Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (5): 774-780.doi: 10.3969/j.issn.2095-4344.0450
Previous Articles Next Articles
Strategies for promoting the differentiation of cardiac stem cells into cardiomyocytes
Wang Lei, Chen Xu-xiang, Wu Quan-hua, Wu Hao, Long Hui-bao, Hou Jing-ying, Wang Tong
- Department of Emergency, Sun Yat-sen Memorial Hospital of Sun Yat-sen University, Guangzhou 510120, Guangdong Province, China
-
Revised:2017-09-14Online:2018-02-18Published:2018-02-18 -
Contact:Wang Tong, M.D., Doctoral supervisor, Professor, Chief physician, Researcher, Department of Emergency, Sun Yat-sen Memorial Hospital of Sun Yat-sen University, Guangzhou 510120, Guangdong Province, Chin -
About author:Wang Lei, Studying for master’s degree, Department of Emergency, Sun Yat-sen Memorial Hospital of Sun Yat-sen University, Guangzhou 510120, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China, No. 81700242; the Science and Technology Plan of Guangdong Province, No. 2017A020215176; the Medical Research Foundation of Guangdong Province, No. A2016264, A2017001
CLC Number:
Cite this article
Wang Lei, Chen Xu-xiang, Wu Quan-hua, Wu Hao, Long Hui-bao, Hou Jing-ying, Wang Tong. Strategies for promoting the differentiation of cardiac stem cells into cardiomyocytes[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(5): 774-780.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
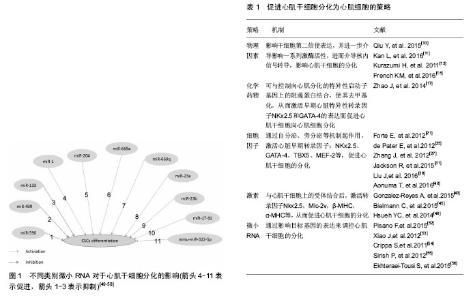
2.1 物理因素 物理因素对干细胞增殖、分化影响的研究逐渐成为热点,通过改变干细胞生长的物理微环境,进而影响干细胞的增殖分化[8]。 研究表明基底硬度是影响干细胞的重要力学因素,其影响CSCs铺展和生长,且CSCs分化能力与基底硬度正相关[9]。合适的基底硬度维持与调节CSCs分化的可能机制:通过RGD或其他整合素受体感知基底硬度,影响干细胞第二信使如cAMP、IP3、DAG表达,并进一步介导影响cAMP/PKA、Akt/PKB等激酶活性,或影响蛋白激酶如RTK表达,以及通过小G蛋白影响MAPK级联反应,进而介导核内信号转导,影响细胞骨架重排及细胞黏附、铺展、迁移[10-11]。 同样,CSCs对胞外机械力刺激也较敏感。对CSCs施加机械牵张力,模拟体内物理微环境,通过干细胞表面受体进行信号传导影响CSCs增殖及分化[12-13]。随着机械牵张力的增加,其分泌炎性细胞因子白细胞介素6、白细胞介素1β以及促血管生成因子血管内皮生长因子及碱性成纤维细胞生长因子增加,并促进了CSCs的分化潜能[14]。胞外机械力会导致G蛋白耦联受体介导的cAMP/PKA、IP3/Ca2+、DAG/PKC途径变化,或通过蛋白激酶如ERK等的磷酸化介导MAPK级联反应,也可通过转化生长因子β受体的活化由Smad蛋白调控基因表达,影响CSCs特异性分化[15]。 CSCs存活于适当的物理微环境刺激下,模拟CSCs体内微环境,高效促进CSCs分化为心肌细胞,为临床治疗心肌梗死、心力衰竭类疾病提供了希望。 2.2 化学药物 各类化学药物广泛被用于定向诱导干细胞的分化,此类诱导剂种类繁多,来源广泛,作用效果稳定显著,故一直应用普遍。但用于诱导CSCs向心肌细胞分化的化学药物目前只有5-氮杂胞苷。DNA甲基化与组蛋白甲基化或去乙酰化作用与基因的调控、细胞的进程以及存活密切相关,基于表观遗传学的调控机制,一些研究者尝试用去甲基化试剂如5-氮杂胞苷或脱乙酰化酶抑制剂如曲古霉素A或丙戊酸来调节细胞的分化。分化诱导剂通过直接或间接与心肌细胞接触可诱导CSCs向心肌分化。5-氮杂胞苷作为一种去甲基化药物,可通过与CSCs内甲基转移酶的共价结合,使细胞内部分甲基化静息基因去甲基化,恢复表达活性[16]。Zhao等[17]发现5-氮杂胞苷可与控制向心肌分化的特异性启动子基因上的阻遏蛋白结合,使其去甲基化,从而激活早期心脏特异性转录因子NKx2.5和GATA-4的表达而促进CSCs向心肌细胞分化。 2.3 细胞因子 细胞因子是调节CSCs分化的重要因素,它们通过一系列信号途径,调控CSCs向心肌细胞分化,并通过自分泌、旁分泌机制起作用[18-19],从而促进心肌梗死后的心肌修复。Liu等[20]研究提示,巨噬细胞能表达高水平的白细胞介素10和转化生长因子β以及较低水平的肿瘤坏死因子α和白细胞介素6,用含有高水平细胞因子的巨噬细胞上清液来培养CSCs,能刺激CSCs表现出较高的增殖、抗缺氧和分化活性。 2.3.1 转化生长因子β 转化生长因子β是一种调控细胞生长的重要细胞因子,具有调控细胞增殖、分化和凋亡等多种生物学功能。转化生长因子β可诱导人源性心脏祖细胞(human cardiomyocyte progenitor cells, hCMPC)分化为心肌细胞,可表达缝隙连接蛋白,并能与成熟心肌细胞一样产生动作电位[21]。另一个研究发现,在转化生长因子β1存在下,心肌球的形成增强[22],并且这种增强能被转化生长因子β1受体抑制剂SB431452急剧阻断;由于转化生长因子β在心肌受到损伤后而被激活,因而可能增加成人心肌祖细胞(cardiac progenitor cells,CPCs,是心肌球的主要组成细胞)在体内或体外的再生潜能。 2.3.2 骨形成蛋白2 骨形成蛋白2与转化生长因子β一样,均属于转化生长因子β超家族成员。骨形成蛋白2的两型受体均不能单独介导信号转导,骨形成蛋白2与Ⅰ型受体结合后,引导Ⅱ型受体与Ⅰ型受体形成受体复合物,进而激活多条下游信号通路,对于干细胞的分化发挥重要的作用。已有证据表明,骨形成蛋白2受体的存在对于CSCs的分化是不可或缺的前提条件[23]。人胚胎干细胞中加入骨形成蛋白2,能促进胚胎干细胞向心肌祖细胞分化[24]。研究者在体外用5-氮杂胞苷诱导Sca-1+心肌祖细胞,使其分化为心肌细胞,诱导后第2周细胞就出现Bmpr1a的高表达,使Bmpr1a 基因突变后心肌祖细胞无法分化为心肌细胞[25]。其功能的发挥主要通过Smads信号通路的参与。Song等[26]研究提示骨形成蛋白2主要诱导Smad 1和Smad 5的磷酸化起作用。它们分别与心脏祖细胞上的受体结合,使Smad蛋白发生磷酸化,并进一步通过级联作用,促进心脏祖细胞的分化。 2.3.3 成纤维细胞生长因子 成纤维细胞生长因子是一种有丝分裂原,具有广泛的促进组织修复与血管形成等作用。成纤维细胞生长因子中,成纤维细胞生长因子2的作用最受关注,成纤维细胞生长因子2能够促进细胞增殖、分化、存活。在人的心脏中,成纤维细胞生长因子2可被多种细胞分泌,包括心肌细胞、纤维母细胞以及平滑肌细胞。成纤维细胞生长因子2通过与其高亲和性的络氨酸激酶受体FGFR-1结合发挥生理作用。成纤维细胞生长因子2与其受体结合后,CSCs内的特异性转录因子Nkx2.5表达增强,进而促进CSCs向心肌细胞分化[27]。另外研究发现移植转染成纤维细胞生长因子2的CSCs,CSCs的生存能力增强[28]。有数据表明成纤维细胞生长因子9可能减弱血管细胞凋亡,激活c-Kit心肌祖细胞,促进其分化,并增强C57BL/6和db/db小鼠新生血管形成,从而改善心脏功能[29]。 2.3.4 胰岛素样生长因子1 胰岛素样生长因子1是一种与胰岛素结构类似的生长因子,在体内可以促进多种细胞增殖、分化,同时能够抑制细胞凋亡和坏死[30]。肝脏是其合成和分泌的主要场所。有研究将转染了胰岛素样生长因子1基因的CSCs移植入心肌梗死小鼠体内,移植的CSCs在梗死区域过表达胰岛素样生长因子1,促进了移植细胞的存活和增殖,减少了细胞凋亡[31]。然而,关于其具体机制一直未得到充分研究。Poudel等[32]猜想可能与胰岛素样生长因子1促进内源性CSCs向心肌细胞分化有关,采用能定向诱导分化为心肌细胞的P19胚胎癌细胞为模型,过表达胰岛素样生长因子1可促进P19胚胎癌细胞向心肌细胞分化。另一项研究说明胰岛素样生长因子1可作为一个极佳的备选因子提高心脏祖细胞向心肌细胞分化的效率[33]。有研究进一步证实了胰岛素样生长因子1和肝细胞生长因子在体内外可以作为细胞移植的佐剂,通过刺激移植的干细胞和内源性CSCs的存活、再生和促进血管新生来进行心肌修复[34]。 2.3.5 肝细胞生长因子 肝细胞生长因子是存在于急性肝损伤动物血浆中的蛋白因子,它能干预细胞的DNA合成,且在肝再生过程中起重要作用,然而越来越多的报道表明肝细胞生长因子不止作用于肝再生,而且对于许多组织和细胞的生长、分化起重要调控作用。近年来,人们研究发现肝细胞生长因子在心肌梗死等缺血性心脏病中表达异常,提示肝细胞生长因子参与心血管疾病的发生发展过程[35-36]。c-Met是原癌基因c-met编码的异二聚体蛋白,是一种膜表面受体,HGF通过与其特异性结合后,使其络氨酸残基磷酸化而发挥生物活性。CSCs也可表达c-Met受体,并且分泌肝细胞生长因子,通过HGF/c-Met信号途径动员CSCs向心肌梗死区迁移、增殖和分化[37]。Liu等[38]研究比较了新生儿、成年和老年大鼠的C-kit+ CSCs和HGF/p-met的表达水平,结果发现随着年龄的增加,HGF/p-met的表达水平和C-kit+CSCs丰度逐渐降低;在心肌梗死后立即将携带肝细胞生长因子基因的腺病毒注射到老年大鼠心肌梗死区域的左心室壁中,发现能促进内源性C-kit+ CSCs的增殖和分化。Savi等[39]研究进一步证实,肝细胞生长因子不仅改善了心肌祖细胞在心肌梗死局部的生存,还可以改善心肌祖细胞治疗后梗死大鼠心脏的电稳定性,并且这种改善与Connexin-43、Connexin-40和Cav1.2表达增加相关,这些因子表达有利于修复心脏中的细胞间耦合和激发扩散。 2.3.6 脱嘌呤/脱嘧啶核酸内切酶/氧化还原因子1(Apurinic/apyrimidinic endonuclease/redox factor 1,APE1) APE1能促进细胞存活并可增强移植细胞的治疗效果。在Aonuma等[40]的研究中,与对照组相比,转染了APE1的心肌祖细胞凋亡减轻,机制在于APE1过表达通过转化生长因子β激活激酶1(TAK1)和NF-κB的活化来抑制心肌祖细胞凋亡。将转染了APE1的心肌祖细胞移植治疗心肌梗死小鼠,与对照组相比,转染了APE1的心肌祖细胞在缺血性心脏中有效存活数量增加,心脏功能明显恢复,心脏炎症和纤维化明显减少。提示心肌祖细胞中的APE1过表达可作为改善心脏细胞治疗的新策略。 2.4 激素 2.4.1 催产素 催产素主要表达在下丘脑,是一种小分子多肽,研究发现催产素不只是一种女性生殖激素,还具有心脏保护作用[41],能够减小大鼠心肌梗死后面积,并且提高心功能和改善心室重塑[42]。研究发现催产素与其受体结合后,通过一系列的信号分子可诱导出心搏细胞,上调α-肌球蛋白重链和心房利钠肽基因的转录表达,进而调控其向心肌细胞分化和发育成熟[43]。已有研究证实催产素可以促进SP CD31-细胞向成熟的心肌细胞分化[44],并参与心脏的生长发育。 2.4.2 脑尿钠肽 Bielmann等[45]发现新生小鼠及成年小鼠的心脏祖细胞中均表达脑尿钠肽的两种受体:NPR-A与NPR-B,脑尿钠肽通过与NPR-A结合促进心脏祖细胞的增殖,通过与NPR-B结合,激活转录因子Nkx2.5,Mlc-2v,β- MHC,α- MHC,进而促进心脏祖细胞的分化发育。 2.4.3 前列腺素E2 Hsueh等[46]研究认为心肌干/祖细胞具有前列腺素E2反应性,前列腺素E2通过EP2受体结合直接调节干细胞的活性,或通过间接调节其体内微环境从而促进干细胞的分化,其研究结果表明前列腺素E2具有很大的心脏再生潜力。 2.5 微小RNA 微小RNA(micro-RNA, miRNA)是一类长度约22个核苷酸,具有高度保守性的内源性单链非编码RNA[47]。最新的研究显示miRNA在心肌细胞分化过程中发挥了重要的作用,对心肌干细胞在心肌修复中的作用也有一定的影响[48]。研究表明心肌细胞中表达大量的miR-1、miR-22、miR-30a、miR-30b、miR-30c、miR-133a、miR-133b和miR-499,而在CSCs中miR-1、miR-133a、miR-133b大量表达[49]。 大量研究表明,miR-1与miR-133在成体CSCs的功能调节中发挥着复杂的作用[50]。Ivey等[51]发现miR-1能促进心脏祖细胞分化,并能使哺乳动物或蝇类的细胞脱离细胞周期不再分裂。然而miR-133能抑制骨骼肌成肌细胞分化,并使其保持持续增殖的状态。 miR-499在心肌细胞中的表达量是在CSCs中表达量的数百倍,心肌内的miR-499通过缝隙连接转移到CSCs中,促进CSCs向心肌细胞分化。通过筛选后认为miR-499的靶基因是Rod1和Sox6。用siRNA-Rod1和siRNA-Sox6抑制Rod1和Sox6表达后,细胞分化的结果与在CSCs中过表达miR-499的实验结果一致,导致Nkx2.5+和GATA4+细胞的数量明显增多,促进CSCs向心肌细胞分化,证明了miR-499可以通过抑制目标基因Rod1和Sox6的表达来调控CSCs的分化[52-55]。 Ekhteraei-Tousi等[56]研究发现,miR-590过表达能够通过降低转化生长因子βR2的表达水平来减弱CDCs的分化。 对于研究稍少一些的miR亚型,如:miR-204,miR-669a,miR-669q,miR-23a,miR-708,miR-23b以及mmu-miR-322-5p,同样也能促进CSCs向心肌细胞分化[53-54,57-58]。 不同类别微小RNA对于心肌干细胞分化的影响,见图1。 除单个独立的miR,一些miRs成簇表达组成基因簇,每个基因簇成员共同转录产生一个多顺反子的转录物,miR-17-92复合物包括miR-17,-18,-19a,-19b,-20a以及-92a[55]。miR-17-92复合物的家族成员能够被骨形成蛋白的一个亚型——骨形成蛋白4所调控。Wang等[11]表明敲除骨形成蛋白4基因会导致miR-17-92复合体表达降低,同时也会导致心肌分化延迟。这些证据均表明miRNAs在调节CSCs的分化过程中所扮演的重要角色。 2.6 其他 数据显示,Sfrp2(一种典型的Wnt抑制剂)治疗可以促进缺血再灌注损伤后心肌祖细胞的分化。用Sfrp2处理心肌祖细胞可抑制其增殖,并将其引入心肌分化[59]。卵泡抑素样蛋白1(Follistatin-like-protein 1,FSTL1)、2,3,5,4’-二苯乙烯-2-O-β-d-葡萄糖苷(2,3,5,4’-tetrahydroxystilbene-2-O-β-d-glucoside, THSG)、神经调节蛋白1(Neuregulin-1,NRG1)、甲状腺素、胚胎干细胞来源的外泌体、旁分泌因子C3orf58 (一种缺氧和Akt诱导的干细胞因子)、胰岛素、二甲基亚砜、过氧化物酶体增殖物激活受体γ等均被不同程度证实与心肌细胞的增殖或其他种类干细胞向心肌细胞的分化有关[60-64],但尚未有研究证实这些蛋白、激素、细胞因子等能促进CSCs在体内外向心肌细胞分化,还有待于更多的研究予以明确。 综上,促进CSCs分化为心肌细胞的重要策略参见表1。"

| [1] Fuentes T, Kearns-Jonker M. Endogenous cardiac stem cells for the treatment of heart failure. Stem Cells Cloning. 2013;6: 1-12.[2] Iancu CB, Iancu D, Ren?ea I, et al. Molecular signatures of cardiac stem cells. Rom J Morphol Embryol. 2015;56(4): 1255-1262.[3] Di Siena S, Gimmelli R, Nori SL, et al. Activated c-Kit receptor in the heart promotes cardiac repair and regeneration after injury. Cell Death Dis. 2016;7(7):e2317.[4] Valente M, Nascimento DS, Cumano A, et al. Sca-1+ cardiac progenitor cells and heart-making: a critical synopsis. Stem Cells Dev 2014;23 (19):2263-2273.[5] Marbán E. Breakthroughs in cell therapy for heart desease: focus on cardiosphere-derived cells. Mayo Clin Proc. 2014; 89(6): 850-858. [6] Makkar RR, Smith RR, Cheng K, et al. Intracoronary cardiosphere-derived cells for heart regeneration after myocardial infarction (CADUCEUS): a prospective, randomised phase 1 trial. Lancet. 2012;379(9819):895-904.[7] Cesselli D, Aleksova A, Mazzega E, et al. Cardiac stem cell aging and heart failure. Pharmacol Res. 2017. pii: S1043-6618(16)30838-6. [Epub ahead of print][8] Koudstaal S, Jansen Of Lorkeers SJ, Gaetani R, et al. Concise review: heart regeneration and the role of cardiac stem cells. Stem Cells Transl Med. 2013;2(6):434-443.[9] Mayfield AE, Tilokee EL, Latham N, et al. The effect of encapsulation of cardiac stem cells within matrix-enriched hydrogel capsules on cell survival, post-ischemic cell retention and cardiac function. Biomaterials. 2014;35(1):133-142.[10] Qiu Y, Bayomy AF, Gomez MV, et al. A role for matrix stiffness in the regulation of cardiac side population cell function. Am J Physiol Heart Circ Physiol. 2015;308(9):H990-997.[11] Kan L, Thayer P, Fan H, et al. Polymer microfiber meshes facilitate cardiac differentiation of c-kit(+) human cardiac stem cells. Exp Cell Res. 2016;347(1):143-152.[12] Mauretti A, Bax NA, van Marion MH, et al. Cardiomyocyte progenitor cell mechanoresponse unrevealed: strain avoidance and mechanosome development. Integr Biol (Camb). 2016;8(9):991-1001.[13] Kurazumi H, Li TS, Takemoto Y, et al. Haemodynamic unloading increases the survival and affects the differentiation of cardiac stem cells after implantation into an infarcted heart. Eur J Cardiothorac Surg. 2014;45(6):976-982.[14] Kurazumi H, Kubo M, Ohshima M, et al. The effects of mechanical stress on the growth, differentiation, and paracrine factor production of cardiac stem cells. PLoS One. 2011;6(12):e28890.[15] French KM, Maxwell JT, Bhutani S, et al. Fibronectin and Cyclic Strain Improve Cardiac Progenitor Cell Regenerative Potential In Vitro. Stem Cells Int. 2016;2016:8364382.[16] Miller JL, Grant PA. The role of DNA methylation and histone modifications in transcriptional regulation in humans. Subcell Biochem. 2013;61:289-317.[17] Zhao J, Feng Y, Yan H, et al. β-arrestin2/miR-155/GSK3β regulates transition of 5'-azacytizine-induced Sca-1-positive cells to cardiomyocytes. J Cell Mol Med. 2014;18(8):1562-1570.[18] Ho YS, Tsai WH, Lin FC, et al. Cardioprotective Actions of TGFβRI Inhibition Through Stimulating Autocrine/Paracrine of Survivin and Inhibiting Wnt in Cardiac Progenitors. Stem Cells. 2016;34(2):445-455.[19] Bao L, Meng Q, Li Y, et al. C-Kit Positive Cardiac Stem Cells and Bone Marrow-Derived Mesenchymal Stem Cells Synergistically Enhance Angiogenesis and Improve Cardiac Function After Myocardial Infarction in a Paracrine Manner. J Card Fail. 2017; 23(5):403-415.[20] Liu L, Jin X, Zhou Z, et al. Enhancement of Anti-Hypoxic Activity and Differentiation of Cardiac Stem Cells by Supernatant Fluids from Cultured Macrophages that Phagocytized Dead Mesenchymal Stem Cells. Int J Mol Sci. 2016;17(7): E1175.[21] Kawaguchi N. Adult cardiac-derived stem cells: differentiation and survival regulators. Vitam Horm. 2011;87:111-125.[22] Forte E, Miraldi F, Chimenti I, et al. TGFβ-dependent epithelial-to-mesenchymal transition is required to generate cardiospheres from human adult heart biopsies. Stem Cells Dev. 2012;21(17):3081-3090.[23] Wang J, Greene SB, Bonilla-Claudio M, et al. Bmp signaling regulates myocardial differentiation from cardiac progenitors through a MicroRNA-mediated mechanism. Dev Cell. 2010; 19(6):903-912.[24] Menasché P, Vanneaux V, Hagège A, et al. Human embryonic stem cell-derived cardiac progenitors for severe heart failure treatment: first clinical case report. Eur Heart J. 2015;36(30): 2011-2017.[25] de Pater E, Ciampricotti M, Priller F, et al. Bmp signaling exerts opposite effects on cardiac differentiation. Circ Res. 2012;110(4): 578-587. [26] Song J, McColl J, Camp E, et al. Smad1 transcription factor integrates BMP2 and Wnt3a signals in migrating cardiac progenitor cells. Proc Natl Acad Sci U S A. 2014;111(20):7337-7342.[27] Zhang J, Liu J, Liu L, et al. The fibroblast growth factor signaling axis controls cardiac stem cell differentiation through regulating autophagy. Autophagy. 2012;8(4):690-691.[28] Zhang J, Liu J, Huang Y, et al. FRS2α-mediated FGF signals suppress premature differentiation of cardiac stem cells through regulating autophagy activity. Circ Res. 2012;110(4):e29-39.[29] Singla D, Wang J. Fibroblast Growth Factor-9 Activates c-Kit Progenitor Cells and Enhances Angiogenesis in the Infarcted Diabetic Heart. Oxid Med Cell Longev. 2016;2016:5810908.[30] Engels MC, Rajarajan K, Feistritzer R, et al. Insulin-like growth factor promotes cardiac lineage induction in vitro by selective expansion of early mesoderm. Stem Cells. 2014;32(6):1493-1502.[31] Jackson R, Tilokee EL, Latham N, et al. Paracrine Engineering of Human Cardiac Stem Cells With Insulin-Like Growth Factor 1 Enhances Myocardial Repair. J Am Heart Assoc. 2015;4(9): e002104.[32] Poudel B, Bilbao D, Sarathchandra P, et al. Increased cardiogenesis in P19-GFP teratocarcinoma cells expressing the propeptide IGF-1Ea. Biochem Biophys Res Commun. 2011;416(3-4):293-299.[33] Jackson R, Tilokee EL, Latham N, et al. Paracrine Engineering of Human Cardiac Stem Cells With Insulin-Like Growth Factor 1 Enhances Myocardial Repair. J Am Heart Assoc. 2015;4(9):e002104. [34] D'Amario D, Cabral-Da-Silva MC, Zheng H, et al. Insulin-like growth factor-1 receptor identifies a pool of human cardiac stem cells with superior therapeutic potential for myocardial regeneration. Circ Res. 2011;108(12):1467-1481.[35] Wang X, Li Q, Hu Q, et al. Intra-myocardial injection of both growth factors and heart derived Sca-1+/CD31- cells attenuates post-MI LV remodeling more than does cell transplantation alone: neither intervention enhances functionally significant cardiomyocyte regeneration. PLoS One. 2014;9(2):e95247.[36] Ellison GM, Torella D, Dellegrottaglie S, et al. Endogenous cardiac stem cell activation by insulin-like growth factor-1/hepatocyte growth factor intracoronary injection fosters survival and regeneration of the infarcted pig heart. J Am Coll Cardiol. 2011;58(9):977-986.[37] Gallo S, Sala V, Gatti S, et al. HGF/Met Axis in Heart Function and Cardioprotection. Biomedicines. 2014;2(4): 247-262.[38] Liu J, Wu P, Wang H, et al. Necroptosis Induced by Ad-HGF Activates Endogenous C-Kit+ Cardiac Stem Cells and Promotes Cardiomyocyte Proliferation and Angiogenesis in the Infarcted Aged Heart. Cell Physiol Biochem. 2016; 40(5):847-860.[39] Savi M, Bocchi L, Rossi S, et al. Antiarrhythmic effect of growth factor-supplemented cardiac progenitor cells in chronic infarcted heart. Am J Physiol Heart Circ Physiol. 2016;310(11):H1622-1648.[40] Aonuma T, Takehara N, Maruyama K, et al. Apoptosis- Resistant Cardiac Progenitor Cells Modified With Apurinic/ Apyrimidinic Endonuclease/Redox Factor 1 Gene Overexpression Regulate Cardiac Repair After Myocardial Infarction. Stem Cells Transl Med. 2016;5(8):1067-1078.[41] Jankowski M, Broderick TL, Gutkowska J. Oxytocin and cardioprotection in diabetes and obesity. BMC Endocr Disord. 2016;16(1):34.[42] Houshmand F, Faghihi M, Zahediasl S. Role of atrial natriuretic Peptide in oxytocin induced cardioprotection. Heart Lung Circ. 2015;24(1):86-93.[43] Gonzalez-Reyes A, Menaouar A, Yip D, et al. Molecular mechanisms underlying oxytocin-induced cardiomyocyte protection from simulated ischemia-reperfusion. Mol Cell Endocrinol. 2015;412:170-181.[44] Danalache BA, Yu C, Gutkowska J, et al. Oxytocin-Gly-Lys-Arg stimulates cardiomyogenesis by targeting cardiac side population cells. J Endocrinol. 2014;220(3):277-289.[45] Bielmann C, Rignault-Clerc S, Liaudet L, et al. Brain natriuretic peptide is able to stimulate cardiac progenitor cell proliferation and differentiation in murine hearts after birth. Basic Res Cardiol. 2015;110(1):455.[46] Hsueh YC, Wu JM, Yu CK, et al. Prostaglandin E? promotes post-infarction cardiomyocyte replenishment by endogenous stem cells. EMBO Mol Med. 2014;6(4): 496-503.[47] Purvis N, Bahn A, Katare R. The Role of MicroRNAs in Cardiac Stem Cells. Stem Cells Int. 2015;2015:194894.[48] Gama-Carvalho M, Andrade J, Brás-Rosário L. Regulation of Cardiac Cell Fate by microRNAs: Implications for Heart Regeneration. Cells. 2014;3(4):996-1026.[49] Hosoda T, Zheng H, Cabral-da-Silva M, et al. Human cardiac stem cell differentiation is regulated by a mircrine mechanism. Circulation. 2011;123(12):1287-1296.[50] Chimenti I, Pagano F, Cavarretta E, et al. Β-blockers treatment of cardiac surgery patients enhances isolation and improves phenotype of cardiosphere-derived cells. Sci Rep. 2016;6:36774.[51] Ivey KN, Srivastava D. MicroRNAs as regulators of differentiation and cell fate decisions. Cell Stem Cell. 2010; 7(1):36-41.[52] Pisano F, Altomare C, Cervio E, et al. Combination of miRNA499 and miRNA133 exerts a synergic effect on cardiac differentiation. Stem Cells. 2015;33(4):1187-1199.[53] Xiao J, Liang D, Zhang H, et al. MicroRNA-204 is required for differentiation of human-derived cardiomyocyte progenitor cells. J Mol Cell Cardiol. 2012;53(6):751-759.[54] Crippa S, Cassano M, Messina G, et al. miR669a and miR669q prevent skeletal muscle differentiation in postnatal cardiac progenitors. J Cell Biol. 2011;193(7):1197-1212.[55] Sirish P, López JE, Li N, et al. MicroRNA profiling predicts a variance in the proliferative potential of cardiac progenitor cells derived from neonatal and adult murine hearts. J Mol Cell Cardiol. 2012;52(1):264-272.[56] Ekhteraei-Tousi S, Mohammad-Soltani B, Sadeghizadeh M, et al. Inhibitory effect of hsa-miR-590-5p on cardiosphere- derived stem cells differentiation through downregulation of TGFB signaling. J Cell Biochem. 2015;116(1): 79-191.[57] Wu Q, Zhan J, Li Y, et al. Differentiation-Associated MicroRNA Alterations in Mouse Heart-Derived Sca-1(+)CD31(-) and Sca-1(+)CD31(+) Cells. Stem Cells Int. 2016;2016:9586751.[58] Deng S, Zhao Q, Zhou X, et al. Neonatal Heart-Enriched miR-708 Promotes Differentiation of Cardiac Progenitor Cells in Rats. Int J Mol Sci. 2016;17(6): E875.[59] Schmeckpeper J, Verma A, Yin L, et al. Inhibition of Wnt6 by Sfrp2 regulates adult cardiac progenitor cell differentiation by differential modulation of Wnt pathways. J Mol Cell Cardiol. 2015;85:215-225.[60] Bareja A, Hodgkinson CP, Payne AJ, et al. HASF (C3orf58) is a novel ligand of the insulin-like growth factor 1 receptor. Biochem J. 2017;474(5):771-780.[61] Lu Q, Liu Y, Wang Y, et al. Rapamycin efficiently promotes cardiac differentiation of mouse embryonic stem cells. Biosci Rep. 2017;37(3): BSR20160552.[62] Kochegarov A, Lemanski LF. New Trends in Heart Regeneration: A Review. J Stem Cells Regen Med. 2016; 12(2):61-68.[63] Peymani M, Ghaedi K, Irani S, et al. Peroxisome Proliferator-Activated Receptor γ Activity is Required for Appropriate Cardiomyocyte Differentiation. Cell J. 2016; 18(2):221-228.[64] Noseda M, Abreu-Paiva M, Schneider MD. The Quest for the Adult Cardiac Stem Cell. Circ J. 2015;79(7):1422-1430. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Li Cai, Zhao Ting, Tan Ge, Zheng Yulin, Zhang Ruonan, Wu Yan, Tang Junming. Platelet-derived growth factor-BB promotes proliferation, differentiation and migration of skeletal muscle myoblast [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1050-1055. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||