Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (9): 1370-1375.doi: 10.3969/j.issn.2095-4344.0466
Previous Articles Next Articles
Effect of 1,25(OH)2D3 on type I collagen secretion in adipose-derived mesenchymal stem cells and its mechanism
Zhai Min1, Hu Xiao-gen2, Liu Hong-lin1, Xu Shi-qing1, Wang Zai1, Zhang Wen-jian1
- 1Institute of Clinical Medical Sciences, 2Department of Plastic Surgery, China-Japan Friendship Hospital, Beijing 100029, China
-
Revised:2018-01-29Online:2018-03-28Published:2018-04-03 -
Contact:Zhang Wen-jian, Ph.D., Master’s supervisor, Professor, Investigator, Institute of Clinical Medical Sciences, China-Japan Friendship Hospital, Beijing 100029, China; Wang Zai, M.D., Associate investigator, Institute of Clinical Medical Sciences, China-Japan Friendship Hospital, Beijing 100029, China -
About author:Zhai Min, Master candidate, Institute of Clinical Medical Sciences, China-Japan Friendship Hospital, Beijing 100029, China -
Supported by:the National Natural Science Foundation of China, No. 81370873, 81402451; the Talent Youth Plan of the China-Japan Friendship Hospital, No. 2015-QNYC-B-06
CLC Number:
Cite this article
Zhai Min, Hu Xiao-gen, Liu Hong-lin, Xu Shi-qing, Wang Zai, Zhang Wen-jian. Effect of 1,25(OH)2D3 on type I collagen secretion in adipose-derived mesenchymal stem cells and its mechanism[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(9): 1370-1375.
share this article
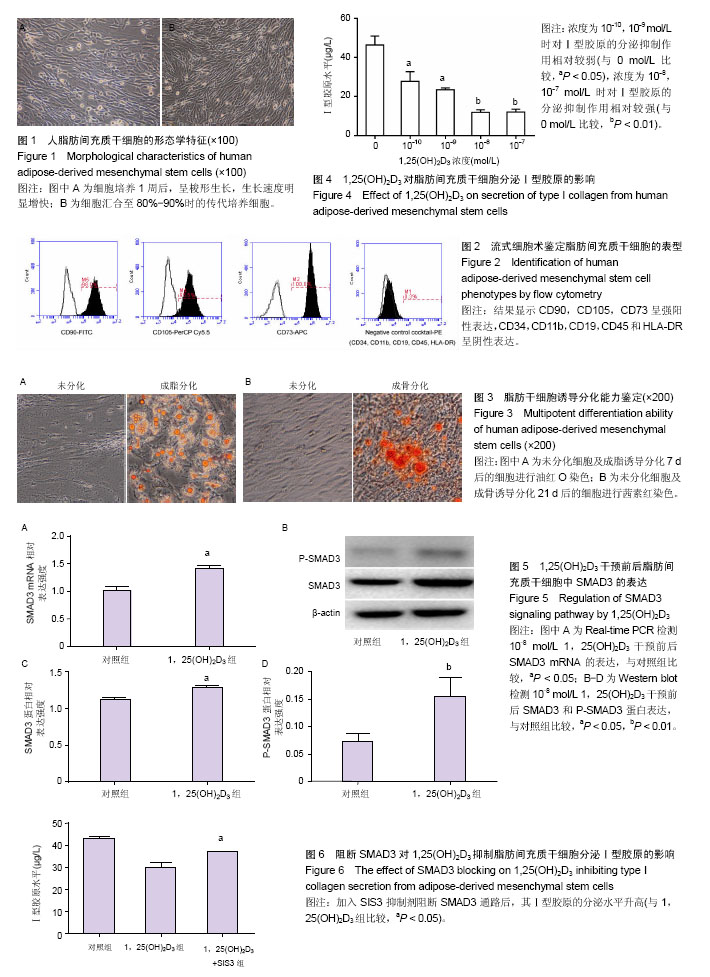
2.1 脂肪间充质干细胞的鉴定结果 人脂肪组织块消化接种培养过夜后细胞贴壁,3 d后逐渐开始快速增殖,细胞呈梭形成纤维样形态,见图1。通过流式细胞术检测发现,该细胞表面强表达CD90,CD105,CD73,不表达CD34,CD11b,CD19,CD45和HLA-DR,见图2,提示该细胞为间充质干细胞,即脂肪间充质干细胞。成脂分化7 d后,油红O染色可见脂滴形成;成骨分化21 d后,茜素红染色可见钙结节形成,见图3。 2.2 1,25(OH)2D3对Ⅰ型胶原分泌的影响 ELISA结果显示,与对照组相比,4个浓度的1,25(OH)2D3对脂肪间充质干细胞分泌Ⅰ型胶原均有明显的抑制作用,并且浓度越高抑制作用越明显(P < 0.01),见图4。 2.3 1,25(OH)2D3对SMAD3表达的影响 Real-time PCR结果显示,10-8 mol/L 1,25(OH)2D3干预后SMAD3 mRNA的表达略有升高(P < 0.05),见图5A。Western blot结果显示,10-8 mol/L 1,25(OH)2D3干预后SMAD3蛋白的表达水平有一定增高(P < 0.05),其结果与PCR结果一致;而P-SMAD3蛋白水平较对照组明显增加(P < 0.01),见图5B-D。 2.4 阻断SMAD3后对1,25(OH)2D3作用的影响 1,25(OH)2D3干预后脂肪间充质干细胞分泌Ⅰ型胶原能力下降,加入SMAD3通路抑制剂SIS3后Ⅰ型胶原分泌水平增高(P < 0.05),表明1,25(OH)2D3可通过SMAD3信号通路影响Ⅰ型胶原的分泌,见图6。"
| [1] Gurtner GC, Werner S, Barrandon Y, et al. Wound repair and regeneration. Nature. 2008;453(7193):314-321. [2] Ho S, Marçal H, Foster LJ. Towards scarless wound healing: a comparison of protein expression between human, adult and foetal fibroblasts. Biomed Res Int. 2014;2014:676493.[3] Rodriguez AM, Elabd C, Amri EZ, et al. The human adipose tissue is a source of multipotent stem cells. Biochimie. 2005; 87(1):125-128.[4] Jung H, Kim HH, Lee DH, et al. Transforming growth factor-beta 1 in adipose derived stem cells conditioned medium is a dominant paracrine mediator determines hyaluronic acid and collagen expression profile. Cytotechnology. 2011;63(1):57-66.[5] Kim WS, Park BS, Sung JH, et al. Wound healing effect of adipose-derived stem cells: a critical role of secretory factors on human dermal fibroblasts. J Dermatol Sci. 2007;48(1): 15-24.[6] Thielitz A, Vetter RW, Schultze B, et al. Inhibitors of dipeptidyl peptidase IV-like activity mediate antifibrotic effects in normal and keloid-derived skin fibroblasts. J Invest Dermatol. 2008; 128(4):855-866.[7] Xing L, Franz MG, Marcelo CL, et al. Amnion-derived multipotent progenitor cells increase gain of incisional breaking strength and decrease incidence and severity of acute wound failure. J Burns Wounds. 2007;7:e5.[8] Jung KH, Song SU, Yi T, et al. Human bone marrow-derived clonal mesenchymal stem cells inhibit inflammation and reduce acute pancreatitis in rats. Gastroenterology. 2011; 140(3):998-1008.[9] Rodbell M. Metabolism of isolated fat cells. II. The similar effects of phospholipase C (Clostridium perfringens alpha toxin) and of insulin on glucose and amino acid metabolism. J Biol Chem. 1966;241(1):130-139.[10] Zuk PA, Zhu M, Mizuno H, et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Eng. 2001;7(2):211-228.[11] Zuk PA, Zhu M, Ashjian P, et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13(12): 4279-4295.[12] Chlupác J, Filová E, Bacáková L. Vascular prostheses: 50 years of advancement from synthetic towards tissue engineering and cell therapy. Rozhl Chir. 2010;89(1):85-94.[13] Ramos TV, Wang T, Maki CB, et al. Adipose stem cell side population in the mouse. J Tissue Eng Regen Med. 2009; 3(6):430-441.[14] Diekman BO, Estes BT, Guilak F. The effects of BMP6 overexpression on adipose stem cell chondrogenesis: Interactions with dexamethasone and exogenous growth factors. J Biomed Mater Res A. 2010;93(3):994-1003.[15] Fraser JK, Wulur I, Alfonso Z, et al. Fat tissue: an underappreciated source of stem cells for biotechnology. Trends Biotechnol. 2006;24(4):150-154.[16] Secretan C, Bagnall KM, Jomha NM. Effects of introducing cultured human chondrocytes into a human articular cartilage explant model. Cell Tissue Res. 2010;339(2):421-427.[17] Ahmed N, Taylor DW, Wunder J, et al. Passaged human chondrocytes accumulate extracellular matrix when induced by bovine chondrocytes. J Tissue Eng Regen Med. 2010; 4(3):233-241.[18] Werdin F, Tenenhaus M, Rennekampff HO. Chronic wound care. Lancet. 2008;372(9653):1860-1862.[19] Goldberg SR, Diegelmann RF. Wound healing primer. Surg Clin North Am. 2010;90(6):1133-1146.[20] Falanga V. Wound healing and its impairment in the diabetic foot. Lancet. 2005;366(9498):1736-1743.[21] Jo DI, Yang HJ, Kim SH, et al. Coverage of skin defects without skin grafts using adipose-derived stem cells. Aesthetic Plast Surg. 2013;37(5):1041-1051.[22] Marino G, Moraci M, Armenia E, et al. Therapy with autologous adipose-derived regenerative cells for the care of chronic ulcer of lower limbs in patients with peripheral arterial disease. J Surg Res. 2013;185(1):36-44.[23] Yun IS, Jeon YR, Lee WJ, et al. Effect of human adipose derived stem cells on scar formation and remodeling in a pig model: a pilot study. Dermatol Surg. 2012;38(10):1678-1688.[24] Corcione A, Benvenuto F, Ferretti E, et al. Human mesenchymal stem cells modulate B-cell functions. Blood. 2006;107(1):367-372.[25] Yun IS, Jeon YR, Lee WJ, et al. Effect of human adipose derived stem cells on scar formation and remodeling in a pig model: a pilot study. Dermatol Surg. 2012;38(10):1678-1688.[26] Welsh J. Cellular and molecular effects of vitamin D on carcinogenesis. Arch Biochem Biophys. 2012;523(1): 107-114.[27] Zhou YS, Liu YS, Tan JG. Is 1, 25-dihydroxyvitamin D3 an ideal substitute for dexamethasone for inducing osteogenic differentiation of human adipose tissue-derived stromal cells in vitro. Chin Med J (Engl). 2006;119(15):1278-1286.[28] Fromigué O, Marie PJ, Lomri A. Differential effects of transforming growth factor beta2, dexamethasone and 1,25-dihydroxyvitamin D on human bone marrow stromal cells. Cytokine. 1997;9(8):613-623.[29] Curtis KM, Aenlle KK, Frisch RN, et al. TAp63γ and ΔNp63β promote osteoblastic differentiation of human mesenchymal stem cells: regulation by vitamin D3 Metabolites. PLoS One. 2015;10(4):e0123642.[30] 沈小辉,杨华清,唐文娟,等. 1α,25-二羟维生素D3对成骨细胞增殖分化、Ⅰ型胶原及核心结合因子α-1基因表达的影响[J]. 实用老年医学,2014,28(6):500-505.[31] 吴学建,程省,陶金刚. 1,25(OH)2VD3对人脐带间充质干细胞增殖及成骨分化的影响[J]. 中华显微外科杂志, 2011, 34(5): 386-389.[32] 柳逸博,王佐林. 浓缩生长因子与1,25二羟基维生素D3对骨髓间充质干细胞增殖与成骨分化的影响[J]. 口腔颌面外科杂志, 2016,26(1):13-18.[33] Potter JJ, Liu X, Koteish A, et al. 1,25-dihydroxyvitamin D3 and its nuclear receptor repress human α1 (I) collagen expression and type I collagen formation. Liver Int. 2013; 33(5):677-686.[34] Bläuer M, Sand J, Laukkarinen J. Physiological and clinically attainable concentrations of 1,25-dihydroxyvitamin D3 suppress proliferation and extracellular matrix protein expression in mouse pancreatic stellate cells. Pancreatology. 2015;15(4):366-371.[35] Ramirez AM, Wongtrakool C, Welch T, et al. Vitamin D inhibition of pro-fibrotic effects of transforming growth factor beta1 in lung fibroblasts and epithelial cells. J Steroid Biochem Mol Biol. 2010;118(3):142-150. [36] Halder SK, Osteen KG, Al-Hendy A. 1,25-dihydroxyvitamin d3 reduces extracellular matrix-associated protein expression in human uterine fibroid cells. Biol Reprod. 2013;89(6):150.[37] Sato M. Upregulation of the Wnt/beta-catenin pathway induced by transforming growth factor-beta in hypertrophic scars and keloids. Acta Derm Venereol. 2006;86(4):300-307.[38] Li Y, Zou N, Wang J, et al. TGF-β1/Smad3 Signaling Pathway Mediates T-2 Toxin-Induced Decrease of Type II Collagen in Cultured Rat Chondrocytes. Toxins (Basel). 2017;9(11): E359. |
| [1] | Li Xuan, Sun Yimin, Li Longbiao, Wang Zhenming, Yang Jing, Wang Chenglin, Ye Ling. Manufacturing of nano-modified polycaprolactone microspheres and its biological effects in dental pulp cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(10): 1602-1608. |
| [2] | Liu Xiaogang, Li Tian, Zhang Duo. Effect and mechanism of the effective components of Chinese medicine on promoting the differentiation of bone marrow mesenchymal stem cells into chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 121-126. |
| [3] | Mai Liping, He Guodong, Chen Shaoxian, Zhu Jiening, Hou Xinghua, Zhang Mengzhen, Li Xiaohong. Expression of aldehyde dehydrogenase 3B1 during human bone marrow mesenchymal stem cells senescence [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 40-44. |
| [4] | Yang Tengyun, Li Yanlin, Liu Dejian, Wang Guoliang, Zheng Zhujun. Chondrogenic differentiation of peripheral blood-derived mesenchymal stem cells induced by transforming growth factor beta 3: a dose-effect relationship [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 45-51. |
| [5] | Huang Tao, Cheng Zhijian, Jia Zhiqiang, Zhao Xiaoguang, Wang Lei, Zhai Wenjing, Zhou Yongxin. Mechanism by which miR-146a regulates osteogenic differentiation of adipose derived mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 70-75. |
| [6] | Jing Jin, Zhao Shandi, Chen Long, Peng Shuanglin, Tang Hui, Guo Daijin, Zeng Xinyi, Xiao Jingang. Repair of calvarial defects in osteoporotic mice by adipose-derived stem cells combined with biphasic calcium phosphate ceramic scaffold [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 90-95. |
| [7] | Cui Shuaishuai, Yang Xiaohong. Effects of miRNA in self-renewal, multidirectional differentiation, fate and function regulation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 101-106. |
| [8] | Kang Yue, Liu Jie. Biological function of circular RNAs in osteogenic differentiation of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 107-112. |
| [9] | Feng Zhiguo, Sun Haibiao, Han Xiaoqiang. Regulation of proliferation, differentiation and apoptosis of bone-related cells by long-stranded non-coding RNA [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 113-120. |
| [10] | Song Huifang, Tan Jiayin, Kang Yi, Li Bin, Bi Zhifei, Long Nü, Xia Zhongnian, Guo Rui. Hypoxic pretreatment enhances the protective effect of aged human bone marrow mesenchymal stem cells conditioned medium against H9C2 oxidative stress damage [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 1-6. |
| [11] | Xie Xingqi, Hu Wei, Tu Guanjun. Bone marrow mesenchymal stem cells-derived exosomes combined with chondroitinase ABC for treating spinal cord injury in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 20-26. |
| [12] | Zhang Xuelei, Luo Gan, Yu Shenghui, Gu Zuchao, Peng Xu, He Xueling, Liu Yan, Zhang Xiaomei. Acellular nerve scaffold combined with bone marrow mesenchymal stem cells and platelet-rich gel for femoral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 27-32. |
| [13] | Zhao Laihe, Xia Bing, Ma Teng, Gao Jianbo, Li Shengyou, Gao Xue, Zheng Yi, Hu Guangwen, Luo Zhuojing, Huang Jinghui. Extracellular matrix of Schwann-like cells induced by bone marrow mesenchymal stem cells promotes axonal regeneration after peripheral nerve injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 33-39. |
| [14] | Shuai Zhiqin, Chen Jiameng, Liu Taotao, Hu Anling, Li Lisheng, Yu Limei, Xu Shangfu. Research status and problems of stem cells and their derived exosomes for prevention and treatment of vascular restenosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 138-144. |
| [15] | Li Zhongkang, Zheng Jiahua, Tian Yanpeng, Huang Xianghua. Latest progress and mechanisms of mesenchymal stem cells on premature ovarian failure [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(1): 145-152. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||