Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (20): 3240-3245.doi: 10.12307/2024.385
Previous Articles Next Articles
The effective components of Chinese medicine combined with scaffold materials promote bone tissue regeneration
Dong Xinyu, Dong Xinyue, Wang Wanting, Fan Haixia, Cheng Huanzhi
- School of Stomatology, Jining Medical University, Jining 272000, Shandong Province, China
-
Received:2023-05-23Accepted:2023-07-04Online:2024-07-18Published:2023-09-11 -
Contact:Cheng Huanzhi, Master, School of Stomatology, Jining Medical University, Jining 272000, Shandong Province, China -
About author:Dong Xinyu, School of Stomatology, Jining Medical University, Jining 272000, Shandong Province, China -
Supported by:Jining Key Research and Development Project, No. 2021YXNS019 (to FHX); Jining Medical University Educational Teaching Research Project, No. Y2020023 (to CHZ)
CLC Number:
Cite this article
Dong Xinyu, Dong Xinyue, Wang Wanting, Fan Haixia, Cheng Huanzhi. The effective components of Chinese medicine combined with scaffold materials promote bone tissue regeneration[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3240-3245.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 支架材料 骨组织工程中理想的载中药支架材料应该满足:①具有良好的生物相容性;②能够填补骨缺损;③拥有载药能力和控释能力;④具有骨传导性。支架材料为特定的细胞提供结构支撑作用,可引导组织再生和控制组织结构。理想的支架材料是中药及其有效成分可以应用到骨组织工程的前提。支架材料从单一生物材料到复合材料的发展,为搭载中药提供了可能性。单一生物材料可以分为天然生物衍生材料、人工合成无机材料、高分子有机合成材料三大类。 天然生物衍生材料有胶原、壳聚糖、藻酸盐、珊瑚骨、天然骨、糖胺聚糖等[9],具有接近人体的网架结构、力学性能和部分活性因子,骨传导性良好,具有促进成骨的作用[10]。壳聚糖有抗微生物活性,可促进血管生成[11]。这些材料来源丰富、制作简便、价格低廉,然而此类材料的纯净度和最终质量很难控制,且机械支撑能力差。 人工合成无机材料主要包括磷酸钙水泥、羟基磷灰石、β-磷酸三钙、生物活性玻璃陶瓷等,其取材便利且制备方法简单,具有良好的骨传导性,在机体中有一定的降解度,且降解产物一般对人体无害。但此类材料有各自不足之处:β-磷酸三钙刚性及脆性高、加工困难[12];生物活性玻璃陶瓷韧性差且不易降解;碳酸钙的机械性能不足;羟基磷灰石的力学强度低于正常骨且无骨诱导作用,只能作为充填材料[13]。 高分子有机合成材料包括聚乳酸、聚甲基丙烯酸甲酯、聚羟基乙酸等,材料性质稳定、可塑性良好、降解周期可以调节,在实验研究中也表现出良好的成骨作用。但是,临床应用的纯高分子有机合成材料骨传导性差,表现出欠佳的力学性能,降解后产生的酸性物质易引起炎症反应,以上缺陷限制了某些高分子有机合成材料在组织工程中的应用[14-15]。 单一生物材料在机械强度、免疫原性、降解度等方面均有各自的缺陷,无法达到骨组织工程对载药支架材料的要求。通过不同生物材料的有机结合,加持新的制作工艺,研究者们制备了不同的复合材料。复合材料在生物降解性、载药性能、抗炎作用以及增强种子细胞黏附、增殖和分化与诱导骨血管化等方面有了改善,为搭载中药有效成分提供了更多的可能。 壳聚糖-β-甘油磷酸盐水凝胶是一种智能药物系统,在室温下呈液体形式,随着温度的升高可变成凝胶[16],其具有三维和高度多孔的结构,可以为骨再生过程中各种细胞的生长和增殖提供合适的环境[17],并且可以存储大量药物,在特定位置以受控速率将其释放到环境中[18];加入不同的成分,可以控制其形成凝胶的时间、温度和pH值。通过在壳聚糖-β-甘油磷酸盐水凝胶中添加明胶、聚乙烯醇和氧化石墨烯等其他化合物,可以改变药物释放速率和凝胶形成时间。柳玉梅[19]将黄连素负载于壳聚糖-β-甘油磷酸盐水凝胶上证明了其控释效果。宁钰等[20]利用溶液铸浇-粒子滤沥法构建载淫羊藿苷/凹凸棒石/Ⅰ型胶原/聚己内酯复合支架,发现其具有多孔性和表面积大的特点,孔隙直径为 1-500 μm,该孔隙有助于细胞的迁入和营养物质的交换,而表面积大则有利于细胞黏附、生长和分化,实验结果表明此支架可以控制药物微量长效释放。纳米材料和3D打印技术的出现使支架材料的各种性能有了进一步提升,可以精细控制孔隙率和内部结构,从而在促进血管化和调整支架降解速度方面取得重大突破[21-22]、载药功能也有一定的提升。 2.2 中药成分 中医药已被世界卫生组织列为有效的全球医学项目,其成分错综复杂,主要通过抗炎、促进早期血管的生成和诱导成骨促进骨再生。与西药相比,中药具有低毒性和多效性。将中药成分依托支架材料在病灶处缓慢释放,可长时间维持有效浓度。与典型的口服给药途径相比,局部给药方式可以避免肝脏和肠道酶的首过效应,降低全身不良反应。中药按照有效活性成分可分为黄酮类化合物、非黄酮类多酚、生物碱、苷等种类。该文对中药的成骨作用及其搭载不同支架材料后在骨组织工程的应用及疗效进行总结。 2.2.1 黄酮类化合物 黄酮类化合物广泛存在于自然界的植物中,属植物次生代谢产物。黄酮类化合物是以黄酮(2-苯基色原酮)为母核而衍生的一类化合物,其中包括黄酮的同分异构体及其氢化和还原产物,也即以C6-C3-C6为基本碳架的一系列化合物。 黄酮类化合物可双向调节成骨细胞:①一方面,通过上调碱性磷酸酶、增加骨形成蛋白的表达诱导成骨细胞的形成。表没食子儿茶素,黄腐酚、淫羊藿苷可以促进矮小相关转录因子2的表达,从而刺激骨髓间充质干细胞的分化[23-25]。橙皮素、水飞蓟素能通过SMAD蛋白通路增加成骨细胞的形成[26-27]。②另一方面,抑制破骨细胞活性。黄酮类药物被证明具有雌激素作用,通过与雌激素受体结合可有效上调成骨细胞分泌骨保护素的含量并减少破骨细胞分化因子,与骨保护素相作用的破骨细胞分化因子无法再与破骨细胞结合,从而抑制破骨细胞的活化[28-30]。另外,槲皮素、淫羊藿苷也可以通过减少自噬抑制破骨细胞的分化[31]。 此外,黄酮类化合物有显著的抗炎能力,是炎性递质(环氧合酶或脂氧合酶)的显著抑制剂,可降低肿瘤坏死因子α、白细胞介素1β、白细胞介素6等炎性因子的水平。黄酮类化合物通过抑制磷脂酶A2、环氧合酶或脂氧合酶的活性来减少类花生酸的产生。黄酮类化合物是磷酸二酯酶、蛋白激酶、组胺释放的抑制剂,也是抗炎活性的基因转录的调节剂,例如:槲皮素可以抑制核因子κB/核苷酸结合寡聚化结构域样受体蛋白3信号通路[32];黄腐酚可通过NRF2抗氧化信号通路和上调下游因子血红素氧合酶1发挥抗炎作用[33]。 类黄酮的供氢能力决定了它们的抗氧化应激特性,此类药物具有显著的自由基清除能力,这主要归因于其中羟基取代基的高反应性,从而能够保护身体免受活性氧的损害。另外,黄酮类化合物的雌激素样作用可以通过非经典信号通路在骨细胞中作为活性氧作用的拮抗剂[34]。类黄酮可促进血管的生成,柚皮苷通过上调趋化因子、趋化因子受体的表达水平激活PI3K/Akt信号通路,从而激发了内皮祖细胞的增殖,促进新生血管的生成[35]。然而,黄腐酚可通过抑制内皮细胞的核因子κB和Akt通路来抑制血管的生成,这对于骨再生的血供起消极作用[36]。 基于黄酮类化合物诱导成骨细胞分化、抑制破骨细胞形成的促成骨作用,以及其显著的抗炎、抗氧化应激的特性,凸显了其在骨缺损的修复中的应用潜能。研究者将类黄酮类中药负载于各种支架材料,证明了载药支架材料在分子水平、细胞水平和组织水平促进成骨的作用较未载药支架材料明显提高(表1)。同时在动物实验中,采用口服黄酮类化合物可促进复合支架材料在骨组织工程中的成骨作用,提示黄酮类化合物及黄酮类化合物复合支架材料在骨组织工程中有明显的促进成骨作用。"

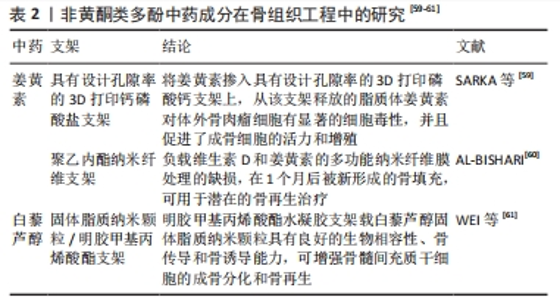
2.2.2 非黄酮类多酚 除了上述黄酮类化合物,多酚中的其他化合物——白藜芦醇和姜黄素有抗炎、抗氧化、促进骨生成的作用[47-48]。白藜芦醇和姜黄素可通过抑制核因子κB通路减少前列腺素2的产生。白藜芦醇可通过激活Sirt1/AMPK(腺嘌呤核糖核苷酸依赖的蛋白激酶)和Nrf2等信号通路来下调氧化酶的表达和活性,减少活性氧的生成[49-50]。姜黄素能降低脂多糖刺激下的大鼠牙龈成纤维细胞中骨保护素/核因子κB受体活化因子配体比率,并减少炎性因子白细胞介素1β和肿瘤坏死因子α的产生[51]。 在骨生成中,白藜芦醇和姜黄素通过雌激素受体依赖机制和与细胞外信号调节激酶激活的耦合来刺激人骨髓间充质干细胞的增殖和成骨细胞分化[52-53]。白藜芦醇可通过SIRT1/FOXO3A轴上调矮小相关转录因子2基因表达来促进人类间充质干细胞的成骨分化[54]。此外,白藜芦醇既可以减少趋化因子巨噬细胞炎性蛋白1α的产生,又能防止破骨细胞前体骨细胞前体细胞的迁移,最终使破骨细胞的形成受到有效抑制[55]。姜黄素能通过早期生长应答因子1促进人类牙周韧带干细胞的成骨分化,并增强人牙周膜干细胞在体内的活力和骨骼修复能力[52, 56]。白藜芦醇能基于间充质干细胞的热休克蛋白70启动子驱动的血管内皮生长因子A诱导促进小鼠模型中的血管生成[57]。但是姜黄素对血管有双重作用,在较低剂量下观察到姜黄素的促血管生成作用,而在较高剂量下抗血管生成[58],这提示在应用姜黄素时需要严格把握药物的浓度。 非黄酮类多酚以白藜芦醇和姜黄素为代表,有抗氧化、促进成骨、促进血管生成的作用。非黄酮类多酚复合支架材料的结合可以起到抗炎杀菌、促进骨组织再生的作用,在临床治疗骨缺损中有着较大的应用潜力(表2)。"


2.2.3 生物碱 生物碱大多数有复杂的环状结构,是中草药中重要的有效成分之一,可以促进间充质干细胞的分化、改善成骨细胞增殖并抑制破骨细胞形成,目前已经成为主流的抗骨质疏松药物,同时具有显著的抗炎作用[62]。由于其促进成骨细胞活化及抗炎作用,已成为骨组织工程研究关注的药物。 黄连素属于生物碱,能抑制炎症的形成和发展,其可以通过相关信号通路降低环氧化酶2和前列腺素E2的合成,并抑制炎症反应中的白细胞介素1、组胺、一氧化氮等的形成。同时,黄连素在传统医学中被广泛用作抗感染药物[63],目前已被证明对多种细菌具有抗菌活性,包括无乳链球菌[64]、胸膜肺炎放线杆菌[65]、不同的葡萄球菌菌株和痢疾志贺氏菌。黄连素还有效防止了表皮葡萄球菌生物膜在钛盘表面上的形成,并且是用于治疗假体周围感染的潜在药剂[66-67]。 黄连素通过降低骨质疏松小鼠血清肿瘤坏死因子α和白细胞介素6的水平来抑制骨吸收[68],还可以通过增加β-连环蛋白/T细胞因子的转录活性促进骨髓间充质干细胞的成骨分化[69],上调磷酸化p38 MAPK(促分裂素原活化蛋白激促分裂素原活化蛋白激酶)的表达促进成骨细胞分化[70];此外,其可以抑制核转录因子核因子κB和蛋白激酶B信号传导,从而抑制破骨细胞的活性[71]。 黄连素同时被发现具有促进血管生成的作用。BANAEI等[72]发现用 10 mg/kg 富含黄连素的提取物(每周5 d通过管饲法)治疗显著减小了心肌梗死的大小,并增加了心肌缺血再灌注损伤大鼠血管生成促进因子的表达,包括重组人源血管内皮生长因子、碱性成纤维细胞生长因子2和 血小板反应素1。ZHU等[73]在心肌梗死损伤小鼠中用黄连素治疗,结果显示可导致miR-29b升高可激活蛋白激酶B(Akt)信号通路,从而促进血管生成和细胞增殖和迁移。研究者目前研究了黄连素与载药支架结合后应用于骨组织工程的效果,证明了其在抑菌和促进成骨方面的作用(表3)。"


2.2.4 苷类 三七总皂苷通过调控PI3K/Akt/mTOR信号通路蛋白的表达影响血管内皮生长因子和缺氧诱导因子1α水平,从而对机体组织新生血管的形成产生影响,促进骨痂组织新生血管的形成[77]。柳毅等[78]将三七总皂苷用于骨缺损的动物模型,相比对照组,实验组模型骨痂面积、外骨痂厚度均得到了有效增加,表明三七总皂苷可能通过诱导骨痂增生从而加快成骨进程;此外,还发现三七总皂苷也可诱导小鼠间充质干细胞向成骨细胞分化。 人参皂苷Rb1可通过上调矮小相关转录因子2基因蛋白表达调节Wnt/β-catenin信号通路,促进成骨细胞分化。研究发现,人参皂苷Rb1通过作用于体外培养的小鼠胚胎成骨细胞前体细胞(MC3T3-E1)可激活其中的骨形成标志物,如矮小相关转录因子2基因、Ⅰ型胶原蛋白、碱性磷酸酶和骨钙素,提高细胞内碱性磷酸酶活性、刺激成骨细胞分化。此外,人参皂苷Rb1也可促进小鼠胚胎成骨细胞前体细胞的矿化[79]。 苷类可以促进机体组织新生血管的形成,有明确的促成骨作用,将苷类中药负载于骨组织工程的支架上,可以促进成骨分化和血管生成,具有良好的骨修复效果,同时提高人牙髓干细胞成骨相关蛋白的表达,促进硬组织的再生(表4)。"

| [1] 梁健,苏睿,宋泉生,等.柚皮苷对骨细胞的作用以及负载柚皮苷的羟基磷灰石复合支架治疗骨缺损的研究[J].医学研究杂志, 2023,52(5):18-20,110. [2] 陈泽驹,冯少龙,刘桂宏,等.基于骨组织工程的骨再生策略[J].中国医药生物技术,2022,17(5):440-444. [3] 刘迪.三七总皂苷调控TGF-β1/Smads信号传导促进牵张成骨新骨形成的机制研究[D].南宁:广西医科大学,2020. [4] ROSETI L, PARISI V, PETRETTA M, et al. Scaffolds for Bone Tissue Engineering: State of the art and new perspectives. Mater Sci Eng C Mater Biol Appl. 2017;78:1246-1262. [5] 关德强,于波,李欣,等.中药促进骨再生的研究进展[J].时珍国医国药,2018,29(4):956-958. [6] 王伟佳,唐硕,马应钧,等.纳米羟基磷灰石复合材料在骨组织工程支架中的研究现状[J].湖南师范大学自然科学学报,2022,45(4):123-129. [7] 陈艺菲,郑赛男,阙林.3D打印复合材料骨组织工程支架及其在颌面骨再生中的研究进展[J].山东医药, 2022,62(25):83-86. [8] 熊伟,袁灵梅,钱国文,等.“补肾壮骨”中药应用于骨组织工程支架修复节段性骨缺损[J].中国组织工程研究,2023,27(21):3438-3444. [9] BROVOLD M, ALMEIDA JI, PLA-PALACIN I, et al. Naturally-Derived Biomaterials for Tissue Engineering Applications. Adv Exp Med Biol. 2018; 1077:421-449. [10] INSUASTI-CRUZ E, SUAREZ-JARAMILLO V, MENA UK, et al. Natural Biomaterials from Biodiversity for Healthcare Applications. Adv Healthc Mater. 2022;11(1):e2101389. [11] VENKATESAN J, KIM SK. Chitosan composites for bone tissue engineering--an overview. Mar Drugs. 2010;8(8):2252-2266. [12] 杨为中,周大利,尹光福,等.骨组织工程支架材料磷酸钙双相生物陶瓷的研究进展[J].硅酸盐学报,2004,32(9):1143-1149. [13] 李慎松,文益民,李志琳,等.骨髓基质干细胞复合羟基磷灰石-磷酸三钙修复骨缺损过程中的微循环变化[J].中国组织工程研究与临床康复,2007,10(35):7050-7052. [14] JING Y, QUAN C, LIU B, et al. A Mini Review on the Functional Biomaterials Based on Poly(lactic acid) Stereocomplex. Polym Rev. 2016;56(2):262-286. [15] KIM DY, KWON DY, KWON JS, et al. Stimuli-Responsive InjectableIn situ-Forming Hydrogels for Regenerative Medicines. Polym Rev. 2015;55(3):207-452. [16] CHENITE A, CHAPUT C, WANG D, et al. Novel injectable neutral solutions of chitosan form biodegradable gels in situ. Biomaterials. 2000;21(21):2155-2161. [17] AHMADI R, DE BRUIJN JD. Biocompatibility and gelation of chitosan-glycerol phosphate hydrogels. J Biomed Mater Res A. 2008;86(3):824-832. [18] SELIKTAR D. Designing cell-compatible hydrogels for biomedical applications. Science. 2012;336(6085):1124-1128. [19] 柳玉梅.负载黄连素的壳聚糖温敏水凝胶的制备及体外抑菌研究[D].南京:南京大学,2020. [20] 宁钰,秦文,任亚辉,等.载淫羊藿苷/凹凸棒石/Ⅰ型胶原/聚己内酯复合支架修复兔胫骨缺损的实验研究[J].中国修复重建外科杂志, 2019,33(9):1181-1189. [21] 丁焕文,涂强,王迎军,等.数字化骨科手术新方法的建立及其临床广泛应用[J].中国骨科临床与基础研究杂志,2010,2(2):92-97. [22] 周静,冯卓卓,罗洁,等.纳米支架材料在骨组织工程应用中的优势[J].临床口腔医学杂志,2018,34(10):633-635. [23] 姜涛,凌翠敏,陈庆真,等.淫羊藿苷通过提高自噬促进成骨细胞分化防治骨质疏松[J].中国组织工程研究,2021,25(17):2643-2649. [24] JEONG HM, HAN EH, JIN YH, et al. Xanthohumol from the hop plant stimulates osteoblast differentiation by RUNX2 activation. Biochem Biophys Res Commun. 2011;409(1):82-89. [25] LIN SY, KANG L, WANG CZ, et al. (-)-Epigallocatechin-3-Gallate (EGCG) Enhances Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells. Molecules. 2018;23(12):3221. [26] XUE D, CHEN E, ZHANG W, et al. The role of hesperetin on osteogenesis of human mesenchymal stem cells and its function in bone regeneration. Oncotarget. 2017;8(13):21031-21043. [27] YING X, SUN L, CHEN X, et al. Silibinin promotes osteoblast differentiation of human bone marrow stromal cells via bone morphogenetic protein signaling. Eur J Pharmacol. 2013;721(1-3):225-230. [28] CHEN C, WU M, LEI H, et al. A Novel Prenylflavonoid Icariside I Ameliorates Estrogen Deficiency-Induced Osteoporosis via Simultaneous Regulation of Osteoblast and Osteoclast Differentiation. ACS Pharmacol Transl Sci. 2023;6(2):270-280. [29] GUO AJ, CHOI RC, ZHENG KY, et al. Kaempferol as a flavonoid induces osteoblastic differentiation via estrogen receptor signaling. Chin Med. 2012;7:10. [30] WU GJ, CHEN KY, YANG JD, et al. Naringin Improves Osteoblast Mineralization and Bone Healing and Strength through Regulating Estrogen Receptor Alpha-Dependent Alkaline Phosphatase Gene Expression. J Agric Food Chem. 2021;69(44):13020-13033. [31] 张波,胡凌云,苟林,等.槲皮素对人骨髓间充质干细胞增殖和成骨分化的影响及分子机制[J].中国骨质疏松杂志,2022,28(12):1765-1769. [32] 张文静.槲皮素对炎症状态下人牙周膜干细胞成骨损伤的改善作用及机制探究[D].济南:山东大学,2021. [33] LEE IS, LIM J, GAL J, et al. Anti-inflammatory activity of xanthohumol involves heme oxygenase-1 induction via NRF2-ARE signaling in microglial BV2 cells. Neurochem Int. 2011;58(2):153-160. [34] WEAVER CM, ALEKEL DL, WARD WE, et al. Flavonoid intake and bone health. J Nutr Gerontol Geriatr. 2012;31(3):239-253. [35] 张敏,张晓明,刘童斌.柚皮苷在骨组织再生领域的应用潜力[J].中国组织工程研究,2023,27(5):787-792. [36] ALBINI A, DELL’EVA R, VENE R, et al. Mechanisms of the antiangiogenic activity by the hop flavonoid xanthohumol: NF-kappaB and Akt as targets. FASEB J. 2006;20(3):527-529. [37] 宁兆荣,郭延伟,李松,等.脂肪干细胞复合载淫羊藿苷支架材料对兔下颌骨缺损的修复[J].实用口腔医学杂志,2013,29(5):611-615. [38] XiA L, LI Y, ZHOU Z, et al. Icariin delivery porous PHBV scaffolds for promoting osteoblast expansion in vitro. Mater Sci Eng C Mater Biol Appl. 2013;33(6):3545-3552. [39] XIE Y, SUN W, YAN F, et al. Icariin-loaded porous scaffolds for bone regeneration through the regulation of the coupling process of osteogenesis and osteoclastic activity. Int J Nanomedicine. 2019;14:6019-6033. [40] JI Y, WANG L, WATTS DC, et al. Controlled-release naringin nanoscaffold for osteoporotic bone healing. Dent Mater. 2014;30(11):1263-1273. [41] YU X, SHEN G, SHANG Q, et al. A Naringin-loaded gelatin-microsphere/nano-hydroxyapatite/silk fibroin composite scaffold promoted healing of critical-size vertebral defects in ovariectomised rat. Int J Biol Macromol. 2021;193(Pt A):510-518. [42] SONG JE, JEON YS, TIAN J, et al. Evaluation of silymarin/duck’s feet-derived collagen/hydroxyapatite sponges for bone tissue regeneration. Mater Sci Eng C Mater Biol Appl. 2019;97:347-355. [43] KHOOBI MM, NADDAF H, HOVEIZI E, et al. Silymarin effect on experimental bone defect repair in rat following implantation of the electrospun PLA/carbon nanotubes scaffold associated with Wharton’s jelly mesenchymal stem cells. J Biomed Mater Res A. 2020;108(9):1944-1954. [44] LEENA RS, VAIRAMANI M, SELVAMURUGAN N. Alginate/Gelatin scaffolds incorporated with Silibinin-loaded Chitosan nanoparticles for bone formation in vitro. Colloids Surf B Biointerfaces. 2017;158:308-318. [45] REN M, WANG X, HU M, et al. Enhanced bone formation in rat critical-size tibia defect by a novel quercetin-containing alpha-calcium sulphate hemihydrate/nano-hydroxyapatite composite. Biomed Pharmacother. 2022;146:112570. [46] PREETHI AM, BELLARE JR. Concomitant Effect of Quercetin- and Magnesium-Doped Calcium Silicate on the Osteogenic and Antibacterial Activity of Scaffolds for Bone Regeneration. Antibiotics (Basel). 2021;10(10):1170. [47] MURGIA D, MAUCERI R, CAMPISI G, et al. Advance on Resveratrol Application in Bone Regeneration: Progress and Perspectives for Use in Oral and Maxillofacial Surgery. Biomolecules. 2019;9(3):94. [48] 王海英,张宇琪,孙昊天,等.姜黄素及其衍生物的作用及机制[J].生理科学进展,2022,53(4):271-275. [49] XIA N, DAIBER A, FORSTERMANN U, et al. Antioxidant effects of resveratrol in the cardiovascular system. Br J Pharmacol. 2017;174(12):1633-1646. [50] TAMAKI N, CRISTINA OR, INAGAKI Y, et al. Resveratrol improves oxidative stress and prevents the progression of periodontitis via the activation of the Sirt1/AMPK and the Nrf2/antioxidant defense pathways in a rat periodontitis model. Free Radic Biol Med. 2014;75:222-229. [51] XIAO CJ, YU XJ, XIE JL, et al. Protective effect and related mechanisms of curcumin in rat experimental periodontitis. Head Face Med. 2018;14(1):12. [52] TAN L, CAO Z, CHEN H, et al. Curcumin reduces apoptosis and promotes osteogenesis of human periodontal ligament stem cells under oxidative stress in vitro and in vivo. Life Sci. 2021;270:119125. [53] DAI Z, LI Y, QUARLES LD, et al. Resveratrol enhances proliferation and osteoblastic differentiation in human mesenchymal stem cells via ER-dependent ERK1/2 activation. Phytomedicine. 2007;14(12):806-814. [54] TSENG PC, HOU SM, CHEN RJ, et al. Resveratrol promotes osteogenesis of human mesenchymal stem cells by upregulating RUNX2 gene expression via the SIRT1/FOXO3A axis. J Bone Miner Res. 2011;26(10):2552-2563. [55] LIANG Z, XUE Y, WANG T, et al. Curcumin inhibits the migration of osteoclast precursors and osteoclastogenesis by repressing CCL3 production. BMC Complement Med Ther. 2020;20(1):234. [56] SHI W, LING D, ZHANG F, et al. Curcumin promotes osteogenic differentiation of human periodontal ligament stem cells by inducting EGR1 expression. Arch Oral Biol. 2021;121:104958. [57] CHEN YB, LAN YW, HUNG TH, et al. Mesenchymal stem cell-based HSP70 promoter-driven VEGFA induction by resveratrol promotes angiogenesis in a mouse model. Cell Stress Chaperones. 2015;20(4):643-652. [58] HOSSAIN DM, BHATTACHARYYA S, DAS T, et al. Curcumin: the multi-targeted therapy for cancer regression. Front Biosci (Schol Ed). 2012;4(1):335-355. [59] SARKAR N, BOSE S. Liposome-Encapsulated Curcumin-Loaded 3D Printed Scaffold for Bone Tissue Engineering. ACS Appl Mater Interfaces. 2019;11(19):17184-17192. [60] AL-BISHARI AM, AL-SHAAOBI BA, AL-BISHARI AA, et al. Vitamin D and curcumin-loaded PCL nanofibrous for engineering osteogenesis and immunomodulatory scaffold. Front Bioeng Biotechnol. 2022;10:975431. [61] WEI B, WANG W, LIU X, et al. Gelatin methacrylate hydrogel scaffold carrying resveratrol-loaded solid lipid nanoparticles for enhancement of osteogenic differentiation of BMSCs and effective bone regeneration. Regen Biomater. 2021;8(5):b44. [62] LI S, LIU X, CHEN X, et al. Research Progress on Anti-Inflammatory Effects and Mechanisms of Alkaloids from Chinese Medical Herbs. Evid Based Complement Alternat Med. 2020;2020:1303524. [63] BOBEREK JM, STACH J, GOOD L. Genetic evidence for inhibition of bacterial division protein FtsZ by berberine. PLoS One. 2010;5(10):e13745. [64] PENG L, KANG S, YIN Z, et al. Antibacterial activity and mechanism of berberine against Streptococcus agalactiae. Int J Clin Exp Pathol. 2015;8(5): 5217-5223. [65] KANG S, LI Z, YIN Z, et al. The antibacterial mechanism of berberine against Actinobacillus pleuropneumoniae. Nat Prod Res. 2015;29(23):2203-2206. [66] WANG X, QIU S, YAO X, et al. Berberine inhibits Staphylococcus epidermidis adhesion and biofilm formation on the surface of titanium alloy. J Orthop Res. 2009;27(11):1487-1492. [67] WANG X, YAO X, ZHU Z, et al. Effect of berberine on Staphylococcus epidermidis biofilm formation. Int J Antimicrob Agents. 2009;34(1):60-66. [68] HE XF, ZHANG L, ZHANG CH, et al. Berberine alleviates oxidative stress in rats with osteoporosis through receptor activator of NF-kB/receptor activator of NF-kB ligand/osteoprotegerin (RANK/RANKL/OPG) pathway. Bosn J Basic Med Sci. 2017;17(4):295-301. [69] TAO K, XIAO D, WENG J, et al. Berberine promotes bone marrow-derived mesenchymal stem cells osteogenic differentiation via canonical Wnt/beta-catenin signaling pathway. Toxicol Lett. 2016;240(1):68-80. [70] LEE HW, SUH JH, KIM HN, et al. Berberine promotes osteoblast differentiation by Runx2 activation with p38 MAPK. J Bone Miner Res. 2008; 23(8):1227-1237. [71] HU JP, NISHISHITA K, SAKAI E, et al. Berberine inhibits RANKL-induced osteoclast formation and survival through suppressing the NF-kappaB and Akt pathways. Eur J Pharmacol. 2008;580(1-2):70-79. [72] BANAEI P, NAZEM F, NAZARI A, et al. Preconditioning Effect of High-Intensity Interval Training (HIIT) and Berberine Supplementation on the Gene Expression of Angiogenesis Regulators and Caspase-3 Protein in the Rats with Myocardial Ischemia-Reperfusion (IR) Injury. Biomed Res Int. 2020;2020:4104965. [73] ZHU ML, YIN YL, PING S, et al. Berberine promotes ischemia-induced angiogenesis in mice heart via upregulation of microRNA-29b. Clin Exp Hypertens. 2017;39(7):672-679. [74] 柳玉梅.负载黄连素的壳聚糖温敏水凝胶的制备及体外抑菌研究[D].南京:南京大学,2020. [75] 汪芳,陈云平,苏香萍.抗菌止血壳聚糖/黄连素多孔干凝胶的制备及其表征[J].中国组织工程研究,2017,21(6):899-905. [76] MA L, YU Y, LIU H, et al. Berberine-releasing electrospun scaffold induces osteogenic differentiation of DPSCs and accelerates bone repair. Sci Rep. 2021;11(1):1027. [77] 张晓军,刘健,万磊,等.基于缺氧诱导因子及PI3K-AKT-mTOR信号通路诱导佐剂关节炎滑膜血管新生的实验观察[J].中国临床保健杂志, 2018,21(1):84-87. [78] 柳毅,陈建治.三七总皂苷及其诱导成骨的试验和机制[J].国际口腔医学杂志,2015,42(1):75-78. [79] KIM HM, KIM DH, HAN HJ, et al. Ginsenoside Re Promotes Osteoblast Differentiation in Mouse Osteoblast Precursor MC3T3-E1 Cells and a Zebrafish Model. Molecules. 2016;22(1):42. [80] LUO P, YU L, LIN Q, et al. Strontium Modified Calcium Sulfate Hemihydrate Scaffold Incorporating Ginsenoside Rg1/Gelatin Microspheres for Bone Regeneration. Front Bioeng Biotechnol. 2020;8:888. [81] CHEN CY, SHIE MY, LEE AK, et al. 3D-Printed Ginsenoside Rb1-Loaded Mesoporous Calcium Silicate/Calcium Sulfate Scaffolds for Inflammation Inhibition and Bone Regeneration. Biomedicines. 2021;9(8):907. [82] 秦宇星,任前贵,沈佩锋,等.组织工程技术治疗骨缺损:应用于临床还有多远?[J].中国组织工程研究,2021,25(29):4703-4708. [83] 陈凯,张超,王路,等.骨组织工程中促进血管化策略的研究进展[J].中国骨伤,2015,28(4):383-388. [84] 余晓宏.云南白药对体内植入人牙周膜成纤维细胞-Bio-Oss Collagen成骨分化的组织学研究[D]. 昆明:昆明医科大学,2014. [85] WU GS, LI HK, ZHANG WD. Metabolomics and its application in the treatment of coronary heart disease with traditional Chinese medicine. Chin J Nat Med. 2019;17(5):321-330. [86] SUO T, WANG H, LI Z. Application of proteomics in research on traditional Chinese medicine. Expert Rev Proteomics. 2016;13(9):873-881. [87] XIN T, ZHANG Y, PU X, et al. Trends in herbgenomics. Sci China Life Sci. 2019; 62(3):288-308. |
| [1] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [2] | Dai Yuexing, Zheng Liqin, Wu Minhui, Li Zhihong, Li Shaobin, Zheng Desheng, Lin Ziling. Effect of vessel number on computational fluid dynamics in vascular networks [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1206-1210. |
| [3] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [4] | Zhang Ya, Mu Qiuju, Wang Zilin, Liu Hongjie, Zhu Lili. Hydrogel loaded with platelet-rich plasma promotes wound healing in diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 690-696. |
| [5] | Lan Weiwei, Yu Yaodong, Huang Di, Chen Weiyi. In vitro degradation behavior of Mg-Zn-Ca alloys [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 717-723. |
| [6] | Zhou Xiaowen, Fu Zuchang, Huang Fei, Ai Jianguo, Zhao Feng. Bone defect blocked by bone cement segmental filling in single-plane tibial bone transport [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 736-740. |
| [7] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [8] | Yin Tong, Yang Jilei, Li Yourui, Liu Zhuoran, Jiang Ming. Application of core-shell structured nanofibers in oral tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 766-770. |
| [9] | Wang Yeyuan, Du Yilang, Yu Dehao, Ning Fengting, Bai Bing. Effect of micro-arc oxidation treatment on biological activity of medical metals [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 771-776. |
| [10] | Wang Jiani, Chen Junyu. Angiogenesis mechanism of metal ions and their application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 804-812. |
| [11] | Yang Yuqing, Chen Zhiyu. Role and application of early transient presence of M1 macrophages in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 594-601. |
| [12] | Zhou Shibo, Guan Jianbin, Yu Xing, Zhao He, Yang Yongdong, Liu Tao. Animal models of femoral bone defects: preparation status and characteristics [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 633-638. |
| [13] | Kong Xiangyu, Wang Xing, Pei Zhiwei, Chang Jiale, Li Siqin, Hao Ting, He Wanxiong, Zhang Baoxin, Jia Yanfei. Biological scaffold materials and printing technology for repairing bone defects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 479-485. |
| [14] | Xu Jing, Lyu Huixin, Bao Xin, Zhang Yi, Wang Yihan, Zhou Yanmin. Application of near infrared responsive hydrogels in tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 486-492. |
| [15] | Dai Jing, Liu Shasha, Shen Mingjing. Exosome-loaded injectable hydrogel for repairing bone defects around implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 347-354. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||