Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (12): 1944-1949.doi: 10.3969/j.issn.2095-4344.0212
Previous Articles Next Articles
Research advances in adiponectin preventing osteoporosis by bone metabolism regulation
Huang Lei-tao1, Wu Xia2, Lai Qi1, Chen Gui-ping1, Liu Yuan1, Zhang Bin1, Dai Min1
- 1Department of Orthopedics, Research Center for Artificial Joint Engineering Technology, 2Department of Cardiothoracic Surgery, the First Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China
-
Received:2018-01-02Online:2018-04-28Published:2018-04-28 -
Contact:Corresponding author: Dai Min, Master, Chief physician, Professor, Department of Orthopedics, Research Center for Artificial Joint Engineering Technology, the First Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China Corresponding author: Zhang Bin, Department of Orthopedics, Research Center for Artificial Joint Engineering Technology, the First Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China -
About author:Huang Lei-tao, Master candidate, Department of Orthopedics, Research Center for Artificial Joint Engineering Technology, the First Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China Wu Xia, Department of Cardiothoracic Surgery, the First Affiliated Hospital of Nanchang University, Nanchang 330006, Jiangxi Province, China Huang Lei-tao and Wu Xia contributed equally to this work. -
Supported by:the National Natural Science Foundation of China, No. 700202002; Jiangxi Provincial Department of Science and Technology-Leading Talent Training Program (555 Engineering), No. 1210025001/5201; the Postgraduate Innovation Foundation of Nanchang University, No. cx2016390; the Postgraduate Innovation Foundation of Jiangxi Province, No. YC2016-S107
CLC Number:
Cite this article
Huang Lei-tao1, Wu Xia2, Lai Qi1, Chen Gui-ping1, Liu Yuan1, Zhang Bin1, Dai Min1. Research advances in adiponectin preventing osteoporosis by bone metabolism regulation[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(12): 1944-1949.
share this article
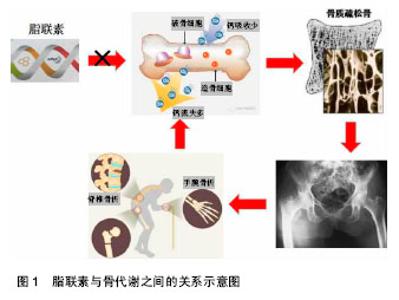
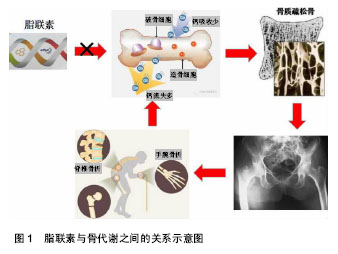
2.1 脂联素及其受体概述 脂联素(adiponectin,APN)又名 Acrp30,adipoQ,ApM1,是目前为止发现的与肥胖呈负相关的脂肪细胞因子之一。在动脉粥样硬化中,脂联素被认为是一种保护因子,而在一些自身免疫性疾病如类风湿性及风湿性关节炎中,则被认为具有前炎性因子的作用[8]。脂联素主要是由成熟的脂肪细胞合成和分泌,具有一定的多功能激素蛋白[9]。人脂联素分子全长包含244个氨基酸,一级序列结构包括4个结构 域[10]:氨基末端的信号序列、一小段非螺旋区、一段22个氨基酸组成的胶原重复序列以及羧基末端球形功能区[11]。基因位于3q27,片段长约17 kb,包含3个外显子和2个内含子组成[12]。脂联素是血浆中最丰富的特异性脂肪细胞因子,其表达量一般是常见激素的3倍,而且在女性中偏高,但研究证实其与昼夜节律及年龄变化无明显关系[13]。血清中脂联素的质量浓度为5-30 μg/L,占总血清蛋白的0.01%以上,脂联素是一个在肥胖患者体内水平下调的脂肪因子[14]。 脂联素以不同形式存在,其存在形式主要因胶原结构域的不同而被区分,形成低、中、高相对分子质量的3种主要聚合物形式[15-17],其中脂联素在体内循环中主要以高分子量形式存在[18-19]。体内脂联素水平受多种因素的影响,研究表明女性血浆中脂联素的水平明显高于男性,且在进入青春期后更加显著,这可能与男性雄激素对脂联素分泌有抑制作用有关[20]。 脂联素存在2种受体,最早是由Yamauchi等[21]在2003年提出来的,他们在用Northern Blot测定法克隆出脂联素受体基因的cDNA全长时,发现其存在两种异构体,后来命名为脂联素受体1,2,其中脂联素受体1分布广泛,主要存在于骨骼肌中;而脂联素受体2 在肝脏中分布最多[22]。脂联素的生物学效应主要是通过脂联素受体1,2来介导的,主要通过激活腺苷酸激酶、促分裂素原活化蛋白激酶和过氧化物酶体增生物激活受体α 配体,最终来促进骨骼肌细胞的脂肪酸氧化和糖吸收,降低体内血脂浓度及葡萄糖水平,是人体的脂质代谢和血糖稳态的调控网络中的重要调节因子[23]。不同受体与不同形式的根据脂联素受体的类型与其结合的亲和力也有所不同,脂联素受体1,2与脂联素单体亲和性较高,不同型式脂联素的生物学作用可能有较大差别[24-25]。许多研究表明,脂联素与胰岛素抵抗、动脉粥样硬化及抗炎作用密切相关[26-30]。在体外动物模型中,脂联素具有改善肥胖相关性疾病的作用,如由肥胖引起人体内胰岛素抵抗、葡萄糖耐量异常、2 型糖尿病等[31-32]。 2.2 骨组织的生理代谢 骨组织代谢是通过成骨细胞和破骨细胞参与的骨形成与骨吸收来实现的,其代谢活动是一个动态平衡过程[1,33]。在人的生长期,骨形成大于骨吸收,骨量呈线性增长,表现为骨皮质增厚,骨松质更密集,这一过程称为骨构建或骨塑形。在成人期,骨生长停止,但骨的形成和吸收仍在继续,处于一种平衡状态,称为骨重建。骨重建开始于骨吸收,随后是骨形成。骨的吸收与形成连续进行,最终使骨能不断地自我修复和适应新的应力要求[11]。正常成熟骨的代谢主要以骨重建方式进行,在骨代谢调节激素和局部细胞因子的协调作用下,骨组织通过体内骨形成与骨吸收之间相互影响及制约,使体内骨转换达到一个动态的平衡状态,以维持人体正常骨的生理代谢效应[34]。骨组织在整个生命活动过程中都具有新陈代谢的活性,骨基质、骨矿物质以及骨代谢生化标志物的变化来反映。其中骨密度是反映骨质疏松程度,预测骨折危险性的重要依据。骨密度是定量评估骨骼强度的一个重要指标,通常使用T值判断骨密度是否正常,它会随着成年后年龄的增长和体内雌激素水平的降低而逐年下降,尤其在女性中表现的更为明显[1]。 2.3 脂联素与成骨、破骨之间的关系 查阅近年来国内外文献发现,脂联素对骨密度具有一定调控作用。通过绝经前后的女性体内脂联素和骨密度检测发现两者之间呈现负相关性,同时与体内体脂含量和非脂肪含量存在负相关,认为脂联素是骨密度的独立预测因子。同样研究表明,脂联素影响骨的代谢,可通过3个不同的途 径[1,35]:①自分泌或旁分泌的途径;②内分泌途径;③通过影响胰岛素信号通路产生作用。脂联素对骨代谢的作用既可以影响成骨细胞的活性,又可以影响破骨细胞的活性,是上述3个途径综合作用的结果[36](图1)。有学者研究发现,在破骨细胞诱导实验中,以不同浓度的脂联素作用细胞,脂联素对破骨细胞的作用呈浓度依赖关系[37],并且其可能通过抑制破骨细胞中生成的关键转录因子——核转录因子κB来起作用的,提示脂联素可能影响破骨细胞的活性来调节骨代谢。有研究通过研究发现,脂联素和瘦素在体内的水平都能通过影响成骨及破骨的活性来调控骨代谢[13],使人体的骨形成和骨吸收处"
| [1] 王磊,李兴.脂联素影响骨代谢致骨质疏松症的研究进展[J].现代药物与临床,2011,26(6):426-429.[2] 鲍晓雪,王娜,李玉坤.肥胖与骨质疏松症关系的研究进展[J].中华临床医师杂志,2015,9(14):2749-53.[3] Zhang Q, Riddle RC, Clemens TL. Bone and the regulation of global energy balance. J Int Med. 2015;277(6):681-689.[4] Gonnelli S, Caffarelli C, Nuti R. Obesity and fracture risk. Clin Cases Miner Bone Metab. 2014;11(1):9-14.[5] Fernandes TAP, Gonçalves LML, Brito JAA. Relationships between bone turnover and energy metabolism. J Diabetes Res. 2017;2017:9021314.[6] Chen T, Wu YW, Lu H, et al. Adiponectin enhances osteogenic differentiation in human adipose-derived stem cells by activating the APPL1-AMPK signaling pathway. Biochem Biophys Res Commun. 2015;461(2):237-242.[7] Ding WX, Dong YB, Ding N, et al. Adiponectin protects rat heart from left ventricular remodeling induced by chronic intermittent hypoxia via inhibition of TGF-β/smad2/3 pathway. J Thorac Dis. 2014;6(9):1278-1284.[8] Francin PJ, Abot A, Guillaume C, et al. Association between adiponectin and cartilage degrandation in human osteoarthritis. Osteoarthritis Cartilage. 2014;22(3):519-526.[9] 方曦,陈琳洁,谢长好,等.脂联素与脊柱关节炎的研究进展[J].中华全科医学,2016,14 (10):1732-1735.[10] Mendez-Sanchez N, Chavez-Tapia NC, Zamora-Valdes D, et al. Adiponectin, structure, function and pathophysiological implications in non-alcoholic fatty liver disease. Mini Rev Med Chem. 2006;6(6):651-656.[11] 陈栖栖,刘钢.脂联素在类风湿关节炎中的作用和进展[J].实用医院临床杂志,2016,13(2):174-177.[12] Wang QP, Li XP, Wang M, et al. Adiponectin exerts its negative effect on bone metabolism via OPG/RANKL pathway: an in vivo study. Endocrine. 2014. DOI: 10.1007 /s12020-014-0216-z.[13] 王峰,郑陆.脂联素、瘦素与运动及骨代谢的关系[J].中国骨质疏松杂志,2016,22(2):221-227.[14] Elda L. Pacheco-Pantoja, Victoria J, et al. Adiponectin receptors are present in RANK-L-induced multinucleated osteoclast-like cells. J Recept Signal Transduct Res. 2013. DOI: 10.3109/10799893.2013.828070.[15] Kanazawa I. Adiponectin in Metabolic Bone Disease. Curr Med Chem. 2012;19:5481-5492.[16] Pajvani UB, Du X, Combs TP, et al. Structure function studies of the adiponectin-secreted hormone Acrp30/adiponectin. Biol Chem. 2003;278(11):9073-9085.[17] Klein-Wieringa IR, Andersen SN, Herb-van Toorn L, et al. Are baseline high molecular weight adiponectin levels associated with radiographic progression in rheumatoid arthritis and osteoarthritis. J Rheumatol. 2014;41(5):853-857.[18] Pajvani U B, Hankins M, Combs T P, et al. Complex distribution, not absolute amount of adiponectin, correlates with thizolidinedione-mediated mprovement in insulin sensitivity. J Biol Chem. 2004;279(13):12152-12162. [19] Kalisz M, Baranowska B, Wolinska-Witort E, et al. Total and high molecular weight adiponectin levels and risk of cardiovascular disease in individuals with high blood glucose levels. Atherosclerosis. 2013;229(1):222-227.[20] Huang ZH, Manickam B, Ryvkin V, et al. PCOS is associated with increased CD11c expression and crown-like structures in adipose tissue and increased central abdominal fat depots independent of obesity. J Clin Endocrinol Metab. 2013;98(1): E17-E24.[21] Yamauchi T, Kamon J, Ito Y, et al. Cloning of adiponectin receptors that mediate antidiabetic metabolic effects. Nature. 2003;423(6941):762-769.[22] Kharroubi I, Rasschaert J, Eizirik DL, et al. Expression of adiponectin receptors in pancreatic beta cells. Biochem Biophys Res Commun. 2003;312(4):1118-1122.[23] Zhang ZL, He JW, Qin YJ, et al. Association between SNP and haplotypes in PPARGC1 and adiponectin genes and bone mineral density in Chinese nuclear families. Acta Pharmacol Sin. 2007; 28(2):287-295.[24] Hangen F, Drevon CA. Activation of nuclear factor-kappaB by high molecular weight and globular adiponectin. Endocrinology. 2007; 148(11):5478-5486.[25] Neumeier M, Weigert J, Schfiffier A, et al. Different effects of adiponectin isoforms in human monocytic cells. J Leukoc Biol. 2006;79(4):803-808.[26] Sonmez A, Dogru T, Yilmaz MI, et al. Adiponectin and insulin resistance in young and healthy smokers. Endocr J. 2006;53(6): 729-734.[27] Koenig W, Khuseyinova N, Baumert J, et al. Serum concentrations of Adiponectin and risk of type 2diabetes mellitus and coronary heart disease in apparently healthy middle-aged men: results from the 18-year follow-up of a large cohort from southern Germany. J Am Coll Cardiol. 2006;48(7):1369-1377.[28] Schulze MB, Shai I, Rimm EB, et al. Adiponectin and future coronary heart disease events among men with type 2 diabetes. Diabetes. 2005;54(2):534-539.[29] Tajtakova M, Petrasova D, Petrovicova J, et al. Adiponectin as a biomarker of clinical manifestation of metabolic syndrome. Endocr Regul. 2006;40(1):15-19.[30] Kadowaki T, Yamauchi T, Kubota N, et al. Adiponectin and AdipoQnectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J Clin Invest. 2006;116(7):1784-1792.[31] Okada-Iwabu M, Yamauchi T, Iwabu M, et al. A small-molecule AdipoR agonist for type 2 diabetes and short life in obesity. Nature. 2013;503:493-499.[32] Okada-Iwabu M, Iwabu M, Ueki K, et al. Perspective of small-molecule adipor agonist for type 2 diabetes and short life in obesity. Diabetes Metab J. 2015;39:363-372.[33] Dimitri P, Rosen C. The Central Nervous System and Bone Metabolism: An Evolving Story. Calcif Tissue Int. 2017;100(5): 476-485.[34] Xu C, Ochi H, Fukuda T, et al. Circadian clock regulates bone resorption in mice. J Bone Miner Res. 2016;31(7):1344-1355.[35] Cao Y, Gomes SA, Rangel EB, et al. S-nitrosoglutathione reductase-dependent PPARγ denitrosylation participates in MSC-derived adipogenesis and osteogenesis. J Clin Invest. 2015;125(4):1679-1691.[36] Martin PJ, Haren N,Ghali O,et al. Adipogenic RNAs are transferred in osteoblasts via bone marrow adipocytes-derived extracellular vesicles (EVs). BMC Cell Biol. 2015;16(1):1-10.[37] Su CM, Lee WL, Hsu CJ, et al. Adiponectin Induces Oncostatin M Expression in Osteoblasts through the PI3K/Akt Signaling Pathway. Adiponectin Induces Oncostatin M Expression in Osteoblasts through the PI3K/Akt Signaling Pathway. Int J Mol Sci. 2016;17:29.[38] 邹晓玲,罗湘杭.脂联素研究进展[J].实用预防医学, 2010,17(11): 2338-2341.[39] Scherer PE, Williams S, Fogliano M, et al. A novel serum protein similar to C1q, produced exclusively in adipocytes. J Biol Chem. 1995;270:26746-26749.[40] Huang CY, Lee CY, Chen MY, et al. Adiponectin increases BMP-2 expression in osteoblasts via AdipoR receptor signaling pathway. J Cell Physiol. 2010;224(2):475-483.[41] Yamaguchi N, Kukita T, Li YJ, et al. Adiponectin inhibits osteoclast formation stimulated by lipopolysaccharide from Actinobacillus actinomycetemcomitans. FEMS Immunol Med Microbiol. 2007;49: 28-34.[42] Berner HS, Lygstadaas Sp, Spahr A, et al. Adiponeetin and its receptors are expressed in bone-forming cells. J Bone. 2004; 35(4):842-849.[43] 袁凌青,罗湘杭,谢辉,等.脂联素对人成骨细胞增殖作用的研究[J].中国骨质疏松杂志,2006,12(2):135-137.[44] Oshima K, Nampei A, Matsuda M, et al.Adiponectin increases bone mass by suppressing osteoclast and activating osteoblast. Biochem Biophys Res Commun. 2005;331(2):520-526.[45] Luo XH, Guo LJ, Yuan LQ, et al. Adiponectin stimulates human osteoblasts proliferation and differentiation via the MAPK signaling pathway. Exp Cell Res. 2005;309(1):99-109.[46] Luo XH, Guo LJ, Xie H, et al. Adiponectin stimulates RANKL and inhibits OPG expression in human osteoblasts through the MAPK signaling pathway. J Bone Miner Res. 2006;21(10):1648-1656.[47] Wang QP, Li XP, Wang, M, et al. Adiponectin exerts its negative effect on bone metabolism via OPG/RANKL pathway: an in vivo study. Endocrine. 2014;47(3):845-853.[48] Lee HW, Kim SY, Kim AY, et al. Adiponectin Stimulates Osteoblast Differentiation Through Induction of COX2 in Mesenchymal Progenitor Cells. Stem Cells. 2009,27(9):2254-2262.[49] Berner HS, Lyngstadaas SP, Spahr A et al. Adiponectin and its receptors are expressed in bone-forming cells. Bone. 2004;35: 842-849.[50] Shinoda Y, Yamaguchi M, Ogata N, et al. Regulation of bone formation by adiponectin through autocrine/paracrine and endocrine pathways. Cell Biochem. 2006;99(1):196-208.[51] Jurimae J, Jurimae T, Plasma adiponectin concentration in healthy pre- and postmenopausal women: relationship with body composition, bone mineral, and metabolic variables. Am J Physiol Endocrinol Metab. 2007;293(1):42-47.[52] Lewiecki EM. RANK ligand inhibition with denosumab for the management of osteoporosis. Expert Opin Biol Ther. 2006;6(10): 1041-1050.[53] Schett G, Hayer S, Zmerina J, et al. Mechanisms of Disease: the link between RANKL and arthritic bone disease. Nat Clin Pract Pheumatol. 2005;1(1):47-54.[54] Simonet WS, Laeey DL, Dunstan CR, et al. Osteporotegerin: a novel secreted pretein involved in the regulation of bone density. Cell. 1997;89(2):309-319.[55] P. Szulc, P.D. Delmas, Biochemical markers of bone turnover: potential use in the investigation and management of postmenopausal osteoporosis. Osteoporos Int. 2008;19(12): 1683-1704.[56] Yamauchi T; Kadowaki T. Physiological and pathophysiological roles of adiponectin and adiponectin receptors in the integrated regulation of metabolic and cardiovascular diseases. Int J Obes. 2008;7;S13-S18.[57] Hong X, Arguelles LM, Tsai HJ, et al. Plasma adipokines, bone mass, and hip geometry in rural Chinese adolescents. J Clin Endocrinol Metab. 2010;95:1644-1652.[58] Kiechl S, Werner P, Knoflach M, et al. The osteoprotegerin/ RANK/RANK-L system: a bone key to vascular disease. Expert Rev Cardiovasc Ther. 2006;4(6):801-811.[59] Kostenuik PJ. Osteoprotegerin and RANKL regulate bone resorption, density, geometry and strength. Curr Opin Pharmacol. 2005;5(6):618-625.[60] Krumbholz G, Junker S, Meier FMP, et al. Response of human rheumatoid arthritis osteoblasts and osteoclasts to adiponectin. Clin Exp Rheumatol. 2017;35(3):406-414.[61] Zhang L, Meng S, Tu Q, et al. Adiponectin ameliorates experimental periodontitis in diet-induced obesity mice. PLoS One. 2014;9(5):e97824. [62] 张平平,向川.骨髓间充质干细胞治疗骨关节炎:可能与未来[J].中国组织工程研究,2014,18(6):968-973.[63] 贾懿劼,田京.干细胞治疗骨质疏松症的可能与可行[J].中国组织工程研究,2012,16(1):148-152. |
| [1] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [2] | Zeng Yanhua, Hao Yanlei. In vitro culture and purification of Schwann cells: a systematic review [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1135-1141. |
| [3] | Xu Dongzi, Zhang Ting, Ouyang Zhaolian. The global competitive situation of cardiac tissue engineering based on patent analysis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(5): 807-812. |
| [4] | Wu Zijian, Hu Zhaoduan, Xie Youqiong, Wang Feng, Li Jia, Li Bocun, Cai Guowei, Peng Rui. Three-dimensional printing technology and bone tissue engineering research: literature metrology and visual analysis of research hotspots [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 564-569. |
| [5] | Chang Wenliao, Zhao Jie, Sun Xiaoliang, Wang Kun, Wu Guofeng, Zhou Jian, Li Shuxiang, Sun Han. Material selection, theoretical design and biomimetic function of artificial periosteum [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 600-606. |
| [6] | Liu Fei, Cui Yutao, Liu He. Advantages and problems of local antibiotic delivery system in the treatment of osteomyelitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 614-620. |
| [7] | Li Xiaozhuang, Duan Hao, Wang Weizhou, Tang Zhihong, Wang Yanghao, He Fei. Application of bone tissue engineering materials in the treatment of bone defect diseases in vivo [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 626-631. |
| [8] | Zhang Zhenkun, Li Zhe, Li Ya, Wang Yingying, Wang Yaping, Zhou Xinkui, Ma Shanshan, Guan Fangxia. Application of alginate based hydrogels/dressings in wound healing: sustained, dynamic and sequential release [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 638-643. |
| [9] | Chen Jiana, Qiu Yanling, Nie Minhai, Liu Xuqian. Tissue engineering scaffolds in repairing oral and maxillofacial soft tissue defects [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(4): 644-650. |
| [10] | Xing Hao, Zhang Yonghong, Wang Dong. Advantages and disadvantages of repairing large-segment bone defect [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(3): 426-430. |
| [11] | Gao Shan, Huang Dongjing, Hong Haiman, Jia Jingqiao, Meng Fei. Comparison on the curative effect of human placenta-derived mesenchymal stem cells and induced islet-like cells in gestational diabetes mellitus rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 3981-3987. |
| [12] | Chen Siqi, Xian Debin, Xu Rongsheng, Qin Zhongjie, Zhang Lei, Xia Delin. Effects of bone marrow mesenchymal stem cells and human umbilical vein endothelial cells combined with hydroxyapatite-tricalcium phosphate scaffolds on early angiogenesis in skull defect repair in rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3458-3465. |
| [13] | Wang Hao, Chen Mingxue, Li Junkang, Luo Xujiang, Peng Liqing, Li Huo, Huang Bo, Tian Guangzhao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Guo Quanyi, Lu Xiaobo. Decellularized porcine skin matrix for tissue-engineered meniscus scaffold [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3473-3478. |
| [14] | Mo Jianling, He Shaoru, Feng Bowen, Jian Minqiao, Zhang Xiaohui, Liu Caisheng, Liang Yijing, Liu Yumei, Chen Liang, Zhou Haiyu, Liu Yanhui. Forming prevascularized cell sheets and the expression of angiogenesis-related factors [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3479-3486. |
| [15] | Liu Chang, Li Datong, Liu Yuan, Kong Lingbo, Guo Rui, Yang Lixue, Hao Dingjun, He Baorong. Poor efficacy after vertebral augmentation surgery of acute symptomatic thoracolumbar osteoporotic compression fracture: relationship with bone cement, bone mineral density, and adjacent fractures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(22): 3510-3516. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||