Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (14): 2239-2244.doi: 10.3969/j.issn.2095-4344.0788
Previous Articles Next Articles
Stem cells combined with three-dimensional scaffolds for the treatment of osteoporotic bone defects
Yuan Wei-jian, Wang Chen-yu, Li Chen, Yang Fan, Wang Zhong-han, Li Zu-hao, Qin Yan-guo, Liu He, Wang Jin-cheng
- Department of Orthopedics, Second Hospital of Jilin University, Chuangchun 130041, Jilin Province, China
-
Received:2018-01-16Online:2018-05-18Published:2018-05-18 -
Contact:Wang Jin-cheng, Chief physician, Professor, Doctoral supervisor, Department of Orthopedics, Second Hospital of Jilin University, Chuangchun 130041, Jilin Province, China -
About author:Yuan Wei-jian, Master candidate, Department of Orthopedics, Second Hospital of Jilin University, Chuangchun 130041, Jilin Province, China -
Supported by:the National Natural Science Foundation of China, No. 81671804; the Natural Science Foundation of Jilin Province, No. 20160101109JC, 20150414006GH, 20150312028ZG, 20130206060GX; the Excellent Doctoral Candidate Incubation Program in Norman Bethune Health Science Center of Jilin University, No. YB201501; the Postgraduate Innovation Research Program of Jilin University, No. 2016041, 2016108
CLC Number:
Cite this article
Yuan Wei-jian, Wang Chen-yu, Li Chen, Yang Fan, Wang Zhong-han, Li Zu-hao, Qin Yan-guo, Liu He, Wang Jin-cheng. Stem cells combined with three-dimensional scaffolds for the treatment of osteoporotic bone defects[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(14): 2239-2244.
share this article
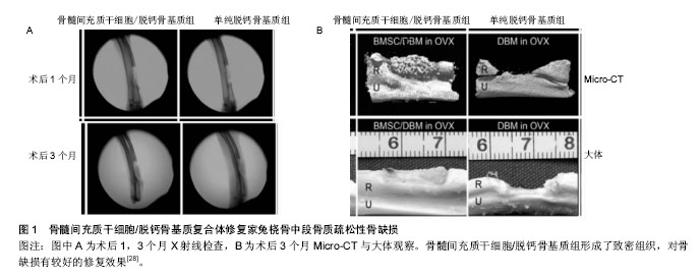
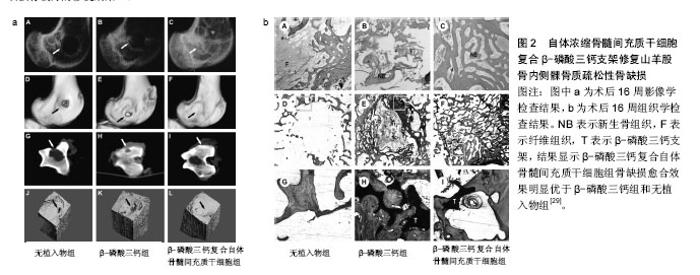
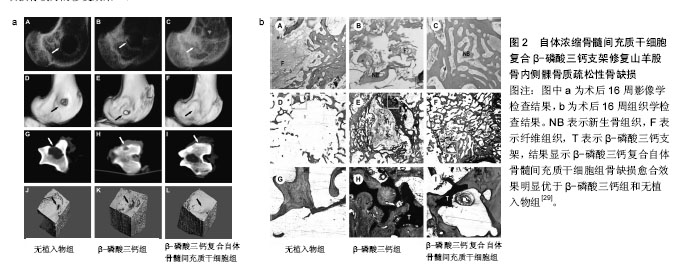
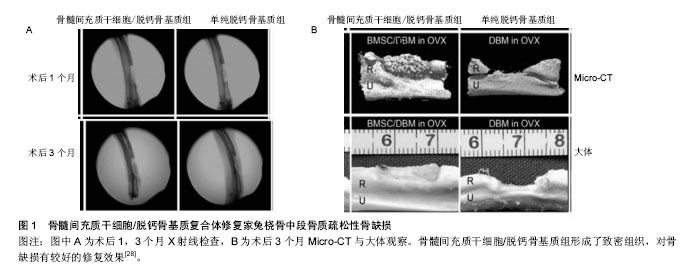
2.1 干细胞复合三维支架的特点 细胞疗法在几十年的疾病治疗过程中已引起了广泛临床关注。由于干细胞具有自我更新和多向分化潜能,在不同的微环境可诱导不同的分化方向,用于修复或再生受损的组织,被认为是作为细胞替代疗法治疗骨科疾病的理想细胞来源。用于骨质疏松治疗的候选干细胞类型包括胚胎干细胞、诱导多能干细胞和间充质干细胞等,但胚胎干细胞和诱导多能干细胞的使用由于伦理问题和基于病毒衍生方法而受到限制[10]。间充质干细胞的使用似乎克服了这样的限制,此外,间充质干细胞可促进成骨细胞的活性,改变骨代谢,使体内的骨形成和骨吸收达到一个新的平衡,因此可从根本上治疗骨质疏松。此外,有研究显示间充质干细胞复合三维支架还可促进骨不连、骨缺损的愈 合[11-14]。 合适的支架能够为修复骨缺损相关的细胞附着和生长提供了一个有利微环境,并提供相应的机械强度,起到对骨缺损的支撑作用,促进愈合的同时不带来额外的不良反应。所以用于植入骨缺损部位的三维支架一般应具有以下特点[15]:①生物相容性:合成支架的材料对复合在支架内的干细胞和相邻的体内组织无免疫原性,不引起排斥,生物相容性好[16];②生物降解性:材料在完成支架作用后能够自动降解,降解时间能够智能化地响应缺损组织的生长速度,使得新生骨组织的形成与支架降解速率达到一个平衡状态[17];③细胞界面:支架应当具有良好的表面活性,促进相关细胞的黏附生长和迁移、增殖、分化,激活细胞特异性基因的表达,维持细胞正常表型,这对于复合其中的干细胞具有较大的促进意义[18];④三维多孔结构:具有三维立体结构的支架,合适的孔隙率和孔隙直径,利于促进细胞的生长和黏附,诱导血管和神经的长入,加速营养成分和代谢产物的交换,达到促进骨缺损修复的作用[19-20];⑤机械强度:支架需要具有一定的机械强度,在植入骨缺损部位后提供一定时间的支持,直至新生骨组织形成后可替代支架的支撑作用后再完成降解[21]。 在现阶段的研究中,可降解水凝胶构建的三维支架研究较为广泛。致孔剂浸出创建多孔支架技术被广泛采用,该技术也被称为溶液浇铸/颗粒沥滤,即将水溶性盐颗粒与可降解聚合物溶液进行混合,再把混合物浇铸到具有理想形状的模具中,然后除去溶剂,将盐颗粒被滤出后即可得到多孔结构。这种制备方法操作简易,并且可通过调节盐颗粒/聚合物比率和颗粒尺寸来获得不同孔径和孔隙率的三维支架[22]。此外,近年来3D打印技术引起了研究者的广泛兴趣。 现阶段,三维支架已被广泛应用于对骨缺损的修 复[23],尤其是以干细胞复合的三维支架,植入后对骨缺损的修复表现出巨大的优势[24-25]。总之,对于骨质疏松性骨缺损,干细胞复合三维支架复合体中的支架,除了提供一定的机械强度外,还为修复相关细胞、因子提供一个有利的再生微环境,复合体更重要的作用的调节骨代谢,促进骨缺损的愈合,起到更好的修复作用[14,26]。 2.2 干细胞复合三维支架的应用 将干细胞-三维支架复合体植入正常骨缺损模型中,已被广泛证明可促进骨缺损的愈合,由于其对骨形成平衡的调节作用,促进新骨形成的机制在理论上也可更好地促进骨质疏松性骨缺损的修复,因此,干细胞复合三维支架在植入骨质疏松性骨缺损部位是一种有潜力的治疗方法,研究者对此进行了一系列实验。目前用于骨质疏松症的细胞疗法中,间充质干细胞是使用最广泛的一种干细胞[27]。 由于干细胞具有自我更新和多向分化潜能,可用于修复或再生受损组织,被认为是作为细胞替代疗法治疗骨科疾病的理想细胞来源。骨髓间充质干细胞作为细胞替代治疗的种子细胞已被广泛研究。Wang等[28]将骨髓间充质干细胞在体外成骨诱导之后复合脱钙骨基质支架,植入卵巢切除骨质疏松家兔桡骨中段1.5 cm长骨缺损中,移植后第1,3个月进行 X射线平片、Micro-CT和组织形态学检查,结果显示骨髓间充质干细胞/脱钙骨基质组形成了致密组织,对骨缺损有较好的修复效果,免疫组织化学检测表明成骨活性增强,与单纯脱钙骨基质相比显著促进了缺损处的骨再生;与非骨质疏松对照组相比,骨质疏松性骨缺损的愈合明显延迟(图1)。Cao等[29]研究证明自体浓缩骨髓间充质干细胞与具有多孔结构的β-磷酸三钙支架相结合,可成功修复骨质疏松性山羊股骨内侧髁临界大小的骨缺损,并建议使用自体富集骨髓间充质干细胞作为治疗骨质疏松性骨缺损的一种快速、安全的策略。术后16周骨缺损影像学检查检查显示,无植入物组可见骨缺损区不透光区极小,几乎没有新骨形成;β-磷酸三钙组虽然在骨缺损边缘有一些不透光区域,但新骨形成的量与无植入物组相比无明显差异;β-磷酸三钙复合自体浓缩骨髓间充质干细胞组缺损区有明显的新生骨形成,与周围组织结合良好,未见明显间隙(图2)。将煅烧骨包裹同种异体骨髓间充质干细胞涂层作为骨组织工程修复的三维支架材料,植入骨质疏松性大鼠颅骨处直径为8 mm的缺损,在成骨细胞分化和矿化形成方面的潜力明显超过煅烧骨-骨髓间充质干细胞简单的组合,同种异体骨髓间充质干细胞涂层在支架材料上存活并参与新骨形成,移植后缺损处新形成的骨具有与周围骨组织相同的厚度,为骨质疏松大面积骨缺损治疗提供了新策略[30]。 与骨髓间充质干细胞相比,脂肪间充质干细胞更容易分离,更丰富,产量更高[31]。自体脂肪间充质干细胞,由于其自身成骨分化,以及通过对骨形态发生蛋白2和骨形态发生蛋白受体IB基因信号通路的激活,促进成骨作用和抑制脂肪生成,促进骨质疏松家兔的骨再生[32]。Cho 等[33]将脂肪间充质干细胞注射到卵巢切除小鼠尾静脉,与仅接受PBS注射的对照组小鼠进行比较,移植4,8周后骨密度明显增加。Pei等[34]将来源于健康大鼠和骨质疏松大鼠的脂肪间充质干细胞复合到聚乳酸-羟基乙酸共聚物支架,并植入骨质疏松性大鼠颅骨缺损处,相对于单纯聚乳酸-羟基乙酸共聚物表现出较高的骨体积密度,尤其是健康大鼠来源的脂肪间充质干细胞效果更显著。"

此外,脐血来源干细胞最近成为一种治疗骨质疏松症的候选干细胞。来自脐血的干细胞拥有独特而宝贵的内在特征:第一,可较容易地通过非侵入性方法获得,而不对供者产生风险;第二,具有低免疫原性潜能;第三,它们含有一个具有较强成骨分化能力的细胞群体[35]。这对干细胞的选择提供了的新的思路。Taiani等[36]将小鼠胚胎干细胞来源成骨细胞复合在一个三维Ⅰ型胶原凝胶支架中,植入骨质疏松性小鼠胫骨骨折缺损部位,移植4周后骨密度、骨小梁体积比和骨小梁数目显著增加,表明实验组显示了一个更好的愈合结果。 一些可由干细胞分泌的生长因子在成骨分化过程扮演着重要角色,如骨形态发生蛋白、转化生长因子β、成纤维生长因子、血管生长因子、类胰岛素生长因子等。因此,三维支架复合的干细胞基质中存在促进成骨的生长因子,有利于促进骨质疏松性骨缺损的愈合[37]。间充质干细胞分泌的血管生长因子a是一种血管生成因子,也参与骨的形成和修复。血管生长因子a结合骨形态发生蛋白9可明显增加骨量和激活成骨活性,这为节段性骨缺损或者骨质疏松性骨折的治疗提供了一种新的潜在策略[38]。一项利用骨形态发生蛋白治疗卵巢切除大鼠骨质疏松性骨折的研究显示,雌激素缺乏的环境可能导致骨形态发生蛋白7敏感性增加,与对照组相比,骨形态发生蛋白治疗组明显增强骨的矿化,达到一个更好的愈合[39]。骨形态发生蛋白通过增强受损的分子和细胞机制,对改善骨折的愈合具有潜在的影响[40]。唐尤超等[41-42]利用人源性骨形态发生蛋白2基因修饰的骨髓间充质干细胞复合珊瑚羟基磷灰石支架修复骨质疏松性下颌骨缺损,结果表明复合支架植入4周后可在支架边缘发现新生骨质,8周后可见相互连接的板状成熟骨基质,与对照组相比,骨形态发生蛋白2基因修饰的骨髓间充质干细胞复合支架组显著增加骨缺损部位的新生骨量。此外,将异基因骨形态发生蛋白6修饰的骨髓间充质干细胞悬浮在纤维蛋白凝胶后,植入猪椎体压缩性骨折缺损中,移植后6周行CT检查即可见缺损处的骨再生,6个月后取出椎体进行体外检测,与单纯纤维蛋白凝胶组相比,治疗组明显促进骨再生。这为骨质疏松最常见的椎体压缩性骨折治疗提供了新的技术,通过局部注射微创方法获得了满意的治疗效果[43]。 骨保护素作为一种可抑制破骨细胞分化的治疗剂而被熟知,现已被广泛应用于慢性骨吸收性疾病的治 疗[44]。另一方面,骨髓间充质干细胞在骨形成中发挥着重要作用。为了抑制过度骨吸收和促进骨形成,Liu等[45]开发了一种新的治疗策略,将骨保护素基因修饰的骨髓间充质干细胞接种在羟基磷灰石支架上,以促进卵巢切除骨质疏松大鼠临界尺寸下颌骨缺损的骨再生,与对照组相比,经修饰的骨髓间充质干细胞骨保护素基因表达水平较高,且能维持骨保护素蛋白高表达状态2周以上;体外骨吸收实验表明骨保护素能抑制破骨细胞分化,抑制破骨细胞介导的骨吸收;影像学及组织学检查结果显示,羟基磷灰石支架复合骨保护素修饰的骨髓间充质干细胞,可明显促进缺损处的骨形成,抑制了破骨细胞活性。这一实验结果证明基因修饰的干细胞复合三维支架对于骨质疏松相关骨缺损的重建具有令人关注的潜力。 虽然干细胞-三维支架复合体植入骨质疏松性骨缺损模型的研究还不是十分广泛,但已引起了研究者极大的兴趣,现有的研究证明这种方法可明显促进骨缺损的愈合,减少并发症(表1)。"

| [1] Teitelbaum SL.Stem Cells and Osteoporosis Therapy.Cell Stem Cell.2010;7(5):553-554.[2] Compston J,Cooper A,Cooper C,et al.UK clinical guideline for the prevention and treatment of osteoporosis. Arch Osteoporos. 2017;12(1):43.[3] Boyce BF,Rosenberg E,de Papp AE,et al.The osteoclast, bone remodelling and treatment of metabolic bone disease. Eur J Clin Invest.2012;42(12):1332-1341.[4] Hemmeler C,Morell S,Amsler F,et al.Screening for osteoporosis following non-vertebral fractures in patients aged 50 and older independently of gender or level of trauma energy-a Swiss trauma center approach.Arch Osteoporos. 2017;12(1):38. [5] Mirsaidi A,Genelin K,Vetsch JR,et al.Therapeutic potential of adipose-derived stromal cells in age-related osteoporosis. Biomaterials.2014;35(26):7326-7335.[6] Cao Q,Zhang JY,Liu HT,et al.The mechanism of anti-osteoporosis effects of 3-hydroxybutyrate and derivatives under simulated microgravity.Biomaterials. 2014;35(28): 8273-8283.[7] Cella L,Oppici A,Arbasi M,et al.Autologous bone marrow stem cell intralesional transplantation repairing bisphosphonate related osteonecrosis of the jaw.Head Face Med.2011;7:16.[8] Abrahamsen B,Eiken P,Eastell R Subtrochanteric and diaphyseal femur fractures in patients treated with alendronate: a register-based national cohort study.J Bone Miner Res.2009;24(6):1095-1102.[9] Wei DL,Jung JS,Yang HL,et al.Nanotechnology Treatment Options for Osteoporosis and Its Corresponding Consequences.Curr Osteoporos Rep.2016;14(5):239-247.[10] Takahashi K,Tanabe K,Ohnuki M,et al.Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell.2007;131(5):861-872.[11] Maiti SK,Ninu AR,Sangeetha P,et al.Mesenchymal stem cells-seeded bio-ceramic construct for bone regeneration in large critical-size bone defect in rabbit.J Stem Cells Regen Med.2016;12(2):87-99.[12] Chen Y,Xu J,Huang Z,et al.An Innovative Approach for Enhancing Bone Defect Healing Using PLGA Scaffolds Seeded with Extracorporeal-shock-wave-treated Bone Marrow Mesenchymal Stem Cells (BMSCs). Sci Rep. 2017; 7:44130.[13] Hsiao HY,Yang SR,Brey EM,et al.Hydrogel Delivery of Mesenchymal Stem Cell-Expressing Bone Morphogenetic Protein-2 Enhances Bone Defect Repair.Plast Reconstr Surg Glob Open.2016;4(8):e838. [14] Mendes Junior D,Domingues JA,Hausen MA,et al.Study of mesenchymal stem cells cultured on a poly(lactic-co-glycolic acid) scaffold containing simvastatin for bone healing.J Appl Biomater Funct Mater. 2017;15(2):e133-e141.[15] 刘凯,文刚,刘日富,等.骨髓间充质干细胞治疗局部骨质疏松[J].中国骨质疏松杂志,2013,19(11):1203-1206.[16] Staff PO.Correction: Decellularized Wharton's Jelly from human umbilical cord as a novel 3D scaffolding material for tissue engineering applications.PloS One. 2017;12(3):e0173827.[17] Flynn L,Prestwich GD,Semple JL,et al.Adipose tissue engineering in vivo with adipose-derived stem cells on naturally derived scaffolds.J Biomed Mater Res A. 2009;89(4): 929-941.[18] Sachar A,Strom TA,Serrano MJ,et al.Osteoblasts responses to three-dimensional nanofibrous gelatin scaffolds.J Biomed Mater Res A.2012;100(11):3029-3041.[19] Hadavi M,Hasannia S,Faghihi S,et al.Novel calcified gum Arabic porous nano-composite scaffold for bone tissue regeneration.Biochem Biophys Res Commun. 2017;488(4): 671-678. [20] He JX,Tan WL,Han QM,et al.Fabrication of silk fibroin/cellulose whiskers-chitosan composite porous scaffolds by layer-by-layer assembly for application in bone tissue engineering.J Mater Sci. 2016;51(9):4399-4410.[21] Wang W,Deng D,Wang B,et al.Comparison of autologous, allogeneic and cell-free scaffold approaches for engineered tendon repair in a rabbit model-A pilot study.Tissue engineering Part A. 2017;23(15-16):750-761. [22] 杨琳,管晓燕,陈黎明,等.智能水凝胶在骨类硬组织再生和修复中的应用[J].中国组织工程研究,2016,20(3):430-434.[23] Maksimkin AV,Senatov FS,Anisimova NY,et al.Multilayer porous UHMWPE scaffolds for bone defects replacement. Mater Sci Eng C Mater Biol Appl. 2017;73:366-372. [24] Lo SC,Li KC,Chang YH,et al.Enhanced critical-size calvarial bone healing by ASCs engineered with Cre/loxP-based hybrid baculovirus.Biomaterials.2017;124:1-11.[25] Nau C,Henrich D,Seebach C,et al.Tissue engineered vascularized periosteal flap enriched with MSC/EPCs for the treatment of large bone defects in rats.Int J Mol Med. 2017; 39(4):907-917.[26] Sangkert S,Kamonmattayakul S,Chai WL,et al.Modified porous scaffolds of silk fibroin with mimicked microenvironment based on decellularized pulp/fibronectin for designed performance biomaterials in maxillofacial bone defect.J Biomed Mater Res A.2016;105(6):1624-1636. [27] Antebi B,Pelled G,Gazit D.Stem cell therapy for osteoporosis. Curr Osteoporos Rep. 2014;12(1):41-47.[28] Wang ZX,Chen C,Zhou Q,et al.The Treatment Efficacy of Bone Tissue Engineering Strategy for Repairing Segmental Bone Defects Under Osteoporotic Conditions.Tissue Eng Part A. 2015;21(17-18):2346-2355.[29] Cao L,Liu G,Gan Y,et al.The use of autologous enriched bone marrow MSCs to enhance osteoporotic bone defect repair in long-term estrogen deficient goats.Biomaterials. 2012;33(20): 5076-5084.[30] Liu Y,Ming L,Luo H,et al.Integration of a calcined bovine bone and BMSC-sheet 3D scaffold and the promotion of bone regeneration in large defects.Biomaterials. 2013;34(38): 9998-10006.[31] Liu HY,Chiou JF,Wu AT,et al.The effect of diminished osteogenic signals on reduced osteoporosis recovery in aged mice and the potential therapeutic use of adipose-derived stem cells.Biomaterials. 2012;33(26):6105-6112.[32] Ye X,Zhang P,Xue S,et al.Adipose-derived stem cells alleviate osteoporosis by enhancing osteogenesis and inhibiting adipogenesis in a rabbit model.Cytotherapy. 2014;16(12): 1643-1655.[33] Cho SW,Sun HJ,Yang JY,et al.Human adipose tissue-derived stromal cell therapy prevents bone loss in ovariectomized nude mouse.Tissue Eng Part A.2012;18(9-10):1067-1078.[34] Pei M,Li J,McConda DB,et al.A comparison of tissue engineering based repair of calvarial defects using adipose stem cells from normal and osteoporotic rats.Bone. 2015; 78:1-10.[35] Ilic D,Miere C,Lazic E.Umbilical cord blood stem cells: clinical trials in non-hematological disorders.Brit Med Bull. 2012; 102(1):43-57.[36] Taiani JT,Buie HR,Campbell GM,et al.Embryonic stem cell therapy improves bone quality in a model of impaired fracture healing in the mouse; tracked temporally using in vivo micro-CT. Bone. 2014;64:263-272.[37] Saeed H,Ahsan M,Saleem Z,et al. Mesenchymal stem cells (MSCs) as skeletal therapeutics - an update. J Biomed Sci. 2016;23:41.[38] Pi CJ,Liang KL,Ke ZY,et al.Adenovirus-mediated expression of vascular endothelial growth factor-a potentiates bone morphogenetic protein9-induced osteogenic differentiation and bone formation.Biol Chem.2016;397(8):765-775.[39] Mathavan N,Tagil M,Isaksson H.Do osteoporotic fractures constitute a greater recalcitrant challenge for skeletal regeneration? Investigating the efficacy of BMP-7 and zoledronate treatment of diaphyseal fractures in an open fracture osteoporotic rat model.Osteoporos Int. 2017;28(2): 697-707. [40] Van Lieshout EM,Alt V.Bone graft substitutes and bone morphogenetic proteins for osteoporotic fractures: what is the evidence? Injury.2016;47 Suppl 1:S43-46.[41] 唐尤超,王远勤,汤炜.基因修饰骨髓间充质干细胞复合珊瑚羟基磷灰石支架材料修复骨质疏松性下颌骨缺损[J].中国组织工程研究与临床康复,2008,12(14):2601-2605.[42] Tang YC,Tang W,Lin YF,et al.Combination of bone tissue engineering and BMP-2 gene transfection promotes bone healing in osteoporotic rats.Cell Biol Int. 2008;32(9): 1150-1157.[43] Pelled G,Sheyn D,Tawackoli W,et al.BMP6-Engineered MSCs Induce Vertebral Bone Repair in a Pig Model: A Pilot Study. Stem Cells Int.2016;2016:6530624.[44] Huo J,Sun X.Effect of Astragalus polysaccharides on ovariectomy-induced osteoporosis in mice. Genet Mol Res. 2016;15(4).doi:10.4238/gmr15049169.[45] Liu X,Bao C,Xu HH,et al.Osteoprotegerin gene-modified BMSCs with hydroxyapatite scaffold for treating critical-sized mandibular defects in ovariectomized osteoporotic rats.Acta Biomater. 2016;42:378-388. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||