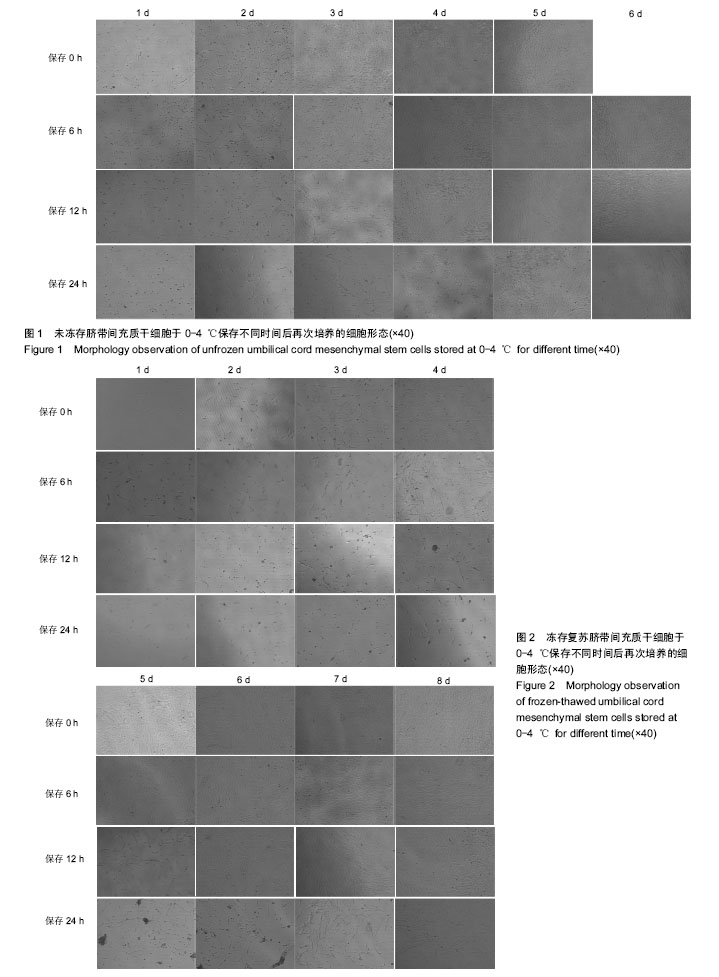
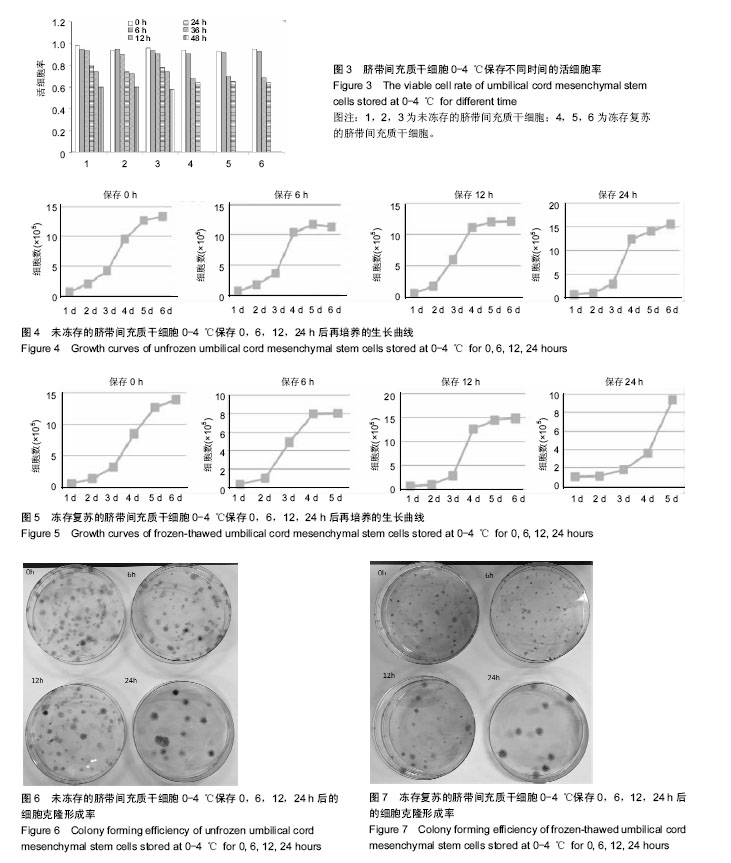
| [1] Semenov OV, Koestenbauer S, Riegel M, et al. Multipotent mesenchymal stem cells from human placenta: critical parameters for isolation and maintenance of stemness after isolation. Am J Obstet Gynecol. 2010;202(2):193.e1-193.e13.[2] Winkler S, Borkham-Kamphorst E, Stock P, et al. Human mesenchymal stem cells towards non-alcoholic steatohepatitis in an immunodeficient mouse model. Exp Cell Res.2014;326(2):230-239.[3] Karahuseyinoglu S, Cinar O, Kilic E, et al. Biology of stem cells in human umbilical cord stroma: in situ and in vitro surveys. Stem Cells. 2007;25(2):319-331.[4] Teixeira FG, Carvalho MM, Sousa N, et al. Mesenchymal stem cells secretome: a new paradigm for central nervous system regeneration. Cell Mol Life Sci. 2013;70(20): 3871-3882.[5] 朱婷,李爱斌,曹景云,等.胎盘间充质干细胞的应用研究[J].中国组织工程研究,2015,19(32):5236-5242.[6] Moodley Y, Atienza D, Manuelpillai U, et al. Human umbilical cord mesenchymal stem cells reduce fibrosis of bleomycin-induced lung injury. Am J Pathol. 2009;175(1): 303-313.[7] 李清,唐笛,时玉舫,等.间充质干细胞与免疫的相互作用:从基础研究到临床应用[J].生命科学,2016,28(8):933-940.[8] Teixeira FG, Carvalho MM, Neves-Carvalho A, et al. Secretome of mesenchymal progenitors from the umbilical cord acts as modulator of neural/glial proliferation and differentiation. Stem Cell Rev. 2015;11(2):288-297.[9] Liu WH, Song FQ, Ren LN, et al. The multiple functional roles of mesenchymal stem cells in participating in treating liver diseases. J Cell Mol Med. 2015;19(3):511-520.[10] Ribeiro A, Laranjeira P, Mendes S, et al. Mesenchymal stem cells from umbilical cord matrix, adipose tissue and bone marrow exhibit different capability to suppress peripheral blood B, natural killer and T cells. Stem Cell Res Ther. 2013; 4(5):125.[11] 袁宝珠,张可华,纳涛. 建立间充质干细胞制剂质量控制评价体系[C].2013年中国药学大会,2013.[12] 中华人民共和国国家卫生和计划生育委员会.《干细胞制剂质量控制及临床前研究指导原则( 试行) 》( 征求意见稿) 解读[J].转化医学杂志,2013,2(6):321-322.[13] 关于征求干细胞临床研究管理办法(试行)意见的函[J].中国组织工程研究,2015,19(10):1640.[14] 中华人民共和国国家卫生和计划生育委员会.《干细胞临床试验研究管理办法( 试行) 》(征求意见稿) 解读[J].转化医学杂志, 2013,2(6):322-323.[15] 周琪. 中国及中国科学院干细胞与再生医学研究概述[J].生命科学,2016,28(8):833-838.[16] 姜天娇,孙金海. 国外干细胞产品监管现状及对我国的启示[J].中国社会医学杂志,2016,33(2):117-120.[17] Daley GQ, Hyun I, Apperley JF, et al. Setting Global Standards for Stem Cell Research and Clinical Translation: The 2016 ISSCR Guidelines. Stem Cell Reports. 2016;6(6): 787-797.[18] Ramdasi S, Sarang S, Viswanathan C. Potential of Mesenchymal Stem Cell based application in Cancer. Int J Hematol Oncol Stem Cell Res. 2015;9(2):95-103.[19] Bárcia RN, Santos JM, Teixeira M, et al. Umbilical cord tissue-derived mesenchymal stromal cells maintain immunomodulatory and angiogenic potencies after cryopreservation and subsequent thawing. Cytotherapy. 2017;19(3):360-370.[20] Li Q, Yao D, Ma J, et al. Transplantation of MSCs in combination with netrin-1 improves neoangiogenesis in a rat model of hind limb ischemia. J Surg Res. 2011;166(1): 162-169.[21] Nejad-Moghaddam A, Ajdary S, Tahmasbpour E, et al. Immunomodulatory Properties of Mesenchymal Stem Cells Can Mitigate Oxidative Stress and Inflammation Process in Human Mustard Lung. Biochem Genet. 2016;54(6):769-783.[22] Tyndall A. Mesenchymal stem cell treatments in rheumatology: a glass half full. Nat Rev Rheumatol. 2014;10(2):117-124.[23] Zhou HX, Liu ZG, Liu XJ,et al.Umbilical cord-derived mesenchymal stem cell transplantation combined with hyperbaric oxygen treatment for repair of traumatic brain injury.Neural Regen Res. 2016;11(1):107-113.[24] Wang D, Huang S, Yuan X, et al. The regulation of the Treg/Th17 balance by mesenchymal stem cells in human systemic lupus erythematosus. Cell Mol Immunol. 2017; 14(5):423-431.[25] Hajivalili M, Pourgholi F, Kafil HS, et al. Mesenchymal Stem Cells in the Treatment of Amyotrophic Lateral Sclerosis. Curr Stem Cell Res Ther. 2016;11(1):41-50.[26] Paulini J, Higuti E, Bastos RM, et al. Mesenchymal Stem Cells as Therapeutic Candidates for Halting the Progression of Diabetic Nephropathy. Stem Cells Int. 2016;2016:9521629.[27] 陈俊宇,王建忠.成骨细胞、破骨细胞与酒精性股骨头坏死的关联性:预防与靶向治疗的新思路[J].中国组织工程研究,2016, 20(46):6963-6969.[28] 张香梅,田中良哉,单保恩. 间充质干细胞应用于类风湿关节炎组织修复的研究进展[J].中国全科医学,2016,19(32): 4014-4018.[29] 顾问,郭雪君,徐卫国.间充质干细胞治疗呼吸系统疾病的研究进展[J].中华肺部疾病杂志,2016,9(1):82-86.[30] Chang Z, Mao G, Sun L, et al. Cell therapy for cerebral hemorrhage: Five year follow-up report. Exp Ther Med. 2016;12(6):3535-3540.[31] Heidemann R, Mered M, Wang DQ, et al. A new seed-train expansion method for recombinant mammalian cell lines. Cytotechnology. 2002;38(1-3):99-108. |