Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (8): 1161-1166.doi: 10.3969/j.issn.2095-4344.0814
Previous Articles Next Articles
Phenylephrine effects on the morphology of osteoblasts and expression of nicotinamide phosphoribosyltransferase under oxidative stress
Zhang Ju, Han Li-chi, Wang Yi-fei, Yan Xiao-xu
- Department of Oral Medicine, Medical College, Dalian University, Dalian 116622, Liaoning Province, China
-
Received:2017-11-18Online:2018-03-18Published:2018-03-18 -
Contact:Han Li-chi, M.D., Master’s supervisor, Associate chief physician, Department of Oral Medicine, Medical College, Dalian University, Dalian 116622, Liaoning Province, China -
About author:Zhang Ju, Department of Oral Medicine, Medical College, Dalian University, Dalian 116622, Liaoning Province, China -
Supported by:the Natural Science Foundation of Liaoning Province Science and Technology Department, No. 201202008; the Teaching Innovation Project of Dalian University in 2016, No. G1-65; the Innovation Education Foundation for the Undergraduates of Dalian University, No. 2014124
CLC Number:
Cite this article
Zhang Ju, Han Li-chi, Wang Yi-fei, Yan Xiao-xu. Phenylephrine effects on the morphology of osteoblasts and expression of nicotinamide phosphoribosyltransferase under oxidative stress[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(8): 1161-1166.
share this article
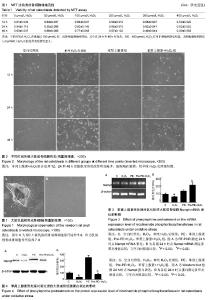
2.1 大鼠乳鼠成骨细胞形态 原代组织块法培养成骨细胞,细胞从组织块边缘长出,5 d后于倒置显微镜下可见,细胞呈三角形、梭形、其他不规则形等多种形态,细胞周围有长短不一的突起相互连接,胞浆丰富、胞核清晰可见(图1A)。传代后细胞生长迅速,5-7 d后进入指数生长期,细胞呈集落式生长,细胞间呈铺路石状汇合,细胞密集处可见复层式生长,此时细胞形态不明显、分界较模糊(图1B)。 2.2 成骨细胞的增殖情况 不同时间点细胞增殖率升高与降低趋势基本一致,H2O2浓度从0 μmol/L分别到 50 μmol/L或100 μmol/L,细胞的增殖率有所增加,之后随着浓度增加至200,300及400 μmol/L细胞增殖率降低(表1)。各组细胞增殖率升高与0 μmol/L比较差异不具有显著性意义;不同时间点H2O2浓度超过100 μmol/L时,成骨细胞增殖率降低,其中在24 h和48 h时间点,300,400 μmol/L增殖率降低明显,其细胞增殖率降低与0 μmol/L比较差异有显著性意义。为有效模拟氧化损伤,应用终浓度为 300 μmol/L的H2O2作为成骨细胞氧化损伤浓度。 2.3 H2O2氧化应激后大鼠成骨细胞形态 于倒置显微镜下观察可见:苯肾上腺素组较空白对照组细胞密度和形态未见明显变化;单纯H2O2处理组细胞正常连接消失、细胞皱缩、呈散在分布。细胞数量减少,随时间增加变化更加明显;苯肾上腺素+H2O2组在处理12,24和48 h后细胞空泡状改变明显减少,细胞略有皱缩,较单纯H2O2处理组轻微(图2)。 2.4 大鼠成骨细胞Nampt mRNA表达水平 苯肾上腺素预处理及H2O2氧化应激处理24 h后,单纯H2O2处理组Nampt mRNA表达较空白对照组减少31.23%(P < 0.05),苯肾上腺素+H2O2组Nampt mRNA表达较空白对照组减少52.51%(P < 0.01),但较单纯H2O2处理组增加206.20% (P < 0.05) (图3)。结果表明,苯肾上腺素预处理可促进氧化应激损伤后大鼠成骨细胞Nampt mRNA的表达。 2.5 大鼠成骨细胞Nampt蛋白表达水平 苯肾上腺素预处理及H2O2氧化应激处理24 h后,单纯H2O2处理组Nampt蛋白表达较空白对照组减少67.98%(P < 0.05);苯肾上腺素+H2O2组Nampt蛋白表达较空白对照组减少23.81% (P < 0.05),但苯肾上腺素+H2O2组Nampt蛋白表达较单纯H2O2处理组增加152.25%(P < 0.05) (图4)。该结果显示,苯肾上腺素预处理氧化应激的成骨细胞可明显上调其Nampt蛋白的表达。"
| [1] Suga S, Goto S, Togari A. Demonstration of Direct Neurite–Osteoclastic Cell Communication In Vitro via the Adrenergic Receptor. J Pharmacol Sci. 2010;112:184-191.[2] Hirai T, Tanaka K, Togari A. alpha1-adrenergic receptor signaling in osteoblasts regulates clock genes and bone morphogenetic protein 4 expression through up-regulation of the transcriptional factor nuclear factor IL-3 (Nfil3)/E4 promoter-binding protein 4 (E4BP4). The Journal of biological chemistry. 2014;289:17174-17183.[3] Kim KA, Kook SH, Song JH,et al.A phenolic acid phenethyl urea derivative protects against irradiation-induced osteoblast damage by modulating intracellular redox state. J Cell Biochem. 2014;115:1877-1887.[4] Domazetovic V, Marcucci G, Iantomasi T, et al. Oxidative stress in bone remodeling: role of antioxidants. Clin Cases Miner Bone. 2017;14:209-216.[5] Hampton MB, Orrenius S. Redox regulation of apoptotic cell death. BioFactors (Oxford, England).1998;8(1-2):1-5.[6] Jin YQ, Li JL, Chen JD, et al. Dalbergioidin (DAL) protects MC3T3-E1 osteoblastic cells against H2O2-induced cell damage through activation of the PI3K/AKT/SMAD1 pathway. Naunyn Schmiedebergs Arch Pharmacol. 2017;390(7):711-720.[7] Lee D, Kook SH, Ji H, et al. N-acetyl cysteine inhibits H2O2-mediated reduction in the mineralization of MC3T3-E1 cells by down-regulating Nrf2/HO-1 pathway. BMB Reports. 2015;48:636-641.[8] 李娟,申广浩,程光,等.组织块法培养SD大鼠的成骨细胞及鉴定[J].中国组织工程研究,2012,16(46):8571-8574.[9] 李丽艳,任塘珂.成骨细胞培养在骨组织工程和运动领域的研究进展[J].中国组织工程研究,2012,16(11):2043-2046.[10] 李菊根,陈建庭,金大地.MTT法分析PDGF对鼠成骨细胞生长作用的影响[J].第一军医大学学报,1998,18(3):69-71.[11] 韩立赤,王雪红,董会,等.苯肾上腺素对大鼠成骨细胞放射损伤的保护作用[J].吉林大学学报,2015,41(6):1113-1117+11.[12] 郭宝磊,杨茂伟,梁单,等.过氧化氢通过线粒体途径诱导成骨细胞凋亡[J].中国老年学杂志,2013,33(22):5661-5663.[13] Wanet A, Arnould T, Najimi M, et al. Connecting Mitochondria, Metabolism, and Stem Cell Fate. Stem Cells Dev. 2015;24: 1957-1971.[14] Mountziaris PM, Spicer PP, Kasper FK,et al. Harnessing and modulating inflammation in strategies for bone regeneration. Tissue Eng Part B Rev. 2011;17(6):393-402.[15] Schett G. Effects of inflammatory and anti-inflammatory cytokines on the bone. Eur J Clin Invest.2011;41:1361-1366.[16] Lacey DC,Simmons PJ,Graves SE,et al. Proinflammatory cytokines inhibitosteogenic differentiation from stem cells: implications for bone repair during inflammation.Osteoarthritis Cartilage.2009;17:735-742.[17] Manolagas SC. From estrogen-centric to aging and oxidative stress: a revised perspective of the pathogenesis of osteoporosis. Endocr Rev. 2010;31:266-300.[18] Banfi G, Iorio EL,Corsi MM.Oxidative stress, free radicals and bone remodeling. Clin Chem Lab Med. 2008;46:1550-1555.[19] Mann V, Huber C, Kogianni G, et al. The antioxidant effect of estrogen and Selective Estrogen Receptor Modulators in the inhibition of osteocyte apoptosis in vitro. Bone.2007;40: 674-684.[20] Baek KH, Oh KW, Lee WY,et al. Association of oxidative stress with postmenopausal osteoporosis and the effects of hydrogen peroxide on osteoclast formation in human bone marrow cell cultures. Calcif Tissue Int. 2010;87:226-235.[21] Ichiseki T, Matsumoto T, Nishino M,et al. Oxidative stress and vascular permeability in steroid-induced osteonecrosis model. J Orthop Sci. 2004;9(5):509-515.[22] Komori T. Cell Death in Chondrocytes, Osteoblasts, and Osteocytes. Int J Mol Sci. 2016;17(12). pii: E2045.[23] Hu K, Olsen BR. Osteoblast-derived VEGF regulates osteoblast differentiation and bone formation during bone repair. J Clin Invest. 2016;126(2):509-526.[24] Lean JM, Jagger CJ, Kirstein B,et al. Hydrogen peroxide is essential for estrogen-deficiency bone loss and osteoclast formation. Endocrinology. 2005;146:728-735.[25] Huang RL, Yuan Y, Tu J, et al. Opposing TNF-α/IL-1β- and BMP-2-activated MAPK signaling pathways converge on Runx2 to regulate BMP-2-induced osteoblastic differentiation. Cell Death Dis. 2014;5:e1187.[26] Pawlowska E, Wysokiński D, Tokarz P, et al. Dexamethasone and 1,25-Dihydroxyvitamin D3 Reduce Oxidative Stress-Related DNA Damage in Differentiating Osteoblasts. Int J Mol Sci. 2014;15:16649-16664.[27] Almubarak S, Nethercott H, Freeberg M, et al. Tissue engineering strategies for promoting vascularized bone regeneration. Bone.2016;83:197-209.[28] Chen CT, Shih YR, Kuo TK,et al.Coordinated changes ofmitochondrial biogenesis and antioxidant enzymes during osteogenicdifferentiation of human mesenchymal stem cells, Stem Cells. 2008;(26):960-968.[29] Wang D,ChristensenK,Chawla G,et al. Franceschi,Isolation and characterization of MC3T3-E1 preosteoblast subclones withdistinct in vitro and in vivo differentiation/mineralization potential. J Bone Miner Res.1999;14(6):893-903.[30] Vasin MV,Ushakov IB,Semenova LA,et al.Radiation protective efficacy of alpha-adrenomimetics during local gamma irradiation of the skin. Radiats Biol Radioecol. 1999;39(2-3): 249-253.[31] Xiang B, Li YJ, Zhao XB, et al. Mechanism of the protective effect of phenylephrine pretreatment against irradiation-induced damage in the submandibular gland. Exp Ther Med. 2013;5:875-879.[32] Tanaka K, Hirai T, Kodama D, et al. alpha1B -Adrenoceptor signalling regulates bone formation through the up-regulation of CCAAT/enhancer-binding protein delta expression in osteoblasts. Br J Pharmacol. 2016;173:1058-1069.[33] Olarescu NC, Ueland T, Lekva T, et al. Adipocytes as a source of increased circulating levels of nicotinamide phosphoribosyltransferase/visfatin in active acromegaly.J Clin Endocrinol Metab. 2012;97(4):1355-1362.[34] He X, He J, Shi Y, et al. Nicotinamide phosphoribosyltransferase (Nampt) may serve as the marker for osteoblast differentiation of bone marrow-derived mesenchymal stem cells. Exp Cell Res. 2017;352:45-52.[35] Li Y, He J, He X, et al. Nampt expression increases during osteogenic differentiation of multi- and omnipotent progenitors.Biochem Biophys Res Commun. 2013;434(1): 117-123.[36] Marie-Charlotte Laiguillon XH, Carole Bougault, et al. Expression and function of visfatin (Nampt), an adipokine-enzyme involved in inflammatory pathways of osteoarthritis. Arthritis Res Ther. 2014;16:1-12.[37] Lin YC, Wu HC, Liao CC, et al. Secretion of one adipokine Nampt/Visfatin suppresses the inflammatory stress-induced NF-kappaB activity and affects Nampt-dependent cell viability in Huh-7 cells. Mediators Inflamm. 2015;2015:392471.[38] Romacho T, Villalobos LA, Cercas E, et al. Visfatin as a novel mediator released by inflamed human endothelial cells. PloS one. 2013;8:e78283.[39] Garten A, Petzold S, Korner A, et al. Nampt: linking NAD biology, metabolism and cancer. Trends Endocrinol Metab. 2009;20(3):130-138.[40] Imai S. Nicotinamide phosphoribosyltransferase (Nampt): A link between NAD biology, metabolism, and diseases. Curr Pharm Des.2009;15:20-28.[41] 吴梦然,张红胜.NAD+和Nampt与SIRT1的研究进展[J].医学综述, 2010;16(7):968-971.[42] Romanello M, Bicego M, Pirulli D, et al.ExtracellularNAD+: a novel autocrine/paracrine signal in osteoblast physiology. Biochem Biophys Res Commun. 2002;(299):424-431.[43] Ling M, Huang P, Islam S, et al. Epigenetic regulation of Runx2 transcription and osteoblast differentiation by nicotinamide phosphoribosyltransferase. Cell Biosci. 2017;7: 27.[44] Xie H, Tang SY, Luo XH, et al. Insulin?like efects of visfatin on human osteoblasts. Calcif Tissue Int. 2007;80:201-210. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [3] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| [4] | Liu Cong, Liu Su. Molecular mechanism of miR-17-5p regulation of hypoxia inducible factor-1α mediated adipocyte differentiation and angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1069-1074. |
| [5] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [6] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [7] | Ma Zetao, Zeng Hui, Wang Deli, Weng Jian, Feng Song. MicroRNA-138-5p regulates chondrocyte proliferation and autophagy [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 674-678. |
| [8] | Wang Yujiao, Liu Dan, Sun Song, Sun Yong. Biphasic calcium phosphate loaded with advanced platelet rich fibrin can promote the activity of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 504-509. |
| [9] | Zhou Jihui, Yao Meng, Wang Yansong, Li Xinzhi, Zhou You, Huang Wei, Chen Wenyao. Influence of novel nanoscaffolds on biological behaviors of neural stem cells and the related gene expression [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 532-536. |
| [10] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [11] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [12] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [13] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [14] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [15] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||