Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (5): 717-723.doi: 10.12307/2024.263
Previous Articles Next Articles
In vitro degradation behavior of Mg-Zn-Ca alloys
Lan Weiwei1, 2, Yu Yaodong1, Huang Di1, 2, Chen Weiyi1, 2
- 1Research Center for Nano-Biomaterials and Regenerative Medicine, College of Biomedical Engineering, Taiyuan University of Technology, Taiyuan 030024, Shanxi Province, China; 2Shanxi-Zheda Institute of Advanced Materials and Chemical Engineering, Taiyuan 030032, Shanxi Province, China
-
Received:2023-03-06Accepted:2023-04-23Online:2024-02-18Published:2023-08-16 -
Contact:Huang Di, PhD, Professor, Master’s supervisor, Research Center for Nano-Biomaterials and Regenerative Medicine, College of Biomedical Engineering, Taiyuan University of Technology, Taiyuan 030024, Shanxi Province, China; Shanxi-Zheda Institute of Advanced Materials and Chemical Engineering, Taiyuan 030032, Shanxi Province, China -
About author:Lan Weiwei, PhD, Lecturer, Research Center for Nano-Biomaterials and Regenerative Medicine, College of Biomedical Engineering, Taiyuan University of Technology, Taiyuan 030024, Shanxi Province, China; Shanxi-Zheda Institute of Advanced Materials and Chemical Engineering, Taiyuan 030032, Shanxi Province, China -
Supported by:National Natural Science Foundation of China, No. 12272253 (to HD), No. 12202302 (to LWW); a grant from Shanxi-Zheda Institute of Advanced Materials and Chemical Engineering, No. 2021SX-AT008 (to HD)
CLC Number:
Cite this article
Lan Weiwei, Yu Yaodong, Huang Di, Chen Weiyi. In vitro degradation behavior of Mg-Zn-Ca alloys[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 717-723.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
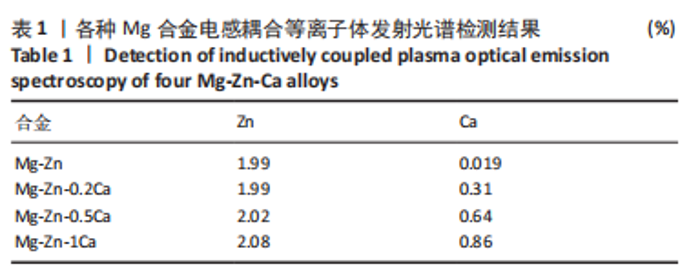
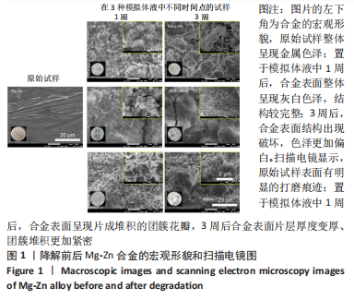
2.1 各种Mg合金结构和成分分析 电感耦合等离子体发射光谱表征结果如表1所示,该结果证实,实际所制备合金成分参数基本符合前期设计参数。图1-4中,左下角所示为上述4种合金初始状态和在3种降解环境下不同时间节点的试样降解整体形貌变化,随着降解时间的推移,Mg合金表面有大量的白色沉积物质生成,并且不同Ca含量的Mg合金在不同降解液体中整体的降解形貌和速率也有所不同,整体来看,试样在生理盐水中降解速度最快,其次为PBS,在Hank’s液中降解速率最慢;在生理盐水中的降解速率快慢为:Mg-Zn < Mg-Zn-0.2Ca < Mg-Zn-0.5Ca < Mg-Zn-1Ca,在PBS和Hank’s液中的降解速率快慢为:Mg-Zn < Mg-Zn-0.2Ca≈Mg-Zn-0.5Ca < Mg-Zn-1Ca。"
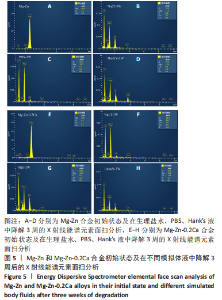
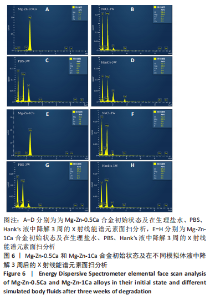
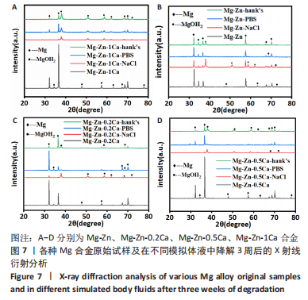
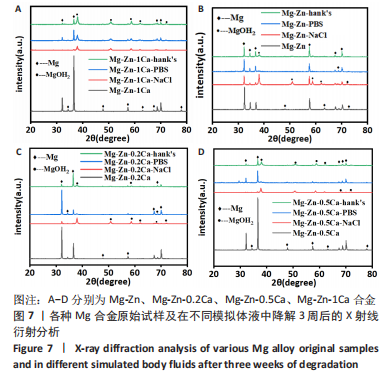
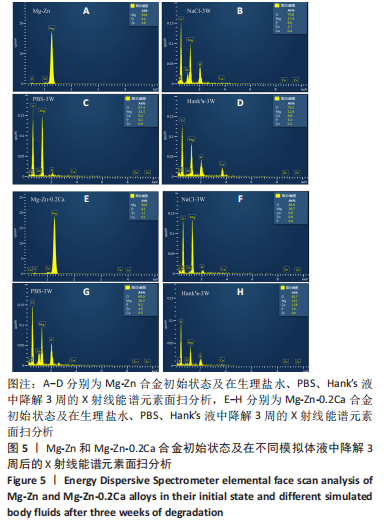
为了进一步明确试样在不同降解节点内的微观结构变化,进行了扫描电镜分析,如图1-4所示,所有样品表面都观察到不规则的纳米级结晶片状结构,但形貌不同,有呈现疏松、多孔相交叉成团簇状,片层卷曲相交连成网状,还有的片层堆叠在一起。 为了阐明这些新生片层物质的成分,对降解3周的各组试样进行了X射线能谱元素分析,从图5,6中可以看到,主要是以O和Mg元素为主,根据Mg合金在体外降解过程中可能发生化学反应推测可能存在MgO和Mg(OH)2。X射线衍射的测试数据也验证了试样降解后的主要物质是Mg(OH)2,见图7所示。另外值得关注的是,在PBS和Hank’s液中检测到了P和Ca元素的生成,其中在Hank’s液中的含量要较高,从中可以推测出样品在降解的同时也有一定的矿化沉积出现。"
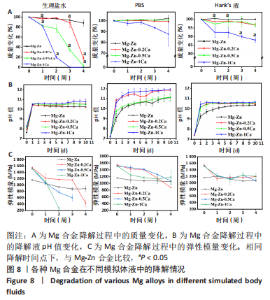
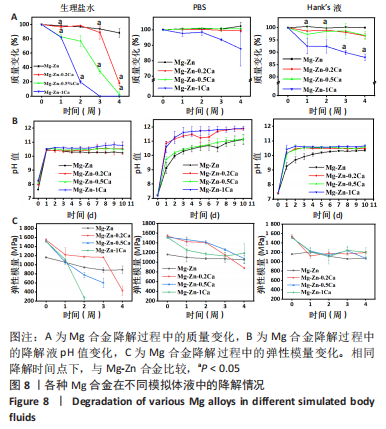
2.2 各种Mg合金在模拟体液中的失重测试结果 为了精确测定4种Mg合金的降解状况,对其质量变化进行了测试和记录(图8A)。整体来看,4种Mg合金随着降解时间的延长整体都有一定的质量损失,其中在生理盐水中降解最快,并且随着时间流逝腐蚀速度加快;在Hank’s液和PBS中降解后试样的质量变化不大,说明在Mg合金基体被腐蚀的同时产生的新物质,也就是发生了矿化。另外,随着Mg合金中钙含量的增加,其腐蚀速率明显加快。 2.3 各种Mg合金在模拟体液中的pH值测试结果 Mg合金作为一种活泼可降解的金属,在复杂人体环境中随着降解的发生会改变其周围环境pH值。根据图8B所示,在短期内(1 d)所有Mg合金样所处的液体环境pH值都在急剧上升,然后趋于平缓,最高pH值介于10.0-11.0之间;整体来看,Mg-Zn合金的pH值改变相较其他有Ca添加合金组略低,随着Ca含量的增加,液体环境pH值上升显著,这种变化与降解速度相对应,试样降解越快释放产物也越多,对周围环境的pH值改变也明显。值得注意的是,相较于其他两种降解环境,Mg合金在PBS中液体环境pH值上升幅度更大,最高为Mg-Zn-1Ca为11.0-12.0。 2.4 各种Mg合金的力学性能表征结果 模如图8C所示,在初始状态下,Mg-Zn合金弹性模量为(1 161±10) MPa,Ca的添加显著增强了Mg合金的力学性能,Mg-Zn-0.2Ca合金弹性模量增加至(1 546±33) MPa,随着钙含量进一步增加,Mg合金的弹性模量有所降低但无显著变化,Mg-Zn-0.5Ca和Mg-Zn-1Ca合金的弹性模量分别为(1 518±28),(1 516±9) MPa,添加Ca元素Mg合金的弹性模量高于Mg-Zn合金(P < 0.05)。随着降解时间的推移,4种Mg合金的力学性能在3种降解环境下均有所降低,其中在生理盐水溶液中的下降最低,Mg-Zn、Mg-Zn-0.2Ca、Mg-Zn-0.5Ca合金降解第3周的弹性模量分别为(882±59),(1 163±7),(602±106) MPa,Mg-Zn-1Ca合金在第3周已经几乎降解完全,降解2周的弹性模量为(268±35) MPa;在PBS和Hank’s液中4种Mg合金的力学性能也均有所降低,但组间比较无明显差异且弹性模量均能保持在初始状态的2/3左右。"
| [1] AGARWAL S, CURTIN J, DUFFY B, et al. Biodegradable magnesium alloys for orthopaedic applications: A review on corrosion, biocompatibility and surface modifications. Mater Sci Eng C. 2016;68:948-963. [2] ZHANG BP, WANG Y, GENG L. Research on Mg-Zn-Ca alloy as degradable biomaterial. Biomaterials. 2011:183-204. doi: 10.5772/23929 [3] XIAO X, ZHU QS, LI YC, et al. In vitro degradation and biocompatibility of Ca-P coated magnesium alloy. Chem Res Chin Univ. 2013;29(2):285-289. [4] XI Z, WU Y, XIANG S, et al. Corrosion resistance and biocompatibility assessment of a biodegradable hydrothermal-coated Mg–Zn–Ca alloy: an in vitro and in vivo study. ACS Omega. 2020;5(9):4548-4557. [5] WANG H, GUAN S, WANG Y, et al. In vivo degradation behavior of Ca-deficient hydroxyapatite coated Mg–Zn–Ca alloy for bone implant application. Colloids Surf. 2011;88(1):254-259. [6] WANG HX, GUAN SK, WANG X, et al. In vitro degradation and mechanical integrity of Mg-Zn-Ca alloy coated with Ca-deficient hydroxyapatite by the pulse electrodeposition process. Acta Biomater. 2010;6(5):1743-1748. [7] MARTYNENKO NS, ANISIMOVA NY, KISELEVSKIY MV, et al. In vitro biodegradation of resorbable magnesium alloys promising for implant development. Sovrem Tekhnologii Med. 2021;12(6):47-52. [8] WU YF, WANG YM, ZHAO DW, etal. In vivo study of micro-arc oxidation coated Mg alloy as a substitute for bone defect repairing: degradation behavior, mechanical properties, and bone respone. Colloids Surf. 2019;181(1):349-359. [9] WAN Y, XIONG G, LUO H, et al. Preparation and characterization of a new biomedical magnesium-calcium alloy. Mater Des. 2008;29(10): 2034-2037. [10] MAO G, JIN X, SUN J, et al. Microalloying design of biodegradable Mg-2Zn-0.05Ca promises improved bone-implant applications. ACS Biomater Sci Eng. 2021;7(6):2755-2766. [11] CHEN Y, DOU J, PANG Z, et al. Improving the corrosion resistance of micro-arc oxidation coated Mg-Zn-Ca alloy. RSC Adv. 2020;10(14): 8244-8254. [12] ZHU Y, ZHAO Q, ZHANG YH, et al. Hydrothermal synthesis of protective coating on magnesium alloy using de-ionized water. Surf Coat Technol. 2012;206(11-12):2961-2966. [13] ZHANG F, MA A, SONG D, et al. Improving in-vitro biocorrosion resistance of Mg-Zn-Mn-Ca alloy in Hank’s solution through addition of cerium. J Rare Earths. 2015;33(1):93-101. [14] PANEMANGALORE DB, SHABADI R, GUPTA M. Corrosion behavior, microstructure and mechanical properties of novel Mg-Zn-Ca-Er alloy for bio-medical applications. Metals. 2021;11(3):519. [15] ABDEL-GAWAD SA, SHOEIB MA. Corrosion studies and micro-structure of Mg-Zn-Ca alloys for biomedical applications. Surf Interfaces. 2019; 14:108-116. [16] CHENG YF, DU WB, LIU K, et al. Mechanical properties and corrosion behaviors of Mg4Zn0.2Mn0.2Ca alloy after long term in vitro degradation. T Nonferr Metal Soc. 2020;30(2):363-372. [17] AMUKARIMI S, MOZAFARI M. Biodegradable magnesium-based biomaterials: An overview of challenges and opportunities. MedComm (2020). 2021;2(2):123-144. [18] 黄艺聪,周盟,姚森,等.可降解生物医用金属在骨折内固定修复应用的研究进展[J].中华骨与关节外科杂志,2021,14(9):811-816. [19] LIN LH, LEE HP, YEH ML. Characterization of a sandwich PLGA-Gallic ACID-PLGA coating on mg alloy ZK60 for bioresorbable coronary artery stents. Materials (Basel). 2020;13(23):5538. [20] TRAN DT, CHEN FH, WU GL, et al. Influence of spin coating and dip coating with gelatin/hydroxyapatite for bioresorbable mg alloy orthopedic implants: in vitro and in vivo studies. ACS Biomater Sci Eng. 2023;9(2):705-718. [21] ZHANG Y, FNENG XH, HUANG QY, et al. Effect of the microstructure parameters on the corrosion characteristics of Mg-Zn-Ca alloy with columnar structure. J Magnes Alloy. 2022. doi.org/10.1016/j.jma. 2021.12.017 [22] WANG J, XU J, HOPKINS C, et al. Biodegradable magnesium based implants in orthopedics—a general review and perspectives. Adv Sci. 2020;7(8):1902443. [23] 崔珠殷,李淑波,付军健,等.晶粒尺寸对生物医用Mg-3Zn-0.2Ca-0.2Mn合金降解行为的影响[J].中国有色金属学报,2023,33(3): 665-677. [24] 张源,郑瑞宁,刘芸,等.生物医用镁合金表面改性涂层研究进展[J].表面技术,2022,51(8):146-155. [25] DONG J, LIN T, SHAO H, et al. Advances in degradation behavior of biomedical magnesium alloys: A review. J Alloys Compd. 2022;908: 164600. [26] ZHANG ZQ, WANG L, ZENG MQ, et al. Biodegradation behavior of micro-arc oxidation coating on magnesium alloy-from a protein perspective. Bioact Mater. 2020;5(2):398-409. [27] KHALILI MA, TAMJID E. Controlled biodegradation of magnesium alloy in physiological environment by metal organic framework nanocomposite coatings. Sci Rep. 2021;11:8645. [28] 吕娟,吴楠,娄洁,等.Mg含量和轧制变形对可降解血管支架用Zn-Mg合金力学性能影响研究[J].金属功能材料,2022,29(3):104-106. [29] CHOUDHARY L, RAMAN RKS, NIE JF. In vitro evaluation of degradation of a calcium phosphate coating on a mg-zn-ca alloy in a physiological environment. Corrosion. 2012;68(6):499-506. [30] LI Z, GU X, LOU S, et al. The development of binary Mg-Ca alloys for use as biodegradable materials within bone. Biomaterials. 2008;29(10):1329-1344. [31] 田亚强,赵冠璋,刘芸,等.生物可降解医用镁合金体内外降解行为研究进展[J].材料工程,2021,49(5):24-37. [32] JEONG YS, KIM WJ. Enhancement of mechanical properties and corrosion resistance of Mg-Ca alloys through microstructure refinement by indirect extrusion. Corros Sci. 2014;82:392-403. [33] YIN ZZ, QI WC, ZENG RC, et al. Advances in coatings on biodegradable magnesium alloys. J Magnes Alloy. 2020;8(1):42-65. [34] WANG Y, FU P, WANG N, et al. Challenges and solutions for the additive manufacturing of biodegradable magnesium implants. Engineering. 2020; 6(11):1267-1275. [35] TSAKIRIS V, TARDEI C, CLICINSCHI FM. Biodegradable Mg alloys for orthopedic implants-A review. J Magnes Alloy. 2021;9(6):1884-1905. [36] MD YUSOP AH, ALSAKKAF A, NOORDIN MA, et al. Degradation-triggered release from biodegradable metallic surfaces. J Biomed Mater Res B Appl Biomater. 2021;109(12):2184-2198. [37] WANG L, WANG C, WU S, et al. Influence of the mechanical properties of biomaterials on degradability, cell behaviors and signaling pathways: current progress and challenges. Biomater Sci. 2020;8(10):2714. [38] SUN Y, ZHANG B, WANG Y, et al. Preparation and characterization of a new biomedical Mg-Zn-Ca alloy. Mater Des. 2012;34:58-64. [39] ZHANG C, LIN J, NGUYEN N, et al. Antimicrobial Bioresorbable Mg-Zn-Ca Alloy for Bone Repair in a Comparison Study with Mg-Zn-Sr Alloy and Pure Mg. ACS Biomater Sci Eng. 2019;6(1):517-538. [40] LIN X, WU S, WU XX, et al. Biodegradable Mg-based alloys: biological implications and restorative opportunities. Int Mater Rev. 2022;68(4):365-403. [41] 张文毓.生物医用金属材料研究现状与应用进展[J].金属世界, 2020(1):7. [42] NAFIKOV RK, KULYASOVA OB, KHUDODODOVA GD, et al. Microstructural assessment, mechanical and corrosion properties of a mg-sr alloy processed by combined severe plastic deformation. Materials (Basel). 2023;16(6):2279. |
| [1] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [2] | Dai Yuexing, Zheng Liqin, Wu Minhui, Li Zhihong, Li Shaobin, Zheng Desheng, Lin Ziling. Effect of vessel number on computational fluid dynamics in vascular networks [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1206-1210. |
| [3] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [4] | Cheng Jinhui, Wu Quan, Peng Min, Huang Changli, Tian Huimin, Li Yang. Preparation and properties of selective laser melting of porous titanium at a low energy density [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 664-668. |
| [5] | Wang Jianchun, Yang Shuqing, Su Xin, Wang Hongyuan. Different contents of B2O3 affect mechanical properties and bioactivity of bioactive glass scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 712-716. |
| [6] | Wei Yuxue, Wang Di, Liu Xiaoqiu. Design, synthesis and properties of oral composite resin monomers with different photoinitiators [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 731-735. |
| [7] | Zhou Xiaowen, Fu Zuchang, Huang Fei, Ai Jianguo, Zhao Feng. Bone defect blocked by bone cement segmental filling in single-plane tibial bone transport [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 736-740. |
| [8] | Zhang Yihai, Shang Peng, Ma Benyuan, Hou Guanghui, Cui Lunxu, Song Wanzhen, Qi Dexuan, Liu Yancheng. Structural design and mechanical property analysis of trabecular scaffold of triply periodic minimal surface with a radial gradient [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 741-746. |
| [9] | Yang Yuqing, Chen Zhiyu. Role and application of early transient presence of M1 macrophages in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 594-601. |
| [10] | Zhou Shibo, Guan Jianbin, Yu Xing, Zhao He, Yang Yongdong, Liu Tao. Animal models of femoral bone defects: preparation status and characteristics [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 633-638. |
| [11] | Dai Jing, Liu Shasha, Shen Mingjing. Exosome-loaded injectable hydrogel for repairing bone defects around implants [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 347-354. |
| [12] | Gao Xueyu, Zhang Wentao, Sun Tianze, Zhang Jing, Li Zhonghai. Application of metal ions in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 439-444. |
| [13] | Long Zhirui, Huang Lei, Xiao Fang, Wang Lin, Wang Xiaobei. Characteristics of hydrogel microspheres in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 472-478. |
| [14] | Kong Xiangyu, Wang Xing, Pei Zhiwei, Chang Jiale, Li Siqin, Hao Ting, He Wanxiong, Zhang Baoxin, Jia Yanfei. Biological scaffold materials and printing technology for repairing bone defects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 479-485. |
| [15] | Wen Xinghua, Ding Huanwen, Cheng Kai, Yan Xiaonan, Peng Yuanhao, Wang Yuning, Liu Kang, Zhang Huiwu. Three-dimensional finite element model analysis of intramedullary nailing fixation design for large femoral defects in Beagle dogs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1371-1376. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||