Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (8): 1178-1183.doi: 10.3969/j.issn.2095-4344.0804
Previous Articles Next Articles
Quantitative peptidomic analysis of the cartilage tissues of different ages
Zhou Zhi-wen, Ju Li, Lou Yue
- Department of Orthopedics, Children’s Hospital of Nanjing Medical University, Nanjing 210008, Jiangsu Province, China
-
Received:2017-11-08Online:2018-03-18Published:2018-03-18 -
Contact:Lou Yue, Professor, Department of Orthopedics, Children’s Hospital of Nanjing Medical University, Nanjing 210008, Jiangsu Province, China -
About author:Zhou Zhi-wen, Master, Physician, Department of Orthopedics Nanjing Children’s Hospital of Nanjing Medical University, Nanjing 210008, Jiangsu Province, China
CLC Number:
Cite this article
Zhou Zhi-wen, Ju Li, Lou Yue. Quantitative peptidomic analysis of the cartilage tissues of different ages[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(8): 1178-1183.
share this article
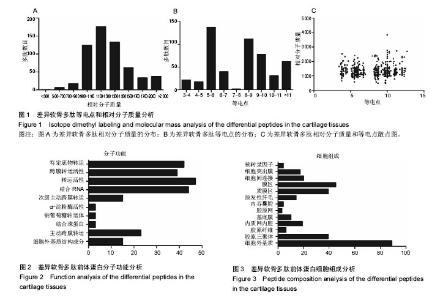
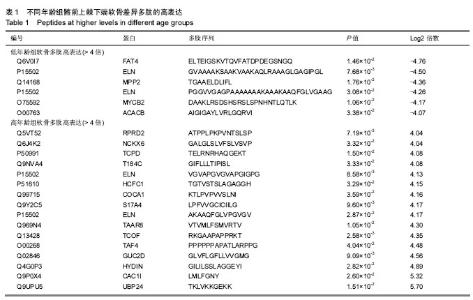
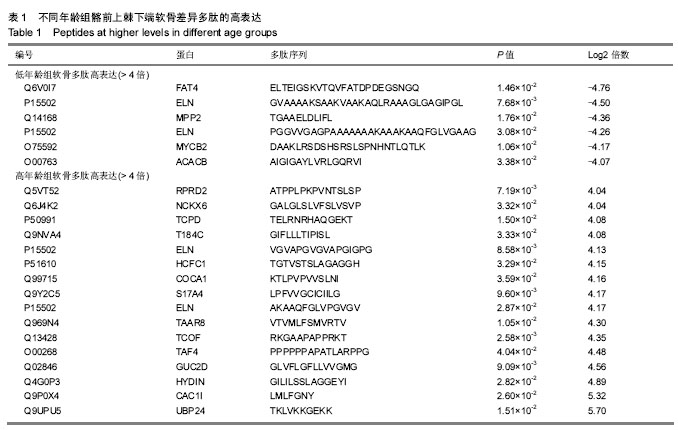
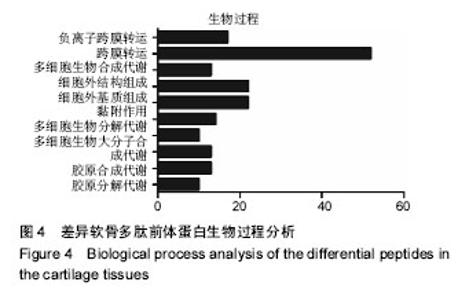
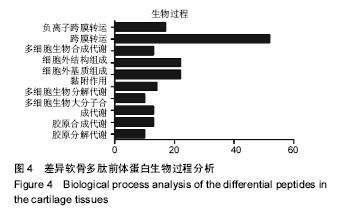
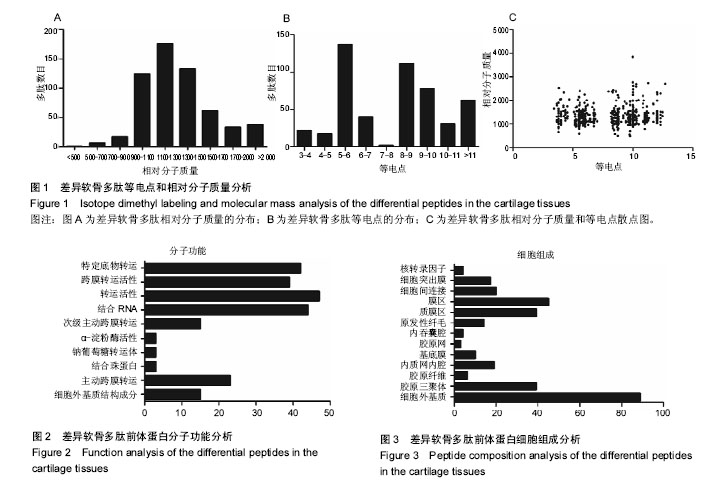
2.1 差异软骨多肽定量分析 两组软骨多肽通过液相色谱-串联质谱分析共有5 027个多肽被鉴定出来,来源于 1 869种大分子蛋白质。差异多肽有588个,来源于428种蛋白质。如表1所示,16个多肽在高年龄组中高表达(大于4倍),6个多肽在低年龄组中高表达(大于4倍)。所有22个高表达多肽具有统计学意义(P < 0.05)。 2.2 差异软骨多肽等电点和相对分子质量分析 首先分析了差异软骨多肽的相对分子质量(图1A)和等电点(图1B),从中可以看出一些分布的不同。并且通过相对分子质量和等电点之间的分布差异可以看出软骨多肽分布的主要区域(图1C)。根据先前的研究[19],由于仪器和数据处理的局限性,液相色谱-串联质谱鉴定的多肽相对分子质量小于3 000。很明显在酸性和碱性pH范围内所识别的多肽聚集在4个区域。 2.3 差异软骨多肽基因本体分析 对鉴定的软骨多肽进行基因本体分析,由P值和FDR共同决定是否有统计学意义。并对潜在的生理功能分类:分子功能(图2),细胞组成(图3),生物过程(图4)。"
| [1] Usami Y, Gunawardena AT, Iwamoto M, et al.Wnt signaling in cartilage development and diseases: lessons from animal studies. Lab Invest.2016;96(2):186-196.[2] Lotz M.Osteoarthritis year 2011 in review: biology. Osteoarthritis Cartilage.2012;20(3):192-196.[3] Clouet J,Vinatier C,Merceron C,et al.From osteoarthritis treatments to future regenerative therapies for cartilage. Drug Discovery Today.2009;14(19-20):913-925.[4] Qi H, Jin M, Duan Y,et al.FGFR3 induces degradation of BMP type I receptor to regulate skeletal development. Biochim Biophys Acta.2014;1843(7):1237-1247.[5] Yuan H, Huang L, Hu X, et al.FGFR3 gene mutation plus GRB10 gene duplication in a patient with achondroplasia plus growth delay with prenatal onset. Orphanet J Rare Dis.2016; 11(1):89.[6] Müller C, Khabut A, Dudhia J, et al.Quantitative proteomics at different depths in human articular cartilage reveals unique patterns of protein distribution. Matrix Biol.2014;40:34-45.[7] Önnerfjord P, Khabut A, Reinholt FP, et al.Quantitative Proteomic Analysis of Eight Cartilaginous Tissues Reveals Characteristic Differences as well as Similarities between Subgroups. J Biol Chem. 2012;287(23):18913-18924.[8] Brodsky B, Persikov AV.Molecular structure of the collagen triple helix. Adv Protein Chem.2005;70:301-339.[9] Bäcklund J, Treschow A, Bockermann R, et al.Glycosylation of type II collagen is of major importance for T cell tolerance and pathology in collagen-induced arthritis. Eur J Immunol. 2002;32(12):3776-3784.[10] Ota KG, Kuratani S.Expression pattern of two collagen type 2 alpha1 genes in the Japanese inshore hagfish (Eptatretus burgeri) with special reference to the evolution of cartilaginous tissue. J Exp Zool B Mol Dev Evol.2010;314(2):157-165.[11] Yeung Tsang K, Wa Tsang S, Chan D, et al.The chondrocytic journey in endochondral bone growth and skeletal dysplasia. Birth Defects Res C Embryo Today. 2014;102(1):52-73.[12] Nishimura R, Hata K, Ono K, et al.Regulation of endochondral ossification by transcription factors. Front Biosci (Landmark Ed).2012;17: 2657-2666.[13] Ballock RT, O'Keefe RJ.Physiology and pathophysiology of the growth plate. Birth Defects Res C Embryo Today.2003; 69(2):123-143.[14] Long F, Ornitz DM.Development of the Endochondral Skeleton. Cold Spring Harb Perspect Biol.2013;5(1):a008334.[15] Hayakawa E, Menschaert G, De Bock PJ, et al.Improving the Identification Rate of Endogenous Peptides Using Electron Transfer Dissociation and Collision-Induced Dissociation. J Proteome Res.2013;12(12):5410-5421.[16] Geho DH, Liotta LA, Petricoin EF, et al.The amplified peptidome: the new treasure chest of candidate biomarkers. Curr Opin Chem Biol.2006;10(1):50-55.[17] Soloviev M, Finch P. Bridging the gap between proteome and metabolome. Proteomics.2006;6(3):744-747.[18] Adermann K, John H, Ständker L, et al.Exploiting natural peptide diversity: novel research tools and drug leads. Curr Opin Biotechnol.2004;15(6):599-606.[19] Shores KS, Knapp DR. Assessment Approach for Evaluating High Abundance Protein Depletion Methods for Cerebrospinal Fluid (CSF) Proteomic Analysis. J Proteome Res. 2007;6(9): 3739-3751.[20] Srinivasan M, Patel MS. Metabolic programming in the immediate postnatal period. Trends Endocrinol Metab.2008; 19(4):146-152.[21] Wallin E, von Heijne G. Genome-wide analysis of integral membrane proteins from eubacterial, archaean, and eukaryotic organisms.Protein Sci. Protein Sci. 1998;7(4): 1029-1038.[22] Mao Y, Kuta A, Crespo-Enriquez I, et al. Dchs1-Fat4 regulation of polarized cell behaviours during skeletal morphogenesis. Nat Commun.2016;7:11469.[23] Kuta A, Mao Y, Martin T, et al. Fat4-Dchs1 signalling controls cell proliferation in developing vertebrae. Development. 2016;143(13):2367-2375.[24] Juarranz Y, Gutierrez-Canas I, Santiago B, et al.Differential expression of vasoactive intestinal peptide and its functional receptors in human osteoarthritic and rheumatoid synovial fibroblasts. Arthritis Rheum.2008;58(4):1086-1095.[25] Jiang W, Gao SG, Chen XG, et al. Expression of synovial fluid and articular cartilage VIP in human osteoarthritic knee: a new indicator of disease severity?. Clin Biochem.2012; 45(18): 1607-1612.[26] von Rechenberg B, Mcilwraith CW, Akens MK, et al. Spontaneous production of nitric oxide (NO), prostaglandin (PGE2) and neutral metalloproteinases (NMPs) in media of explant cultures of equine synovial membrane and articular cartilage from normal and osteoarthritic joints. Equine Vet J.2000;32(2):140-150.[27] Hernanz A, Medina S, de Miguel E, et al. Effect of calcitonin gene-related peptide, neuropeptide Y, substance P, and vasoactive intestinal peptide on interleukin-1beta, interleukin-6 and tumor necrosis factor-alpha production by peripheral whole blood cells from rheumatoid arthritis and osteoarthritis patients. Regul Pept.2003;115(1):19-24.[28] Matsukawa N, Grzesik W J, Takahashi N, et al. The natriuretic peptide clearance receptor locally modulates the physiological effects of the natriuretic peptide system. Proc Natl Acad Sci U S A.1999;96(13):7403-7408.[29] Yasoda A, Komatsu Y, Chusho H, et al. Overexpression of CNP in chondrocytes rescues achondroplasia through a MAPK-dependent pathway. Nat Med.2004;10(1):80-86.[30] Krejci P, Masri B, Fontaine V, et al. Interaction of fibroblast growth factor and C-natriuretic peptide signaling in regulation of chondrocyte proliferation and extracellular matrix homeostasis. J Cell Sci.2005;118(Pt 21):5089-5100.[31] Amizuka N, Henderson JE, Hoshi K, et al. Programmed cell death of chondrocytes and aberrant chondrogenesis in mice homozygous for parathyroid hormone-related peptide gene deletion. Endocrinology.1996;137(11):5055-5067.[32] Fan Y, Jianying F, Chenyan L, et al. Influence on Indian hedgehog-parathyroid hormone-like related protein pathway induced by altered masticatory loading in the condylar cartilage of growing rabbits.Hua Xi Kou Qiang Yi Xue Za Zhi. 2017;35(2):127-132.[33] Kong L, Zhao Y P, Tian Q Y, et al. Extracellular matrix protein 1, a direct targeting molecule of parathyroid hormone-related peptide, negatively regulates chondrogenesis and endochondral ossification via associating with progranulin growth factor. The FASEB Journal.2016;30(8):2741-2754.[34] Zhang H, Wang H, Zeng C, et al. mTORC1 activation downregulates FGFR3 and PTH/PTHrP receptor in articular chondrocytes to initiate osteoarthritis.Osteoarthritis Cartilage. 2017;25(6):952-963.[35] Fischer J, Ortel M, Hagmann S, et al. Role of PTHrP(1-34) Pulse Frequency Versus Pulse Duration to Enhance Mesenchymal Stromal Cell Chondrogenesis.J Cell Physiol. 2016;231(12):2673-2681.[36] Moore ER, Jacobs CR. The primary cilium as a signaling nexus for growth plate function and subsequent skeletal development. J Orthop Res. 2017 Sep 13. doi: 10.1002/jor.23732. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Wu Xun, Meng Juanhong, Zhang Jianyun, Wang Liang. Concentrated growth factors in the repair of a full-thickness condylar cartilage defect in a rabbit [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1166-1171. |
| [3] | Li Jiacheng, Liang Xuezhen, Liu Jinbao, Xu Bo, Li Gang. Differential mRNA expression profile and competitive endogenous RNA regulatory network in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1212-1217. |
| [4] | Geng Qiudong, Ge Haiya, Wang Heming, Li Nan. Role and mechanism of Guilu Erxianjiao in treatment of osteoarthritis based on network pharmacology [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1229-1236. |
| [5] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [6] | He Xiangzhong, Chen Haiyun, Liu Jun, Lü Yang, Pan Jianke, Yang Wenbin, He Jingwen, Huang Junhan. Platelet-rich plasma combined with microfracture versus microfracture in the treatment of knee cartilage lesions: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(6): 964-969. |
| [7] | Liu Xin, Yan Feihua, Hong Kunhao. Delaying cartilage degeneration by regulating the expression of aquaporins in rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 668-673. |
| [8] | Deng Zhenhan, Huang Yong, Xiao Lulu, Chen Yulin, Zhu Weimin, Lu Wei, Wang Daping. Role and application of bone morphogenetic proteins in articular cartilage regeneration [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 798-806. |
| [9] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [10] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [11] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [12] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [13] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [14] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [15] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||