Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (25): 4041-4046.doi: 10.3969/j.issn.2095-4344.0961
Previous Articles Next Articles
Oligodendrocyte progenitor cell transplantation in combination with miconazole for myelin repair in mice with leukodystrophy
Wu Cheng-jun1, Su Xue-wen2, Wang Zhao-yan1, Yang Yin-xiang1, Luan Zuo1
- 1Department of Pediatrics, Navy General Hospital of PLA, Beijing 100048, China; 2Department of Pediatrics, Inner Mongolia People’s Hospital, Hohhot 010017, Inner Mongolia Autonomous Region, China
-
Revised:2018-04-04Online:2018-09-08Published:2018-09-08 -
Contact:Luan Zuo, Master, Chief physician, Department of Pediatrics, Navy General Hospital of PLA, Beijing 100048, China -
About author:Wu Cheng-jun, Master candidate, Department of Pediatrics, Navy General Hospital of PLA, Beijing 100048, China -
Supported by:the National Key Research and Development Plan for Stem Cell Research and Transformation, No. 2017YFA0104200; the National Natural Science Foundation of China, No. 81471486
CLC Number:
Cite this article
Wu Cheng-jun, Su Xue-wen, Wang Zhao-yan, Yang Yin-xiang, Luan Zuo. Oligodendrocyte progenitor cell transplantation in combination with miconazole for myelin repair in mice with leukodystrophy[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(25): 4041-4046.
share this article
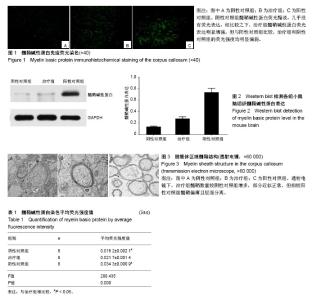
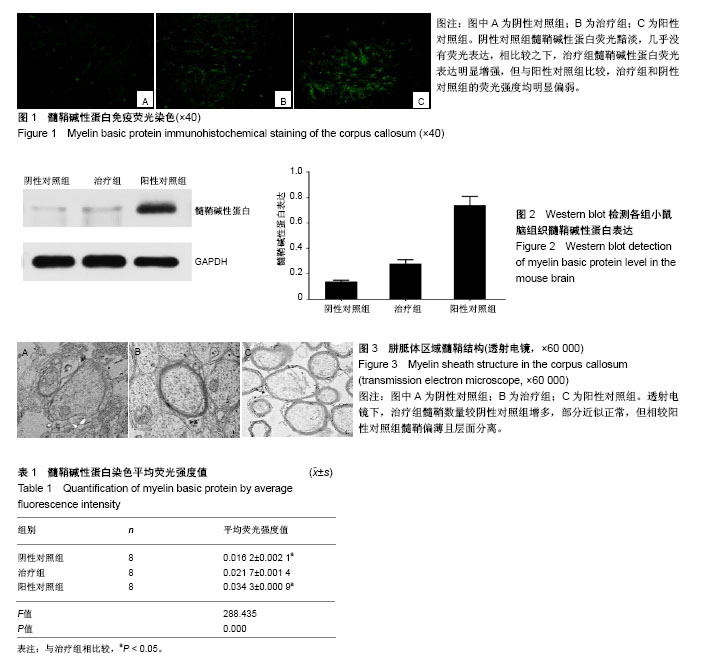
2.1 实验动物大体观察结果 OPCs、PBS的立体定位移植进展顺利,进针、细胞注射均无阻滞,期间小鼠反应平稳,拔针后观察针孔附近无出血、无液体渗漏等情况发生,术后小鼠无不良反应,苏醒后活动如常。咪康唑、生理盐水的腹腔注射过程顺利,注射完毕留针2 min,拔针后观察小鼠腹部表面未见出血、液体渗漏等情况发生。观察期内小鼠正常长大,无提前死亡、无不良疾病等现象发生。 2.2 免疫荧光染色结果 MBP免疫荧光染色观察结果见图1。阴性对照组MBP荧光黯淡,几乎没有荧光表达,相比较之下,治疗组MBP荧光表达明显增强,但与阳性对照组比较,治疗组和阴性对照组的荧光强度均明显偏弱。荧光定量分析结果见表1,与阴性对照组比较,治疗组的平均荧光强度值明显增高,差异有显著性意义(P < 0.05);但与阳性对照组比较,治疗组的平均荧光强度值明显降低,差异有显著性意义(P < 0.05)。 2.3 Western blot检测MBP表达量 取各组小鼠脑组织进行Western blot检测,结果见图2。MBP蛋白表达量(以目的条带和内参条带吸光度值比值表示)在3组间均有明显差异。治疗组的MBP蛋白表达量(0.28±0.03)比阴性对照组(0.14±0.01)高,且比阳性对照组(0.74±0.07)低,差异有显著性意义。结果显示OPCs治疗可增加shi-/-鼠的MBP蛋白表达量,提示其有助于改善脱髓鞘小鼠的脑白质髓鞘形成不良状况,但不足以完全修复髓鞘,与MBP免疫荧光染色结果相一致。 2.4 透射电镜观察结果 透射电镜观察小鼠胼胝体区域的髓鞘结构,见图3。阴性对照组罕见髓鞘结构,轴突形态不规则,板层排列松散,紊乱,与轴索间隙宽大,髓鞘厚度明显降低;治疗组髓鞘数量较阴性对照组明显增多,轴突形态规则,部分髓鞘厚度增加且板层排列紧密,近似正常髓鞘,但多数髓鞘厚度仍明显减低;阳性对照组髓鞘明显增多,所有髓鞘均形态均匀一致,厚度增加,髓鞘边界清晰,板层排列紧密,无松散、紊乱现象。结果提示OPCs治疗可促进脱髓鞘小鼠的髓鞘再生,但尚不足以恢复正常髓鞘结构。"
| [1] Yang E, Prabhu SP. Imaging manifestations of the leukodystrophies, inherited disorders of white matter. Radiol Clin North Am. 2014;52(2):279-319.[2] Leite CC, Lucato LT, Santos GT, et al. Imaging of adult leukodystrophies. Arq Neuropsiquiatr. 2014;72(8):625-632.[3] Turón-Viñas E, Pineda M, Cusí V, et al. Vanishing white matter disease in a spanish population. J Cent Nerv Syst Dis. 2014;6:59-68.[4] Helman G, Van Haren K, Bonkowsky JL, et al. Disease specific therapies in leukodystrophies and leukoencephalopathies. Mol Genet Metab. 2015;114(4):527-536.[5] Raff MC, Abney ER, Cohen J, et al. Two types of astrocytes in cultures of developing rat white matter: differences in morphology, surface gangliosides, and growth characteristics. J Neurosci. 1983;3(6):1289-1300.[6] Miranda CO, Brites P, Mendes Sousa M, et al. Advances and pitfalls of cell therapy in metabolic leukodystrophies. Cell Transplant. 2013;22(2):189-204.[7] Najm FJ, Madhavan M, Zaremba A, et al. Drug-based modulation of endogenous stem cells promotes functional remyelination in vivo. Nature. 2015;522(7555):216-220.[8] Wang C, Luan Z, Yang Y, et al. High purity of human oligodendrocyte progenitor cells obtained from neural stem cells: suitable for clinical application. J Neurosci Methods. 2015;240:61-66.[9] Bunge RP. Glial cells and the central myelin sheath. Physiol Rev. 1968;48(1):197-251.[10] Filley CM, Fields RD. White matter and cognition: making the connection. J Neurophysiol. 2016;116(5):2093-2104.[11] Lesca G, Vanier MT, Creisson E, et al. X-linked adrenoleukodystrophy in a female proband: clinical presentation, biological diagnosis and family consequences. Arch Pediatr. 2005;12(8):1237-1240.[12] van der Knaap MS, Bugiani M. Leukodystrophies: a proposed classification system based on pathological changes and pathogenetic mechanisms. Acta Neuropathol. 2017;134(3): 351-382.[13] Keough MB, Yong VW. Remyelination therapy for multiple sclerosis. Neurotherapeutics. 2013;10(1):44-54.[14] Pouwels PJ, Vanderver A, Bernard G, et al. Hypomyelinating leukodystrophies: translational research progress and prospects. Ann Neurol. 2014;76(1):5-19.[15] Raff MC, Abney ER, Cohen J, et al. Two types of astrocytes in cultures of developing rat white matter: differences in morphology, surface gangliosides, and growth characteristics. J Neurosci. 1983;3(6):1289-1300.[16] Hughes EG, Kang SH, Fukaya M, et al. Oligodendrocyte progenitors balance growth with self-repulsion to achieve homeostasis in the adult brain. Nat Neurosci. 2013;16(6): 668-676.[17] El Waly B, Macchi M, Cayre M, et al. Oligodendrogenesis in the normal and pathological central nervous system. Front Neurosci. 2014;8:145.[18] Bergles DE, Richardson WD. Oligodendrocyte Development and Plasticity. Cold Spring Harb Perspect Biol. 2015;8(2): a020453.[19] Yeung MS, Zdunek S, Bergmann O, et al. Dynamics of oligodendrocyte generation and myelination in the human brain. Cell. 2014;159(4):766-774.[20] Nave KA, Werner HB. Myelination of the nervous system: mechanisms and functions. Annu Rev Cell Dev Biol. 2014; 30:503-533.[21] Buller B, Chopp M, Ueno Y, et al. Regulation of serum response factor by miRNA-200 and miRNA-9 modulates oligodendrocyte progenitor cell differentiation. Glia. 2012; 60(12):1906-1914.[22] Wootla B, Watzlawik JO, Denic A, et al. The road to remyelination in demyelinating diseases: current status and prospects for clinical treatment. Expert Rev Clin Immunol. 2013;9(6):535-549.[23] McMurran CE, Kodali S, Young A, et al. Clinical implications of myelin regeneration in the central nervous system. Expert Rev Neurother. 2018;18(2):111-123.[24] Kremer D, Küry P, Dutta R. Promoting remyelination in multiple sclerosis: current drugs and future prospects. Mult Scler. 2015;21(5):541-549.[25] Deshmukh VA, Tardif V, Lyssiotis CA, et al. A regenerative approach to the treatment of multiple sclerosis. Nature. 2013; 502(7471):327-332.[26] Mi S, Pepinsky RB, Cadavid D. Blocking LINGO-1 as a therapy to promote CNS repair: from concept to the clinic. CNS Drugs. 2013;27(7):493-503.[27] Sun JJ, Ren QG, Xu L, et al. LINGO-1 antibody ameliorates myelin impairment and spatial memory deficits in experimental autoimmune encephalomyelitis mice. Sci Rep. 2015;5:14235.[28] Czepiel M, Boddeke E, Copray S. Human oligodendrocytes in remyelination research. Glia. 2015;63(4):513-530.[29] Osorio MJ, Rowitch DH, Tesar P, et al. Concise Review: Stem Cell-Based Treatment of Pelizaeus-Merzbacher Disease. Stem Cells. 2017;35(2):311-315.[30] Chen LX, Ma SM, Zhang P, et al. Neuroprotective effects of oligodendrocyte progenitor cell transplantation in premature rat brain following hypoxic-ischemic injury. PLoS One. 2015; 10(3):e0115997.[31] Kobayashi Y, Okada Y, Itakura G, et al. Pre-evaluated safe human iPSC-derived neural stem cells promote functional recovery after spinal cord injury in common marmoset without tumorigenicity. PLoS One. 2012;7(12):e52787.[32] Hatch MN, Schaumburg CS, Lane TE, et al. Endogenous remyelination is induced by transplant rejection in a viral model of multiple sclerosis. J Neuroimmunol. 2009;212 (1-2):74-81.[33] Pouya A, Satarian L, Kiani S, et al. Human induced pluripotent stem cells differentiation into oligodendrocyte progenitors and transplantation in a rat model of optic chiasm demyelination. PLoS One. 2011;6(11):e27925.[34] Roach A, Boylan K, Horvath S, et al. Characterization of cloned cDNA representing rat myelin basic protein: absence of expression in brain of shiverer mutant mice. Cell. 1983; 34(3):799-806.[35] Xu WJ,Lakshman N,Morshead CM.Stem cells in the adult CNS revealed: examining their regulation by myelin basic protein.Neural Regen Res.2016;11(12):1916-1917. [36] Pedraza L, Fidler L, Staugaitis SM, et al. The active transport of myelin basic protein into the nucleus suggests a regulatory role in myelination. Neuron. 1997;18(4):579-589.[37] Weil MT, Möbius W, Winkler A, et al. Loss of Myelin Basic Protein Function Triggers Myelin Breakdown in Models of Demyelinating Diseases. Cell Rep. 2016;16(2):314-322.[38] Kelly CM, Precious SV, Scherf C, et al. Neonatal desensitization allows long-term survival of neural xenotransplants without immunosuppression. Nat Methods. 2009;6(4):271-273. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Xie Yang, Lü Zhiyu, Zhang Shujiang, Long Ting, Li Zuoxiao. Effects of recombinant adeno-associated virus mediated nerve growth factor gene transfection on oligodendrocyte apoptosis and myelination in experimental autoimmune encephalomyelitis mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(23): 3678-3683. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||