Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (32): 5097-5103.doi: 10.12307/2024.496
Previous Articles Next Articles
Effect and mechanism of low intensity pulsed ultrasound on early angiogenesis in rats with acute tendon injury
Liu Xueli1, Shen Li1, Bi Wenguang1, Mou Yang1, Li Sen2
- 1School of Rehabilitation, Sichuan Vocational College of Health and Rehabilitation, Zigong 643000, Sichuan Province, China; 2Department of Spine Surgery, Nanjing Drum Tower Hospital Affiliated to Nanjing University Medicine School, Nanjing 210000, Jiangsu Province, China
-
Received:2023-08-16Accepted:2023-09-25Online:2024-11-18Published:2023-12-28 -
Contact:Li Sen, MD, Chief physician, Department of Spine Surgery, Nanjing Drum Tower Hospital Affiliated to Nanjing University Medicine School, Nanjing 210000, Jiangsu Province, China -
About author:Liu Xueli, Master, Rehabilitation therapist, School of Rehabilitation, Sichuan Vocational College of Health and Rehabilitation, Zigong 643000, Sichuan Province, China -
Supported by:Research Project of Zigong Municipal Health Commission, No. 21zd001 (to LXL)
CLC Number:
Cite this article
Liu Xueli, Shen Li, Bi Wenguang, Mou Yang, Li Sen. Effect and mechanism of low intensity pulsed ultrasound on early angiogenesis in rats with acute tendon injury[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5097-5103.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
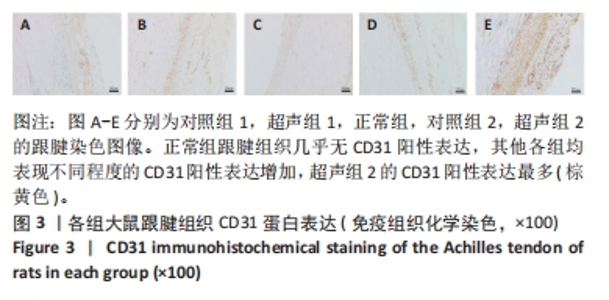
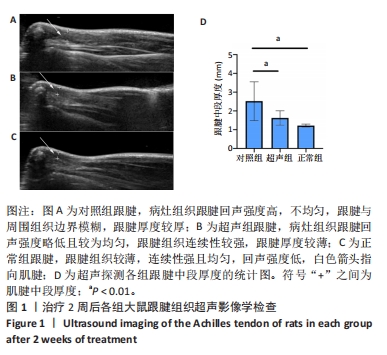
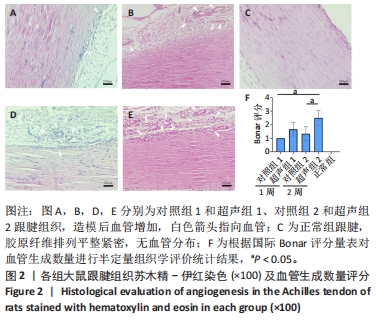
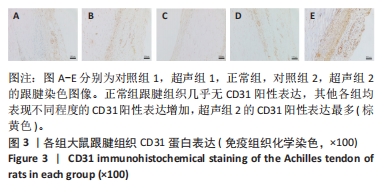
正常组肌腱组织(图2C)胶原纤维排列平整紧密,肌腱细胞呈纺锤形并平行于纤维,腱周组织少且几乎无血管,其他各组肌腱组织(图2A,B,D,E)均有不同程度的细胞增多、炎性细胞浸润、胶原纤维排列紊乱和血管分布改变。参照国际Bonar评分量表对血管生成数量进行半定量分析,结果显示:与对照组相比,超声组的新生血管数量较同期增多,其中超声组2的新生血管数量显著高于对照组2,差异有显著性意义(P < 0.05);而超声组1与对照组1之间差异无显著性意义(P > 0.05,图2F)。 2.4 免疫组织化学检测血管标志物CD31的蛋白表达水平 各组大鼠跟腱组织免疫组化结果见图3,血管标记物CD31主要分布于腱周的血管内皮组织,部分分布于腱内外膜和肌腱细胞。正常组跟腱组织(图3C)几乎无CD31的阳性表达,对照组和超声组均表现不同程度的CD31蛋白阳性表达增加。与对照组相比,超声组跟腱组织中CD31阳性表达增加,并随着时间呈上升趋势,可见超声组2新生血管数量增多最为明显。"
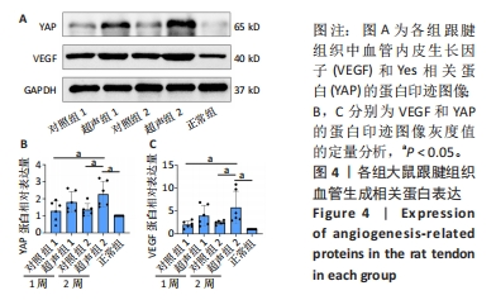
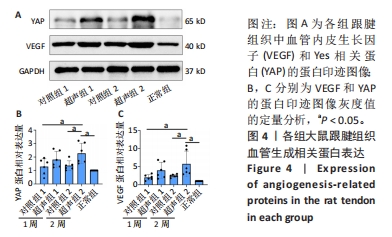
2.5 Western blot检测大鼠跟腱组织中VEGF、YAP的蛋白表达 Western blot检测结果见图4。与正常组相比,经Ⅰ型胶原酶造模的各组受损肌腱中的VEGF、YAP蛋白表达量均有不同程度的增加。其中,超声组的VEGF蛋白表达量均高于同期对照组,但超声组1与对照组1相比,VEGF蛋白表达量无显著性差异(P > 0.05),超声组2与对照组2和正常组相比均明显升高,差异有显著性意义(P < 0.05)。此外,超声组和对照组随时间均有上升趋势,2周时VEGF蛋白表达水平高于1周时,但差异无显著性意义(P > 0.05)。检测Hippo信号通路中关键效应因子YAP蛋白表达水平发现,超声组2明显高于对照组2,差异有显著性意义(P < 0.05);超声组1高于对照组1,但差异无显著性意义(P > 0.05)。"

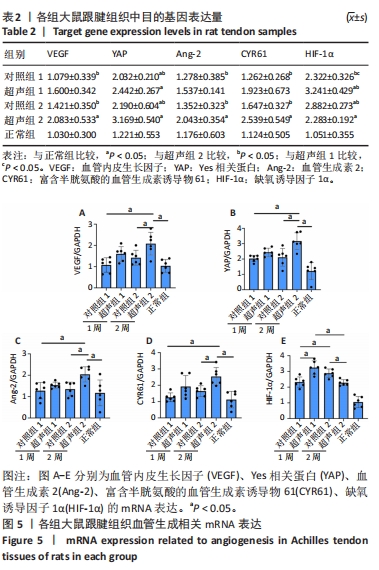
检测Hippo信号通路中关键效应因子YAP的mRNA表达量,发现造模后所有组的YAP基因表达量相比于正常组均有增加。各组间变化与VEGF表达趋势一致,超声组2的YAP基因表达量显著高于对照组2,差异有显著性意义(P < 0.05)。对YAP下游的Ang-2和CYR61的mRNA表达量进行统计,与同期对照组相比,超声组均有增加,但仅超声组2与对照组2之间差异有显著性意义(P < 0.05)。 对比超声组和对照组自身随时间的变化,结果显示随着肌腱愈合过程,超声组2和对照组2的VEGF、YAP、Ang-2和CYR61的mRNA表达量较超声组1和对照组1均有上升趋势,但差异均无显著性意义(P > 0.05)。 与其他因子mRNA表达量不同的是,LIPUS干预1周后超声组1的HIF-1α mRNA表达量相比于对照组1显著上升,差异有显著性意义(P < 0.05);然而,LIPUS干预2周后超声组2的HIF-1α mRNA表达量相比于对照组2显著降低,且差异有显著性意义(P < 0.05);对照组2的HIF-1α mRNA表达量相比于对照组1显著增加(P < 0.05),而超声组2相比于超声组1显著降低(P < 0.05)。"

| [1] NOURISSAT G, BERENBAUM F, DUPREZ D. Tendon injury: From biology to tendon repair. Nat Rev Rheumatol. 2015;11:223-233. [2] RIBBANS WJ, SEPTEMBER AV, COLLINS M. Tendon and Ligament Genetics: How Do They Contribute to Disease and Injury? A Narrative Review. Life (Basel). 2022;12(5):663. [3] SNEDEKER JG, FOOLEN J. Tendon injury and repair - A perspective on the basic mechanisms of tendon disease and future clinical therapy. Acta Biomater. 2017;63:18-36. [4] ACKERMAN JE, BEST KT, MUSCAT SN, et al. Metabolic Regulation of Tendon Inflammation and Healing Following Injury. Curr Rheumatol Rep. 2021;23(3):15. [5] LIU X, ZHU B, LI Y, et al. The Role of Vascular Endothelial Growth Factor in Tendon Healing. Front Physiol. 2021;12:766080. [6] YING ZM, LIN T, YAN SG. Low-intensity pulsed ultrasound therapy: a potential strategy to stimulate tendon-bone junction healing. J Zhejiang Univ Sci B. 2012;13(12):955-963. [7] ZHANG N, CHOW SK, LEUNG KS, et al. Ultrasound as a stimulus for musculoskeletal disorders. J Orthop Translat. 2017;9:52-59. [8] LU H, CHEN C, QU J, et al. Initiation Timing of Low-Intensity Pulsed Ultrasound Stimulation for Tendon-Bone Healing in a Rabbit Model. Am J Sports Med. 2016;44(10):2706-2715. [9] DE OLIVEIRA PERRUCINI PD, POLI-FREDERICO RC, DE ALMEIDA PIRES-OLIVEIRA DA, et al. Anti-Inflammatory and Healing Effects of Pulsed Ultrasound Therapy on Fibroblasts. Am J Phys Med Rehabil. 2020;99(1):19-25. [10] LAI WC, IGLESIAS BC, MARK BJ, et al. Low-Intensity Pulsed Ultrasound Augments Tendon, Ligament, and Bone-Soft Tissue Healing in Preclinical Animal Models: A Systematic Review. Arthroscopy. 2021; 37(7):2318-2333. [11] LI Y, LI W, LIU X, et al. Effects of Low-Intensity Pulsed Ultrasound in Tendon Injuries. J Ultrasound Med. 2023;42(9):1923-1939. [12] APTE RS, CHEN DS, FERRARA N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell. 2019;176(6):1248-1264. [13] AZAD T, JANSE VAN RENSBURG HJ, LIGHTBODY ED, et al. A LATS biosensor screen identifies VEGFR as a regulator of the Hippo pathway in angiogenesis. Nat Commun. 2018;9(1):1061. [14] BOOPATHY GTK, HONG W. Role of Hippo Pathway-YAP/TAZ Signaling in Angiogenesis. Front Cell Dev Biol. 2019;7:49. [15] HUANG Y, PAN M, SHU H, et al. Vascular endothelial growth factor enhances tendon-bone healing by activating Yes-associated protein for angiogenesis induction and rotator cuff reconstruction in rats. J Cell Biochem. 2020;121(3):2343-2353. [16] DELCOMBEL R, JANSSEN L, VASSY R, et al. New prospects in the roles of the C-terminal domains of VEGF-A and their cooperation for ligand binding, cellular signaling and vessels formation. Angiogenesis. 2013;16(2):353-371. [17] SOLCHAGA LA, BENDELE A, SHAH V, et al. Comparison of the effect of intra-tendon applications of recombinant human platelet-derived growth factor-BB, platelet-rich plasma, steroids in a rat achilles tendon collagenase model. J Orthop Res. 2014;32(1):145-150. [18] TORRES-SILVA R, LOPES-MARTINS RA, BJORDAL JM, et al. The low level laser therapy (LLLT) operating in 660 nm reduce gene expression of inflammatory mediators in the experimental model of collagenase-induced rat tendinitis. Lasers Med Sci. 2015;30(7):1985-1990. [19] TSAI YP, CHANG CW, LEE JS, et al. Direct radiofrequency application improves pain and gait in collagenase-induced acute achilles tendon injury. Evid Based Complement Alternat Med. 2013;2013:402692. [20] UEDA Y, INUI A, MIFUNE Y, et al. Molecular changes to tendons after collagenase-induced acute tendon injury in a senescence-accelerated mouse model. BMC Musculoskelet Disord. 2019;20(1):120. [21] MAFFULLI N, LONGO UG, FRANCESCHI F, et al. Movin and Bonar scores assess the same characteristics of tendon histology. Clin Orthop Relat Res. 2008;466(7):1605-1611. [22] LIMA KMME, COSTA JÚNIOR JFS, PEREIRA WCA, et al. Assessment of the mechanical properties of the muscle-tendon unit by supersonic shear wave imaging elastography: a review. Ultrasonography. 2018; 37(1):3-15. [23] ZABRZYŃSKA M, GRZANKA D, ZIELIŃSKA W, et al. The Bonar Score in the Histopathological Assessment of Tendinopathy and Its Clinical Relevance-A Systematic Review. Medicina (Kaunas). 2021;57(4):367. [24] KING CM, VARTIVARIAN M. Achilles Tendon Rupture Repair: Simple to Complex. Clin Podiatr Med Surg. 2023;40(1):75-96. [25] GORICK CM, CHAPPELL JC, PRICE RJ. Applications of Ultrasound to Stimulate Therapeutic Revascularization. Int J Mol Sci. 2019;20(12): 3081. [26] KORNTNER S, LEHNER C, GEHWOLF R, et al. Limiting angiogenesis to modulate scar formation. Adv Drug Deliv Rev. 2019;146:170-189. [27] WU F, NERLICH M, DOCHEVA D. Tendon injuries: Basic science and new repair proposals. EFORT Open Rev. 2017;2(7):332-342. [28] ZHANG F, LIU H, STILE F, et al. Effect of vascular endothelial growth factor on rat Achilles tendon healing. Plast Reconstr Surg. 2003;112(6): 1613-1619. [29] HALL K, RAN S. Regulation of tumor angiogenesis by the local environment. Front Biosci (Landmark Ed). 2010;15(1):195-212. [30] COPELAND K, PURVIS AR. A Retrospective Chart Review of Chronic Wound Patients Treated with Topical Oxygen Therapy. Adv Wound Care (New Rochelle). 2017;6(5):143-152. [31] TEMPFER H, TRAWEGER A. Tendon Vasculature in Health and Disease. Front Physiol. 2015;6:330. [32] 王增涛,郝丽文,李桂石. Wistar大鼠解剖图谱[M].济南:山东科学技术出版社,2009. [33] FARCIC TS, BALDAN CS, CATTAPAN CG, et al. Treatment time of ultrasound therapy interferes with the organization of collagen fibers in rat tendons. Braz J Phys Ther. 2013;17(3):263-271. [34] FARCIC TS, BALDAN CS, MACHADO AFP, et al. Collagen Fibers in the Healing Process of Rat Achilles Tendon Rupture Using Different Times of Ultrasound Therapy. Adv Wound Care (New Rochelle). 2018; 7(4):114-120. [35] JEREMIAS JÚNIOR SL, CAMANHO GL, BASSIT AC, et al. Low-intensity pulsed ultrasound accelerates healing in rat calcaneus tendon injuries. J Orthop Sports Phys Ther. 2011;41(7):526-531. [36] PEACH CJ, MIGNONE VW, ARRUDA MA, et al. Molecular Pharmacology of VEGF-A Isoforms: Binding and Signalling at VEGFR2. Int J Mol Sci. 2018;19(4):1264. [37] LAI F, WANG J, TANG H, et al. VEGF promotes tendon regeneration of aged rats by inhibiting adipogenic differentiation of tendon stem/progenitor cells and promoting vascularization. FASEB J. 2022;36(8): e22433. [38] WU JB, TANG YL, LIANG XH. Targeting VEGF pathway to normalize the vasculature: an emerging insight in cancer therapy. Onco Targets Ther. 2018;11:6901-6909. [39] BUDEL SJ, PENNING MM, PENNING LC. Hippo signaling pathway in companion animal diseases, an under investigated signaling cascade. Vet Q. 2021;41(1):172-180. [40] DEY A, VARELAS X, GUAN KL. Targeting the Hippo pathway in cancer, fibrosis, wound healing and regenerative medicine. Nat Rev Drug Discov. 2020;19(7):480-494. [41] WU Z, GUAN KL. Hippo Signaling in Embryogenesis and Development. Trends Biochem Sci. 2021;46(1):51-63. |
| [1] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [2] | Wang Yeyuan, Du Yilang, Yu Dehao, Ning Fengting, Bai Bing. Effect of micro-arc oxidation treatment on biological activity of medical metals [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 771-776. |
| [3] | Wang Jiani, Chen Junyu. Angiogenesis mechanism of metal ions and their application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 804-812. |
| [4] | Zhang Ya, Mu Qiuju, Wang Zilin, Liu Hongjie, Zhu Lili. Hydrogel loaded with platelet-rich plasma promotes wound healing in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 690-696. |
| [5] | Yang Yuqing, Chen Zhiyu. Role and application of early transient presence of M1 macrophages in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 594-601. |
| [6] | Maisituremu·Heilili, Zhang Wanxia, Nijiati·Nuermuhanmode, Maimaitituxun·Tuerdi. Effect of intraarticular injection of different concentrations of ozone on condylar histology of rats with early temporomandibular joint osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 505-509. |
| [7] | Xiong Yang, Zhou Shibo, Yu Xing, Bi Lianyong, Yang Jizhou, Wang Fengxian, Qu Yi, Yang Yongdong, Zhao Dingyan, Zhao He, Qiu Ziye, Jiang Guozheng. Molecular biological mechanism of acquired heterotopic ossification [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(30): 4881-4888. |
| [8] | Li Sijin, Feng Xiaoteng, Wang Yiru, Liu Ping. Macrophage-specific promoter SP146-C1 enhances vascular endothelial growth factor C expression in atherosclerotic mice [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4202-4208. |
| [9] | He Bo, He Zhijun, Liu Tao, Ma Suilu, Wei Xiaotao, Wang Weiwei. Action mechanism and advantages of mesenchymal stem cells for treating flap ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(25): 4065-4071. |
| [10] | Dong Xinyu, Dong Xinyue, Wang Wanting, Fan Haixia, Cheng Huanzhi. The effective components of Chinese medicine combined with scaffold materials promote bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3240-3245. |
| [11] | Wang Raokaijuan, Zhao Lixing. Application of deferoxamine in bone tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3272-3280. |
| [12] | Li Chengming, Xue Dongling, Yang Xinyu, Xiao Chi, Cui Daping. Mechanism of Chinese medicine for promoting blood circulation and removing blood stasis combined with platelet-rich plasma to improve steroid-induced necrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 288-294. |
| [13] | Fang Yuan, Kang Zhijie, Wang Haiyan, Li Xiaohe, Zhang Kai. Visualization analysis of vascular stimulating scaffolds in bone tissue engineering#br# [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2708-2715. |
| [14] | Gao Li, Liu Liu, Ren Wenyan, Liu Xue, Wang Yiyu. Influential mechanism of graphene and its derivatives on angiogenesis and vascularized bone [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2716-2722. |
| [15] | Guo Yuxin, Wang Hao, Li Mingqi, Chen Yueying, Pan Juhong, Huang Xin, Wang Zhiwen, Zhou Qing. Ultrasound-optimized hydrogel scaffold used to promote transdermal delivery of gold nanoparticles [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2330-2337. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||