Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (17): 2708-2715.doi: 10.12307/2024.373
Previous Articles Next Articles
Visualization analysis of vascular stimulating scaffolds in bone tissue engineering
Fang Yuan1, Kang Zhijie1, Wang Haiyan2, Li Xiaohe2, Zhang Kai3
- 1School of Basic Medical Sciences, Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China; 2Department of Human Anatomy, School of Basic Medicine, Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China; 3Department of Orthopedics, Second Hospital of Ulanqab, Ulanqab 012000, Inner Mongolia Autonomous Region, China
-
Received:2023-04-25Accepted:2023-06-02Online:2024-06-18Published:2023-12-15 -
Contact:Wang Haiyan, Master, Professor, Master’s supervisor, Department of Human Anatomy, School of Basic Medicine, Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China Li Xiaohe, PhD, Professor, Doctoral supervisor, Department of Human Anatomy, School of Basic Medicine, Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China -
About author:Fang Yuan, Master candidate, Attending physician, School of Basic Medical Sciences, Inner Mongolia Medical University, Hohhot 010000, Inner Mongolia Autonomous Region, China -
Supported by:Health Science and Technology Project of Inner Mongolia Autonomous Region, No. 202201188 (to WHY); Inner Mongolia Natural Science Foundation, No. 2021MS08086 (to WHY); “Achievement Transformation” Project of Inner Mongolia Medical University, No. YKD2020CGZH009 (to WHY); Inner Mongolia Natural Science Foundation, No. 2020MS08124 (to LXH); Youth Innovation and Entrepreneurship Talent Project of “Grassland Talents” Project in Inner Mongolia Autonomous Region, No. 2020 (to LXH); Follow-up Research Project of Inner Mongolia Medical University, No. 2019 (to LXH); Zhiyuan Talent Project of Inner Mongolia Medical University in 2021 (to LXH); 2021 Key Scientific Research Project of Inner Mongolia Medical University, No. YKD2021ZD001 (to LXH); Development Plan for Innovation Teams in Higher Education Institutions of Inner Mongolia Department of Education, No. NMGIRT2227 (to LXH); Basic Research Project of Ulanqab City, No. 2021JC321 (to ZK)
CLC Number:
Cite this article
Fang Yuan, Kang Zhijie, Wang Haiyan, Li Xiaohe, Zhang Kai. Visualization analysis of vascular stimulating scaffolds in bone tissue engineering[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2708-2715.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
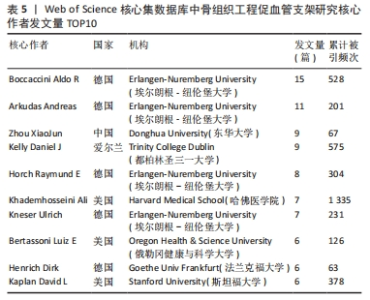
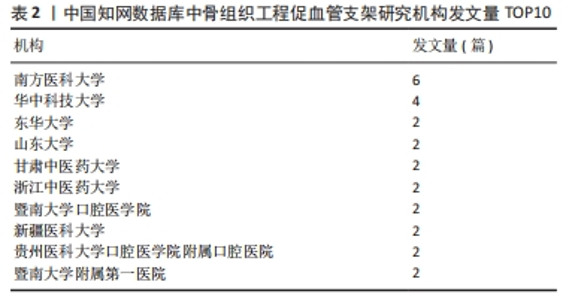
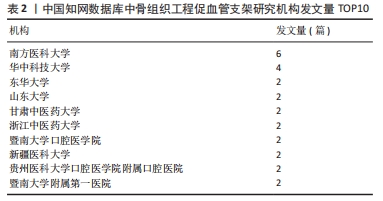
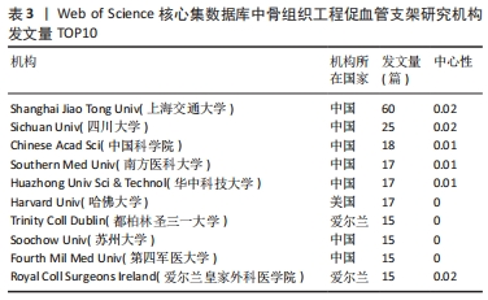
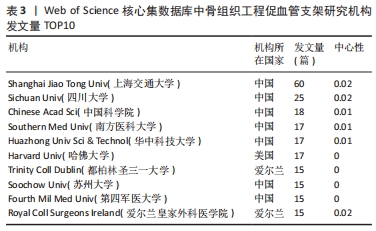
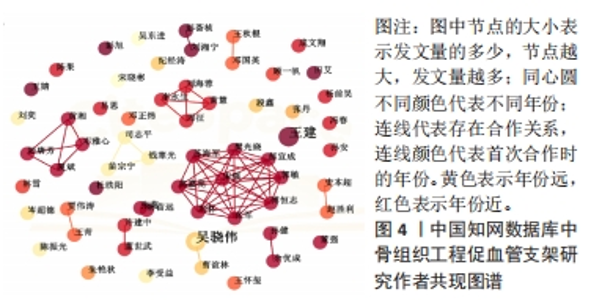
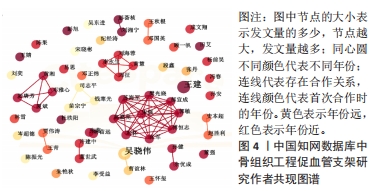
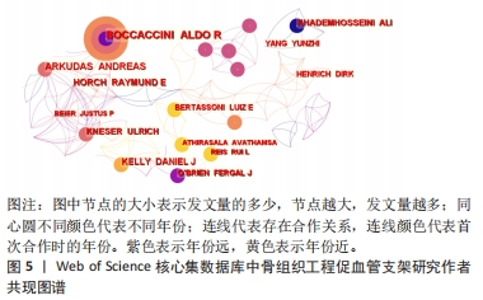
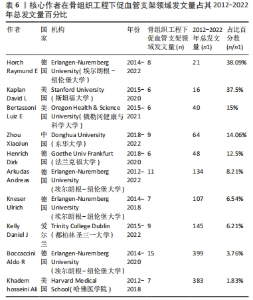
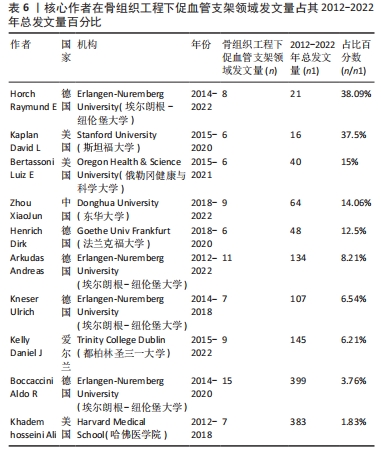
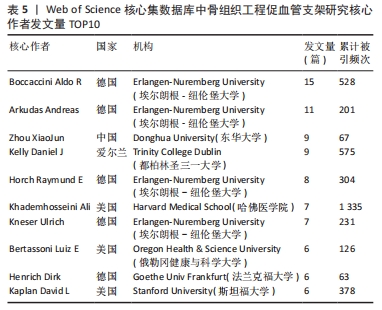
核心作者是指某一学科或专业领域中,发文量多、被引率高和影响力大的杰出作者。通过对核心作者研究方向的分析,可以了解骨组织工程下促血管支架领域的发展脉络。根据核心作者计算公式,对中国知网和Web of Science核心集数据库文献进行计算,中国知网数据计算得N=1.06,取整数为2篇,核心作者共有2位,分别是吴骁伟和王健。Web of Science核心集数据库数据计算得N=2.89,取整数为3篇,核心作者共有249位。 骨组织工程下促血管支架领域发文量排名前10位的核心作者中,发文数量最多的为德国Erlangen-Nuremberg University的Boccaccini Aldo R,共发表15篇文章,其他依次为Arkudas Andreas(11篇)、Zhou XiaoJun(9篇)、Kelly Daniel J(9篇)、Horch Raymuend E(8篇)、Khademhosseini Ali(7篇)、Kneser Ulrich(7篇)、Bertassoni Luiz E(6篇)、Henrich Dirk(6篇)、Kaplan David L(6篇)。累计被引次数最多的为Harvard Medical School的Khademhosseini Ali,累计被引1 335次,其他依次为Kelly Daniel J(575次)、Boccaccini Aldo R(528次)、Kaplan David L(378次)、Horch Raymund E(304次)、Kneser Ulrich(231次)、Arkudas Andreas (201次)、Bertassoni Luiz E(126次)、Zhou XiaoJun(67次)、Henrich Dirk(63次),见表5。"

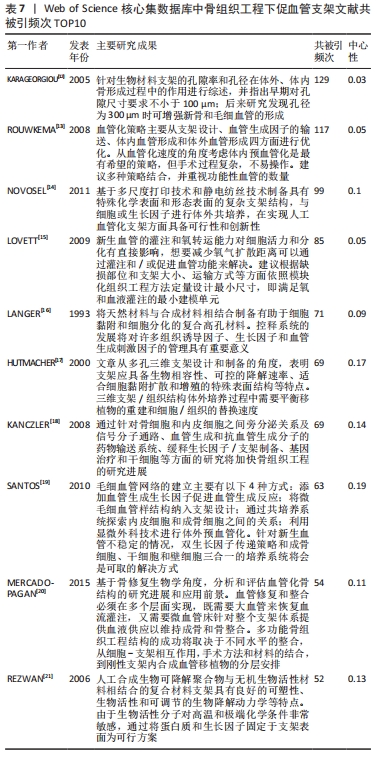
2.5 文献共被引分析 利用CiteSpace 6.1.R2绘制出Web of Science核心集数据库文献共被引知识图谱,得到节点数为474,连线数为778的文献共被引知识图谱,见图6。统计文献共被引次数最多的10篇文章(均为综述)[12-21],见表7。通过对表中10篇文献的主要内容进行分析,目前骨组织工程促血管支架领域的关注点主要分以下几类:①血管化策略:支架设计、血管生成因子的输送、体外共培养、体内预血管化;②技术方面:3D打印、静电纺丝、血管移植及血管融合;③机制方面:移植物的重建和细胞/组织的替换速度、内皮细胞和成骨细胞之间的关系、骨细胞和内皮细胞之间旁分泌关系及信号分子通路、血管生成和抗血管生成分子;④其他方面:孔隙率、氧梯度、血液灌注、营养供应及生物降解性。因中国知网数据库无法导出参考文献信息,因此文章未对中国知网数据库文献进行共被引分析。"


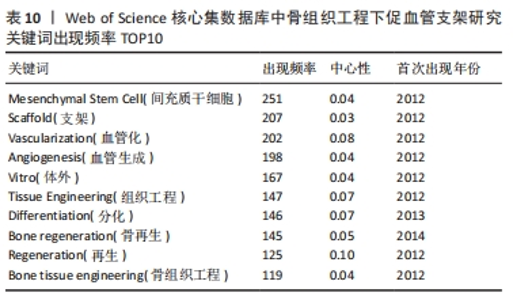
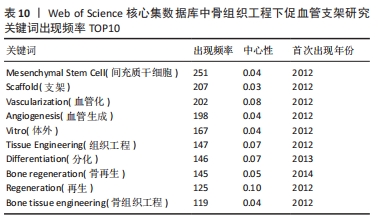
将Web of Science核心集数据库文献数据导入CiteSpace 6.1.R2,参数设置同上,得到节点数为277,连线数为1 995的关键词图谱,见图8。其中关键词“mesenchymal stem cell”出现频率最高,为251次,中心性为0.04。其他依次为“scaffold”(207次)、“vascularization”(202次)、“angiogenesis”(198次)、“vitro”(167次)、“tissue engineering”(147次)、“differentiation”(146次)、“bone regeneration”(145次)、“regeneration”(125次)、“bone tissue engineering”(119次),见表10。"

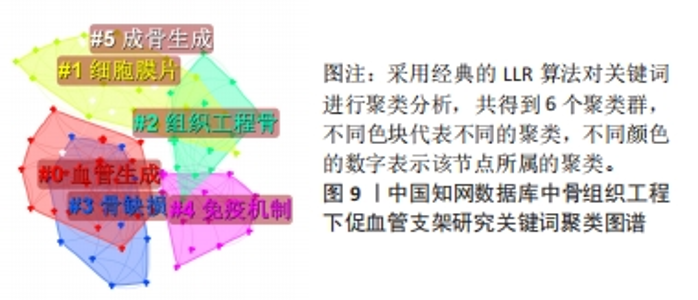
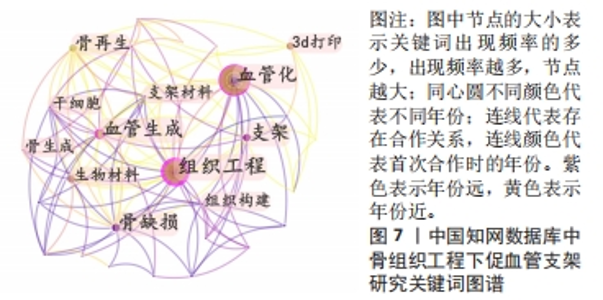
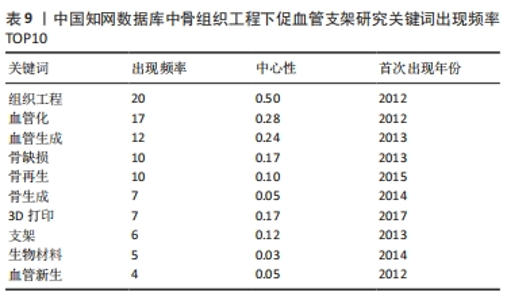
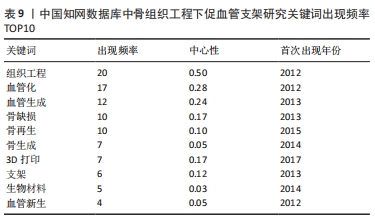
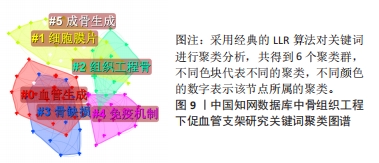
对比国内外关键词图谱及关键词频率表,发现国内外研究热点有所不同。中国从2013年开始使用支架及生物材料对骨缺损、骨生成及骨再生方面进行血管化和血管生成的研究;而3D打印在中国首次出现于2017年。国外的研究方向更趋向于细胞层面,以间充质干细胞(mesenchymal stem cell)与不同支架(scaffold)相融合,研究其在体内或体外(vitro)的骨再生(bone regeneration)、细胞分化(differentiation)、血管化(vascularization)或血管生成(angiogenesis)的能力。 2.7.2 关键词聚类 CiteSpace 6.1.R2 的聚类分析功能将具有相似特征的关键词划分至同一组中,可以了解一个学科领域的发展历程、各时期主要研究方向以及最新的研究热点。在中国知网数据库关键词图谱基础上,制作关键词聚类图谱,得到了Q:0.452 1(Q > 0.3表明可视化图谱中聚类结构显著)、S:0.806 6(S > 0.7表明聚类结果令人信服且关联性强)的关键词聚类图谱[32],共得到关键词聚类标签6个,分别为血管生成(0.756)、细胞膜片(0.834)、组织工程骨(0.866)、骨缺损(0.854)、免疫机制(0.893)及成骨生成(0.769),见图9。"
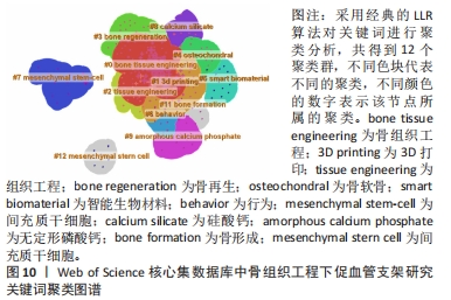
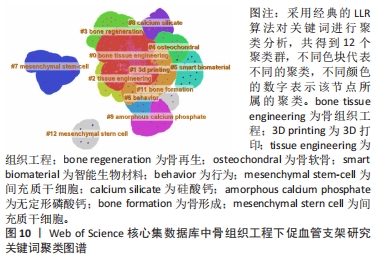
在Web of Science核心集数据库关键词图谱基础上,制作关键词聚类图谱,得到Q:0.587 3(Q > 0.3表明可视化图谱中聚类结构显著),S为0.832 9(S > 0.7表明聚类结果令人信服且关联性强)的图谱,共得到12个聚类标签,分别为“bone tissue engineering”(0.719),“3D printing”(0.579),“tissue engineering”(0.887),“bone regeneration”(0.957),“osteochondral”(0.949),“smart biomaterial”(0.993),“behavior”(0.97),“mesenchymal stem-cell”(0.987),“calcium silicate”(0.983),“amorphous calcium phosphate”(0.988),“bone formation”(0.996),“mesenchymal stern cell”(1),见图10。"

中国知网数据库关键词突现图中共出现10个关键词,其中材料方面主要为水凝胶和生物材料,细胞方面主要为干细胞。Web of Science核心集数据库关键词突现图中显示排名前20的关键词,早期焦点主要集中于“骨髓基质细胞、基因表达、生物相容性、骨形态发生蛋白诱导成骨、外周血、预制、祖细胞、缺氧、人骨髓、血管生长因子”;后期则集中于“透明质酸、复合材料、骨形态发生蛋白2、聚已酸内酯、干细胞、3D打印、间充质干细胞、壳聚糖”。由此可见,初期促血管支架的研究主要利用骨髓基质细胞、祖细胞等种子细胞进行分化诱导,或者在体外预制血管后植入支架当中,对血管生长因子、基因表达和生物相容性进行研究。后期研究的方法除了继续使用干细胞一类的种子细胞进行研究外,加大了对材料方面促血管作用的研究,如透明质酸、聚己酸内酯和壳聚糖等。除此之外,3D打印技术也于2017年开始应用于促血管支架领域,且为当下热点之一。"

| [1] HUANG EE, ZHANG N, GANIO EA, et al. Differential dynamics of bone graft transplantation and mesenchymal stem cell therapy during bone defect healing in a murine critical size defect. J Orthop Translat. 2022;36(8):64-74. [2] SCHEMITSCH EH. Size Matters: defining critical in bone defect size. J Orthop Trauma. 2017;31(10):S20-S22. [3] 罗卓荆,毕龙.中国骨缺损修复的成就与展望[J].空军军医大学学报,2022,43(4):263-267. [4] KASHIRINA A, YAO YT, LIU YJ, et al. Biopolymers as bone substitutes: a review. Biomater Sci. 2019;7(10):3961-3983. [5] 苑博,王智巍,唐一钒,等.3D打印技术构建不同比例聚己内酯-磷酸三钙支架及其体外诱导成骨性能[J].中国组织工程研究,2019,23(6):821-826. [6] VAN VOSS MRH, VESUNA F, BOL GM, et al. Targeting mitochondrial translation by inhibiting DDX3: a novel radiosensitization strategy for cancer treatment. Oncogene. 2018;37(1):63-74. [7] ALMUBARAK S, NETHERCOTT H, FREEBERG M, et al. Tissue engineering strategies for promoting vascularized bone regeneration. Bone. 2016;83(2):197-209. [8] 栾春娟,侯海燕,王贤文.国际科技政策研究热点与前沿的可视化分析[J].科学学研究,2009,27(2):240-243. [9] 陈悦,陈超美,刘则渊,等.CiteSpace知识图谱的方法论功能[J].科学学研究,2015, 33(2):242-253. [10] 汪德根,陈田,王金莲,等.1980-2009年国内外旅游研究比较[J].地理学报,2011, 66(4):535-548. [11] 于良楠,柯尊清.中国文化安全研究的热点主题与前沿——基于CiteSpace软件的可视化分析[J].中国文化产业评论,2021,30(1):130-146 [12] KARAGEORGIOU V, KAPLAN D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials. 2005;26(27):5474-5491. [13] ROUWKEMA J, RIVRON NC, VAN BLITTERSWIJK CA. Vascularization in tissue engineering. Trends Biotechnol. 2008;26(8):434-441. [14] NOVOSEL EC, KLEINHANS C, KLUGER PJ. Vascularization is the key challenge in tissue engineering. Adv Drug Deliv Rev. 2011;63(4):300-311. [15] LOVETT M, LEE K, EDWARDS A, et al. Vascularization strategies for tissue engineering. Tissue Eng Part B Rev. 2009;15(3):353-370. [16] LANGER R, VACANTI JP. Tissue engineering. Science. 1993;260(5110):920-926. [17] HUTMACHER DW. Scaffolds in tissue engineering bone and cartilage. Biomaterials. 2000,21(24):2529-2543. [18] KANCZLER JM, OREFFO RO. Osteogenesis and angiogenesis: the potential for engineering bone. Eur Cell Mater. 2008;15(2):100-114. [19] SANTOS MI, REIS RL.Vascularization in bone tissue engineering: physiology,current strategies,major hurdles and future challenges. Macromol Biosci. 2010;10(1):12-27. [20] MERCADO-PAGÁN ÁE, STAHL AM, SHANJANI Y, et al. Vascularization in bone tissue engineering constructs. Ann Biomed Eng. 2015;43(3):718-729. [21] REZWAN K, CHEN QZ, BLAKER JJ, et al. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials. 2006;27(18): 3413-3431. [22] WU C, ZHOU Y, XU M, et al. Copper-containing mesoporous bioactive glass scaffolds with multifunctional properties of angiogenesis capacity, osteostimulation and antibacterial activity. Biomaterials. 2013;34(2):422-433. [23] SPILLER KL, NASSIRI S, WITHEREL CE, et al. Sequential delivery of immunomodulatory cytokines to facilitate the M1-to-M2 transition of macrophages and enhance vascularization of bone scaffolds. Biomaterials. 2015;37(1):194-207. [24] TANG D, TARE RS, YANG LY, et al. Biofabrication of bone tissue: approaches, challenges and translation for bone regeneration. Biomaterials. 2016;83(3):363-382. [25] DE WITTE TM, FRATILA-APACHITEI LE, ZADPOOR AA, et al. Bone tissue engineering via growth factor delivery: from scaffolds to complex matrices. Regen Biomate. 2018; 5(4):197-211. [26] ABBASI N, HAMLET S, LOVE RM, et al. Porous scaffolds for bone regeneration. J Sci Adv Mater Dev. 2020;5(1):1-9. [27] WU C, CHANG J. Multifunctional mesoporous bioactive glasses for effective delivery of therapeutic ions and drug/growth factors. J Control Release. 2014;193(11):282-295. [28] XU HHK, WANG P, WANG L, et al. Calcium phosphate cements for bone engineering and their biological properties. Bone Res. 2017;5(1):1-19. [29] KARGOZAR S, BAINO F, HAMZEHLOU S, et al. Bioactive glasses: sprouting angiogenesis in tissue engineering. Trends Biotechnol. 2018;36(4):430-444. [30] DIOMEDE F, MARCONI GD, FONTICOLI L, et al. Functional relationship between osteogenesis and angiogenesis in tissue regeneration. Int J Mol Sci. 2020;21(9):32-42. [31] WU L, GU Y, LIU L, et al. Hierarchical micro/nanofibrous membranes of sustained releasing VEGF for periosteal regeneration. Biomaterials. 2019;227(1):119555. [32] 董佳乐,魏远好,张洪武.基于知识图谱的骨缺损治疗可视化分析[J].国组织工程研究,2022,26(18):2906-2913. [33] HSU EL, STOCK SR. Growth factors,carrier materials, and bone repair. Handb Exp Pharmacol. 2020;262:121-56. [34] BOSE S, ROY M, BANDYOPADHYAY A. Recent advances in bone tissue engineering scaffolds. Trends Biotechnol. 2012;30(10):546-554. [35] CHEN YC, LIN RZ, QI H, et al. Functional human vascular network generated in photocrosslinkable gelatin methacrylate hydrogels. Adv Funct Mater. 2012;22(10):2027-2039. [36] ZHU H, MONAVARI M, ZHENG K, et al. 3D bioprinting of multifunctional dynamic nanocomposite bioinks incorporating cu-doped mesoporous bioactive glass nanoparticles for bone tissue engineering. Small. 2022;18(12):2104996. [37] TWOHIG C, HELSINGA M, MANSOORIFAR A, et al. A dual-ink 3D printing strategy to engineer pre-vascularized bone scaffolds in-vitro. Mater Sci Eng C Mater Biol Appl. 2021;123(3):111976. [38] MANZINI BM, MACHADO LMR, NORITOMI PY, et al. Advances in Bone tissue engineering: a fundamental review. J Biosci. 2021;46(1):1-18. [39] DALY AC, PITACCO P, NULTY J, et al. 3D printed microchannel networks to direct vascularisation during endochondral bone repair. Biomaterials. 2018;162(1):34-46. [40] FITZPATRICK V, MARTIN-MOLDES Z, DECK A, et al. Functionalized 3D-printed silk-hydroxyapatite scaffolds for enhanced bone regeneration with innervation and vascularization. Biomaterials. 2021;276(9):120995. [41] 贺丹.骨髓间充质干细胞在RGD改良壳聚糖基可注射纳米复合凝胶中的血管化研究[D].成都:西南交通大学,2019. [42] 孙安,毕翔宇,韩祥祯,等.联合应用血管内皮生长因子及血小板衍生生长因子BB干预骨髓间充质干细胞的血管化及增殖能力[J].中国组织工程研究,2020,24(1):1-6. [43] 周琦琪,韩祥祯,张文静,等.碱性成纤维细胞生长因子诱导大鼠骨髓间充质干细胞膜片成骨及成血管分化[J].中国组织工程研究,2021,25(1):1-5. [44] 晏金超.3D打印挤出沉积制备金属阳离子掺杂HA骨支架研究[D].南昌:南昌大学, 2020. [45] ZHOU X, QIAN Y, CHEN L, et al. Flowerbed-inspired biomimetic scaffold with rapid internal tissue infiltration and vascularization capacity for bone repair. ACS Nano. 2023; 17(5):5140-5156. [46] CHENG WX, LIU YZ, MENG XB, et al. PLGA/β-TCP composite scaffold incorporating cucurbitacin B promotes bone regeneration by inducing angiogenesis. J Orthop Transl. 2021;31(11):41-51. [47] XU H, WANG C, LIU C, et al. Cotransplantation of mesenchymal stem cells and endothelial progenitor cells for treating steroid-induced osteonecrosis of the femoral head. Stem Cells Transl Med. 2021;10(5):781-796. [48] WINKLER S, MUTSCHALL H, BIGGEMANN J, et al. Human umbilical vein endothelial cell support bone formation of adipose-derived stem cell-loaded and 3D-printed osteogenic matrices in the arteriovenous loop model. Tissue Engineering Part A. 2021;27(5-6):413-423. [49] ZHANG Y, XIE Y, HAO Z, et al. Umbilical mesenchymal stem cell-derived exosome-encapsulated hydrogels accelerate bone repair by enhancing angiogenesis. ACS Appl Mater Interfaces. 2021;13(16):18472-18487. [50] PIARD C, LUTHCKE R, KAMALITDINOV T, et al. Sustained delivery of vascular endothelial growth factor from mesoporous calcium‐deficient hydroxyapatite microparticles promotes in vitro angiogenesis and osteogenesis. J Biomed Mater Res A. 2021;109(7):1080-1087. [51] BAYER EA, JORDAN J, ROY A, et al. Programmed platelet-derived growth factor-BB and bone morphogenetic protein-2 delivery from a hybrid calcium phosphate/alginate scaffold. Tissue Eng Part A. 2017;23(23-24):1382-1393. [52] NOVAIS A, LESIEUR J, SADOINE J, et al. Priming dental pulp stem cells from human exfoliated deciduous teeth with fibroblast growth factor-2 enhances mineralization within tissue-engineered constructs implanted in craniofacial bone defects. Stem Cells Transl Med. 2019;8(8):844-857. [53] LEE J, HUH SJ, SEOK JM, et al. Surface engineering of 3D-printed scaffolds with minerals and a pro-angiogenic factor for vascularized bone regeneration. Acta Biomater. 2022; 140(3):730-744. [54] WEI X, ZHOU W, TANG Z, et al. Magnesium surface-activated 3D printed porous PEEK scaffolds for in vivo osseointegration by promoting angiogenesis and osteogenesis. Bioact Mater. 2023;20(5):16-28. [55] HOEMANN CD, GONZÁLEZ JR, GUZMÁN-MORALES J, et al. Chitosan coatings with distinct innate immune bioactivities differentially stimulate angiogenesis, osteogenesis and chondrogenesis in poly-caprolactone scaffolds with controlled interconnecting pore size. Bioact Mater. 2022;10(9):430-442. [56] LI T, PENG M, YANG Z, et al. 3D-printed IFN-gamma-loading calcium silicate-beta-tricalcium phosphate scaffold sequentially activates M1 and M2 polarization of macrophages to promote vascularization of tissue engineering bone. Acta Biomater. 2018;71(3):96-107. [57] CHEN S, WANG H, LIU D, et al. Early osteoimmunomodulation by mucin hydrogels augments the healing and revascularization of rat critical-size calvarial bone defects. Bioactive Materials. 2023;25(2):176-188. [58] BISWAS L, CHEN J, DE ANGELIS J, et al. Lymphatic vessels in bone support regeneration after injury. Cell. 2023;186(2):382-397. |
| [1] | Li Zikai, Zhang Chengcheng, Xiong Jiaying, Yang Xirui, Yang Jing, Shi Haishan. Potential effects of ornidazole on intracanal vascularization in endodontic regeneration [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(在线): 1-7. |
| [2] | Lai Pengyu, Liang Ran, Shen Shan. Tissue engineering technology for repairing temporomandibular joint: problems and challenges [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(在线): 1-9. |
| [3] | Yuan Weibo, Liu Chan, Yu Limei. Potential application of liver organoids in liver disease models and transplantation therapy [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(8): 1684-1692. |
| [4] | Sun Yuting, Wu Jiayuan, Zhang Jian. Physical factors and action mechanisms affecting osteogenic/odontogenic differentiation of dental pulp stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1531-1540. |
| [5] | Yu Ting, Lyu Dongmei, Deng Hao, Sun Tao, Cheng Qian. Icariin pretreatment enhances effect of human periodontal stem cells on M1-type macrophages [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1328-1335. |
| [6] | Yang Zhihang, Sun Zuyan, Huang Wenliang, Wan Yu, Chen Shida, Deng Jiang. Nerve growth factor promotes chondrogenic differentiation and inhibits hypertrophic differentiation of rabbit bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1336-1342. |
| [7] | Hu Taotao, Liu Bing, Chen Cheng, Yin Zongyin, Kan Daohong, Ni Jie, Ye Lingxiao, Zheng Xiangbing, Yan Min, Zou Yong. Human amniotic mesenchymal stem cells overexpressing neuregulin-1 promote skin wound healing in mice [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1343-1349. |
| [8] | Jin Kai, Tang Ting, Li Meile, Xie Yuan. Effects of conditioned medium and exosomes of human umbilical cord mesenchymal stem cells on proliferation, migration, invasion, and apoptosis of hepatocellular carcinoma cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1350-1355. |
| [9] | Li Dijun, Jiu Jingwei, Liu Haifeng, Yan Lei, Li Songyan, Wang Bin. Three-dimensional gelatin microspheres loaded human umbilical cord mesenchymal stem cells for chronic tendinopathy repair [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1356-1362. |
| [10] | Lou Guo, Zhang Min, Fu Changxi. Exercise preconditioning for eight weeks enhances therapeutic effect of adipose-derived stem cells in rats with myocardial infarction [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1363-1370. |
| [11] | Liu Qi, Li Linzhen, Li Yusheng, Jiao Hongzhuo, Yang Cheng, Zhang Juntao. Icariin-containing serum promotes chondrocyte proliferation and chondrogenic differentiation of stem cells in the co-culture system of three kinds of cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1371-1379. |
| [12] | Huang Ting, Zheng Xiaohan, Zhong Yuanji, Wei Yanzhao, Wei Xufang, Cao Xudong, Feng Xiaoli, Zhao Zhenqiang. Effects of macrophage migration inhibitory factor on survival, proliferation, and differentiation of human embryonic stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1380-1387. |
| [13] | Aikepaer · Aierken, Chen Xiaotao, Wufanbieke · Baheti. Osteogenesis-induced exosomes derived from human periodontal ligament stem cells promote osteogenic differentiation of human periodontal ligament stem cells in an inflammatory microenvironment [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1388-1394. |
| [14] | Zhang Zhenyu, Liang Qiujian, Yang Jun, Wei Xiangyu, Jiang Jie, Huang Linke, Tan Zhen. Target of neohesperidin in treatment of osteoporosis and its effect on osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1437-1447. |
| [15] | Xie Liugang, Cui Shuke, Guo Nannan, Li Aoyu, Zhang Jingrui. Research hotspots and frontiers of stem cells for Alzheimer’s disease [J]. Chinese Journal of Tissue Engineering Research, 2025, 29(7): 1475-1485. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||