Chinese Journal of Tissue Engineering Research ›› 2018, Vol. 22 ›› Issue (4): 606-611.doi: 10.3969/j.issn.2095-4344.0098
Previous Articles Next Articles
Tissue-engineered heart: electrophysiological properties and long-term safety
Tong Cai-ling1, 2, 3, Li Ming-hui2, 3, Qi Zhong-quan2, 3
- 1School of Life Science, 2Organ Transplantation Institute, Xiamen University, Xiamen 361102, Fujian Province, China; 3the Key Laboratory of Organ and Tissue Regeneration of Fujian Province, Xiamen 361102, Fujian Province, China
-
Received:2017-09-17Online:2018-02-08Published:2018-02-08 -
Contact:Qi Zhong-quan, M.D., Professor, Organ Transplantation Institute, Xiamen University, Xiamen 361102, Fujian Province, China; the Key Laboratory of Organ and Tissue Regeneration of Fujian Province, Xiamen 361102, Fujian Province, China -
About author:Tong Cai-ling, M.D., School of Life Science, Organ Transplantation Institute, Xiamen University, Xiamen 361102, Fujian Province, China; the Key Laboratory of Organ and Tissue Regeneration of Fujian Province, Xiamen 361102, Fujian Province, China Li Ming-hui, Studying For master’s degree, Organ Transplantation Institute, Xiamen University, Xiamen 361102, Fujian Province, China; the Key Laboratory of Organ and Tissue Regeneration of Fujian Province, Xiamen 361102, Fujian Province, China Tong Cai-ling and Li Ming-hui contributed equally to this work.
CLC Number:
Cite this article
Tong Cai-ling1, 2, 3, Li Ming-hui2, 3, Qi Zhong-quan2, 3. Tissue-engineered heart: electrophysiological properties and long-term safety[J]. Chinese Journal of Tissue Engineering Research, 2018, 22(4): 606-611.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 心脏支架材料 良好的支架材料是构建组织工程心脏的基础。一个理想的组织工程心脏支架材料要具备以下特征:①要具有良好的生物相容性。生物材料最终应用于人体内,具有免疫反应的组织工程材料会被受体排斥,造成移植失败,给患者带来极大痛苦。②要具有合适的表面结构,良好的生物材料要有较好地细胞亲和性,这样种子细胞才能通过识别材料表面位点定位在细胞表面,实现在支架材料内的进一步增殖、分化、形成器官。材料表面要光滑,具有抗凝血作用,可以保证器官的长时间存活。另外支架材料要具有合适的孔径大小,可以增大细胞贴附面积,可供细胞进行物质交换及废物的排出。③具有一定的力学强度和柔韧性。力学强度可以使材料在细胞培养接种过程中保持完整的形态结构;柔韧性可以使支架材料和机体有机的贴合在一起。 近年来,组织工程中作为细胞支架的生物材料选择主要趋向于天然高分子材料、合成高分子材料和去细胞心脏支架材料3种(表1)。 2.2 去细胞支架材料的制备及评估 去细胞是指用去污剂将组织或器官中的细胞去除,获得一个保留完整细胞外基质的组织或器官支架,并维持其原有的三维空间结构、生化组成和生物活性。要得到良好的去细胞心脏支架,选择一个能够将细胞去除完全,且维持原有组织或器官完整的支架系统和机械强度的去污剂至关重要。Ott等[6]将大鼠心脏置于改良的Langendorff装置上,通过冠状动脉灌注来对比SDS、PEG和Triton-X100三种化学去垢剂的脱细胞程度。结果显示,SDS的洗脱效果明显优于PEG和Triton-X100,并且洗脱时间短,效率高。目前"

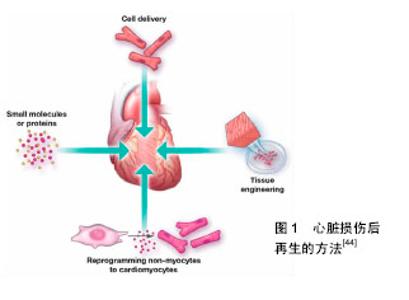
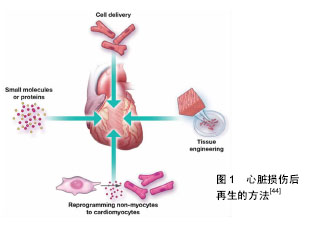
SDS是心脏组织脱细胞最常用的灌注液。除此化学方法外,物理洗脱法和酶消化法也可达到去细胞的目的。前者如声波降解法、搅拌法等通常与化学洗脱法一起进行,达到去除破碎细胞成分的效果[22]。但由于心脏的脉管系统复杂且脆弱,故通常不会采用此法以免破坏支架的完整性。酶消化法主要是利用胰酶、胶原酶和核酸酶等的酶解作用破坏细胞膜蛋白进而脱去细胞。酶具有很强的特异性,所以不同的细胞和材料组成,在消化过程中酶的选择和作用时间也要做出相应改变,选择正确的酶处理方可体现其高效性。此外,酶的选择和处理不当还会导致胶原蛋白和纤维组成被破坏,得到不完整的心脏支架。 Rieder等[23]通过完全去除细胞的效率及再细胞化的潜力两方面比较了3种猪心脏瓣膜的脱细胞方法,发现使用0.25%叔辛基苯基聚氧乙烯/脱氧胆酸钠和核酸酶消化可得到一个再细胞化效果良好的支架。Hussein等[24]总结并分析了多种常用的物理、化学和生物洗脱液的作用原理及优缺点,可以为细胞洗脱液的初步选择提供参考。2015年,Papalamprou等[25]在其研究中通过抗原清除的方法构建细胞外基质支架,同时采用亲水性和亲脂性的异种抗原以分段的方式除去心脏组织中心肌细胞和非心肌细胞的抗原成分和细胞组成,也可成功保持原有心脏组织的结构、生化特性和力学特性,且后续实验结果表明,将小鼠或人的间充质干细胞接种至以抗原清除法构建的心脏支架进行再细胞化,细胞迁移较为深入,显示其具有良好的生物兼容性。而通过变性去污剂SDS洗涤得到的心脏支架再细胞化后较多细胞分布在外周区域,体内实验也显示移植后伴有长期的慢性炎症反应[26]。去细胞方法虽多,但还需要在实验中不断摸索,积累经验,找到最简单有效的去除方法和试剂作用的最佳浓度和时间,方可达到理想的洗脱效果。 理想的心脏去细胞支架是无毒性、无致病性、无免疫原性的。去细胞后心脏支架内细胞残留情况和心脏支架的完整性及各项指标也是组织工程心脏成败的关键。去细胞支架材料的成分主要包括胶原蛋白、肌原蛋白、纤连蛋白和层粘连蛋白等[27],可用染色分析其细胞残留情况,扫描电子显微镜观察其形态结构,用Western blot或质谱分析等技术对各成分做定量分析等,可通过各个方向的抗拉伸实验评估整个支架的弹性及强度,同时灌注有色染料可清晰观察到心脏支架内的大血管及脉管系统是否完整。Ott等[6]用苏木精-伊红染色和免疫荧光染色等评估脱细胞程度,发现经SDS洗脱12 h又经Triton-X100和洗涤液灌注后心脏支架内没有检测出细胞存在。通过组织学方法如苏木精-伊红染色来判断细胞是否清除完全是目前最常用的手段之一,还可以通过扫描电子显微镜观察脱细胞程度及残留组分。细胞未洗脱彻底会增强心脏支架的免疫原性,与后期种植的异种细胞不相容,移植到体内后会引起受体的免疫排斥反应,使组织工程心脏失去意义。同时也要注意,心脏组织脱细胞完全后需用洗涤液灌流冲洗,以防止洗脱液对支架结构的毒性作用。且避免洗脱液在支架内滞留时间过长,持续消化过度,破坏其纤维和胶原组成,降低支架机械强度,增大移植手术难度。 近年来,人们在多种组织和器官中对去细胞支架材料也进行了系列研究,并评估了去细胞支架作为3维支架的可能性[28]。在软骨组织工程领域中,细胞外基质来源支架被已证明是很好的软骨组织工程仿生支架,被应用于临床实践中。虽然接种细胞后支架会出现萎缩,但可通过配合交联剂使用或其他物理化学方法来改进,克服缺陷[29]。杨立信等[30]将去细胞瓣叶植入SD大鼠背部皮下,按照不同的包埋时间取出瓣叶进行一系列组化实验,以此来评价去细胞支架的免疫原性及炎性反应。后期也可通过器官移植等体内实验来分析支架免疫原性的高低。 2.3 种子细胞的选取 正常的心脏组织由多种细胞组成,心肌细胞是其主要的工作细胞。成熟的心肌细胞在体内增殖能力较低,心肌细胞的获取成为组织工程心脏构建的难题。 近年来有人指出,心肌细胞要不断搏动以维持心脏的泵血功能,这也许是阻碍心肌细胞同时收缩与分裂的原因之一,并认为心肌细胞的增殖可以通过调控某些基因的表达来实现[31]。 Matsuyama等[32]发现敲除了新生鼠心肌细胞连接蛋白43的表达基因,p38 MAPK通路的抑制剂和表达上调的成纤维细胞生长因子能够刺激心肌细胞的增殖,也有研究结果表明抑制或过表达体内某些基因可以促进心肌细胞增殖[33]。 另外有研究显示,心肌细胞干性的丧失是由于高氧环境中DNA损伤所致,低氧环境有利于心肌干细胞干性的维持[34]。但鲜有体外成功获得大量心肌细胞的报道。种子细胞作为组织工程心脏的关键,应同时满足易于获取、分化程度低、增殖能力强、免疫原性低等条件。所以,作为生命之源的干细胞成为组织工程器官构建中种子细胞的优秀候选者,主要因为干细胞具有以下特征: (1)具有自我更新能力。干细胞通过对称和非对称有丝分裂产生两种不同命运的细胞,一种细胞继续保持原有干细胞的功能,另一种为具有特定功能的细胞。子代干细胞可以不断的分裂下去并保持着干细胞的生物特性。这种分裂方式对维持机体器官的稳定性起到了非常重要的作用。 (2)具有多向分化性。胚胎干细胞具有全能分化性,能够分化为各谱系的细胞和特定组织干细胞,在特定环境中发育成完整的个体,也可用于临床细胞移植治疗疾病。但它存在着移植免疫排斥的限制和伦理学的困扰不能广泛获取和应用。成体干细胞虽只能在体外有限扩增,分化效率低,但这类细胞存在于宿主体内,可直接从患者自身获取,无移植排斥也无伦理道德的束缚。 (3)具有高度增殖能力和相对静止期。干细胞在体内可以连续分化增殖数代,也可以在较长时间内处于静止状态。当机体器官受损时,干细胞处于相对活跃的状态,可以增殖数代来修复受损的组织部位。 (4)干细胞的分化需要特定的微环境。在与不同的细胞接触或不同激素、试剂作用下,干细胞向着不同的方向进行特异性分化。 间充质干细胞作为干细胞家族的重要成员之一因其来源广泛,提取和体外培养技术较成熟,易于扩增和纯化等优势,被认为是最有希望应用于临床治疗的一类种子细胞。间充质干细胞来源于发育早期的中胚层和外胚层,可取自全身结缔组织和器官间质中,脐血、肌肉、脂肪、骨骼等都有间充质干细胞存在,以成人骨髓组织中含量最多。除来源丰富外,间充质干细胞还具有低免疫原性、无MHC限制性、体外扩增迅速且稳定、损伤去化作用及归巢功能、免疫调节和组织修复能力强等特点[35],是一类理想的种子细胞。 2011年Stubbs等[36]利用干细胞修复心脏。也有人将经过培养的间充质干细胞植入小鼠心肌梗死模型中,发现能有效减少心梗面积和心肌纤维化[37-38]。除骨髓来源的间充质干细胞外,脂肪组织中间充质干细胞的应用也日渐广泛。它在体内细胞基数大,且可通过微创等更安全的手段获得,具有比骨髓间充质干细胞更强的增殖能力和更稳定的增殖活性,比骨髓间充质干细胞有更大的优越性[39]。间充质干细胞可以沿着胶原纤维等胞外机制四处爬行[40],这也是它作为去细胞支架种子细胞的重要原因之一。 另有研究发现,用含有5-aza胞苷的培养基培养可成功在体外将间充质干细胞诱导成心肌细胞并检测到动作电位[41-43],证实了在调节因子的作用下,间充质干细胞可以成功分化为心肌细胞。近年来,心肌干细胞的研究[44],也为临床上终末期心脏病的治疗和受损心脏的修复带来了新的希望(图1)。"
| [1] Mozaffarian D, Benjamin EJ, Go AS, Arnett DK,et al. Heart disease and stroke statistics--2015 update: a report from the American Heart Association. Circulation, United States: 2015; 131(4): e29-322.[2] 姚震,陈林. 我国心血管疾病现状与展望[J]. 海南医学,2013,24(13): 1873-1876.[3] Cahn F, Kyriakides TR. Generation of an artificial skin construct containing a non-degradable fiber mesh: a potential transcutaneous interface.Biomed Mater. 2008;3(3): 34110.[4] Nemeno-Guanzon JG, Lee S, Berg JR, et al.Trends in Tissue Engineering for Blood Vessels. J Biomed Biotechnol. 2012;2012: 956345.[5] Ling C, Li Q, Brown ME, et al. Bioengineered vocal fold mucosa for voice restoration. Sci Transl Med.2015;7(314): 314ra187.[6] Ott HC, Matthiesen TS, Goh SK, et al. Perfusion-decellularized matrix: using nature’s platform to engineer a bioartificial heart. Nature Med.2008;14(2):213-221.[7] Uzarski JS, Xia Y, Belmonte JC, et al. New strategies in kidney regeneration and tissue engineering.Curr Opin Nephrol Hypertens. 2014;23(4): 399-405.[8] Palakkan AA, Hay DC, Anil Kumar PR, et al.Liver tissue engineering and cell sources: issues and challenges.Liver Int. 2013;33(5):666-676.[9] Kador KE, Montero RB, Venugopalan P, et al. Tissue engineering the retinal ganglion cell nerve fiber layer.Biomaterials. 2013; 34(17):4242-4250.[10] Atala A.Tissue engineering of human bladder. Br Med Bull. 2011; 97:81-104.[11] Petersen TH, Calle EA, Zhao L, et al.Tissue-engineered lungs for in vivo implantation.Science. 2010;329(5991):538-541.[12] Hu XJ, Dong NG, Liu JP, et al.Status on Heart Transplantation in China.Chin Med J (Engl). 2015 Dec 5;128(23):3238-3242.[13] Li Z, Zhang M.Chitosan-alginate as scaffolding material for cartilage tissue engineering.J Biomed Mater Res A. 2005;75(2):485-493.[14] Glowacki J, Mizuno S. Collagen scaffolds for tissue engineering. Biopolymers.2008; 89(5): 338-344.[15] Zhu J, Marchant RE.Design properties of hydrogel tissue-engineering scaffolds.Expert Rev Med Devices. 2011; 8(5):607-626.[16] 徐志云,张宝仁. 组织工程心脏瓣膜的研究现状与进展[J]. 第二军医大学学报, 2003, 24(3): 233-235.[17] Hoang Thi TT, Lee JS, Lee Y, et al. Enhanced Cellular Activity in Gelatin-Poly(Ethylene Glycol) Hydrogels without Compromising Gel Stiffness.Macromol Biosci. 2016;16(3):334-340.[18] Alghazali KM, Nima ZA, Hamzah RN, et al.Bone-tissue engineering: complex tunable structural and biological responses to injury, drug delivery, and cell-based therapies. Drug Metab Rev. 2015;47(4):431-454.[19] Karunaratne A, Xi L, Bentley L, et al. Multiscale alterations in bone matrix quality increased fragility in steroid induced osteoporosis. Bone. 2016;84:15-24.[20] Kim SY, Hwang JY, Shin US. Preparation of nano/macroporous polycaprolactone microspheres for an injectable cell delivery system using room temperature ionic liquid and camphene.J Colloid Interface Sci. 2016;465:18-25[21] 王圣,李温斌.去细胞组织工程心脏瓣膜研究现状与展望[J]. 中国医疗器械信息,2009,15(2):14-17.[22] Bai R,Liu HL.Heart decellularized matrix application in cardiac tissue engineering.Jiefangjun Yixueyuan Xuebao.2015;7:1-4.[23] Rieder E, Kasimir MT, Silberhumer G, et al. Decellularization protocols of porcine heart valves differ importantly in efficiency of cell removal and susceptibility of the matrix to recellularization with human vascular cells. J Thorac Cardiovasc Surg. 2004; 127(2):399-405.[24] Hussein KH, Park KM, Kang KS, et al.Biocompatibility evaluation of tissue-engineered decellularized scaffolds for biomedical application.Mater Sci Eng C Mater Biol Appl. 2016;67:766-778.[25] Papalamprou A, Griffiths LG. Cardiac Extracellular Matrix Scaffold Generated Using Sarcomeric Disassembly and Antigen Removal.Ann Biomed Eng. 2016;44(4):1047-1060.[26] Papalamprou A, Chang CW, Vapniarsky N,et al. Xenogeneic cardiac extracellular matrix scaffolds with or without seeded mesenchymal stem cells exhibit distinct in vivo immunosuppressive and regenerative properties. Acta Biomaterialia.2016;45:155-168.[27] Li Q, Uygun BE, Geerts S, et al.Proteomic Analysis of Naturally-Sourced Biological Scaffolds. Biomaterials.2015;75: 37-46.[28] HERRMANN F E M, LEHNER A, HOLLWECK T et al. In vitro biological and mechanical evaluation of various scaffold materials for myocardial tissue engineering.J Biomed Mater Res A. 2014; 102(4):958-966.[29] 肖统光. 细胞外基质来源支架在软骨组织工程中的应用[J]. 中国组织工程研究,2016,20(38):5737-5744.[30] 杨立信.骨髓间质干细胞构建组织工程心脏瓣膜的实验研究[J]. 第二军医大学.2004.[31] 黄胜兰,吴敬波. 心肌细胞增殖的研究进展[J]. 现代临床医学, 2011, 37(6):9-11.[32] Matsuyama D, Kawahara K.Proliferation of neonatal cardiomyocytes by connexin43 knockdown via synergistic inactivation of p38 MAPK and increased expression of FGF1. Basic Res Cardiol. 2009;104(6):631-642.[33] Ebelt H, Zhang Y, Köhler K, et al. Directed expression of dominant-negative p73 enables proliferation of cardiomyocytes in mice.J Mol Cell Cardiol. 2008;45(3):411-419.[34] Puente BN, Kimura W, Muralidhar SA, et al.The oxygen-rich postnatal environment induces cardiomyocyte cell-cycle arrest through DNA damage response. Cell.2014;157(3): 565-579.[35] Chamberlain G, Fox J, Ashton B,et al.Concise Review: Mesenchymal Stem Cells: Their Phenotype, Differentiation Capacity, Immunological Features, and Potential for Homing. Stem Cells.2007; 25(11):2739-2749.[36] Stubbs SL, Crook JM, Morrison WA, et al.Toward Clinical Application of Stem Cells for Cardiac RegenerationHeart Lung Circ. 2011;20(3):173-179.[37] Sun L, Cui M, Wang Z,et al. Mesenchymal stem cells modified with angiopoietin-1 improve remodeling in a rat model of acute myocardial infarction.Biochem Biophys Res Commun. 2007; 357(3):779-784.[38] de Macedo Braga LM, Lacchini S, Schaan BD, et al.In situ delivery of bone marrow cells and mesenchymal stem cells improves cardiovascular function in hypertensive rats submitted to myocardial infarction.J Biomed Sci. 2008;15(3):365-374.[39] 王福科,刘流,李俊男,等.间充质干细胞在组织工程中的应用进展[J].中国组织工程研究与临床康复,2010,14(36): 6800-6804.[40] Crisan M, Yap S, Casteilla L, et al.A Perivascular Origin for Mesenchymal Stem Cells in Multiple Human Organs. Cell Stem Cell.2008;3(3): 301-313.[41] Kocher AA, Schuster MD, Szabolcs MJ, et al. Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function.Nature Med. 2001;7(4): 430-436.[42] Antonitsis P, Ioannidou-Papagiannaki E, Kaidoglou A, et al. In vitro cardiomyogenic differentiation of adult human bone marrow mesenchymal stem cells. The role of 5-azacytidine.Interact Cardiovasc Thorac Surg. 2007;6(5):593-597.[43] Carvalho PH, Daibert AP, Monteiro BS, et al. Differentiation of adipose tissue-derived mesenchymal stem cells into cardiomyocytes. Arq Bras Cardiol. 2013;100(1):82-89.[44] Garbern JC, Lee RT. Cardiac stem cell therapy and the promise of heart regeneration.Cell Stem Cell. 2013;12(6):689-698.[45] Kang H, Lu S, Peng J, et al.In vivo construction of tissue-engineered cartilage using adipose-derived stem cells and bioreactor technology.Cell Tissue Bank. 2015;16(1):123-133.[46] Guyette JP, Charest JM, Mills RW, et al. Bioengineering Human Myocardium on Native Extracellular Matrix.Circ Res. 2016;118(1): 56-72.[47] Weymann A, Patil NP, Sabashnikov A, et al.Bioartificial heart: a human-sized porcine model--the way ahead.PLoS One. 2014 ; 9(11):e111591.[48] Tao ZW, Mohamed M, Hogan M, et al.Establishing the Framework for Fabrication of a Bioartificial Heart.ASAIO J. 2015;61(4): 429-436.[49] Hirt MN, Boeddinghaus J, Mitchell A, et al.Functional improvement and maturation of rat and human engineered heart tissue by chronic electrical stimulation.J Mol Cell Cardiol. 2014;74: 151-161.[50] He Q, Johnston J, Zeitlinger J.ChIP-nexus enables improved detection of in vivo transcription factor binding footprints. HHS Public Access.2015;33(4): 395–401.[51] 温华知. 腺病毒介导的Sema3A基因对大鼠心肌梗死后交感神经重构和电重构的影响[D]. 武汉大学, 2010.[52] Alteköester AK, Harvey RP.Bioengineered FSTL1 Patches Restore Cardiac Function Following Myocardial Infarction.Trends Mol Med. 2015;21(12):731-733.[53] Rimmbach C, Jung JJ, David R.Generation of Murine Cardiac Pacemaker Cell Aggregates Based on ES-Cell-Programming in Combination with Myh6-Promoter-Selection.J Vis Exp. 2015;(96): e52465. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Liu Zhichao, Zhang Fan, Sun Qi, Kang Xiaole, Yuan Qiaomei, Liu Genzhe, Chen Jiang. Morphology and activity of human nucleus pulposus cells under different hydrostatic pressures [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1172-1176. |
| [5] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [6] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [7] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [8] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [9] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [10] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [11] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [12] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [13] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [14] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [15] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||