Chinese Journal of Tissue Engineering Research ›› 2019, Vol. 23 ›› Issue (2): 165-171.doi: 10.3969/j.issn.2095-4344.0692
Titanium-copper alloys with nanotubular coatings increase antibacterial abilities and osteoblast functions
Zhang Lei1, Zhang Hangzhou2, Allieu Kamara3
- 1First Department of Orthopedics, Third Affiliated Hospital of Jinzhou Medical University, Jinzhou 121001, Liaoning Province, China; 2Department of Sports Medicine/Joint Surgery, First Affiliated Hospital of China Medical University, Shenyang 110001, Liaoning Province, China; 3China Medical University, Shenyang 110001, Liaoning Province, China
-
Online:2019-01-18Published:2019-01-18 -
Contact:Allieu Kamara, China Medical University, Shenyang 110001, Liaoning Province, China -
About author:Zhang Lei, Attending physician, First Department of Orthopedics, Third Affiliated Hospital of Jinzhou Medical University, Jinzhou 121001, Liaoning Province, China -
Supported by:the National Natural Science Foundation of China, No. 81671811; the National Natural Science Foundation of China (Youth Foundation), No. 81501857 (to ZHZ); Liaoning Provincial Reform Project for Key Clinical Diagnosis and Treatment, No. LNCCC-A03-2014; the Natural Science Foundation of Liaoning Province, No. LK201642; Special Science and Technology Research Project for Population and Health in Shenyang, No. F15-139-9-23
CLC Number:
Cite this article
Zhang Lei, Zhang Hangzhou, Allieu Kamara. Titanium-copper alloys with nanotubular coatings increase antibacterial abilities and osteoblast functions[J]. Chinese Journal of Tissue Engineering Research, 2019, 23(2): 165-171.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
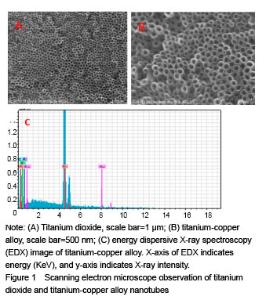
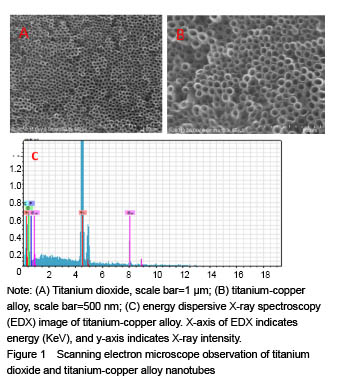
Scanning electron microscope observation of titanium dioxide and titanium-copper alloy nanotubes Figure 1A and B shows regular arrangements of produced titanium dioxide nanotubes and titanium-copper alloy nanotubes of about 80 nm in diameter under the scanning electron microscope. The nanotubes were perpendicular to the titanium base. Figure 1C shows the EDX analysis of the chemical composition of titanium-copper nanotube surface substances under the scanning electron microscope."

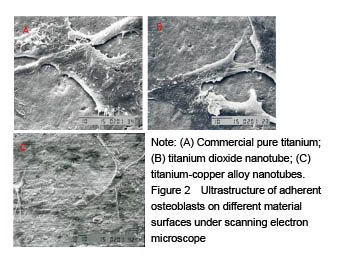
Adhesion and proliferation of mouse osteoblasts on different material surfaces Figure 2A-C shows the proliferation condition of mouse osteoblasts on commercial titanium, titanium dioxide nanotubes and titanium-copper alloy nanotubes after 24 hours of culture. The images show good osteoblasts morphology on the titanium nanotubes and titanium copper alloy nanotubes (Figure 2B and C). A large number of cellular pseudopods were attached to the compounds, indicating that titanium-copper alloy nanotubes have good biocompatibility. Cell count also showed that titanium-copper alloy nanotubes and titanium dioxide nanotubes had significantly better osteoblasts proliferation capacity than the ordinary titanium metal."
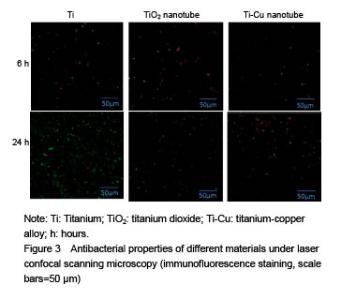
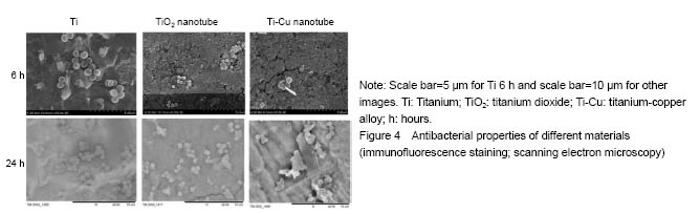
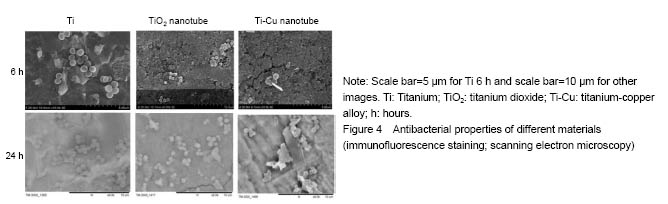
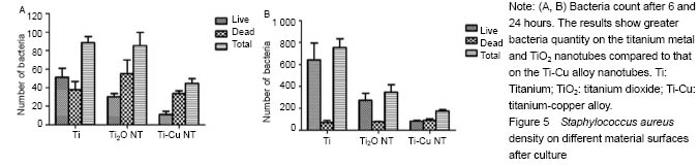
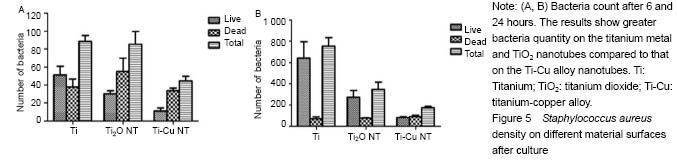
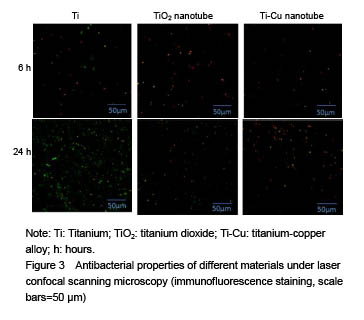
Antibacterial property testing Staphylococcus aureus was co-cultured with titanium-copper alloy nanotubes, titanium dioxide nanotubes and titanium for 6 and 24 hours respectively, and confocal laser scanning microscope (Figure 3) and scanning electron microscope (Figure 4) were then used for bacterial adhesion. There was greater bacteria quantity on the titanium metal and titanium dioxide nanotubes compared to that on the titanium-copper alloy nanotubes (Figure 5). This shows that titanium-copper alloy nanotubes not only have good biocompatibility, but also reduce bacterial adhesion."
| [1] Weiss RJ, Thorsell M, Stark A, et al. 2- to 9-year outcome of stemmed total knee arthroplasty. Similar failure rates in patients when used primary or as a revision. Acta Orthop. 2014;85(6):609-613. [2] Dale H, Hallan G, Hallan G, et al. Increasing risk of revision due to deep infection after hip arthroplasty. Acta Orthop. 2009;80(6):639-645. [3] Martínez-Pastor JC, Maculé-Beneyto F, Suso-Vergara S. Acute infection in total knee arthroplasty: diagnosis and treatment. Open Orthop J. 2013;7:197-204. [4] Crowninshield RD, Rosenberg AG, Sporer SM. Changing demographics of patients with total joint replacement. Clin Orthop Relat Res. 2006;443:266-272. [5] Donlan RM, Costerton JW. Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev. 2002;15(2):167-193. [6] Gilbert P, Collier PJ, Brown MR. Influence of growth rate on susceptibility to antimicrobial agents: biofilms, cell cycle, dormancy, and stringent response. Antimicrob Agents Chemother. 1990;34(10): 1865-1868. [7] Donlan RM. Biofilms and device-associated infections. Emerg Infect Dis. 2001;7:277-281. [8] Park J, Bauer S, Schmuki P, von der Mark K. Narrow window in nanoscale dependent activation of endothelial cell growth and differentiation on TiO2 nanotube surfaces. Nano Lett. 2009;9(9): 3157-3164. [9] Park J, Bauer S, von der Mark K, et al. Nanosize and vitality: TiO2 nanotube diameter directs cell fate. Nano Lett. 2007;7(6):1686-1691. [10] Popat KC, Leoni L, Grimes CA, et al. Influence of engineered titania nanotubular surfaces on bone cells. Biomaterials 2007;28:3188-3197. [11] Brammer KS, Oh S, Cobb CJ, et al. Improved bone-forming functionality on diameter-controlled TiO(2) nanotube surface. Acta Biomater. 2009;5(8):3215-3223. [12] Bjursten LM, Rasmusson L, Oh S, et al. Titanium dioxide nanotubes enhance bone bonding in vivo. J Biomed Mater Res A. 2010;92(3): 1218-1224. [13] Zhang HZ, Sun Y, Tian A, et al. Improved antibacterial activity and biocompatible on the vancomycin-loaded TiO2 nanotubes: in vivo and in vitro studies, International J Nanomed 2013;8:4379-4389. [14] Cook JL, Scott RD, Long WJ. Late hematogenous infections after total knee arthroplasty: experience with 3013 consecutive total knees. J Knee Surg. 2007;20(1):27-33. [15] Schmalzried TP, Amstutz HC, Au MK, et al. Etiology of deep sepsis in total hip arthrosplasty. The significance of hematogenous and recurrent infections. Clin Orthop Rel Res. 1992;280:200-207.[16] Zhang E, Li F, Wang H, et al. A new antibacterial titanium-copper sintered alloy: Preparation and antibacterial property. Mater Sci Eng C Mater Biol Appl. 2013;33(7):4280-4287. [17] Tian XB, Wang ZM, Yang SQ, et al. Antibacterial copper-containing titanium nitride films produced by dual magnetron sputtering, Surf Coat Technol. 2007;201:8606-8609.[18] Secinti KD, Ayten M, Kahilogullari G, et al. Antibacterial effects of electrically activated vertebral implants. J Clin Neurosci 2008;15(4): 434-439. [19] Wan YZ, Xiong GY, Liang H, et al. Modi?cation of medical metals by ion implantation of copper. Appl Surf Sci. 2007;253:9426-9429. [20] Colon G, Ward BC, Webster TJ. Increased osteoblast and decreased Staphylococcus epidermidis functions on nanophase ZnO and TiO2. J Biomed Mater Res A. 2006;78(3):595-604. [21] Melaiye AYW. Silver and its application as an antimicrobial agent. Expert Opin Ther Pat. 2005;15:125-130. [22] Zheng X, Chen Y, Xie Y, et al. Antibacterial property and biocompatibility of plasma sprayed hydroxyapatite/silver composite coatings. Repentu Jishu Zazhi. 2009;18:463.[23] Azam A, Ahmed AS, Oves M, et al. Size-dependent antimicrobial properties of CuO nanoparticles against Gram-positive and -negative bacterial strains. Int J Nanomedicine. 2012;7:3527-3535. [24] Tsao N, Luh TY, Chou CK, et al. In vitro action of carboxyfullerene. J Antimicrob Chemother. 2002;49(4):641-649. [25] Ercan B, Taylor E, Alpaslan E et al. Diameter of titanium nanotubes influences anti-bacterial efficacy. Nanotechnology. 2011;22(29):295102. [26] Helmus MN, Gibbons DF, Cebon D. Biocompatibility: meeting a key functional requirement of next-generation medical devices. Toxicol Pathol. 2008;36(1):70-80. [27] Puckett SD, Taylor E, Raimondo T, et al. The relationship between the nanostructure of titanium surfaces and bacterial attachment. Biomaterials. 31:706-713. [28] Oh SH, Finõnes RR, Daraio C, et al. Growth of nano-scale hydroxyapatite using chemically treated titanium oxide nanotubes. Biomaterials. 2005;26(24):4938-4943. [29] Kim HM, Himeno T, Kawashita M, et al. Surface potential change in bioactive titanium metal during the process of apatite formation in simulated body fluid. J Biomed Mater Res. 2003;67A(4):1305-1309.[30] Qian T, Wang Y. Micro/nano-fabrication technologies for cell biology. Med Biol Eng Comput. 2010;48(10):1023-1032. [31] Meyer U, Büchter A, Wiesmann HP, et al. Basic reactions of osteoblasts on structured material surfaces. Eur Cell Mater. 2005;9: 39-49. [32] Beutner R, Michael J, Schwenzer B, et al. Biological nano-functionalization of titanium-based biomaterial surfaces: a flexible toolbox. J R Soc Interface. 2010;6;7 Suppl 1:S93-S105.[33] Popat KC, Eltgroth M, Latempa TJ, et al. Decreased Staphylococcus epidermis adhesion and increased osteoblast functionality on antibiotic-loaded titania nanotubes. Biomaterials. 2007;28(32):4880-4888. [34] Ren L, Wong HM, Yan CH, et al. Osteogenic ability of Cu-bearing stainless steel. J Biomed Mater Res B Appl Biomater. 2015; 103(7):1433-1444.[35] Stoodley P, Ehrlich GD, Sedghizadeh PP, et al. Orthopaedic biofilm infections. Curr Orthop Pract. 2011;22(6):558-563. [36] Hetrick EM, Schoenfisch MH. Reducing implant-related infections: active release strategies. Chem Soc Rev. 2006;35(9):780-789. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Li Xingping, Xiao Dongqin, Zhao Qiao, Chen Shuo, Bai Yiguang, Liu Kang, Feng Gang, Duan Ke. Preparation and properties of copper-loaded antibacterial functional film on titanium surface [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 553-557. |
| [5] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [6] | Shi Xiaoxiu, Mao Shilong, Liu Yang, Ma Xingshuang, Luo Yanfeng. Comparison of tantalum and titanium (alloy) as orthopedic materials: physical and chemical indexes, antibacterial and osteogenic ability [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 593-599. |
| [7] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [8] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [9] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [10] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [11] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [12] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [13] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [14] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [15] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||