Promoting Angiogenesis of Mesenchymal Stem Cells-Review. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2017;25(4):1267-1270.[24] Thakral S, Wolf A, Beilman GJ, et al. Development and in vivo evaluation of a novel lyophilized formulation for the treatment of hemorrhagic shock. Int J Pharm. 2018;537(1-2):162-171.[25] Vigvári S, Sipos D, Solt J, et al. Faecal microbiota transplantation for Clostridium difficile infection using a lyophilized inoculum from non-related donors: A case series involving 19 patients. Acta Microbiol Immunol Hung. 2017:1-10.[26] Yang YZ, Zhang XY, Fang LJ, et al. Role of Exosomes in the Cross-Talk between Leukemia Cells and Mesenchymal Stem Cells -Review. Zhongguo Shi Yan Xue Ye Xue Za Zhi. 2017;25(4): 1255-1258.[27] Maitani Y, Aso Y, Yamada A, et al. Effect of sugars on storage stability of lyophilized liposome/DNA complexes with high transfection efficiency. Int J Pharm. 2008;356(1-2):69-75.[28] Jizomoto H, Hirano K. Encapsulation of drugs by lyophilized empty dipalmitoylphosphatidylcholine liposomes: effect of calcium ion. Chem Pharm Bull (Tokyo). 1989;37(11):3066-3069.[29] Huang X, Karabudak A, Comber JD, et al. A novel immunization approach for dengue infection based on conserved T cell epitopes formulated in calcium phosphate nanoparticles. Hum Vaccin Immunother. 2017;13(11):2612-2625.[30] Lin LS, Kayasuga-Kariya Y, Nakamura S, et al. Co-lyophilized Aspirin with Trehalose Causes Less Injury to Human Gastric Cells and Gastric Mucosa of Rats. Dig Dis Sci. 2016;61(8):2242-2251.[31] Kayasuga-Kariya Y, Iwanaga S, Fujisawa A, et al. Dermal cell damage induced by topical application of non-steroidal anti-inflammatory drugs is suppressed by trehalose co-lyophilization in ex vivo analysis. J Vet Med Sci. 2013;75(12): 1619-1622. [32] Zhou C, Cleland D, Snell J, et al. Formation of Stable Nanobubbles on Reconstituting Lyophilized Formulations Containing Trehalose. J Pharm Sci. 2016;105(7):2249-2253.[33] Lee J, Lin EW, Lau UY, et al. Trehalose glycopolymers as excipients for protein stabilization. Biomacromolecules. 2013;14(8): 2561-2569.[34] Zhao J, Wang S, Bao J, et al. Trehalose maintains bioactivity and promotes sustained release of BMP-2 from lyophilized CDHA scaffolds for enhanced osteogenesis in vitro and in vivo. PLoS One. 2013;8(1):e54645.[35] 权国波,章金刚,韩颖. 人红细胞冰冻干燥保存研究的进展[J]. 中国实验血液学杂志, 2006,14(1): 191-196.[36] Smith JL, Lepping MD, Rule DM, et al. Evidence for Field-Evolved Resistance of Striacosta albicosta (Lepidoptera: Noctuidae) to Cry1F Bacillus thuringiensis Protein and Transgenic Corn Hybrids in Ontario, Canada. J Econ Entomol. 2017;110(5):2217-2228.[37] Mensink MA, Šibík J, Frijlink HW, et al. Thermal Gradient Mid- and Far-Infrared Spectroscopy as Tools for Characterization of Protein Carbohydrate Lyophilizates. Mol Pharm. 2017;14(10):3550-3557.[38] Dégardin K, Desponds A, Roggo Y. Protein-based medicines analysis by Raman spectroscopy for the detection of counterfeits. Forensic Sci Int. 2017;278:313-325.[39] Pérez-Boza J, Lion M, Struman I. Exploring the RNA landscape of endothelial exosomes. RNA. 2018;24(3):423-435.[40] Qi J, Zhou Y, Jiao Z, et al. Exosomes Derived from Human Bone Marrow Mesenchymal Stem Cells Promote Tumor Growth Through Hedgehog Signaling Pathway. Cell Physiol Biochem. 2017;42(6): 2242-2254.[41] Zheng T, Wu X, Wei X, et al. The release and transmission of amyloid precursor protein via exosomes. Neurochem Int. 2018;114: 18-25.[42] Munson P, Shukla A. Exosomes: Potential in Cancer Diagnosis and Therapy. Medicines (Basel). 2015;2(4):310-327.[43] Silva M, Melo SA. Non-coding RNAs in Exosomes: New Players in Cancer Biology. Curr Genomics. 2015;16(5):295-303.[44] 中药、天然药物免疫毒性(过敏性、光变态反应)研究的技术指导原则[DB/OL]. https://wenku.baidu.com/view/ ef76d775aeaad1f347933f45.html,2015-10-14/2018-02-12.[45] Almeida EV, De Brito SL. Alkaline degradation of lyophilized DMSA prior to labeling with 99mTc: Identification and development of the degradation pathway by HPLC and MS. Nucl Med Biol. 2018;57: 20-27.[46] Ventimiglia LN, Alonso MA. Biogenesis and Function of T Cell-Derived Exosomes. Front Cell Dev Biol. 2016;4:84.[47] Shenoda BB, Ajit SK. Modulation of Immune Responses by Exosomes Derived from Antigen-Presenting Cells. Clin Med Insights Pathol. 2016;9(Suppl 1):1-8.[48] Chen Y, Tang Y, Long W, et al. Stem Cell-Released Microvesicles and Exosomes as Novel Biomarkers and Treatments of Diseases. Stem Cells Int. 2016;2016:2417268. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||