Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (5): 801-806.doi: 10.3969/j.issn.2095-4344.0454
Previous Articles Next Articles
Stem cell-derived exosomes in regenerative medicine: research progress and applications
Zhang En-guo1, 2, Chen Shang-ya2, 3, Yang Ye1, 2, Du Zhong-jun2, Shao Hua2
- 1School of Medicine and Life Sciences, University of Jinan-Shandong Academy of Medical Sciences, Jinan 250062, Shandong Province, China; 2Shandong Academy of Occupational Health and Occupational Medicine, Shandong Academy of Medical Sciences, Jinan 250062, Shandong Province, China; 3Shandong University of Traditional Chinese Medicine, Jinan 250355, Shandong Province, China
-
Revised:2017-09-21Online:2018-02-18Published:2018-02-18 -
Contact:Shao Hua, Investigator, Doctoral supervisor, Shandong Academy of Occupational Health and Occupational Medicine, Shandong Academy of Medical Sciences, Jinan 250062, Shandong Province, China; Du Zhong-jun, Master’s supervisor, Shandong Academy of Occupational Health and Occupational Medicine, Shandong Academy of Medical Sciences, Jinan 250062, Shandong Province, China -
About author:Zhang En-guo, Master candidate, School of Medicine and Life Sciences, University of Jinan-Shandong Academy of Medical Sciences, Jinan 250062, Shandong Province, China; Shandong Academy of Occupational Health and Occupational Medicine, Shandong Academy of Medical Sciences, Jinan 250062, Shandong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81470145, 81602893; the Natural Science Foundation of Shandong Province, No. ZR2015YL049; Shandong Provincial Medical and Health Science and Technology Development Program, No. 2016WS0540; Innovation Project of Shandong Academy of Medical Sciences
CLC Number:
Cite this article
Zhang En-guo, Chen Shang-ya, Yang Ye, Du Zhong-jun, Shao Hua. Stem cell-derived exosomes in regenerative medicine: research progress and applications[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(5): 801-806.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
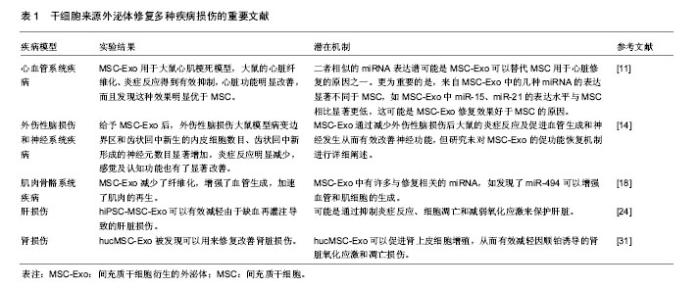
2.1 干细胞源外泌体 1983年,外泌体首次在绵羊网织红细胞中被发现,随后在1987年,Johnstone等将其命名为“exosome” [3-4]。外泌体是一种纳米级尺寸的膜性小囊泡,其直径在30-150 nm之间,多种细胞在正常及病理状态下均可分泌外泌体。外泌体含有多种生物活性因子,包括与其来源细胞相似的生长因子、细胞因子等蛋白质,以及脂质体、miRNA等活性物质。值得一提的是,研究发现其内容物因来源的细胞类型及所处的微环境不同而有不同程度的表达差异[5-8]。不同来源的外泌体虽然内容物有不同程度差异,但通常含有一套相同的蛋白质来调节膜骨架的动态性及膜融合,如与骨架相关的蛋白(β-肌动蛋白、肌球蛋白和微管蛋白)和细胞代谢相关物质(甘油醛-3-磷酸脱氢酶)。此外还含有细胞内信号传导蛋白如β-catenin、Wnt,参与细胞间通讯的蛋白如白细胞介素1b和肿瘤坏死因子α等[9]。而再生医学是一门主要研究机体修复再生的新兴交叉学科,可以理解为利用生物学及组织工程学来寻找有效可行的治疗方法以促进机体自我修复与再生,或者构建移植新的组织或器官以维持、改善、修复受损机体。近年来,干细胞来源的外泌体已被鉴定为细胞间信息交流的潜在载体,可以修复由于创伤及病理性导致的机体损伤,从而作为一种“无细胞”治疗策略为再生医学领域的机体损伤修复、保护提供一种新的思路。 2.2 干细胞源外泌体在再生医学领域的研究 2.2.1 干细胞源外泌体在心血管系统中的研究 最近的研究表明,间充质干细胞(mesenchymal stem cells, MSC)释放的细胞外囊泡(MSC-EV)具有和MSC一样促进血管生成的能力,Bian课题组[10]将MSC-EV用于急性心肌梗死大鼠模型,发现大鼠的血流恢复显著改善,梗死面积明显减少,表明MSC-EV至少能通过促进血管形成保护心脏组织,可以推断MSC-EV的促血管生成活性在保护缺血性心脏损伤中发挥至关重要的作用。但该研究并未对MSC-EV的具体组成成分进行分析。在另一项MSC和MSC衍生的外泌体(MSC-Exo)相关研究中,Shao等[11]研究者将MSC-Exo用于大鼠心肌梗死模型,大鼠的心脏纤维化、炎症反应得到有效抑制,心脏功能明显改善,而且发现这种效果明显优于MSC。为了进一步阐明其潜在机制,研究者进行了miRNA序列分析和几项体外实验,显示MSC-Exo的治疗减少了炎症和纤维化,抑制了成纤维细胞的转化和H9c2细胞的凋亡,保留了心肌梗死后的心肌功能。miRNA测序显示,MSC-Exo和MSC具有部分相似的miRNA表达谱,二者负调节心脏功能的miR-378、miR-130和miR-34表达相对较低,而正调节心脏功能的miR-24、miR-29表达相对较高,二者相似的miRNA表达谱可能是MSC-Exo可以替代MSC用于心脏修复的原因之一。更为重要的是,研究还发现来自MSC-Exo中的几种miRNA的表达显著不同于MSC,如MSC-Exo中miR-15、miR-21的表达水平与MSC相比显著降低,这可能是MSC-Exo修复效果好于MSC的原因。之前的研究发现抑制miR-15的表达水平可以有效预防心脏缺血性损伤[12],这项研究基本表明MSC-Exo可以单独用来促进心脏修复并且其修复受损心肌的效果还优于MSC。 2.2.2 干细胞源外泌体在外伤性脑损伤和神经系统中的研究 外伤性脑损伤是一种导致残疾和死亡的严重疾病,在过去一段时间内虽然进行许多基于神经保护的实验,但其适用性和有效性没有得到广泛证实,因此,迫切寻找一种对外伤性脑损伤有效可行的治疗方法。最近的研究表明MSC-Exo可以促进神经突重塑和神经功能恢复,对外伤性脑损伤和中枢神经系统疾病有明显的改善效果。Xin等[13]研究者首次报道了MSC-Exo系统用于大鼠脑卒中模型后的促神经血管重塑和功能恢复效果。研究者对大脑中动脉闭塞大鼠尾静脉注射MSC-Exo后,其轴突密度突触泡蛋白阳性区沿着纹状体和皮质的缺血边界显著增加。研究证实MSC-Exo可以增强其神经突重塑,有希望成为脑卒中的一种新型的无细胞治疗手段。随后,Zhang等[14]提出一个新的假说,他们发现给予MSC-Exo后,外伤性脑损伤大鼠模型病变边界区和齿状回中新生的内皮细胞数目、齿状回中新形成的神经元数目显著增加,炎症反应明显减少,感觉及认知功能也有了显著改善。该研究首次证明了MSC-Exo通过减少外伤性脑损伤大鼠的炎症反应及促进血管生成和神经发生从而有效改善神经功能,但研究未对MSC-Exo的促功能恢复机制进行详细阐述。在其他干细胞来源的外泌体方面,Lopez-Verrilli等[15]研究者发现月经血源性间充质干细胞衍生的外泌体(MenSC-Exo)对皮质神经元中最长神经突的生长有良好的促进效果,MenSC-Exo的生长刺激效应以及对脑皮质神经突生长的优异效应可以为治疗神经系统疾病提供潜在治疗策略。还有一些研究用抗炎症、神经保护分子和干细胞源外泌体组合用于神经的保护,Kalani 等[16]发现用姜黄素附载胚胎干细胞源外泌体(MESC-Exo)可以对小鼠缺血再灌注损伤模型的神经血管进行修复。缺血再灌注损伤小鼠模型系统给予MESC-Exo后,水肿、梗死体积和星形胶质细胞中的胶质纤维酸性蛋白表达水平显著降低,而且减轻了血管炎症,恢复了血管内皮紧密连接蛋白水平,改善了内皮血管结合蛋白如VE-钙黏蛋白的表达水平。对于神经系统疾病,其神经系统修复的重要机制之一可能是轴突重塑,而MSC-Exo对轴突生长的分子机制尚未阐明,不过最近的一项研究有助于更好解释其作用机制。Zhang等[17]研究者在使用微流体培养原代皮质神经元时,发现MSC-Exo有促进轴突生长的效应,并且在MSC-Exo中,作为miRNA蛋白之一的氩-2(Ago-2)蛋白,如果降低其表达水平则消除了外泌体对轴突的这种生长效应。更为重要的是,研究发现携带升高miR-17-92簇的MSC-Exo激活了PTEN/mTOR信号通路,可以进一步地增强轴突生长效应。所以此研究揭示了携带miRNA定制的MSC-Exo可以将miRNA传递进入受体神经元,激活其目标信号,促进轴突进一步生长,为增强轴突生长的分子机制提出了新见解。 2.2.3 干细胞源外泌体在肌肉骨骼系统中的研究 对于骨骼肌系统,之前的一些研究报道发现,MSC治疗虽然能促进大鼠损伤后的骨骼肌再生,但细胞并未分化为骨骼肌纤维,这可能是MSC通过除肌原性分化之外的其他机制促进骨骼肌的再生。因此,研究者越来越重视其旁分泌机制中外泌体的调控作用。Nakamura等[18]报道了MSC-Exo可以通过增强血管生成来促进肌肉的再生,作用的机制可能是miR-494参与调控。体外实验显示,MSC条件培养基(MSC-CM)、MSC-Exo两组中的Myod1和Myog表达水平显著高于无外泌体的对照组,MSC-Exo和MSC-CM促进了C2C12增殖和分化。体内实验显示MSC-Exo组的纤维化面积显著小于对照组,而毛细血管密度明显大于对照组。此外研究者对3组使用人脐静脉内皮细胞进行了迁移测定,结果发现MSC-CM和MSC-Exo组的人脐静脉内皮细胞的管长度显著大于对照组。研究证实MSC-Exo减少了纤维化,增强了血管生成,加速了肌肉的再生。更为重要的是该研究鉴定了MSC-Exo中许多与修复相关的miRNA,如发现了miR-494可以增强血管和肌细胞的生成。同样,在miRNA方面,Furuta等[19]研究者发现MSC-Exo促进骨折愈合可能是因为其内含的miRNA参与介导。研究者对小鼠股骨骨折模型给予MSC-Exo后,发现其外泌体中骨修复相关的细胞因子:基质细胞衍生因子1(SDF-1)、单核细胞趋化蛋白1(MCP-1)、单核细胞趋化蛋白3(MCP-3)的表达水平明显低于无外泌体组,推断MSC-Exo修复骨骼愈合的过程可能不仅仅归因于这几种因子,还可能由外泌体的其他组分如miRNA参与介导。在软骨修复方面,Zhang等[20]研究者首次证明了来源于人胚胎间充质干细胞衍生的外泌体对软骨修复的功效。Qi课题组[21]为了有效解决由于创伤、肿瘤切除和严重感染等引起的骨损伤这一难题,他们利用源自人诱导多能干细胞的间充质干细胞分泌的外泌体(hiPSC-MSC-Exo)包含MSC和iPSC二者优点的特性,探索其对来自卵巢切除大鼠骨髓间充质干细胞增殖和成骨分化的效果。研究发现hiPSC-MSC-Exo促进了细胞增殖,增强了碱性磷酸酶的活性,上调了大鼠模型中成骨细胞相关基因的mRNA和蛋白表达。该研究证实了hiPSC-MSC-Exo可以显著刺激卵巢切除大鼠骨髓间充质干细胞的增殖和成骨分化,并且表明这种效果会随着外泌体浓度的增加而增强,不过并未报道其具体的分子机制。 2.2.4 干细胞源外泌体在肝损伤中的研究 肝损伤通常发生在各种原因的慢性损伤中,目前没有非常理想的治疗方式。大量研究显示MSC-Exo可以对肝损伤发挥保护作用。Yan课题组[22]发现人脐带间充质干细胞衍生的外泌体(hucMSC-Exo)可以有效挽救由四氯化碳诱导的肝衰竭,并且证实hucMSC-Exo衍生的谷胱甘肽过氧化物酶1是介导肝脏氧化损伤修复的重要因素,可以有效地减少氧化应激和凋亡以保护肝脏。在肝再生方面,王伟伟等[23]研究者报道将人诱导多潜能干细胞(hiPSC-MSC)衍生的外泌体用于肝切除70%大鼠模型,发现肝组织坏死程度减轻,天冬氨酸转氨酶和血谷氨酸转氨酶水平升高,证实hiPSC-MSC外泌体可以有效促进肝切除70%大鼠的肝修复和再生。在保护肝缺血再灌注损伤方面,最近Nong等[24]研究者第一次评估了hiPSC-MSC-Exo在肝脏缺血再灌注损伤大鼠模型中的保护作用。研究发现系统地给予hiPSC-MSC-Exo后,肝细胞损伤标记如天冬氨酸氨基转移酶、丙氨酸氨基转移酶和炎症标记物如白细胞介素6、肿瘤坏死因子α的表达水平显著降低,肝细胞坏死和窦状充血现象也明显被抑制。此外,实验组的肝组织中凋亡标记物如胱天蛋白酶3表达水平降低,氧化标记物如超氧化物歧化酶、谷胱甘肽和谷胱甘肽过氧化物酶水平显著高于对照组。该研究证实了hiPSC-MSC-Exo可以有效减轻由于缺血再灌注导致的肝脏损伤,其机制可能是通过抑制炎症反应、细胞凋亡和减弱氧化应激来保护肝脏。在抗凋亡能力方面,Chen课题组报道了MenSC-Exo在D-GalN/LPS诱导的暴发性肝衰竭小鼠中的抗凋亡潜力[25]。研究发现MenSC-Exo可以表达ICAM-1、白细胞介素6、白细胞介素8、Axl、血管生成素以及IGFBP-6等细胞因子。其中,白细胞介素6是一种促存活的细胞因子,可以有效促进肝细胞增殖[26];Axl可以通过多种信号通路调节细胞的增殖和凋亡[27];血管生成素主要由肝脏产生,可以促进细胞增殖和细胞存活[28];IGFBP-6可以有效防止成纤维细胞衰老及凋亡[29]。MenSC-Exo中这些细胞因子可以有效抑制细胞凋亡,从而潜在地改善肝功能。在抗病毒方面,Qian课题组[30]报道脐带间充质干细胞衍生的外泌体(uMSC-Exo)在对抗丙型肝炎病毒时表现出有效的抗病毒活性。研究发现其抗病毒的过程主要由uMSC-Exo所释放的一系列miRNA如miR-145、miR-199a和miR-221介导,这些物质可以显著抑制丙型肝炎病毒的RNA复制。另外研究指出,将uMSC-Exo与干扰素α或者特拉匹韦联合使用时,抗病毒的效果表现出协同效应,可以明显增强抗丙型肝炎病毒的能力,这一发现可以为寻找抗丙型肝炎病毒最优治疗方法提供了理论指导。 2.2.5 干细胞源外泌体在肾损伤中的研究 肾损伤或急性肾损伤是以肾脏排泄性质障碍或丧失为特征的一类疾病。干细胞来源的外泌体被很多研究者发现可以用来修复改善肾脏损伤。Zhou等[31]研究者将hucMSC-Exo用于顺铂诱导的急性肾损伤大鼠模型,发现8-羟脱氧鸟苷(8-OhdG)、caspase-3和凋亡家族蛋白Bax等损伤因子呈现出低水平的表达,研究初步证实hucMSC-Exo可以促进肾上皮细胞增殖,从而有效地减轻因顺铂诱导的肾脏氧化应激和凋亡损伤。干细胞来源的外泌体介导的miRNA在多种损伤修复中都发挥重要的调控作用,在肾损伤保护方面也不例外。最近,Wang课题组发现携带miR-let7c的MSC可以高效率地将miR-let7c递送至小鼠模型的受损肾细胞[32],并下调肾脏中基质金属蛋白酶9、转化生长因子β1及其受体的表达水平。研究基本证实miR-let7c-MSC可以有效地抑制肾脏纤维化,减轻小鼠模型的肾脏损伤。同样在抑制肾纤维化方面,He等[33]报道MSC-Exo和MSC均可以有效防止肾切除引起的肾损伤,并且可以减轻剩余肾脏的纤维化。在保护肾脏免受缺血再灌注损伤方面,王汝霖等[34]研究者发现来源于骨髓间充质干细胞的外泌体可以通过减少肾小管上皮细胞凋亡并且激活PI3K-Akt通路促进其增殖,从而有效发挥保护作用;Lin等[35]报道使用脂肪干细胞(ADSC)和ADSC-Exo联合治疗大鼠急性缺血再灌注损伤的效果明显优于两者单独使用。在治疗糖尿病肾病方面,Jiang等[36]研究报道源自尿衍生干细胞的外泌体(USC-Exo)可能通过抑制细胞凋亡、促进血管再生和细胞存活来有效预防因糖尿病肾病引起的肾损伤。研究发现USC-Exo可以抑制caspase-3过表达,减轻肾小管上皮细胞凋亡,促进肾小球内皮细胞的增殖。此外USC-Exo还包含转化生长因子β1、血管生成素等一些与血管再生、细胞存活有关的潜在因素。 2.2.6 干细胞源外泌体在其他修复方面的研究 在修复肺损伤方面,Aliotta等[37]研究者发现MSC-Exo可以在肺血管重塑中发挥重要调控作用,并且可以逆转野百合碱诱导的肺动脉高压,为治疗肺动脉高压带来希望。在修复皮肤损伤方面,Hu等[38]报道利用ADSC-Exo可以通过优化成纤维细胞促进皮肤伤口愈合。Yuan等[39]研究者报道人尿来源干细胞衍生的外泌体通过促进血管新生加速皮肤伤口的愈合。在抑制瘢痕形成方面,Fang等[40]报道uMSC-Exo,尤其是其内含的miRNA(如miR-21、miR-23a、miR-125b和miR-145)可以通过抑制TGF-β2/smad2信号通路有效抑制肌成纤维细胞形成,揭示了miRNA可能是uMSC-Exo抑制肌成纤维细胞生成和预防瘢痕形成的关键因素。在改善视网膜损伤方面,Yu等[41]研究者报道MSC-Exo可以通过下调单核细胞趋化蛋白1水平从而有效改善激光诱导的视网膜损伤。 干细胞来源外泌体修复多种疾病损伤的重要文献见表1。"
| [1] Abumaree M, Al Jumah M, Pace RA, et al. Immunosuppressive properties of mesenchymal stem cells. Stem Cell Rev. 2012;8(2): 375-392.[2] 王琎,陈建英. 细胞外囊泡研究新进展[J]. 中国组织工程研究, 2017, 21(4): 621-626.[3] Pan BT, Johnstone RM. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell. 1983;33(3):967-978.[4] Johnstone RM, Adam M, Hammond JR, et al. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987; 262(19):9412-9420.[5] Im H, Shao H, Park YI, et al. Label-free detection and molecular profiling of exosomes with a nano-plasmonic sensor. Nat Biotechnol. 2014;32(5):490-495.[6] Robbins PD, Morelli AE. Regulation of immune responses by extracellular vesicles. Nat Rev Immunol. 2014;14(3):195-208.[7] Lee JK, Park SR, Jung BK, et al. Exosomes derived from mesenchymal stem cells suppress angiogenesis by down-regulating VEGF expression in breast cancer cells. PLoS One. 2013;8(12):e84256.[8] Roccaro AM, Sacco A, Maiso P, et al. BM mesenchymal stromal cell-derived exosomes facilitate multiple myeloma progression. J Clin Invest. 2013;123(4):1542-1555.[9] Derkus B, Emregul KC, Emregul E. A new approach in stem cell research-Exosomes: Their mechanism of action via cellular pathways. Cell Biol Int. 2017;41(5):466-475.[10] Bian S, Zhang L, Duan L, et al. Extracellular vesicles derived from human bone marrow mesenchymal stem cells promote angiogenesis in a rat myocardial infarction model. J Mol Med (Berl). 2014;92(4):387-397.[11] Shao L, Zhang Y, Lan B, et al. MiRNA-Sequence Indicates That Mesenchymal Stem Cells and Exosomes Have Similar Mechanism to Enhance Cardiac Repair. Biomed Res Int. 2017;2017:4150705.[12] Hullinger TG, Montgomery RL, Seto AG, et al. Inhibition of miR-15 protects against cardiac ischemic injury. Circ Res. 2012;110(1): 71-81.[13] Xin H, Li Y, Cui Y, et al. Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J Cereb Blood Flow Metab. 2013;33(11):1711-1715.[14] Zhang Y, Chopp M, Meng Y, et al. Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury. J Neurosurg. 2015;122(4):856-867.[15] Lopez-Verrilli MA, Caviedes A, Cabrera A, et al. Mesenchymal stem cell-derived exosomes from different sources selectively promote neuritic outgrowth. Neuroscience. 2016;320:129-139.[16] Kalani A, Chaturvedi P, Kamat PK, et al. Curcumin-loaded embryonic stem cell exosomes restored neurovascular unit following ischemia-reperfusion injury. Int J Biochem Cell Biol. 2016;79:360-369.[17] Zhang Y, Chopp M, Liu XS, et al. Exosomes Derived from Mesenchymal Stromal Cells Promote Axonal Growth of Cortical Neurons. Mol Neurobiol. 2017;54(4):2659-2673.[18] Nakamura Y, Miyaki S, Ishitobi H, et al. Mesenchymal-stem- cell-derived exosomes accelerate skeletal muscle regeneration. FEBS Lett. 2015;589(11):1257-1265.[19] Furuta T, Miyaki S, Ishitobi H, et al. Mesenchymal Stem Cell-Derived Exosomes Promote Fracture Healing in a Mouse Model. Stem Cells Transl Med. 2016;5(12):1620-1630.[20] Zhang S, Chu WC, Lai RC, et al. Exosomes derived from human embryonic mesenchymal stem cells promote osteochondral regeneration. Osteoarthritis Cartilage. 2016;24(12):2135-2140.[21] Qi X, Zhang J, Yuan H, et al. Exosomes Secreted by Human-Induced Pluripotent Stem Cell-Derived Mesenchymal Stem Cells Repair Critical-Sized Bone Defects through Enhanced Angiogenesis and Osteogenesis in Osteoporotic Rats. Int J Biol Sci. 2016;12(7):836-849.[22] Yan Y, Jiang W, Tan Y, et al. hucMSC Exosome-Derived GPX1 Is Required for the Recovery of Hepatic Oxidant Injury. Mol Ther. 2017;25(2):465-479.[23] 王伟伟, 艾开兴. HiPSC-MSC外泌体促进大鼠肝再生修复作用的研究[J]. 外科理论与实践, 2016,21(3):212-216.[24] Nong K, Wang W, Niu X, et al. Hepatoprotective effect of exosomes from human-induced pluripotent stem cell-derived mesenchymal stromal cells against hepatic ischemia-reperfusion injury in rats. Cytotherapy. 2016;18(12):1548-1559.[25] Chen L, Xiang B, Wang X, et al. Exosomes derived from human menstrual blood-derived stem cells alleviate fulminant hepatic failure. Stem Cell Res Ther. 2017;8(1):9.[26] Hong F, Kim WH, Tian Z, et al. Elevated interleukin-6 during ethanol consumption acts as a potential endogenous protective cytokine against ethanol-induced apoptosis in the liver: involvement of induction of Bcl-2 and Bcl-x(L) proteins. Oncogene. 2002;21(1):32-43.[27] D'Arcangelo D, Gaetano C, Capogrossi MC. Acidification prevents endothelial cell apoptosis by Axl activation. Circ Res. 2002;91(7): e4-12.[28] Li S, Hu GF. Emerging role of angiogenin in stress response and cell survival under adverse conditions. J Cell Physiol. 2012;227(7): 2822-2826.[29] Micutkova L, Diener T, Li C, et al. Insulin-like growth factor binding protein-6 delays replicative senescence of human fibroblasts. Mech Ageing Dev. 2011;132(10):468-479.[30] Qian X, Xu C, Fang S, et al. Exosomal MicroRNAs Derived From Umbilical Mesenchymal Stem Cells Inhibit Hepatitis C Virus Infection. Stem Cells Transl Med. 2016;5(9):1190-1203.[31] Zhou Y, Xu H, Xu W, et al. Exosomes released by human umbilical cord mesenchymal stem cells protect against cisplatin-induced renal oxidative stress and apoptosis in vivo and in vitro. Stem Cell Res Ther. 2013;4(2):34.[32] Wang B, Yao K, Huuskes BM, et al. Mesenchymal Stem Cells Deliver Exogenous MicroRNA-let7c via Exosomes to Attenuate Renal Fibrosis. Mol Ther. 2016;24(7):1290-1301.[33] He J, Wang Y, Sun S, et al. Bone marrow stem cells-derived microvesicles protect against renal injury in the mouse remnant kidney model. Nephrology (Carlton). 2012;17(5):493-500.[34] 王汝霖,许明,戎瑞明,等. 骨髓间充质干细胞来源外泌体对大鼠肾脏缺血再灌注损伤保护作用的研究[C].杭州:2013中国器官移植大会论文汇编, 2013.[35] Lin KC, Yip HK, Shao PL, et al. Combination of adipose-derived mesenchymal stem cells (ADMSC) and ADMSC-derived exosomes for protecting kidney from acute ischemia-reperfusion injury. Int J Cardiol. 2016;216:173-185.[36] Jiang ZZ, Liu YM, Niu X, et al. Exosomes secreted by human urine-derived stem cells could prevent kidney complications from type I diabetes in rats. Stem Cell Res Ther. 2016;7:24.[37] Aliotta JM, Pereira M, Wen S, et al. Exosomes induce and reverse monocrotaline-induced pulmonary hypertension in mice. Cardiovasc Res. 2016;110(3):319-330.[38] Hu L, Wang J, Zhou X, et al. Exosomes derived from human adipose mensenchymal stem cells accelerates cutaneous wound healing via optimizing the characteristics of fibroblasts. Sci Rep. 2016;6:32993.[39] Yuan H, Guan J, Zhang J, et al. Exosomes secreted by human urine-derived stem cells accelerate skin wound healing by promoting angiogenesis in rat. Cell Biol Int. 2017;41(8):933.[40] Fang S, Xu C, Zhang Y, et al. Umbilical Cord-Derived Mesenchymal Stem Cell-Derived Exosomal MicroRNAs Suppress Myofibroblast Differentiation by Inhibiting the Transforming Growth Factor-β/SMAD2 Pathway During Wound Healing. Stem Cells Transl Med. 2016;5(10):1425-1439.[41] Yu B, Shao H, Su C, et al. Exosomes derived from MSCs ameliorate retinal laser injury partially by inhibition of MCP-1. Sci Rep. 2016;6:34562. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Zhengdong, Huang Na, Chen Jingxian, Zheng Zuobing, Hu Xinyu, Li Mei, Su Xiao, Su Xuesen, Yan Nan. Inhibitory effects of sodium butyrate on microglial activation and expression of inflammatory factors induced by fluorosis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1075-1080. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||