Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (17): 2702-2707.doi: 10.12307/2024.474
Previous Articles Next Articles
Low-temperature condensation deposition method for 3D printing of bone tissue engineering poly-L-lactic acid/pearl powder composite scaffold
Gang Fangli1, Shi Rui1, Ma Chunyang2, Xiao Yi1
- 1Department of Biology, Xinzhou Teachers University, Xinzhou 034000, Shanxi Province, China; 2School of Biological and Medical Engineering, Beihang University, Beijing 100083, China
-
Received:2023-07-21Accepted:2023-09-02Online:2024-06-18Published:2023-12-15 -
Contact:Gang Fangli, PhD, Lecturer, Department of Biology, Xinzhou Teachers University, Xinzhou 034000, Shanxi Province, China -
About author:Gang Fangli, PhD, Lecturer, Department of Biology, Xinzhou Teachers University, Xinzhou 034000, Shanxi Province, China -
Supported by:Shanxi Applied Basic Research Project for Youth Science Research, No. 20210302124286 (to GFL); Fund of Key Laboratory of Advanced Materials of Ministry of Education, No. ADV21-16 (to GFL); Xinzhou Basic Science and Technology Research Project, No. 20220505 (to SR)
CLC Number:
Cite this article
Gang Fangli, Shi Rui, Ma Chunyang, Xiao Yi. Low-temperature condensation deposition method for 3D printing of bone tissue engineering poly-L-lactic acid/pearl powder composite scaffold[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(17): 2702-2707.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
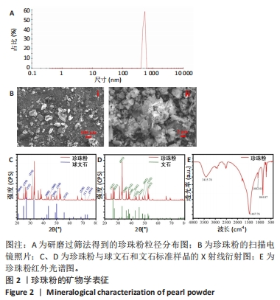
将珍珠研磨成粉与左旋聚乳酸一起溶于1,4-二氧六环制备墨水,在此基础上探索左旋聚乳酸/珍珠粉复合支架的低温冷凝3D打印工艺。首先,对研磨过筛法得到的珍珠粉进行矿物学表征。从图2A可以看到,过筛后的珍珠粉粒径绝大部分分布在400-600 nm之间。扫描电镜图像显示(图2B),制备的珍珠粉颗粒尺寸大小不一,存在少许直径100 μm左右的大颗粒,将图像进一步放大,可以看到较完整的珍珠粉呈片状,部分颗粒直径在2 μm以上,也有一部分粉末状的颗粒直径在1 μm以下,处于纳米级。为了明确所制备珍珠粉中的矿物成分,分别对粉晶进行X射线衍射和红外光谱表征。通过与球文石、文石标准样品的X射线衍射图谱对比(图2C、D),发现珍珠粉中004、110、112的峰位与标准球文石对应,111、021、012的峰位与标准文石相对应,表明该珍珠粉中的主要矿物成分包括球文石和文石,主要化学成分是CaCO3。此外,张刚生等[29]的研究曾指出,生物成因文石的面外弯曲振动频率范围在862-865.1 cm-1处,而无机成因文石的频率振动明显小于生物成因文石,在852.17-858 cm-1之间。文中珍珠粉的红外测试结果显示(图2E),该特征吸收峰对应振动频率为864.07 cm-1,因此判断其为生物成因文石。"

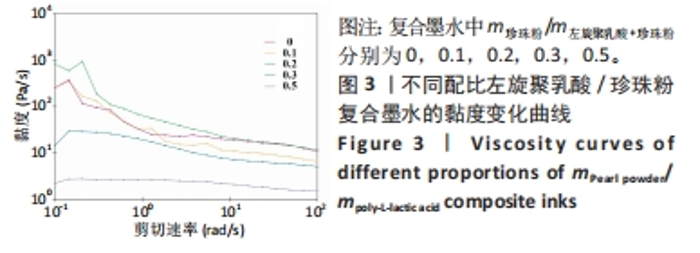
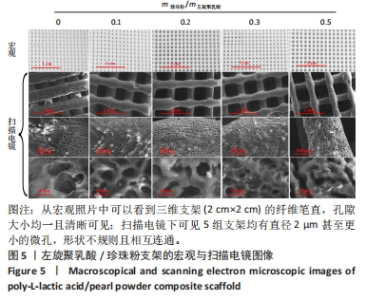
2.2 左旋聚乳酸/珍珠粉复合支架的3D打印 配制了5种不同比例(m珍珠粉/m左旋聚乳酸+珍珠粉=0,0.1,0.2,0.3,0.5)的左旋聚乳酸/珍珠粉墨水,并通过黏度和剪切流变学性能进行测试来确定复合支架的3D打印工艺。由于左旋聚乳酸在1,4-二氧六环中的溶解度较低,所以需要将左旋聚乳酸研磨成粉,并在加热(不超过50 ℃)和不断搅拌的情况下制备墨水。如图3所示,所有墨水的黏度都随着剪切速率的增加呈现出短暂的上升再大幅度下降的趋势,即在低剪切速率下存在剪切增稠现象。因此,鉴于左旋聚乳酸基复合墨水的这种特性,在打印时采用了高压气动推进结合喷嘴高速移动的策略,以避开具有剪切增稠现象的低剪切速率范围,并调整接收温度以使支架能够正常冷凝成型。在实际打印过程中,墨水从喷嘴中挤出后黏到低温的接收台上,会形成一个未冷冻结晶到逐渐冷冻再到完全冷冻结晶并固定形状的过程,但是在这个过程中喷嘴移动速度过快或者料筒内气压值过大都会导致墨水还未完全冷冻凝固而结构坍塌,反之则会导致墨水被冷冻结晶至喷嘴处形成堵塞。因此,墨水黏度、料筒内气压值和喷嘴的移动速度必须相互匹配才能保证良好的三维支架形貌。除此之外,接收台的温度也会影响打印效果。温度过低会导致墨水迅速冷凝而发生变形,温度过高墨水又无法快速成型。经过多次实验发现铝片接收台的温度设置为-2 ℃至-3 ℃最适合左旋聚乳酸基材料的打印。"
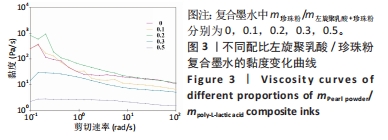
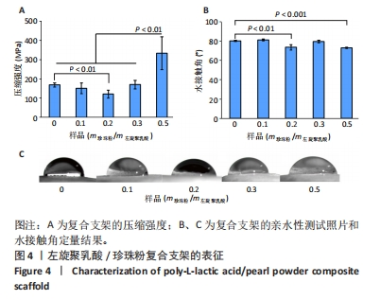
在不同墨水流变学性能测试中发现,随着珍珠粉含量的增加,墨水黏度先略有上升然后下降。这可能是少量珍珠粉的加入会增加墨水黏度,但是材料总浓度不变的情况下珍珠粉的含量增加则左旋聚乳酸含量减少,而墨水的黏度主要依靠左旋聚乳酸提供,所以墨水的黏度会下降。这样的墨水制备策略能够保持最终形成的支架质量不变,但是由于左旋聚乳酸含量的下降会导致墨水黏度明显降低。墨水黏度过低不利于3D打印的进行,因此在实际的墨水制备时应保持左旋聚乳酸含量不变,只改变珍珠粉的含量。该文后续实验所用墨水均以保持左旋聚乳酸含量不变为原则制备,而且在保持喷嘴直径、气压以及喷嘴移速一定的情况下这几种支架打印效果不受黏度变化的影响。 2.3 左旋聚乳酸/珍珠粉复合支架的材料学表征 由于该多孔支架在受力时会被压扁而不出现断裂,因此该文将支架被压到原来高度1/2处时所承受的压强作为其压缩强度。结果显示5组支架都表现出了高抗压性能(> 100 MPa),尤其是珍珠粉0.5组支架的压缩强度高达332.6 MPa,与其他组支架相比差异有显著性意义(P < 0.05),见图4A。 亲水性测试显示,左旋聚乳酸支架中掺杂珍珠粉的含量对其亲水性影响不呈比例(图4B、C)。珍珠粉具有良好的亲水性,在高温熔融法打印的支架中有效提高了支架的亲水性[29],出现这种情况的原因可能是低温冷凝法打印的支架中珍珠粉被左旋聚乳酸包裹而难以与水接触,因而对支架整体的亲水性影响并不规律。"

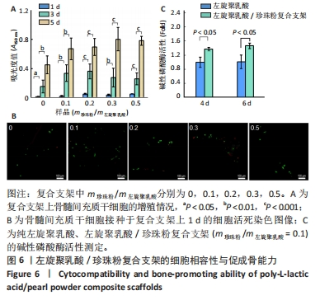
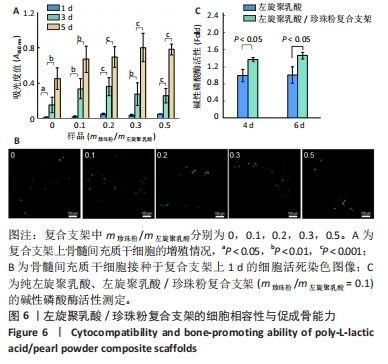
2.4 左旋聚乳酸/珍珠粉复合支架的细胞相容性 如图6A所示,5种支架上的细胞数目都随着培养时间的增加而显著增加,接种细胞后第1天,珍珠粉0.2组、珍珠粉0.3组和珍珠粉0.5组支架上的细胞数量明显多于珍珠粉0组;接种细胞后第3,5天,各组细胞开始快速增殖,珍珠粉0.1组、珍珠粉0.2组、珍珠粉0.3组、珍珠粉0.5组培养3,5 d的细胞增殖均快于珍珠粉0组(P < 0.05)。 支架上的细胞染色结果也进一步证实了材料良好的细胞相容性(图6B)。在细胞培养1 d后,5组支架上的细胞数量较少,但绝大部分为典型的绿色活细胞影像,红色死细胞很少。这可能归因于左旋聚乳酸疏水的特性导致黏附早期存留在支架纤维上的细胞量较少,这些细胞多沿着相互交叉的支架纤维的走行而轴向分布,少部分细胞也会聚集在支架孔隙内。 2.5 左旋聚乳酸/珍珠粉复合支架的促成骨能力 考虑到珍珠粉复合支架的促成骨分化性能已经被多篇论文所证实[30-32],此处仅以珍珠粉含量最少(m珍珠粉/m左旋聚乳酸=0.1)的三维支架与纯左旋聚乳酸支架,测定其成骨早期指标碱性磷酸酶活性作辅助验证。如图6C所示,成骨诱导4,6 d后,与珍珠粉0组相比,珍珠粉0.1组碱性磷酸酶活性显著升高(P < 0.05)。"
| [1] ATALA A, KASPER FK, MIKOS AG. Engineering complex tissues. Sci Transl Med. 2012; 4(160):112-160. [2] ANNAMALAI RT, HONG X, SCHOTT NG, et al. Injectable osteogenic microtissues containing mesenchymal stromal cells conformally fill and repair critical-size defects. Biomaterials. 2019;208:32-44. [3] 杨思敏,王新卫.自体骨移植修复骨缺损的临床研究进展[J].中国疗养医学,2019, 28(9):945-948. [4] AZI ML, APRATO A, SANTI I, et al. Autologous bone graft in the treatment of post-traumatic bone defects: a systematic review and meta-analysis. BMC Musculoskel Dis. 2016;17(1):465. [5] 马武秀,程迅生.自体骨移植修复骨缺损的研究进展[J].中国骨与关节损伤杂志, 2011,26(6):574-576. [6] SHIBUYA N, JUPITER DC. Bone graft substitute: allograft and xenograft. Clin Podiatr Med Surg. 2015;32(1):21-34. [7] BRACEY DN, JINNAH AH, WILLEY JS, et al. Investigating the osteoinductive potential of a decellularized xenograft bone substitute. Cells Tissues Organs. 2019;207(2):97-113. [8] SU X, WANG T, GUO S. Applications of 3D printed bone tissue engineering scaffolds in the stem cell field. Regen Ther. 2021;16:63-72. [9] WANG S, SHI Z, LIU L, et al. The design of Ti6Al4V Primitive surface structure with symmetrical gradient of pore size in biomimetic bone scaffold. Mater Design. 2020; 193:108830. [10] WU S, LEI L, BAO C, et al. An injectable and antibacterial calcium phosphate scaffold inhibiting Staphylococcus aureus and supporting stem cells for bone regeneration. Mat Sci Eng C. 2021;120:111688. [11] YAZDANIAN M, TABESH H, HOUSHMAND B, et al. Fabrication and properties of βTCP/Zeolite/Gelatin scaffold as developed scaffold in bone regeneration: in vitro and in vivo studies. Biocybern Biomed Eng. 2020;40(4):1626-1637. [12] RASTOGI P, KANDASUBRAMANIAN B. Review of alginate-based hydrogel bioprinting for application in tissue engineering. Biofabrication. 2019;11(4):042001. [13] AHSAN SM, THOMAS M, REDDY KK, et al. Chitosan as biomaterial in drug delivery and tissue engineering. Int J Biol Macromol. 2018;110:97-109. [14] ABDOLLAHIYAN P, OROOJALIAN F, MOKHTARZADEH A, et al. Hydrogel-based 3D bioprinting for bone and cartilage tissue engineering. Biotechnol J. 2020;15(12):e2000095. [15] SHUAI C, YANG W, FENG P, et al. Accelerated degradation of HAP/PLLA bone scaffold by PGA blending facilitates bioactivity and osteoconductivity. Bioact Mater. 2021;6(2):490-502. [16] CHEN VJ, MA PX. The effect of surface area on the degradation rate of nano-fibrous poly (L-lactic acid) foams. Biomaterials. 2006;27(20):3708-3715. [17] HUANG Q, LIU Y, OUYANG Z, et al. Comparing the regeneration potential between PLLA/Aragonite and PLLA/Vaterite pearl composite scaffolds in rabbit radius segmental bone defects. Bioact Mater. 2020;5(4):980-989. [18] WU X, STROLL S, LANTIGUA D, et al. Eggshell particle-reinforced hydrogels for bone tissue engineering: an unconventional approach. Biomater Sci. 2019;7:2675-2685. [19] 廖军,徐普.纳米淡水珍珠粉对成骨相关基因表达的影响[J].中国组织工程研究, 2022,26(27):4325-4329. [20] CHEN X, PENG LH, CHEE SS, et al. Nanoscaled pearl powder accelerates wound repair and regeneration in vitro and in vivo. Drug Dev Ind Pharm. 2019;45(6):1009-1016. [21] MA C, JIANG L, WANG Y, et al. 3D printing of conductive tissue engineering scaffolds containing polypyrrole nanoparticles with different morphologies and concentrations. Materials. 2019;12(15):2491. [22] GANG F, YAN H, MA C, et al. Robust magnetic double-network hydrogels with self-healing, MR imaging, cytocompatibility and 3D printability. Chem Commun. 2019; 55(66):9801-9804. [23] 张伟蒙,汪杰,胡晶.3D打印骨组织支架孔隙结构对支架性能影响的研究进展[J].中国塑料,2022,36(12):155-166. [24] OSTROVIDOV S, SALEHI S, COSTANTINI M, et al. 3D bioprinting in skeletal muscle tissue engineering. Small. 2019;15(24):1805530. [25] DADHICH P, DAS B, PAL P, et al. A simple approach for an eggshell-based 3D-printed osteoinductive multiphasic calcium phosphate scaffold. ACS Appl Mater Interfaces. 2016;8(19):11910-11924. [26] LUO W, ZHANG S, LAN Y, et al. 3D printed porous polycaprolactone/oyster shell powder (PCL/OSP) scaffolds for bone tissue engineering. Mater Res Express. 2018;5(4):045403. [27] DU X, YU B, PEI P, et al. 3D printing of pearl/CaSO4 composite scaffolds for bone regeneration. J Mater Chem B. 2018;6(3):499-509. [28] GUO F, WANG E, YANG Y, et al. A natural biomineral for enhancing the biomineralization and cell response of 3D printed polylactic acid bone scaffolds. Int J Biol Macromol. 2023;242:124728. [29] 张刚生,李浩璇.生物成因与无机成因文石的FTIR光谱区别[J].矿物岩石,2006, 26(1):3-6. [30] 张楷,徐普.珍珠粉复合支架在骨修复的研究进展[J].口腔颌面修复学杂志,2022, 23(4):297-315. [31] 李娜,徐普,陈灼庚,等.纳米珍珠粉人工骨修复兔股骨远端缺损[J].中南大学学报(医学版),2020,45(6):684-692. [32] 廖军,徐普.纳米淡水珍珠粉对成骨细胞成骨活性的影响[J].医学研究杂志,2022, 51(2):123-127. [33] CHEN R, PYE JS, LI J, et al. Multiphasic scaffolds for the repair of osteochondral defects: Outcomes of preclinical studies. Bioact Mater. 2023;27:505-545. [34] 韦宗辰,郗悦玮,翁云宣.聚乳酸基复合骨组织修复材料的研究现状及进展[J].中国塑料,2021,35(9):136-146. [35] YUE M, LIU Y, ZHANG P, et al. Integrative analysis reveals the diverse effects of 3D stiffness upon stem cell fate. Int J Mol Sci. 2023;24(11):9311. [36] ENGLER AJ, SEN S, SWEENEY HL, et al. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126(4):677-689. [37] WEI Q, WANG S, HAN F, et al. Cellular modulation by the mechanical cues from biomaterials for tissue engineering. Biomater Transl. 2021;2(4):323-342. [38] WU L, PEI X, ZHANG B, et al. 3D-printed HAp bone regeneration scaffoldsenable nano-scale manipulation of cellular mechanotransduction signals. Chem Eng J. 2022; 455:140699. [39] LU Q, DIAO J, WANG Y, et al. 3D printed pore morphology mediates bone marrow stem cell behaviors via RhoA/ROCK2 signaling pathway for accelerating bone regeneration. Bioact Mater. 2023;26:413-424. [40] 邵云菲,王卉,朱怡然,等.基于丝素蛋白材料构建骨组织修复支架的三维多孔结构体系的研究进展[J].合成生物学,2022,3(4):795-809. |
| [1] | Wang Menghan, Qi Han, Zhang Yuan, Chen Yanzhi. Three kinds of 3D printed models assisted in treatment of Robinson type II B2 clavicle fracture [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1403-1408. |
| [2] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [3] | Wang Wen, Zheng Pengpeng, Meng Haohao, Liu Hao, Yuan Changyong. Overexpression of Sema3A promotes osteogenic differentiation of dental pulp stem cells and MC3T3-E1 [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 993-999. |
| [4] | Ning Tianliang, Wang Kun, Wang Lingbiao, Han Pengfei. Finite element analysis on correction effect of varus foot orthosis based on the three-point force principle [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 891-899. |
| [5] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [6] | Xu Xiaodong, Zhou Jiping, Zhang Qi, Feng Chen, Zhu Mianshun, Shi Hongcan. 3D printing process of gelatin/oxidized nanocellulose skin scaffold with high elastic modulus and high porosity [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 398-403. |
| [7] | Yang Jie, Hu Haolei, Li Shuo, Yue Wei, Xu Tao, Li Yi. Application of bio-inks for 3D printing in tissue repair and regenerative medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 445-451. |
| [8] | Kong Xiangyu, Wang Xing, Pei Zhiwei, Chang Jiale, Li Siqin, Hao Ting, He Wanxiong, Zhang Baoxin, Jia Yanfei. Biological scaffold materials and printing technology for repairing bone defects [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 479-485. |
| [9] | Zhang Jie, Xiao Tianjiao, Li Li, Kang Jiabing, Zhan Jifan, Wei Yan, Tian Ai. Interleukin-4 regulates macrophage polarization and osteogenic differentiation of bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(25): 3960-3966. |
| [10] | Han Yue, Wang Yufei, Liu Wanqing, Dong Ming, Niu Weidong. Effects of icariin on proliferation and differentiation of MC3T3-E1 cells in an inflammatory environment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3709-3714. |
| [11] | Liu Yuhang, Sun Ruifen, Mu Rigen Jiya, Wang Xing, Li Zhijun, Liu Yanan, Hao Yunteng, Cai Yongqiang, Zhang Shaojie, Li Kun. Development of a three-dimensional digital children’s acupuncture point visualization system of Mongolian medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3223-3228. |
| [12] | Song Yue, Shu Qing, Jia Shaohui, Tian Jun. Photobiomodulation-induced osteogenic differentiation of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 3069-3075. |
| [13] | Gou Qiutong, Luo Wenhao, Wang Pin, Lan Yuyan, Liu Min, Huang Haixia. Berberine promotes osteogenic differentiation of bone marrow mesenchymal stem cells in a high-glucose environment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 2974-2980. |
| [14] | Xie Ting, Liu Tingting, Zeng Xuehui, Li Yamin, Zhou Panghu, Yi Nianhua. Vitamin D3 attenuates high-glucose exposure-induced oxidative stress to promote osteogenic differentiation of human umbilical cord mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(19): 2981-2987. |
| [15] | Dong Hanqing, Wu Xing, Xu Pengcheng, Wang Qingwen, Zhang Zhisheng, Zhao Jianyong. 3D printing precise positioning guided ulnar groove plasty for treatment of cubital tunnel syndrome [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(18): 2825-2829. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||