Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (23): 3692-3698.doi: 10.12307/2024.418
Previous Articles Next Articles
Frontier hot trends in ischemic stroke and vascular regeneration based on bibliometric analysis
Xia Tianqing1, Rong Mengwei1, Dan Cunyan1, Yang Ting2, Ding Zhibin2, Song Lijuan1, 3, Ma Cungen1
- 1The Key Research Laboratory of Benefiting Qi for Acting Blood Circulation Method to Treat Multiple Sclerosis of State Administration of Traditional Chinese Medicine/Research Center of Neurobiology, Shanxi University of Chinese Medicine, Jinzhong 030619, Shandong Province, China; 2Department of Neurology, Third Hospital of Shanxi Medical University (Shanxi Bethune Hospital, Shanxi Academy of Medical Sciences, Tongji Shanxi Hospital), Taiyuan 030032, Shanxi Province, China; 3Department of Neurosurgery, Sinopharm Tongmei General Hospital, Datong 037003, Shanxi Province, China
-
Received:2023-06-01Accepted:2023-07-20Online:2024-08-18Published:2023-09-13 -
Contact:Ma Cungen, MD, Professor, Doctoral supervisor, The Key Research Laboratory of Benefiting Qi for Acting Blood Circulation Method to Treat Multiple Sclerosis of State Administration of Traditional Chinese Medicine/Research Center of Neurobiology, Shanxi University of Chinese Medicine, Jinzhong 030619, Shandong Province, China -
About author:Xia Tianqing, Master candidate, The Key Research Laboratory of Benefiting Qi for Acting Blood Circulation Method to Treat Multiple Sclerosis of State Administration of Traditional Chinese Medicine/Research Center of Neurobiology, Shanxi University of Chinese Medicine, Jinzhong 030619, Shandong Province, China -
Supported by:the National Natural Science Foundation of China, Nos. 82004028 (to SLJ) and 81473577 (to MCG); China Postdoctoral Science Foundation, No. 2020M680912 (to SLJ); “Zhang Zhongjing Inheritance and Innovation Special Project” of the National Administration of Traditional Chinese Medicine, No. GZY-KJS-2022-048-1 (to SLJ); Shanxi Provincial Science and Technology Innovative Talent Young Team Project, No. 202204051001028 (to SLJ); Shanxi University of Chinese Medicine 2022 Scientific and Technological Innovation Team Project, No. 2022TD2010 (to MCG and SLJ); Medical Science and Technology Leading Team Project of Shanxi Provincial Health Commission, No. 2020TD05 (to MCG); National Regional TCM Medical Center Cardiovascular Special Fund for Shanxi University of Chinese Medicine Affiliated Hospital, No. XGZX202115 (to SLJ); Shanxi University of Chinese Medicine Young Scientist Cultivation Project, No. 2021PY-QN-09 (to SLJ); Shanxi University of Chinese Medicine Discipline Construction Fund, No. 2023XKJS-02 (to MCG)
CLC Number:
Cite this article
Xia Tianqing, Rong Mengwei, Dan Cunyan, Yang Ting, Ding Zhibin, Song Lijuan, Ma Cungen. Frontier hot trends in ischemic stroke and vascular regeneration based on bibliometric analysis[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3692-3698.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
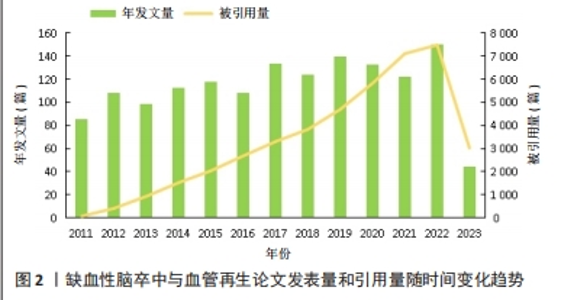
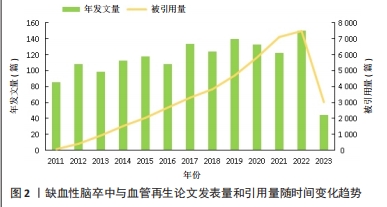
2.1 文章发表量和引用量分析 2011-2023年期间,文章发表量和文章引用量总体上都呈上升趋势,但文章发表量在2013年、2016年和2021年分别有不同程度的回落,在2017年、2019年和2022年出现了发表量的峰值,分别为134,140和151篇(图2)。2017-2019年期间关于MicroRNA(miRNA)的研究是该领域的发文热点,其中2017年最具代表性的是来自阿瓦士容迪沙布尔医科大学KHOSHNAM团队[21]的文章,该作者及其团队就miRNA在脑卒中后的治疗作用机制(包括促进血管新生和神经修复方面)进行了综述,发表后被引用了120次,为治疗缺血性脑卒中指引了新的方向。2019年,来自南京医科大学DU及其团队[22]在之前科研人员的研究基础上进行了新的实验,实验结果揭示了miR-191通过靶向VEZF1抑制血管生成并加剧了缺血性脑损伤的新作用,证明miR-191可作为急性缺血性脑卒中的生物标志物或治疗靶点,对miRNA与缺血性脑卒中之间的作用机制进展实现了新的突破。2022年,此领域文章的发表量迎来了近10年以来的最高值,有关脑缺血损伤后具有促进神经保护和神经恢复的生物活性药材天然化合物的一篇文章被引用的次数最多,为中药促进缺血性脑卒中后的血管新生和神经恢复提供了有力参考依据[23]。从引用量来看,该领域相关文献的总被引量呈不断上升趋势,从2011年的86篇迅速增加到2022年的151篇,有了非常大的增长,说明相关科研人员对这一领域的兴趣持续上涨。此外,从2011年起,干细胞对缺血性脑卒中的治疗和神经血管修复机制也成为关注焦点[24-27]。在保持进一步深入探究的基础上,中药单体成分治疗缺血性脑卒中的作用机制也成为了新的热点[28-29],证明这10年来中国相关科研人员在为促进中医药发展上取得了巨大的成就,使得中医药在现代医学领域得以飞速发展,逐渐被世界所认可。预测在缺血性脑卒中和血管再生这一领域发表及引用的文章,未来几十年在全世界范围内将会有更大数量的增长和飞跃。"
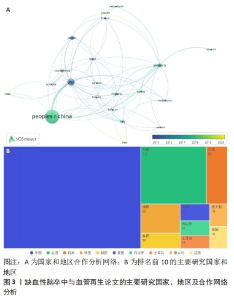
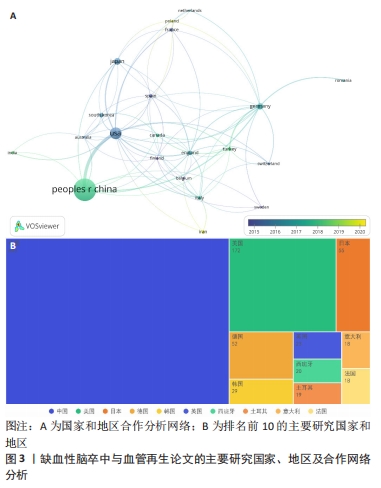
2.2 国家和地区分布及合作特点 在2011-2023年期间,共有56个国家及地区参与了该领域的研究并为相关科研论文的发表做出贡献。在1 484篇论文当中,排名前三的国家和发文量分别为中国(643篇)、美国(172篇)、日本(55篇),见图3。其中,中国的发文量排到了第一位,占总体发文量的43.33%,远远领先于其他国家,说明中国近10年来在缺血性脑卒中和血管再生的科研领域处于核心地位,十分重视此方面的研究,并且论文和科研成果的理论创新和实用价值均走在世界前列。排名第二为美国的172篇,剩余其他国家的发文量均在100篇以下,说明其他国家对此领域研究的重视程度和投入并不是很高。此外,利用VOSviewer对国家之间发文的合作和时间进行了可视化分析(图3A),其中节点的大小表示该国家发文量的总强度,而连接线的粗细代表了国家之间合作的强度。通过图谱发现合作关系最紧密的国家是中国和美国,一共有过60次合作,建立合作关系最多的国家是美国,一共与19个国家均有合作,与多个发达国家合作甚多,并且发达国家之间的合作更为紧密。"
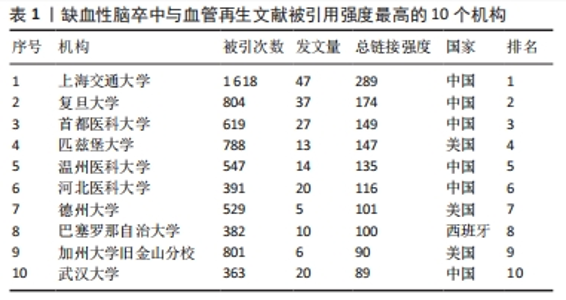
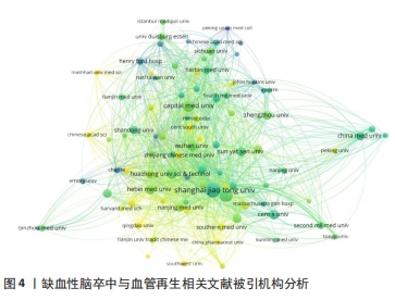
2.3 被引机构分析 首先,借助可视化工具——VOSviewer分析了高被引机构。选择“Citation”作为分析类型,“Orgnization”作为分析单位,并将最低发文数量设置为5,默认最低引用为0。在所有已发表文献的机构中,91个机构符合设定标准。总链接强度最大的前十个机构分别为上海交通大学(文献数量=47,引用量=1 618,总链接强度=283)、复旦大学(文献数量=37,引用量=804,总链接强度=174)、首都医科大学(文献数量=27,引用量=804,总链接强度174)、匹兹堡大学(文献数量=13,引用量=788,总链接强度=147)、温州医科大学(文献数量=14,引用量=547,总链接强度=135)、河北医科大学(文献数量=20,引用量=391,总链接强度=116)、德州大学(文献数量=5,引用量=529,总链接强度=101)、巴塞罗那自治大学(文献数量=10,引用量=382,总链接强度=100)、加州大学旧金山分校(文献数量=6,引用量=801,总链接强度=90)、武汉大学(文献数量=20,引用量=363,总链接强度=89),见表1。发文量的多少意味着此机构在缺血性脑卒中和血管再生方面研究的兴趣程度,发文量多即代表机构的研究人员在此方面进行了持续的研究并已有了许多实质性的进展,而链接强度则是一个综合性的指标,是文献数量和引用量的综合评价,引用量即代表着被其他研究机构认可的程度和可信度,总链接强度高意味着不仅此机构在研究领域取得了非常多的成果,并且研究成果得到了各个机构的认可,文章的质量和数量均得到了保障。因此,总链接强度比发文量更加具有参考意义。"
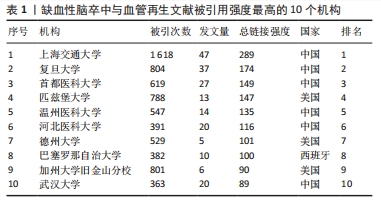

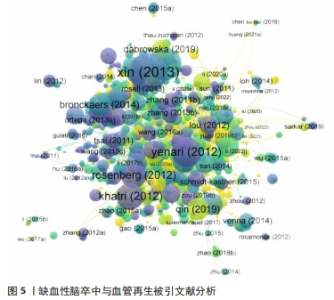
表2列出了近10年被引次数最高的10篇文献,其中被引次数最多的、排在第一位的文章是发表在《Journal of Cerebral Blood Flow and Metabolism》期刊上来自XIN HQ的“Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats”,总共被引用了643次,是其他科研人员引用的热点文章,作者对多能间充质基质细胞的外泌体通过促进神经和血管再生治疗脑卒中这一方向进行实验和论述[35],取得很大的进展和成果,为此领域的研究奠定了非常好的基础。被引量排名第二的文章“Extracellular Vesicles Improve Post-Stroke Neuroregeneration and Prevent Postischemic Immunosuppression”作者为DOEPPNER TR,总共被引用了467次,文章介绍了源自干细胞(如外泌体)的细胞外囊泡和脑缺血后神经与血管再生的关系,通过动物实验证实了细胞外囊泡确实可以改善缺血后神经损伤和脑重塑情况[36],不仅为临床提供了证据,也为后续此领域大量科研人员进一步研究提供了技术和理论指导。被引量排名第三的文章是作者YENARI MA发表的“Neuroprotective mechanisms of hypothermia in brain ischaemia”,是一篇论文综述,主要聚焦于脑缺血后低温对神经系统的影响展开相关综述[37],对之前科研人员的研究成果进行了系统的总结,为后面继续深入探究此方面的研究者进一步提供了理论参考依据。在被引量排名靠前的文章里,低温对缺血性脑卒中血管再生方面的影响以不同的角度和方式被实验和论述,已成为此领域的主要研究热点。"

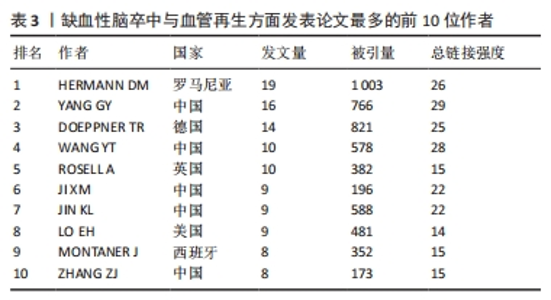
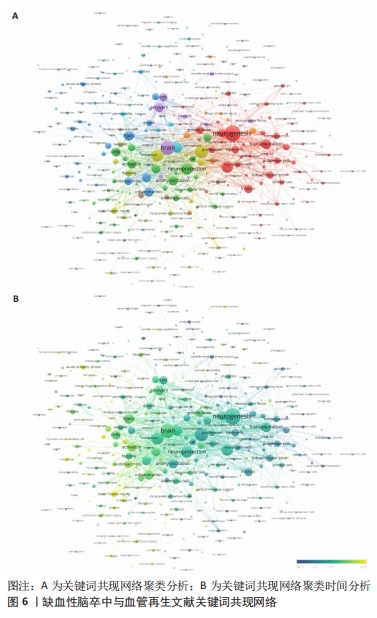
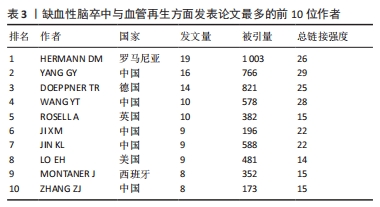
其中,红色聚类共71个关键词,主要聚焦于缺血性脑卒中的治疗与神经功能恢复。有研究表明,在神经保护的过程中血管再生起着非常关键的作用[38],因此神经功能的恢复与血管再生密不可分。关键词“neurogenesis(神经再生)”“functional recovery (功能恢复)”“transplantation (移植)”“growth factor (生长因子)”和“in-vitro (体外)”均能体现两者的紧密联系。 绿色聚类共70个关键词,其中包含“neuroprotection (神经保护)”“artery occlusion (动脉闭塞)”“mechanisms (机制)”“activation (激活)”和“cells (细胞)”,说明研究者注重在缺血性脑卒中发生作用机制方面的研究。在缺血性脑卒中发生后,受损脑实质中的内源性反应有着非常复杂的机制,有证据表明血管内皮生长因子在脑卒中后有着特定的机制功能,是脑卒中后的功能恢复中必不可少的一部分[39]。因此,研究者们大多将重点放在了脑卒中发生的作用机制上。 深蓝色聚类共56个关键词,显示了近些年在治疗方法上是从哪些具体方面发挥作用,其中热点关键词有“injury (损伤)”“apoptosis (细胞凋亡)”“inflammation (炎症)”和“oxidative stress(氧化应激)”等。炎症反应在脑卒中的发病机制中有着至关重要的作用,有研究表明,各种炎性细胞和促炎细胞因子通过炎症相关信号通路在脑卒中发生后促进了血管生成[40]。同样,通过抑制细胞凋亡的方式也可以对血管生成起到促进作用,同时可以预防缺血性脑卒中的发生[41]。并且已有研究证实,氧化应激的良好调节促进着血管的再生和组织的修复[42]。说明在治疗缺血性脑卒中的作用机制方面,抑制细胞凋亡、抗炎、抗氧化应激等方面是研究者们展开研究的焦点。 黄色聚类共41个关键词,此模块主要针对缺血性脑卒中发生后血管再生的机制,比如高频关键词“vegf(内皮生长因子)”“neovascularization (血管生成)”“hypoxia(缺氧)”“inhibition(抑制)”和“erythropoietin (促红细胞生成素)”,促进血管再生被认为是脑卒中后最希望的治疗策略之一,而血管内皮生长因子对脑缺血后神经血管的重塑有着十分积极的影响[43]。这些关键词显示了在脑卒中后血管生成的过程中重要的生物组成部分。 紫色聚类共37个关键词,主要分析了动物实验中与大脑神经恢复有关的信息,例如关键词“brain(大脑)”“recovery (康复)”“rats(大鼠)”“neurotrophic factor(神经营养因子)”和“neurons (神经元)”,神经营养因子是一种对神经具有保护作用的物质,相关研究表明,神经营养因子对促进大鼠脑卒中后的神经恢复有着积极作用,而这种作用是通过激活血管内皮细胞因子通路增加总血管的表面积来实现的[44]。这说明研究者在进行此领域研究时不仅更倾向于用大鼠进行动物实验,还注重缺血性脑卒中发生后大脑神经再生的机制研究。 浅蓝色聚类共有34个关键词,此聚类专注于分析缺血性脑卒中发生后大脑的情况,如 “blood-brain-barrier(血脑屏障)”“tissue-plasminogen activator(纤溶酶原激活剂)”“permeability(渗透率)”“MRI(核磁共振)”和“blood flow(血流量)”,作为实验是否成功的标准。促进血管生成的因素有很多,而其中血管生成素样蛋白1作为血管生成素相关蛋白家族的一员被证实在小鼠脑缺血后可以更好地保持血管的完整性,进而减少血脑屏障的损伤和水肿程度[45]。因此,大脑的各项指标也成为实验对照组之间分析的基础,大脑血流量是否增加、血脑屏障是否遭到破坏以及观察大脑核磁共振图像判断阻塞情况是否好转,也成为了此领域科研人员十分注重的部分。 橙色聚类共有28个关键词,出现频率较高的关键词有“in-vivo(体内)”“mesenchymal stem-cells(间质干细胞)”“gene-expression(基因表达)”“traumation brain injury(脑损伤)”和“vascular- permeability (血管渗透性)”,此聚类从微观角度展示了研究人员在此领域不仅注重体内动物实验,也会通过体外的细胞实验来进行研究,细胞实验也是缺血性脑卒中和血管再生研究实验中不可或缺的一部分。 棕色聚类共有19个关键词,此聚类聚焦于大鼠大脑中动脉闭塞实验的情况,其中高频率词有“matrix metalloproteinases(基质金属蛋白酶)”“rat model(大鼠模型)”“middle cerebral-artery(大脑中动脉)”“cerebral-artery occlusion(脑动脉闭塞)”和“gene therapy(基因治疗)”,说明在对缺血性脑卒中和血管再生方面展开研究时,多数的研究者都会选择构建大鼠大脑中动脉模型进行动物实验,并且效果良好,是研究者们在此领域偏爱的实验动物模型。 此外,通过时间图发现浅黄色的节点关键词代表近些年的研究热点,其中包含“reperfusion injury (再灌注损伤)”和“microRNA(微小核糖核酸)”,缺血性脑卒中发生后缺血再灌注损伤是不可避免的情况,因此研究者聚焦在这方面的防治是有迹可循的,并且符合正常研究思路。而microRNA的研究热潮也符合前面文章发表量中关于近年的研究热点方向情况。 2.6 发文作者分析 表3列出了在此领域发文量最多前10位的作者,在这期间他们一共发表了112篇论文,占论文总数的7%,并被各研究领域大量引用。其中,有5位来自中国,有2位来自美国,有1位来自罗马尼亚,有1位来自德国,还有1位来自英国。有一半是来自中国的作者,中国作者总共发文量为56篇,占前10位作者发文总量的48%,证明中国各机构和科研人员在此方面表现出了极大的兴趣,并且为论文的发表做出了非常大的贡献。而发文量最多的作者是来自罗马尼亚的HERMANN DM,他总共发表了19篇文章,文章被引用了1 003次,总连接强度为26,是唯一一位论文被引量超过1 000次的作者,由此证明他的文章和科研成果被各大研究机构和科研人员所认可,他的主要研究方向聚集在血管内皮生长因子在缺血性脑卒中发生后的作用机制上[46-48],与此相关的文章发表数量居首位,但他被引用最多的文章却是围绕细胞外囊泡改善脑卒中后神经再生的主题撰写的[49],总共被引用了466次,说明其他研究人员也对这个方向表现出了极大的兴趣,在之后也发表了许多相关的论文。"
| [1] ZHOU M, WANG H, ZHU J, et al. Cause-specific mortality for 240 causes in China during 1990-2013: a systematic subnational analysis for the Global Burden of Disease Study 2013. Lancet. 2016;387(10015):251-272. [2] 王励,肖雪梅,许志斌,等.出血性与缺血性脑卒中住院患者危险因素比较分析[J].慢性病学杂志,2014,15(7):526-528. [3] 李云云,屈洪党.脑出血的诊断与治疗[J].中华全科医学,2019,17(2):171-172. [4] 李卉,汤永红.脑卒中病因研究进展[J].现代诊断与治疗,2020,31(21):3380-3382. [5] 马腾云,高玉元,马桂贤,等.缺血性脑卒中脑动脉血栓病理研究及应用进展[J].中华老年心脑血管病杂志,2021,23(12):1339-1341. [6] GBD 2016 DALYs and HALE Collaborators. Global, regional, and national disability-adjusted life-years (DALYs) for 333 diseases and injuries and healthy life expectancy (HALE) for 195 countries and territories, 1990-2016: a systematic analysis for the Global Burden of Disease Study 2016 . Lancet. 2017;390(10100): 1260-1344. [7] RANDOLPH SA. Ischemic Stroke. Workplace Health Saf. 2016;64(9):444. [8] 马林,巢宝华,曹雷,等.2007-2017年中国脑卒中流行趋势及特征分析[J].中华脑血管病杂志(电子版),2020,14(5):253-258. [9] 张艾嘉,王爽,王萍,等.缺血性脑卒中的病理机制研究进展及中医药防治[J].中国实验方剂学杂志,2020,26(5):227-240. [10] MA R, XIE Q, LI H, et al. l-Borneol Exerted the Neuroprotective Effect by Promoting Angiogenesis Coupled With Neurogenesis via Ang1-VEGF-BDNF Pathway. Front Pharmacol. 2021;12:641894. [11] LUNARDI BACCETTO S, LEHMANN C. Microcirculatory Changes in Experimental Models of Stroke and CNS-Injury Induced Immunodepression. Int J Mol Sci. 2019; 20(20):5184. [12] WANG SN, WANG Z, XU TY, et al. Cerebral Organoids Repair Ischemic Stroke Brain Injury. Transl Stroke Res. 2020;11(5):983-1000. [13] LIU Z, CHOPP M. Astrocytes, therapeutic targets for neuroprotection and neurorestoration in ischemic stroke. Prog Neurobiol. 2016;144:103-120. [14] HAO L, ZOU Z, TIAN H, et al. Stem cell-based therapies for ischemic stroke. Biomed Res Int. 2014;2014:468748. [15] Liman TG, Endres M. New vessels after stroke: postischemic neovascularization and regeneration. Cerebrovasc Dis. 2012;33(5):492-499. [16] 邱均平.文献计量学[M].北京:科学技术文献出版社,1988. [17] 刁敬东,农晓琳.文献计量学在学术期刊评价中的应用研究[J].医学信息, 2018,31(23):16-18. [18] 陈莹,常静玲,李新龙,等.基于CNKI的近10年中医药治疗缺血性脑卒中研究的可视化分析[J].时珍国医国药,2021,32(11):2803-2806. [19] 黄茂茂,胡月,王彬川,等.缺血性脑卒中康复近10年国际文献计量学及可视化分析[J].中国组织工程研究,2021,25(23):3725-3733. [20] 陈娟.干细胞移植在缺血性脑卒中治疗中的应用[J].中国组织工程研究, 2012,16(19):3576-3583. [21] KHOSHNAM SE, WINLOW W, FARBOOD Y, et al. Emerging Roles of microRNAs in Ischemic Stroke: As Possible Therapeutic Agents. J Stroke. 2017;19(2):166-187. [22] DU K, ZHAO C, WANG L, et al. MiR-191 inhibit angiogenesis after acute ischemic stroke targeting VEZF1. Aging (Albany NY). 2019;11(9):2762-2786. [23] ZHU T, WANG L, WANG LP, et al. Therapeutic targets of neuroprotection and neurorestoration in ischemic stroke: Applications for natural compounds from medicinal herbs. Biomed Pharmacother. 2022;148:112719. [24] HORIE N, PEREIRA MP, NIIZUMA K, et al. Transplanted stem cell-secreted vascular endothelial growth factor effects poststroke recovery, inflammation, and vascular repair. Stem Cells. 2011;29(2):274-285. [25] BAO X, WEI J, FENG M, et al. Transplantation of human bone marrow-derived mesenchymal stem cells promotes behavioral recovery and endogenous neurogenesis after cerebral ischemia in rats. Brain Res. 2011;1367:103-113. [26] LINDVALL O, KOKAIA Z. Stem cell research in stroke: how far from the clinic? Stroke. 2011;42(8):2369-2375. [27] MACKIE AR, LOSORDO DW. CD34-positive stem cells: in the treatment of heart and vascular disease in human beings. Tex Heart Inst J. 2011;38(5):474-485. [28] LI L, GAN H, JIN H, et al. Astragaloside IV promotes microglia/macrophages M2 polarization and enhances neurogenesis and angiogenesis through PPARγ pathway after cerebral ischemia/reperfusion injury in rats. Int Immunopharmacol. 2021;92:107335. [29] SHI YH, ZHANG XL, YING PJ, et al. Neuroprotective Effect of Astragaloside IV on Cerebral Ischemia/Reperfusion Injury Rats Through Sirt1/Mapt Pathway. Front Pharmacol. 2021;12:639898. [30] CHEN J, ZHANG X, LIU X, et al. Ginsenoside Rg1 promotes cerebral angiogenesis via the PI3K/Akt/mTOR signaling pathway in ischemic mice. Eur J Pharmacol. 2019;856:172418. [31] LI XF, ZHANG XJ, ZHANG C, et al. Ulinastatin protects brain against cerebral ischemia/reperfusion injury through inhibiting MMP-9 and alleviating loss of ZO-1 and occludin proteins in mice. Exp Neurol. 2018;302:68-74. [32] LI Y, ZHANG X, CUI L, et al. Salvianolic acids enhance cerebral angiogenesis and neurological recovery by activating JAK2/STAT3 signaling pathway after ischemic stroke in mice. J Neurochem. 2017;143(1):87-99. [33] SONG D, ZHANG X, CHEN J, et al. Wnt canonical pathway activator TWS119 drives microglial anti-inflammatory activation and facilitates neurological recovery following experimental stroke. J Neuroinflammation. 2019;16(1):256. [34] WANG M, LI Y, ZHANG R, et al. Adiponectin-Transfected Endothelial Progenitor Cells Have Protective Effects After 2-Hour Middle-Cerebral Artery Occlusion in Rats With Type 2 Diabetes Mellitus. Front Neurol. 2021;12:630681. [35] XIN H, LI Y, CUI Y, et al. Systemic administration of exosomes released from mesenchymal stromal cells promote functional recovery and neurovascular plasticity after stroke in rats. J Cereb Blood Flow Metab. 2013;33(11):1711-1715. [36] DOEPPNER TR, HERZ J, GÖRGENS A, et al. Extracellular Vesicles Improve Post-Stroke Neuroregeneration and Prevent Postischemic Immunosuppression. Stem Cells Transl Med. 2015;4(10):1131-1143. [37] YENARI MA, HAN HS. Neuroprotective mechanisms of hypothermia in brain ischaemia. Nat Rev Neurosci. 2012;13(4):267-278. [38] SEEVINCK PR, DEDDENS LH, DIJKHUIZEN RM. Magnetic resonance imaging of brain angiogenesis after stroke. Angiogenesis. 2010;13(2):101-111. [39] NAVARATNA D, GUO S, ARAI K, et al. Mechanisms and targets for angiogenic therapy after stroke. Cell Adh Migr. 2009;3(2):216-223. [40] ZHU H, ZHANG Y, ZHONG Y, et al. Inflammation-Mediated Angiogenesis in Ischemic Stroke. Front Cell Neurosci. 2021;15:652647. [41] XIA CF, YIN H, YAO YY, et al. Kallikrein protects against ischemic stroke by inhibiting apoptosis and inflammation and promoting angiogenesis and neurogenesis. Hum Gene Ther. 2006;17(2):206-219. [42] HUANG YJ, NAN GX. Oxidative stress-induced angiogenesis. J Clin Neurosci. 2019; 63:13-16. [43] HERMANN DM, ZECHARIAH A. Implications of vascular endothelial growth factor for postischemic neurovascular remodeling. J Cereb Blood Flow Metab. 2009;29(10):1620-1643. [44] GAO B, DENG J, ZHANG X, et al. Effects of mesencephalic astrocyte-derived neurotrophic factor on cerebral angiogenesis in a rat model of cerebral ischemia. Neurosci Lett. 2020;715:134657. [45] LAI DM, LI H, LEE CC, et al. Angiopoietin-like protein 1 decreases blood brain barrier damage and edema following focal cerebral ischemia in mice. Neurochem Int. 2008;52(3):470-477. [46] ZECHARIAH A, ELALI A, DOEPPNER TR, et al. Vascular endothelial growth factor promotes pericyte coverage of brain capillaries, improves cerebral blood flow during subsequent focal cerebral ischemia, and preserves the metabolic penumbra. Stroke. 2013;44(6):1690-1697. [47] MA Y, ZECHARIAH A, QU Y, et al. Effects of vascular endothelial growth factor in ischemic stroke. J Neurosci Res. 2012;90(10):1873-1882. [48] REITMEIR R, KILIC E, REINBOTH BS, et al. Vascular endothelial growth factor induces contralesional corticobulbar plasticity and functional neurological recovery in the ischemic brain [published correction appears in Acta Neuropathol. 2015 Oct;130(4):603]. Acta Neuropathol. 2012;123(2):273-284. [49] DOEPPNER TR, HERZ J, GÖRGENS A, et al. Extracellular Vesicles Improve Post-Stroke Neuroregeneration and Prevent Postischemic Immunosuppression. Stem Cells Transl Med. 2015;4(10):1131-1143. [50] HU X, DE SILVA TM, CHEN J, et al. Cerebral Vascular Disease and Neurovascular Injury in Ischemic Stroke. Circ Res. 2017;120(3):449-471. [51] ERGUL A, VALENZUELA JP, FOUDA AY, et al. Cellular connections, microenvironment and brain angiogenesis in diabetes: Lost communication signals in the post-stroke period. Brain Res. 2015;1623:81-96. [52] GREENBERG DA. Poststroke angiogenesis, pro: making the desert bloom. Stroke. 2015;46(5):e101-e102. [53] BACIGALUPPI M, PLUCHINO S, PERUZZOTTI-JAMETTI L, et al. Delayed post-ischaemic neuroprotection following systemic neural stem cell transplantation involves multiple mechanisms. Brain. 2009;132(Pt 8):2239-2251. [54] ZHENG W, HONMOU O, MIYATA K, et al. Therapeutic benefits of human mesenchymal stem cells derived from bone marrow after global cerebral ischemia. Brain Res. 2010;1310:8-16. [55] HSIEH JY, WANG HW, CHANG SJ, et al. Mesenchymal stem cells from human umbilical cord express preferentially secreted factors related to neuroprotection, neurogenesis, and angiogenesis. PLoS One. 2013;8(8):e72604. [56] SCHEIBE F, KLEIN O, KLOSE J, et al. Mesenchymal stromal cells rescue cortical neurons from apoptotic cell death in an in vitro model of cerebral ischemia. Cell Mol Neurobiol. 2012;32(4):567-576. [57] TATE CC, FONCK C, MCGROGAN M, et al. Human mesenchymal stromal cells and their derivative, SB623 cells, rescue neural cells via trophic support following in vitro ischemia. Cell Transplant. 2010;19(8):973-984. [58] QIU L, CAI Y, GENG Y, et al. Mesenchymal stem cell-derived extracellular vesicles attenuate tPA-induced blood-brain barrier disruption in murine ischemic stroke models. Acta Biomater. 2022;154:424-442. [59] LIU C, YANG TH, LI HD, et al. Exosomes from bone marrow mesenchymal stem cells are a potential treatment for ischemic stroke. Neural Regen Res. 2023; 18(10):2246-2251. [60] BHASIN A, SRIVASTAVA MV, KUMARAN SS, et al. Autologous mesenchymal stem cells in chronic stroke. Cerebrovasc Dis Extra. 2011;1(1):93-104. [61] WU F, YANG Z, LI G. Role of specific microRNAs for endothelial function and angiogenesis. Biochem Biophys Res Commun. 2009;386(4):549-553. [62] JEYASEELAN K, LIM KY, ARMUGAM A. MicroRNA expression in the blood and brain of rats subjected to transient focal ischemia by middle cerebral artery occlusion. Stroke. 2008;39(3):959-966. [63] REDELL JB, LIU Y, DASH PK. Traumatic brain injury alters expression of hippocampal microRNAs: potential regulators of multiple pathophysiological processes. J Neurosci Res. 2009;87(6):1435-1448. [64] DHARAP A, BOWEN K, PLACE R, et al. Transient focal ischemia induces extensive temporal changes in rat cerebral microRNAome. J Cereb Blood Flow Metab. 2009;29(4):675-687. [65] LIU DZ, TIAN Y, ANDER BP, et al. Brain and blood microRNA expression profiling of ischemic stroke, intracerebral hemorrhage, and kainate seizures. J Cereb Blood Flow Metab. 2010;30(1):92-101. [66] 钟迪,张舒婷,吴波.《中国急性缺血性脑卒中诊治指南2018》解读[J].中国现代神经疾病杂志,2019,19(11):897-901. [67] 王珊珊,吴远华.中医药防治缺血性脑卒中的现代分子生物学研究进展[J].中国民间疗法,2022,30(22):113-116. [68] LI J, LI C, SUBEDI P, et al. Light Alcohol Consumption Promotes Early Neurogenesis Following Ischemic Stroke in Adult C57BL/6J Mice. Biomedicines. 2023;11(4):1074. [69] LIU Z, LIU M, JIA G, et al. Long-term intermittent fasting improves neurological function by promoting angiogenesis after cerebral ischemia via growth differentiation factor 11 signaling activation. PLoS One. 2023;18(3):e0282338. [70] ZHANG Y, ZHAO X, GUO C, et al. The Circadian System Is Essential for the Crosstalk of VEGF-Notch-mediated Endothelial Angiogenesis in Ischemic Stroke. Neurosci Bull. 2023. doi: 10.1007/s12264-023-01042-9. [71] ZHANG W, HAN L, WEN Y, et al. Electroacupuncture reverses endothelial cell death and promotes angiogenesis through the VEGF/Notch signaling pathway after focal cerebral ischemia-reperfusion injury. Brain Behav. 2023;13(3):e2912. |
| [1] | Sun Yukang, Song Lijuan, Wen Chunli, Ding Zhibin, Tian Hao, Ma Dong, Ma Cungen, Zhai Xiaoyan. Visualization analysis of stem cell therapy for myocardial infarction based on Web of Science in recent ten years [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1143-1148. |
| [2] | Wan Jun, Bai Yanjie, Wang Yan, Chen Shuying, Chen Limin, Xiao Yuqian, Sun Kexin. Mechanism of action and related signaling pathways of long non-coding RNAs in neuroimmuno-inflammatory response after ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3265-3271. |
| [3] | Xu Kangli, An Lanhua, Zhang Jinsheng, Du Xiaoyan, Yin Lele, Zhang Xixian. Research hotspots and frontiers of functional magnetic resonance imaging in treatment of ischemic stroke by traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(11): 1789-1796. |
| [4] | Ying Chunmiao, Pan Xiaolong, Liu Feixiang, Chen Na, Fan Feiyan, Zhang Yunke. Effect of traditional Chinese medicine and compounds for supplementing qi and activating blood circulation and inducing resuscitation on regulating stem cells to promote nerve repair of acute ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 121-130. |
| [5] | Yang Ting, Ding Zhibin, Jiang Nan, Han Hongxia, Hou Miaomiao, Ma Cungen, Song Lijuan, Li Xinyi. Astrocytes regulate glial scar formation in cerebral ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 131-138. |
| [6] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [7] | Wang Shaona, Liu Feixiang, Ying Chunmiao, Gao Chen, Zhang Yunke. Action mechanism of traditional Chinese medicine combined with bone marrow mesenchymal stem cells in regulating blood-brain barrier after cerebral ischemia reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(33): 5377-5384. |
| [8] | Wei Zhihui, Liu Feixiang, Pan Xiaolong, Sun Shibiao, Zhang Yunke. Mechanism of Rehmannia glutinosa combined with mesenchymal stem cells in treating ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(19): 3070-3076. |
| [9] | Song Peng, Liu Yizheng, Lei Zhen. Vascular regeneration of the prefabricated flap using expanded polytetrafluoroethylene membrane barrier material [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(12): 1914-1919. |
| [10] | Li Yong, Yuan Jianmei, Lu Danni, Ren Mihong, Deng Bowen, Wang Jiajun, Ma Rong, Xie Qian, Li Jinxiu, Xu Zhuo, Wang Jian. Comparison of cerebral microcirculation perfusion in rat models of middle cerebral artery occlusion prepared through common carotid artery insertion and external carotid artery insertion [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(11): 1683-1691. |
| [11] | Hu Fei, Wang Jie. Safety and efficacy of mesenchymal stem cells in the treatment of ischemic stroke: a meta-analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(1): 76-82. |
| [12] | Li Zhiyi, He Pengcheng, Bian Tianyue, Xiao Yuxia, Gao Lu, Liu Huasheng. Bibliometric and visualized analysis of ferroptosis mechanism research [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1202-1209. |
| [13] | Fan Yongfu, Su Kaiqi, Yuan Jie, Nie Chenchen, Ruan Xiaodi, Duan Zhaoyuan, Feng Xiaodong. Relationship between iron metabolism and ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(32): 5223-5228. |
| [14] | Yan Nan, Wu Yanlong, Tang Xiaohui, Zhang Xiaoyan, Wang Hui, Yang Tianze, Zhou Maochun, Wang Zhengdong, Yang Xiaoxia. Bone marrow mesenchymal stem cells may alleviate brain damage caused by the microglial overactivation in the cortex around ischemic site of stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3790-3795. |
| [15] | Chen Na, Wang Xiaohan, Zhang Yunke. Effect and mechanism of mesenchymal stem cells on aging-related ischemic stroke [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3914-3920. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||