Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (30): 4913-4920.doi: 10.3969/j.issn.2095-4344.0938
A meta-analysis of repair materials used in cranioplasty
Chen Zhi-qian, Mu Xiong-zheng
- Department of Plastic Surgery, Huashan Hospital of Fudan University, Shanghai 200040, China
-
Received:2018-03-29Online:2018-10-28Published:2018-10-28 -
Contact:Mu Xiong-zheng, Professor, Chief physician, Doctoral supervisor, Department of Plastic Surgery, Huashan Hospital of Fudan University, Shanghai 200040, China -
About author:Chen Zhi-qian, Master candidate, Department of Plastic Surgery, Huashan Hospital of Fudan University, Shanghai 200040, China -
Supported by:Foundation of Shanghai Hospital Development Center for Emerging Frontier Technology Projects, No. SHDC12014113
CLC Number:
Cite this article
Chen Zhi-qian, Mu Xiong-zheng. A meta-analysis of repair materials used in cranioplasty[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(30): 4913-4920.
share this article

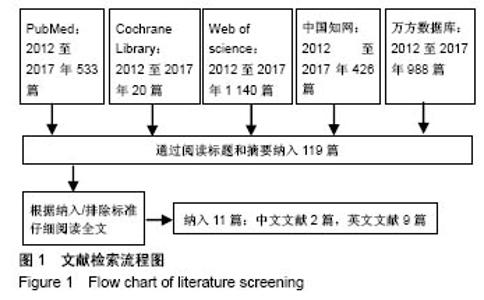
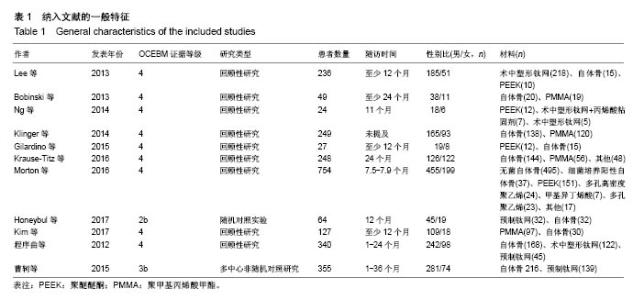
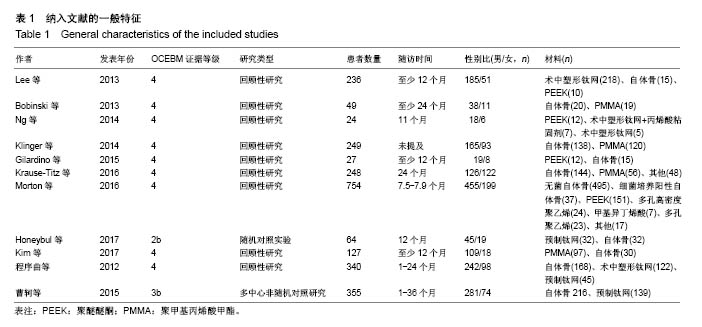
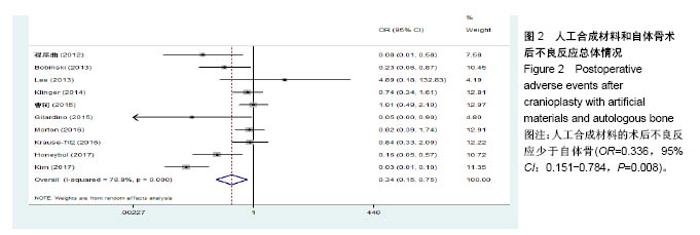
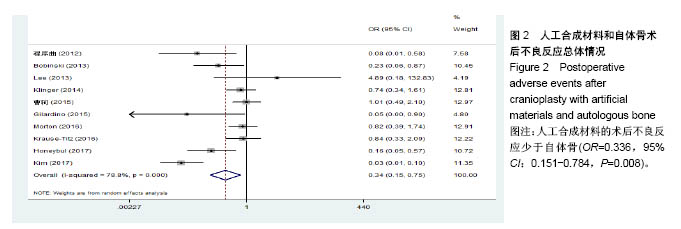
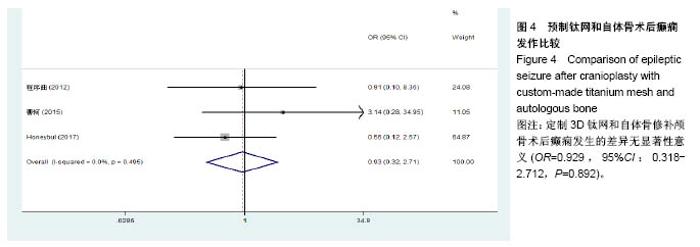
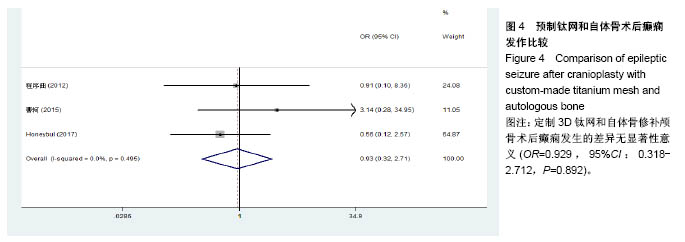
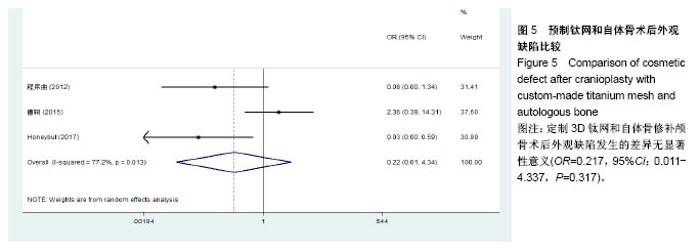
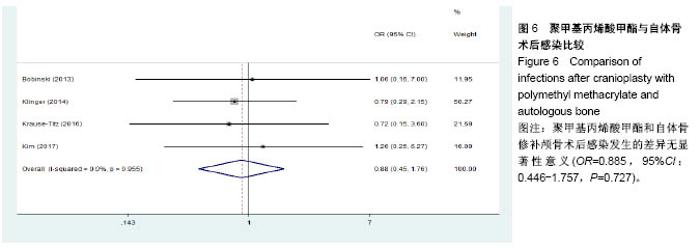
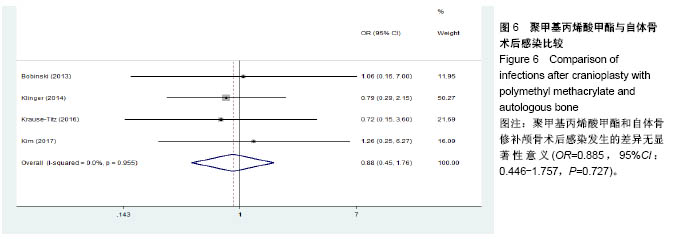
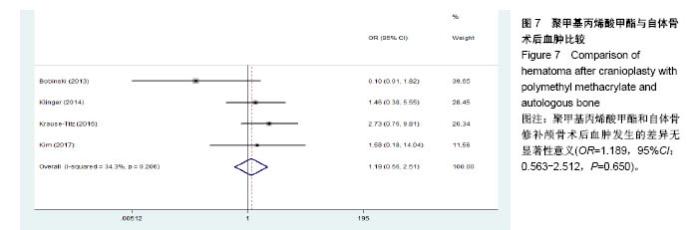
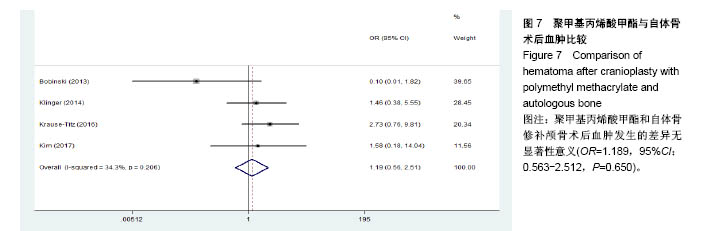
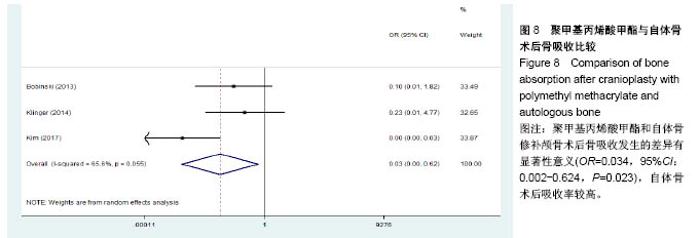
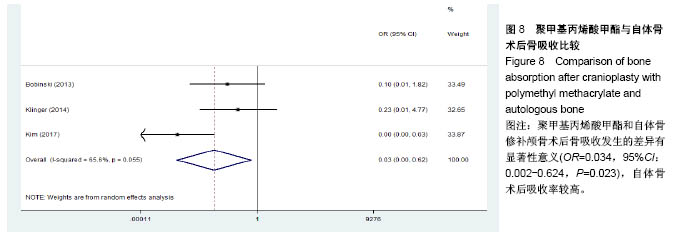
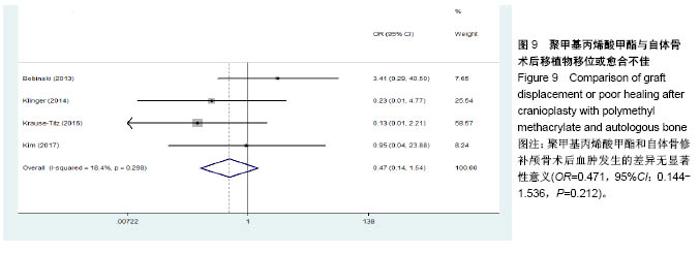
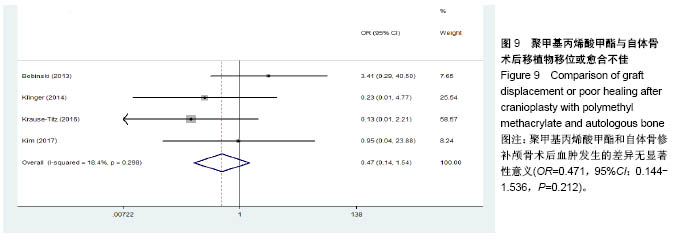
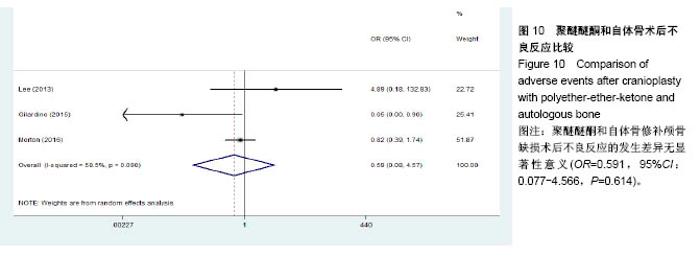
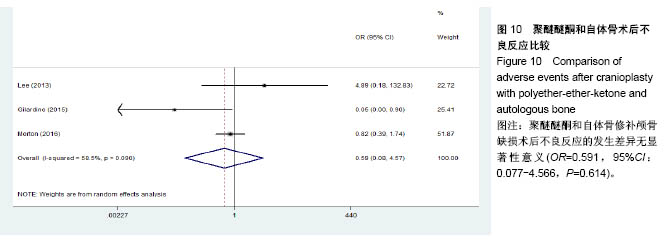
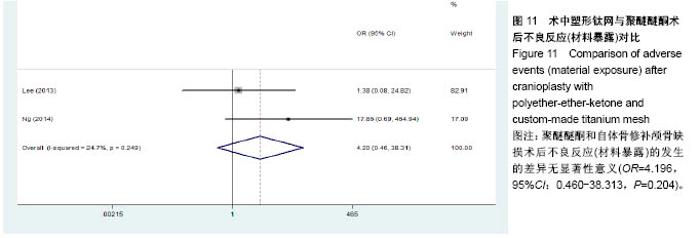
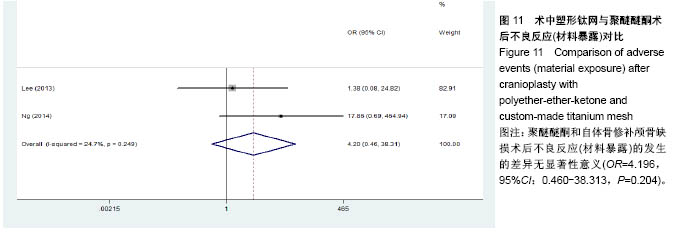
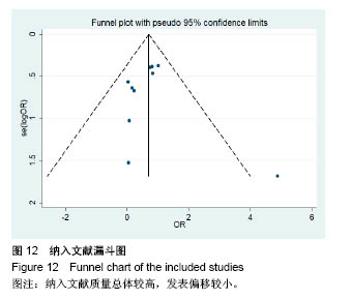
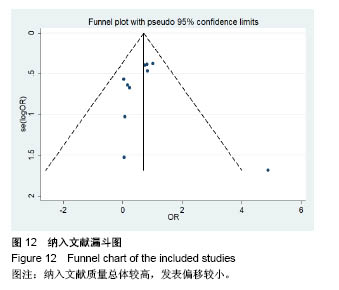
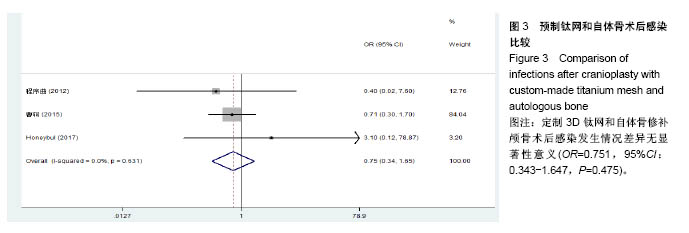
2.3.2 预制钛网和自体骨术后不良反应比较 术后感染:共有3个研究报告了预制钛网与自体骨术后感染的情况(图3)[13-14,21],各研究间无明显异质性(P=0.631,I2=0.0%),采用固定效应模型分析;Meta分析结果显示:定制3D钛网和自体骨修补颅骨术后感染发生情况差异无显著性意义(OR=0.751,95%CI:0.343-1.647,P=0.475)。 术后癫痫:共3个研究报告了预制钛网和自体骨术后癫痫发作的情况(图4)[13-14,21],各研究间无明显异质性(P=0.495,I2=0.0%),采用固定效应模型分析;Meta分析结果显示:定制3D钛网和自体骨修补颅骨术后癫痫发生的差异无显著性意义(OR=0.929,95%CI:0.318-2.712,P= 0.892)。 术后外观缺陷:共3个研究报告了预制钛网和自体骨术后癫痫发作的情况(图5)[13-14,21],各研究间存在异质性(P=0.013,I2=77.2%),采用随机效应模型分析;Meta分析结果显示:定制3D钛网和自体骨修补颅骨术后外观缺陷发生的差异无显著性意义(OR=0.217,95%CI:0.011- 4.337,P=0.317)。 2.3.3 PMMA和自体骨术后不良反应比较 术后感染:共4个研究报告了PMMA和自体骨术后癫痫发作的情况(图6)[1,16,18,22],各研究间无明显异质性(P=0.955,I2=0.0%),采用固定效应模型分析;Meta分析结果显示:PMMA和自体骨修补颅骨术后感染发生的差异无显著性意义(OR=0.885,95%CI:0.446-1.757,P=0.727)。 术后血肿(包括硬膜外血肿、硬膜下血肿、颅内血肿):共4个研究报告了PMMA和自体骨术后血肿发生的情况(图7)[1,16,18,22],各研究间无明显异质性(P=0.206,I2=34.3%),采用固定效应模型分析;Meta分析结果显示:PMMA和自体骨修补颅骨术后血肿发生的差异无显著性意义(OR=1.189,95%CI:0.563-2.512,P=0.650)。 术后骨吸收:共3个研究报告了PMMA和自体骨术后骨吸收的情况(图8)[16,18,22],各研究间存在异质性(P=0.055,I2=65.6%),采用随机效应模型分析;Meta分析结果显示:PMMA和自体骨修补颅骨术后骨吸收发生的差异有显著性意义(OR=0.034,95%CI:0.002-0.624,P=0.023);结果提示自体骨术后吸收率较高。 术后移植物移位或愈合不佳:共4个研究报告了PMMA和自体骨术后移植物移位或者愈合不佳的情况(图9)[1,16,18,22],各研究间无明显异质性(P=0.298,I2=18.4%),采用固定效应模型分析;Meta分析结果显示:PMMA和自体骨修补颅骨术后血肿发生的差异无显著性意义(OR=0.471,95%CI:0.144-1.536,P=0.212)。 2.3.4 PEEK和自体骨术后不良反应 3个研究报道了PEEK与自体骨术后不良反应的比较(包括感染、骨吸收、材料暴露等不良反应)(图10)[15,19-20];各研究间存在异质性(P=0.090,I2=58.5%),故采用随机效应模型分析;Meta分析结果显示:PEEK和自体骨修补颅骨缺损术后不良反应的发生的差异无显著性意义(OR=0.591,95%CI:0.077-4.566,P=0.614)。 2.3.5 术中塑形钛网和PEEK术后不良反应比较 共2个研究报道了术中塑形钛网与PEEK术后材料暴露的比较(图11)[15,17];各研究间无明显异质性(P=0.249,I2=24.7%),故采用固定效应模型分析;Meta分析结果显示:PEEK和自体骨修补颅骨缺损术后(材料暴露)的发生差异无显著性意义(OR=4.196,95%CI:0.460-38.313,P=0.204)。虽然结果提示无显著性意义,但是仔细分析数据发现两组的PEEK组均无材料暴露,而两组的术中塑形钛网材料暴露率分别为40.00%和5.96%,说明术中塑形钛网由于难以完全与颅骨贴合,暴露率较高。 2.3.6 对森林图的解读 OR为比值比,图中菱形表示合并后比值比区间,若其与OR=1相交,则两组之间差异无显著性意义;若在OR=1的左侧,表示实验组比对照组并发症少;反之,则结论相反。 2.4 发表偏倚分析 总体发表偏倚漏斗图见图12,所纳入研究较为集中于漏斗图顶端,且较为对称,可见该系统评价质量总体较高,发表偏倚较小。"
| [1] Krause-Titz UR, Warneke N, Freitag-Wolf S, et al. Factors influencing the outcome (GOS) in reconstructive cranioplasty. Neurosurg Rev. 2016;39(1):133-139.[2] Kurland DB, Khaladj-Ghom A, Stokum JA, et al. Complications associated with decompressive craniectomy: a systematic review. Neurocrit Care. 2015;23:292-304.[3] Yamaura A, Makino H. Neurological deficits in the presence of the sinking skin flap following decompressive craniectomy. Neurol Med Chir (Tokyo). 1977;17(1Pt1):43-53.[4] Kasprzak P, Ormezowska E, Jaskólski D. Cranioplasty as the return-to-work factor-112 patients with cranial defects treated in the Department of Neurosurgery at the Medical University Of Lodz. Int J Occup Med Environ Health. 2017;30(5):803-809.[5] 刁云锋,杨细平.几种颅骨修补材料的临床应用及不良反应防治[J].中国组织工程研究与临床康复,2011,15(16):2985-2989.[6] 王玖,郑新瑞,费舟.颅骨修补时机和应用材料的研究进展[J].临床神经外科杂志,2016,13(5):397-400.[7] Smith DM, Cooper GM, Afifi AM, et al. Regenerative surgery in cranioplasty revisited. Plast Reconstr Surg. 2011;128(5):1053-1060.[8] Khader BA, Towler MR. Materials and techniques used in cranioplasty fixation: a review. Mater Sci Eng. 2016;66:315-322.[9] Zanaty M, Chalouhi N, Starke RM, et al. Complications following cranioplasty: incidence and predictors in 348 cases. J Neurosurg. 2015;123(1):182-188.[10] Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Br Med J. 2009;6(7):e1000100.[11] OCEBM Levels of Evidence Working Group. The Oxford Levels of Evidence 2. Oxford Cent Evidence-Based Med 2012. 2015.[12] Wells G, Shea B, O’Connell D, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa Heal Res Inst 2006. 2016.[13] 程序曲,赵富文,楮荣涛,等.自体骨瓣、普通钛网、预塑二维钛网修补颅骨缺损比较[J].中国临床神经外科杂志,2012,17(1):24-26.[14] 曹轲,李宗正,刘玉飞,等.自体骨与钛网两种颅骨修补方案的疗效分析[J].中华神经外科杂志,2015,31(9):918-922.[15] Lee L, Ker J, Quah BL, et al. A retrospective analysis and review of an institution's experience with the complications of cranioplasty. Br J Neurosurg. 2013;27(5):629-635.[16] Bobinski L, Koskinen LD, Lindvall P. Complications following cranioplasty using autologous bone or polymethylmethacrylate- Retrospective experience from a single center. Clin Neurol Neurosurg. 2013;115(9):1788-1791.[17] Ng ZY, Ang WJJ, Nawaz I. Computer-Designed Polyetheretherketone Implants Versus Titanium Mesh (±Acrylic Cement) in Alloplastic Cranioplasty. J Craniofac Surg. 2014;25(2):e185-e189.[18] Klinger DR, Madden C. Autologous and acrylic cranioplasty: a review of 10 years and 258 cases. World Neurosurg. 2014;82(3-4):e525-e530.[19] Gilardino MS, Karunanayake M, Al-Humsi T, et al. A Comparison and cost analysis of cranioplasty techniques. J Craniofac Surg. 2015;26(1):113-117.[20] Morton RP, Abecassis IJ, Hanson JF, et al. Predictors of infection after 754 cranioplasty operations and the value of intraoperative cultures for cryopreserved bone flaps. J Neurosurg. 2016;125(3):766-770.[21] Honeybul S, Morrison DA, Ho KM, et al. A randomized controlled trial comparing autologous cranioplasty with custom-made titanium cranioplasty. J Neurosurg. 2017;126(1):81-90.[22] Kim SH, Kang DS, Cheong JH, et al. Comparison of complications following cranioplasty using a sterilized autologous bone flap or polymethyl methacrylate. Korean J Neurotrauma. 2017;13(1):15.[23] Sakamoto S, Eguchi K, Kiura Y, et al. CT perfusion imaging in the syndrome of the sinking skin flap before and after cranioplasty. Clin Neurol Neurosurg. 2006;108(6):583-585.[24] Di Stefano C, Rinaldesi M L, Quinquinio C, et al. Neuropsychological changes and cranioplasty: a group analysis. Brain Inj. 2016;30(2):164-171.[25] Paredes I, Castaño AM, Cepeda S, et al. The effect of cranioplasty on cerebral hemodynamics as measured by perfusion computed tomography and doppler ultrasonography. J Neurotrauma. 2016; 33(17):1586-1597.[26] Gooch MR, Gin GE, Kenning TJ, et al. Complications of cranioplasty following decompressive craniectomy: analysis of 62 cases. Neurosurg Focus. 2009;26(6):E9.[27] Schwarz F, Dünisch P, Walter J, et al. Cranioplasty after decompressive craniectomy: is there a rationale for an initial artificial bone-substitute implant? A single-center experience after 631 procedures. J Neurosurg. 2016;124(3):710-715.[28] Schoekler B, Trummer M. Prediction parameters of bone flap resorption following cranioplasty with autologous bone. Clin Neurol Neurosurg. 2014;120:64-67.[29] Chang TJ, Choi JW, Ra YS, et al. Changes in graft thickness after skull defect reconstruction with autogenous split calvarial bone graft. J Craniofac Surg. 2014;25(4):1241-1244.[30] Corliss B, Gooldy T, Vaziri S, et al. Complications after in vivo and ex vivo autologous bone flap storage for cranioplasty: a comparative analysis of the literature. World Neurosurg. 2016;96:510-515.[31] Kim JS, Park IS, Kim SK, et al. Analysis of the risk factors affecting the surgical site infection after cranioplasty following decompressive craniectomy. Korean J Neurotrauma. 2015;11(2):100.[32] Tsang AC, Hui VK, Lui W, et al. Complications of post-craniectomy cranioplasty: Risk factor analysis and implications for treatment planning. J Clin Neurosci. 2015;22(5):834-837.[33] Riordan MA, Simpson VM, Hall WA. Analysis of factors contributing to infections after cranioplasty: a single-institution retrospective chart review. World Neurosurg. 2016;87:207-213.[34] Thesleff T, Lehtimaki K, Niskakangas T, et al. Cranioplasty with adipose-derived stem cells, beta-tricalcium phosphate granules and supporting mesh: six-year clinical follow-up results. Stem Cells Transl Med. 2017;6(7):1576-1582.[35] Di Bella C, Farlie P, Penington AJ. Bone regeneration in a rabbit critical-sized skull defect using autologous adipose-derived cells. Tissue Eng Part A. 2008;14(4):483-490.[36] Roldán JC, Schulz P. BMP-7 preserves surface integrity of degradable-ceramic cranioplasty in a göttingen minipig model. Plast Reconstr Surg. 2017;5(3):e1255.[37] Smith DM, Cooper GM, Afifi A M, et al. Regenerative Surgery in Cranioplasty Revisited. Plast Reconstr Surg. 2011;128(5):1053-1060.[38] Sun M, He Y, Zhou T, et al. Adipose extracellular matrix/stromal vascular fraction gel secretes angiogenic factors and enhances skin wound healing in a murine model. Biomed Res Int. 2017;2017:1-11.[39] Liao HT, Marra KG. Application of platelet-rich plasma and platelet-rich fibrin in fat grafting: basic science and literature review. Tissue Eng. 2014;20(4):267-276. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [12] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [13] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [14] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| [15] | Liu Liyong, Zhou Lei. Research and development status and development trend of hydrogel in tissue engineering based on patent information [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3527-3533. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||