Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (20): 3265-3271.doi: 10.12307/2024.350
Previous Articles Next Articles
Mechanism of action and related signaling pathways of long non-coding RNAs in neuroimmuno-inflammatory response after ischemic stroke
Wan Jun1, Bai Yanjie2, Wang Yan2, Chen Shuying1, Chen Limin1, Xiao Yuqian1, Sun Kexin1
- 1Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China; 2The First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, Henan Province, China
-
Received:2023-05-18Accepted:2023-06-15Online:2024-07-18Published:2023-09-11 -
Contact:Bai Yanjie, Associate chief physician, Master’s supervisor, the First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou 450000, Henan Province, China -
About author:Wan Jun, Master candidate, Henan University of Chinese Medicine, Zhengzhou 450046, Henan Province, China -
Supported by:Special Key Course of TCM Scientific Research in Henan Province, No. 20-21ZY1009 (to BYJ); Henan Province Traditional Chinese Medicine Inheritance and Innovation Talent Project (Zhongjing Project) for TCM Discipline Top Talents, No. CZ0237-08 (to BYJ); Henan Provincial Science and Technology Research Plan, No. 222102310529 (to BYJ); National Clinical Research Base of Traditional Chinese Medicine of Henan Provincial Health Commission, No. 2022JDZX005 (to BYJ); Henan Province TCM Top Talent Training Project Special Topic, No. 2022ZYBJ07 (to BYJ)
CLC Number:
Cite this article
Wan Jun, Bai Yanjie, Wang Yan, Chen Shuying, Chen Limin, Xiao Yuqian, Sun Kexin. Mechanism of action and related signaling pathways of long non-coding RNAs in neuroimmuno-inflammatory response after ischemic stroke[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3265-3271.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
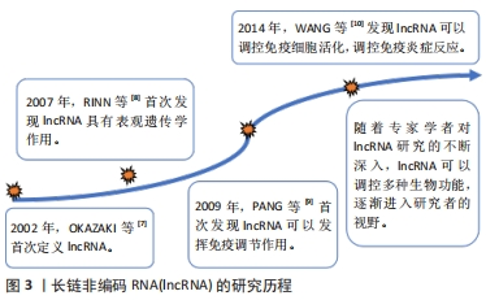
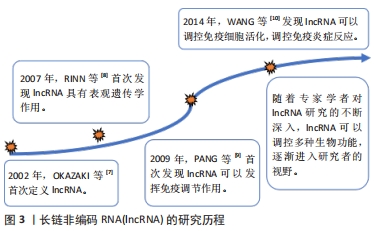
2.1 lncRNA的概述 2.1.1 lncRNA的研究历程 lncRNA是一类存在于细胞核或者细胞质中的长度为200多个核苷酸的全转录本[6]。在2002年,OKAZAKI等[7]在鉴定小鼠的所有全长非编码RNA转录本时,首次提出lncRNA的概念,但因为其没有蛋白质编码的潜力,所以被认为是基因组在进化过程中出现的没有其他生物学功能的基因组,不被大家所关注。之后有学者发现lncRNA HOTAIR能够与蛋白复合体抑制复合物2相互作用发挥表观遗传的作用[8]。随后,越来越多的研究表明lncRNA具有重要的调节功能,特别是在调节细胞分化的表观遗传过程方面。此外,有研究者通过基因组检测发现在CD8+T细胞中存在许多随着淋巴细胞分化或活化而动态变化的lncRNA,此研究打开了lncRNA调控免疫功能的大门[9]。lncRNA在许多生物过程中发挥着重要作用,但是在调节免疫细胞分化过程始终没有定论。所以WANG等[10]通过研究发现在人的树突状细胞中lncRNA可以与细胞质中的转录激活因子3结合,进而促进其磷酸化调控树突状细胞的分化,这项研究为lncRNA在免疫系统的调节中提供了新的方向。随着各位专家学者对lncRNA的深入研究,其在各类疾病中的调控作用正被逐步地挖掘,目前LncRNA逐渐成为各位研究者讨论的热点话题。lncRNA的研究历程见图3。"

2.1.2 lncRNA的功能 随着研究的不断深入,发现lncRNA可能具有较短的开放阅读框架,能够与蛋白质、染色质或者RNA自身的相互作用来调节各种细胞功能。研究人员根据lncRNA与邻近编码区基因组的相对位置将其分为6类[11]:由正义编码链外显子转录而成,通常与蛋白质编码基因覆盖或重叠的正义lncRNA、由编码反义蛋白的基因转录而来的反义lncRNA、两个基因之间的基因组间隔中的基因间lncRNA、由第二转录本的内含子形成的内含子lncRNA、从相反方向转录,相距约1 kb的双向lncRNA和来自蛋白质编码基因的增强子区域的增强子lncRNA。此外lncRNA除了可以充当支架,还可以从核酸或蛋白质靶标中募集或阻隔效应蛋白,并且在调控基因程序中发挥关键作用[12]。 lncRNA在生物学中的功能可以归纳为以下3种机制[13-15]:①作为基因组结构和转录的主要调节因子,参与造血基因表达网络和造血干细胞功能的调节;②参与mRNA加工、转录后控制和蛋白活性调控;③充当关键信号转导介质,参与细胞核结构域的组织功能。此外,lncRNA作为基因间转录、翻译和表观遗传水平的调控序列,在中枢神经系统中表达显著,有效调控缺血性脑卒中的发生发展[16]。近年来,亦有研究证明lncRNA除了调控表观遗传学水平、与免疫细胞相互作用外,还可以作为神经胶质细胞和神经元之间的通讯工具,有效维持中枢神经系统微环境稳态[17]。"

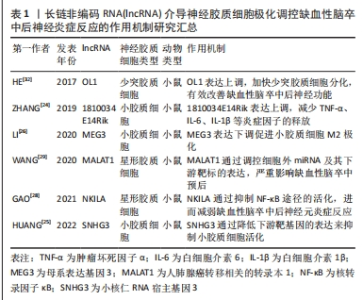
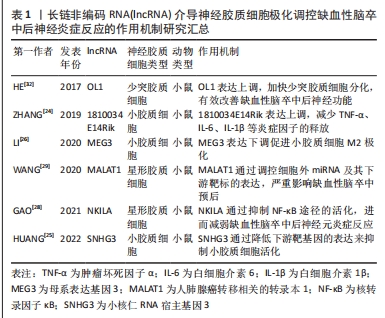
2.2 lncRNA作用神经胶质细胞调控缺血性脑卒中后神经免疫炎症级联反应 缺血性脑卒中发病机制的主要特征是神经炎症反应。在缺血性脑卒中早期易触发常驻炎症细胞的快速活化、炎性细胞因子和细胞间核因子易位等机制进而诱导机体神经免疫炎症级联反应,进一步加重脑组织损伤[18]。 根据最新的研究发现,lncRNA可以通过多种方式参与缺血性脑卒中后神经免疫炎症反应。一方面,lncRNA可以调节炎症和免疫细胞的活化和分化,从而影响炎症反应的程度和持续时间;另一方面,lncRNA还可以在缺血性脑损伤后调节细胞凋亡和自噬等过程,从而影响神经元的存活和功能恢复[19-20]。 神经胶质细胞包括小胶质细胞、星形胶质细胞和少突胶质细胞,是中枢神经系统梗死周围环境的主要组成部分,与缺血性卒中后免疫炎症调节密切相关[21]。神经和免疫系统控制全身机体稳态,并以相互协调的方式对各种类型的组织损伤作出反应,特别是缺血性脑组织损伤。脑缺血导致神经元细胞死亡或浸润周围免疫细胞,从而诱发影响卒中后功能预后的神经炎症[22]。研究显示,某些lncRNA通过介导神经胶质细胞的极化触发神经免疫轴的激活,进而直接或间接影响缺血性脑卒中的启动、传播和消退阶段。下文将主要阐述lncRNA如何通过调控小胶质细胞、星形胶质细胞及少突胶质细胞等免疫细胞活化抑制缺血性脑卒中后神经免疫炎症级联反应,进而发挥缺血性脑卒中后神经保护作用。 2.2.1 lncRNA与小胶质细胞 小胶质细胞被称为大脑中的先天免疫细胞,其激活形式包括促炎M1表型和抗炎M2表型。在缺血性脑卒中早期,激活M1型小胶质细胞通过分泌促炎细胞因子如肿瘤坏死因子α和白细胞介素6等损伤神经元功能;随着疾病的发展,激活M2型小胶质细胞促进修复抗炎反应,保护脑组织损伤。 lncRNA是调节小胶质细胞极化和激活的重要调控因子,小胶质细胞活化被认为是神经炎症的重要标志物[23]。一些lncRNA通过改变小胶质细胞M1/M2极化状态参与缺血性脑卒中的发生发展。有研究表明,lncRNA-1810034E14Rik在氧糖剥夺诱导的小胶质细胞中显著降低,在体外和体内实验中过表达1810034E14Rik可减少缺血性脑卒中模型小鼠梗死体积,降低神经元中炎性细胞因子的表达,这表明lncRNA-1810034E14Rik能够发挥抑制小胶质细胞活化、减轻神经炎症的作用[24]。HUANG等[25]通过动物实验发现,在脑缺血再灌注损伤模型小鼠中lncRNA小核仁RNA宿主基因3(small nucleolar RNA host gene 3,SNHG3)通过降低组蛋白去乙酰化酶3 (histone deacetylase 3,HDAC3)表达来抑制小胶质细胞活化和肿瘤坏死因子α、白细胞介素6和白细胞介素1β等炎症因子的释放,减弱脑卒中后炎症反应。此外,lncRNA母系表达基因3(maternally expressed gene 3,MEG3)在大脑中动脉闭塞再灌注小鼠脑组织和脑缺血再灌注损伤诱导的神经元细胞中高度表达。MEG3可以结合Krüppel样因子4(Krüppel-like factor 4,KLF4)并抑制其蛋白进程。当MEG3缺失时通过靶向KLF4增强小胶质细胞M2极化,最终MEG3的抑制缓解了缺血性脑卒中后神经炎症的损伤[26]。因此,缺血性脑卒中后活化的小胶质细胞可产生神经毒性和神经保护的双重作用,lncRNA通过调控小胶质细胞极化抑制神经免疫炎症级联机制改善缺血性脑卒中后神经功能。 2.2.2 lncRNA与星形胶质细胞 星形胶质细胞是在中枢神经系统中占比最大的神经胶质细胞。脑卒中后星形胶质细胞呈现出对早期脑组织保护至关重要的反应状态,反应性星形胶质细胞亚群的化学遗传消融会显著损害血管和细胞外基质重塑,进而使血管通透性加剧,诱导持续的细胞死亡和运动功能障碍[27]。在缺血性脑卒中发展过程中lncRNA通过不同的调控形式影响星形胶质细胞的增殖、活化,进而抑制神经炎症反应对脑组织发挥保护作用。GAO等[28]通过动物实验构建缺血性脑卒中小鼠模型实验发现与核转录因子κB相互作用的lncRNA NKILA(NF-kappaB interacting lncRNA)可以阻断核转录因子κB途径的激活,抑制星形胶质细胞增殖以及神经元炎症反应。此外,还有研究发现lncRNA人肺腺癌转移相关的转录本1(metastasis-associated lung adenocarcinoma transcript 1,MALATl)在脑缺血再灌注损伤处理的小鼠星形胶质细胞中显著上调,有趣的是体外缺血再灌注条件下MALAT1通过竞争性结合miR-145来调控下游靶标血清水通道蛋白4的表达,进一步加重脑组织损伤[29]。这些提示lncRNA可能通过作用星形胶质细胞显著抑制卒中后神经炎症反应,在缺血性脑卒中功能障碍恢复中发挥重要作用。 2.2.3 lncRNA与少突胶质细胞 少突胶质细胞是中枢神经系统的髓磷脂形成细胞,也是神经元的终身伴侣,对髓鞘产生和保持轴突的完整性至关重要[30]。研究人员发现lncRNA在少突胶质细胞前体细胞与神经干细胞分化过程中起到一定的调节作用,并且可能参与少突胶质发生过程[31]。HE等[32]研究了OL谱系细胞中的lncRNA转录组,发现lncRNA OL1过表达时可促进发育中大脑的少突胶质细胞分化,但是lncRNA OL1的失活将严重影响缺血缺氧脑组织损伤后髓鞘形成并诱导髓鞘再生功能障碍。目前有关lncRNA调控少突胶质细胞作用于缺血性脑卒中后神经炎症的机制的研究相对较少,但毋庸置疑的是lncRNA参与缺血性脑卒中后诱导的神经炎症免疫应答反应有望成为缺血性脑卒中治疗策略的新突破靶点。文章总结了lncRNA介导神经胶质细胞极化调控缺血性脑卒中后神经炎症反应的作用机制进展见表1。"
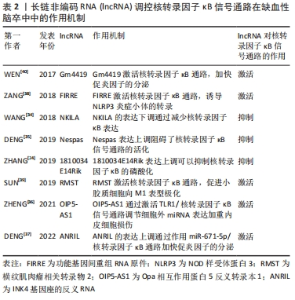
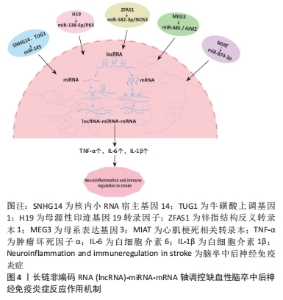
2.3 lncRNA调节缺血性脑卒中后神经免疫炎症级联反应的相关信号通路 2.3.1 核转录因子κB信号通路 核转录因子κB家族由RelA (p65)、RelB、c-Rel、核转录因子κB1 (p50)和核转录因子κB2(p52)5个成员组成。在正常生理情况下处于失活状态,当机体受到外界刺激时诱发肿瘤坏死因子α、白细胞介素炎症因子等的大量释放或者机体遭受细胞核损伤时,DNA结合的异二聚体和同二聚体由于失去与抑制性蛋白IκB的联系而积聚在细胞核中,并诱导靶基因启动转录过程[33]。核转录因子κB是一个涉及多种生物过程的转录因子家族,包括炎症、凋亡、增殖和发育。作为炎症免疫信号级联的组成部分,核转录因子κB信号通路被认为是正常生理学中炎症反应的中心调节因子,在缺血性脑卒中神经免疫炎症级联反应中发挥重要作用。 在缺血再灌注损伤诱导的脑卒中模型中lncRNA NKILA下调可以抑制模型小鼠海马神经元中的核转录因子κB信号传导,诱发炎症因子分泌,进而介导随后的神经元死亡[34]。DENG等[35]通过体内体外研究证明在缺血性脑卒中海马神经细胞中lncRNA Nespas的表达上调,Nespas过表达抑制了炎性细胞因子的分泌和核转录因子κB信号通路的激活,这说明lncRNA Nespas在缺血性脑卒中的小鼠中发挥抗炎和抑制细胞凋亡的作用。除此之外,lncRNA-1810034E14Rik在缺血性脑卒中中同样发挥显著的抗炎作用,其机制是lncRNA-1810034E14Rik过表达可以显著抑制海马神经元中小胶质细胞的活化进而抑制p65的磷酸化,进而减少炎症因子的释放[24]。以上这些lncRNA通过抑制核转录因子κB信号通路的激活,进一步抑制神经炎症级联反应,有效缓解脑卒中后神经功能损伤。 内皮细胞损伤触发神经炎症反应也是缺血性脑卒中发病的主要原因。lncRNA Opa相互作用蛋白5反义转录本1 (opa-interacting protein 5 antisense transcript 1,OIP5-AS1)在氧化低密度脂蛋白诱导的内皮细胞损伤中发挥重要作用。Zheng等[36]发现lncRNA OIP5-AS1通过激活TLR1/核转录因子κB信号通路调节miR-98-5p介导的高迁移率族蛋白B1来加速低密度脂蛋白诱导的内皮细胞损伤。还有其他研究人员探索发现miR-671-5p是lncRNA INK4基因座的反义RNA(antisense non-coding RNA in the INK4 locus,ANRIL)的直接靶标之一,核转录因子κB是miR-671-5p的下游因子。在缺血性脑卒中模型小鼠实验中,lncRNA ANRIL的表达量下调通过调节miR-671-5p/核转录因子κB通路减少了核转录因子κB、促炎细胞因子(白细胞介素1β、白细胞介素6和肿瘤坏死因子α)的表达,缓解了缺血性脑卒中诱导的神经炎症[37]。 有研究证明,lncRNA 功能基因间重组RNA原件(functional intergenic repeating RNA element,FIRRE)不仅可以促进核转录因子κB通路活化,而且活化的核转录因子κB也可以增加缺氧处理的脑小胶质细胞中的FIRRE表达上调。因此,FIRRE和核转录因子κB相互作用共同促进NOD样受体蛋白3炎症小体的转录,从而诱发炎症因子释放,进一步加重脑损伤[38]。SUN等[39]通过建立缺血性脑卒中细胞模型发现lncRNA 横纹肌肉瘤相关转录物2(rhabdomyosarcoma 2-associated transcript,RMST)通过转化生长因子β激活激酶1介导的核转录因子κB通路与异质核糖核蛋白K竞争性相互作用,促进了缺血再灌注损伤诱导的小胶质细胞M1极化,加重缺血性脑卒中后的神经炎症反应。此外多数证据表示过度活化的小胶质细胞分泌大量的炎性细胞因子诱导炎症发生,不利于缺血性脑卒中后的神经功能恢复。例如高表达的lncRNA Gm4419通过调控IκBα的磷酸化状态,导致肿瘤坏死因子α、白细胞介素1β和白细胞介素6转录激活的细胞核核转录因子κB水平增加,进一步阻碍了脑小胶质细胞缺血再灌注诱导的神经元损伤[40]。综上所述,核转录因子κB信号通路是触发缺血性脑卒中后的神经免疫炎症级联反应的核心通路,lncRNA通过调控核转录因子κB磷酸化能够有效缓解缺血性脑卒中后炎症损伤。LncRNA调控核转录因子κB信号通路在缺血性脑卒中疾病中的作用机制见表2。"
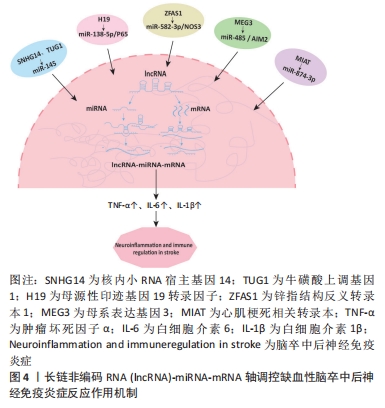
2.3.2 lncRNA-miRNA-mRNA轴 lncRNA作为miRNA的前体或宿主,通过竞争性结合或海绵效应直接或间接影响miRNA表达。同时miRNA反过来通过靶向mRNA的3′端结合位点的不完全互补调节靶基因的转录和表观遗传水平[41]。还有一些lncRNA与mRNA结合,从而直接与miRNA竞争,破坏miRNA对mRNA的转录修饰作用。因此lncRNA-miRNA-mRNA轴有助于调控疾病的发生发展。近年来越来越多的研究表明,缺血性卒中后特异性miRNA的抗炎或促炎作用受到lncRNA的高度调控,lncRNA-miRNA-mRNA轴在缺血性脑卒中神经免疫炎症级联反应中发挥至关重要的作用。 多项研究发现,miR-145过表达具有抑制缺血性卒中后炎症损伤的潜力。QI等[42]通过动物实验发现敲低lncRNA 核内小RNA宿主基因14 (small nucleolar RNA host gene 14,SNHG14)可使miR-145-5p的表达上调,而miR-145-5p过表达可以逆转由lncRNA SNHG14表达过量诱导的海马神经元中相关蛋白的增加并降低了脑小胶质细胞分泌的肿瘤坏死因子α和氮氧化合物的表达。lncRNA牛磺酸上调基因1(taurine upregulated gene 1,TUG1)也能够直接与miR-145a-5p结合通过核转录因子κB途径调控炎症反应,具体机制是lncRNA TUG1能够负向调控miR-145a-5p表达激活核转录因子κB磷酸化在缺血性脑卒中早期通过调节小胶质细胞极化和炎症细胞因子的产生,减弱卒中后神经炎症反应[43]。同时lncRNA TUG1也可以在卒中后通过miR-145a-5p/TLR4轴促进NLRP3炎症小体依赖性焦亡,加重缺血性脑卒中疾病发展[44]。 lncRNA MEG3可以通过此调控轴介导缺血性脑卒中期间炎症小体缺失的调节机制。在缺血性脑卒中模型小鼠的神经细胞中lncRNA MEG3作为分子海绵抑制miR-485的表达,黑色素瘤缺乏因子2(absent in melanoma 2,AIM2)作为miR-485的直接下游靶标促进卒中后细胞焦亡和炎症的发生。当敲低lncRNA MEG3表达时可逆转靶向miR-485 / AIM2轴介导的焦亡过程[45]。此外,lncRNA家族母源性印迹基因19转录因子(maternally expressed transcript 19,H19)在卒中后脑组织中高度表达,参与缺血性脑卒中后神经炎症的调控。Li等[46]研究发现lncRNA H19对miR-138-5p的表达有负调控作用。H19通过调节miR-138-5p和p65的表达,促进缺血性脑卒中模型小鼠的炎症反应并改善其神经功能。其次,在缺血性脑卒中患者血清中lncRNA心肌梗死相关转录本(myocardial infarction associated transcript,MIAT)和白细胞介素1β上调,沉默MIAT、白细胞介素1β或促进miR-874-3p表达可改善脑卒中患者的行为活动和神经功能[47]。此外与H19和MIAT表达不同的是,lncRNA锌指结构反义转录本1(zinc finger protein antisense chain 1,ZFAS1)在缺血性脑卒中患者血浆中显著下调。ZFAS1可作为miR-582-3p的分子海绵负向调节miR-582-3p/NOS3轴的表达,抑制肿瘤坏死因子α、白细胞介素1β和一氧化氮的释放,防止神经元损伤并调节炎症发生[48]。由此可知,lncRNA-miRNA-mRNA轴通过调控不同分子机制参与缺血性脑卒中后神经炎症过程,有效改善神经功能。lncRNA-miRNA-mRNA轴调控缺血性脑卒中后神经免疫炎症反应作用机制见图4。"
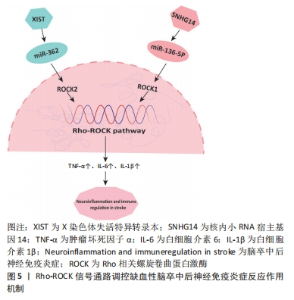
2.3.3 Rho-ROCK信号通路 RhoA是一种小的鸟苷三磷酸酶(guanosine triphosphatases,GTPase)蛋白,当与二磷酸鸟苷(guanosine diphosphate,GDP)结合时,Rho不活跃,而通过鸟嘌呤交换因子(guanine exchange factors,GEP)与GTP结合时Rho变得活跃,随后激活下游效应物Rho相关螺旋卷曲蛋白激酶(Rho-associated coiled helix kinase,ROCK)[49]。ROCK是一个丝氨酸/苏氨酸激酶家族,包括ROCK1和ROCK2两种亚型,其中ROCK2转录本在大脑中表达丰富[50]。缺血性脑卒中后ROCK激活通过刺激神经炎症导致急性期脑组织损伤。一方面ROCK通过内皮源性一氧化氮合酶还原介导p-选择素和细胞间黏附分子1等的过表达;另一方面,用ROCK抑制剂治疗培养的小胶质细胞可降低白细胞介素6和肿瘤坏死因子α等促炎因子表达,并增加白细胞介素10等抗炎因子的释放量[51]。 例如在缺血性脑卒中小鼠的缺血半暗带中lncRNA X染色体失活特异转录本(X-inactive specific transcript,XIST)表达升高,抑制XIST显著阻碍细胞凋亡的发生,降低了脑卒中小鼠的神经损伤,其机制可能是XIST的耗竭通过miR-362/ROCK2轴减弱了缺血性脑卒中诱导的神经损伤和炎症介质的释放[52]。此外,lncRNA SNHG14也可以通过调控ROCK通路介导卒中后神经炎症反应。ZHONG等[53]建立缺血性脑卒中动物模型发现lncRNA SNHG14在脑卒中小鼠的血清中表达上调,作为miR-136-5p的分子海绵,能够正向调控ROCK1的表达。SNHG14通过提高ROCK1的表达来促进大脑神经损伤和炎症反应,同时降低缺血性脑卒中诱导的miR-136-5p水平。因此,Rho-ROCK信号通路有望成为调节缺血性脑卒中后炎症的一个有前途的靶点通路。Rho-ROCK信号通路调控缺血性脑卒中后神经免疫炎症反应作用机制见图5。"

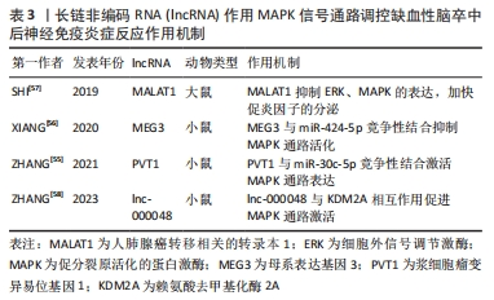
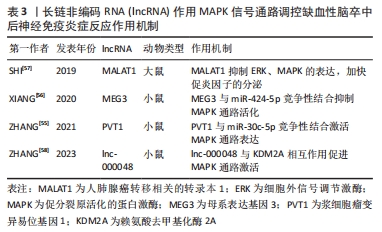
2.3.4 MAPK信号通路 促分裂原活化的蛋白激酶(mitogen-activated protein kinase,MAPK)信号通路是一个级联磷酸化过程,可参与调控细胞多种生理病理过程,是介导缺血性脑卒中后神经炎症级联反应的重要信号通路[54]。lncRNA通过调控MAPK信号通路的磷酸化进而影响缺血性脑卒中后炎症过程,有效缓解缺血性脑卒中疾病的发生发展。例如 lncRNA浆细胞瘤变异易位基因1(plasmacytoma variant translocation 1,PVT1)在经过脑缺血再灌注治疗的小鼠大脑皮质神经元中上调,随后作为竞争性内源性RNA与miR-30c-5p竞争,从而负向调节其内源性靶标ROCK2,最终导致MAPK信号的激活,增加了脑卒中后炎症危险[55]。lncRNA MEG3在缺血性脑卒中模型小鼠中高度表达,XIANG等[56]发现敲低MEG3通过抑制MAPK途径增加了miR-424-5p的表达,进而减少缺血性脑卒中小鼠梗死体积并改善神经功能。此外lncRNA MALAT1也被探究出可能通过阻滞ERK/MAPK信号通路促进炎性细胞因子的分泌,最终提高缺血性脑卒中大鼠的神经元凋亡水平,加重脑组织损伤[57]。 最新研究结果显示,lnc-000048通过与赖氨酸去甲基化酶2A相互作用降低了组蛋白去甲基化酶的活性,间接调节了MAP2K2的细胞外信号调节激酶磷酸化,最终通过MAPK途径激活炎症反应[58]。因此靶向lnc-000048 /KDM1A/MAP2K2/ERK轴可能是预防缺血性脑卒中疾病的重要手段。lncRNA作用MAPK信号通路调控缺血性脑卒中后神经免疫炎症反应作用机制见表3。"

2.3.5 其他信号通路 研究发现有其他lncRNA也可以通过介导蛋白激酶B(protein kinase B,PKB,又称AKT)信号通路、细胞外信号调节激酶(extracellular signal-regulated kinase,ERK)信号通路等调控缺血性脑卒中后神经免疫炎症级联反应。 例如,AKT通路已被证明参与缺血性脑卒中后的神经炎症调节。GAO等[59]发现当机体受到缺血缺氧等外界刺激时,血浆中lncRNA FAL1的表达显著降低。但lncRNA FAL1的过表达抑制了由缺氧复氧诱导的白细胞介素6、单核细胞趋化蛋白1的分泌,并通过调节PAK1/AKT信号通路保护脑微血管内皮细胞免受炎症损伤。同时敲低lncRNA MALAT1调控miR-126的表达并通过激活PI3K/Akt通路也能够对脑微血管内皮细胞发挥保护作用[60]。此外在脑缺血再灌注损伤模型血清中lncRNA核富集转录本1(Nuclear enriched abundant transcript 1,NEAT1)的表达显著增加,抑制lncRNA NEAT1的表达可以降低AKT / STAT3途径的活性并促进小胶质细胞向M1表型的极化,最终减弱炎症反应的发生[61]。总之,lncRNA通过靶向AKT通路有效改善卒中后神经炎症损伤。 ERK通路也是参与缺血性脑卒中后神经免疫炎症反应的重要途径。有研究发现lncRNA MIAT通过EGR1-ELK1-ERK途径机械调节颈动脉平滑肌细胞表型向促炎巨噬细胞样细胞的转变,增加发生缺血性脑卒中等血管性疾病的风险[62]。另外GUO等[63]研究表明,沉默lncRNA SNHG15可以通过作用miR-18a并激活ERK信号通路降低促炎细胞因子肿瘤坏死因子α和白细胞介素1β水平,减弱缺血性脑卒中后神经炎症反应。综上所述,lncRNA可以通过与不同的信号通路相互作用来调控神经炎症机制,这为治疗缺血性脑卒中提供了很好的实验依据和思路。"

| [1] CAPRIO FZ, SOROND FA. Cerebrovascular disease: primary and secondary stroke prevention. Med Clin North Am. 2019;103(2):295-308. [2] Global, regional, and national burden of stroke and its risk factors, 1990-2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Neurol. 2021; 20(10):795-820. [3] PLUTA R, JANUSZEWSKI S, CZUCZWAR SJ. Neuroinflammation in post-ischemic neurodegeneration of the brain: friend, foe, or both. Int J Mol Sci. 2021;22(9):4405. [4] JURCAU A, SIMION A. Neuroinflammation in cerebral ischemia and ischemia/reperfusion injuries: from pathophysiology to therapeutic strategies. Int J Mol Sci. 2021; 23(1):14. [5] CHEN Z, WU H, ZHANG M. Long non-coding RNA: an underlying bridge linking neuroinflammation and central nervous system diseases. Neurochem Int. 2021;148: 105101. [6] BEGOLLI R, SIDERIS N, GIAKOUNTIS A. LncRNAs as chromatin regulators in cancer: from molecular function to clinical potential. Cancers (Basel). 2019;11(10):1524. [7] OKAZAKI Y, FURUNO M, KASUKAWA T, et al. Analysis of the mouse transcriptome based on functional annotation of 60,770 full-length cDNAs. Nature. 2002;420(6915):563-573. [8] RINN JL, KERTESZ M, WANG JK, et al. Functional demarcation of active and silent chromatin domains in human HOX loci by noncoding RNAs. Cell. 2007;129(7):1311-1323. [9] PANG KC, DINGER ME, MERCER TR, et al. Genome-wide identification of long noncoding RNAs in CD8+ T cells. J Immunol. 2009;182(12):7738-7748. [10] WANG P, XUE Y, HAN Y, et al. The STAT3-binding long noncoding RNA lnc-DC controls human dendritic cell differentiation. Science. 2014;344(6181):310-313. [11] ZHU L, LI N, SUN L, et al. Non-coding RNAs: the key detectors and regulators in cardiovascular disease. Genomics. 2021;113(1 Pt 2):1233-1246. [12] ALVAREZ-DOMINGUEZ JR, LODISH HF. Emerging mechanisms of long noncoding RNA function during normal and malignant hematopoiesis. Blood. 2017;130(18):1965-1975. [13] QIU Y, XU M, HUANG S. Long noncoding RNAs: emerging regulators of normal and malignant hematopoiesis. Blood. 2021;138(23):2327-2336. [14] SLABY O, LAGA R, SEDLACEK O. Therapeutic targeting of non-coding RNAs in cancer. Biochem J. 2017;474(24):4219-4251. [15] LIN C, YANG L. Long noncoding RNA in cancer: wiring signaling circuitry. Trends Cell Biol. 2018;28(4):287-301. [16] ZHANG X, WANG W, ZHU W, et al. Mechanisms and functions of long non-coding RNAs at multiple regulatory levels. Int J Mol Sci. 2019;20(22):5573. [17] WANG ZY, WEN ZJ, XU HM, et al. Exosomal noncoding RNAs in central nervous system diseases: biological functions and potential clinical applications. Front Mol Neurosci. 2022;15:1004221. [18] DAIDONE M, CATALDI M, PINTO A, et al. Non-coding RNAs and other determinants of neuroinflammation and endothelial dysfunction: regulation of gene expression in the acute phase of ischemic stroke and possible therapeutic applications. Neural Regen Res. 2021;16(11):2154-2158. [19] GHAFOURI-FARD S, SHOOREI H, TAHERI M. Non-coding RNAs participate in the ischemia-reperfusion injury. Biomed Pharmacother. 2020;129:110419. [20] TRIPATHI S, SHREE B, MOHAPATRA S, et al. The Expanding regulatory mechanisms and cellular functions of long non-coding RNAs (lncRNAs) in neuroinflammation. Mol Neurobiol. 2021;58(6):2916-2939. [21] XU S, LU J, SHAO A, et al. Glial cells: role of the immune response in ischemic stroke. Front Immunol. 2020;11:294. [22] LI S, CAO Y, ZHANG H, et al. Construction of lncRNA-mediated ceRNA network for investigating immune pathogenesis of ischemic stroke. Mol Neurobiol. 2021;58(9):4758-4769. [23] SCHIRMER L, VELMESHEV D, HOLMQVIST S, et al. Neuronal vulnerability and multilineage diversity in multiple sclerosis. Nature. 2019;573(7772):75-82. [24] ZHANG X, ZHU XL, JI BY, et al. LncRNA-1810034E14Rik reduces microglia activation in experimental ischemic stroke. J Neuroinflammation. 2019;16(1):75. [25] HUANG D, CAO Y, ZU T, et al. Interference with long noncoding RNA SNHG3 alleviates cerebral ischemia-reperfusion injury by inhibiting microglial activation. J Leukoc Biol. 2022;111(4):759-769. [26] LI T, LUO Y, ZHANG P, et al. LncRNA MEG3 regulates microglial polarization through KLF4 to affect cerebral ischemia-reperfusion injury. J Appl Physiol (1985). 2020;129(6):1460-1467. [27] WILLIAMSON MR, FUERTES CJA, DUNN AK, et al. Reactive astrocytes facilitate vascular repair and remodeling after stroke. Cell Rep. 2021;35(4):109048. [28] GAO W, NING Y, PENG Y, et al. LncRNA NKILA relieves astrocyte inflammation and neuronal oxidative stress after cerebral ischemia/reperfusion by inhibiting the NF-κB pathway. Mol Immunol. 2021;139:32-41. [29] WANG H, ZHENG X, JIN J, et al. LncRNA MALAT1 silencing protects against cerebral ischemia-reperfusion injury through miR-145 to regulate AQP4. J Biomed Sci. 2020; 27(1):40. [30] BARATEIRO A, BRITES D, FERNANDES A. Oligodendrocyte development and myelination in neurodevelopment: molecular mechanisms in health and disease. Curr Pharm Des. 2016;22(6):656-679. [31] DONG X, CHEN K, CUEVAS-DIAZ DURAN R, et al. Comprehensive identification of long non-coding RNAs in purified cell types from the brain reveals functional lncRNA in OPC fate determination. PLoS Genet. 2015;11(12):e1005669. [32] HED, WANG J, LU Y, et al. lncRNA functional networks in oligodendrocytes reveal stage-specific myelination control by an lncOL1/Suz12 complex in the CNS. Neuron. 2017;93(2):362-378. [33] CILDIR G, LOW KC, TERGOANKAR V. Noncanonical NF-κB Signaling in health and disease. Trends Mol Med. 2016;22(5):414-429. [34] WANG M, JIANG YM, XIA LY, et al. LncRNA NKILA upregulation mediates oxygen glucose deprivation/re-oxygenation-induced neuronal cell death by inhibiting NF-κB signaling. Biochem Biophys Res Commun. 2018;503(4):2524-2530. [35] DENG Y, CHEN D, WANG L, et al. Silencing of long noncoding RNA nespas aggravates microglial cell death and neuroinflammation in ischemic stroke. Stroke. 2019;50(7): 1850-1858. [36] ZHENG Z, ZHANG G, LIANG X, et al. LncRNA OIP5-AS1 facilitates ox-LDL-induced endothelial cell injury through the miR-98-5p/HMGB1 axis. Mol Cell Biochem. 2021; 476(1):443-455. [37] DENG L, JIANG J, CHEN S, et al. Long non-coding RNA ANRIL downregulation alleviates neuroinflammation in an Ischemia Stroke Model via Modulation of the miR-671-5p/NF-κB pathway. Neurochem Res. 2022;47(7):2002-2015. [38] ZANG Y, ZHOU X, WANG Q, et al. LncRNA FIRRE/NF-kB feedback loop contributes to OGD/R injury of cerebral microglial cells. Biochem Biophys Res Commun. 2018;501(1): 131-138. [39] SUN XL, WANG ZL, WU Q, et al. LncRNA RMST activates TAK1-mediated NF-κB signaling and promotes activation of microglial cells via competitively binding with hnRNPK. IUBMB Life. 2019;71(11):1785-1793. [40] WEN Y, YU Y, FU X. LncRNA Gm4419 contributes to OGD/R injury of cerebral microglial cells via IκB phosphorylation and NF-κB activation. Biochem Biophys Res Commun. 2017;487(4):923-929. [41] XIONG W, QU Y, CHEN H, et al. Insight into long noncoding RNA-miRNA-mRNA axes in myocardial ischemia-reperfusion injury: the implications for mechanism and therapy. Epigenomics. 2019;11(15):1733-1748. [42] QI X, SHAO M, SUN H, et al. Long non-coding RNA SNHG14 promotes microglia activation by regulating miR-145-5p/PLA2G4A in cerebral infarction. Neuroscience. 2017;348:98-106. [43] WANG H, LIAO S, LI H, et al. Long non-coding RNA TUG1 sponges mir-145a-5p to regulate microglial polarization after oxygen-glucose deprivation. Front Mol Neurosci. 2019;12:215. [44] YAO M, LUO Y, LI H, et al. LncRNA Tug1 contributes post-stroke NLRP3 inflammasome-dependent pyroptosis via miR-145a-5p/Tlr4 axis. Mol Neurobiol. 2022;59(11):6701-6712. [45] LIANG J, WANG Q, LI JQ, et al. Long non-coding RNA MEG3 promotes cerebral ischemia-reperfusion injury through increasing pyroptosis by targeting miR-485/AIM2 axis. Exp Neurol. 2020;325:113139. [46] LI H, TANG C, WANG D. LncRNA H19 promotes inflammatory response induced by cerebral ischemia-reperfusion injury through regulating the miR-138-5p-p65 axis. Biochem Cell Biol. 2020;98(4):525-536. [47] ZHANG S, ZHANG Y, WANG N, et al. Long non-coding RNA MIAT impairs neurological function in ischemic stroke via up-regulating microRNA-874-3p-targeted IL1B. Brain Res Bull. 2021;175:81-89. [48] ZHANG Y, ZHANG Y. lncRNA ZFAS1 improves neuronal injury and inhibits inflammation, oxidative stress, and apoptosis by sponging miR-582 and upregulating NOS3 expression in cerebral ischemia/reperfusion injury. Inflammation. 2020;43(4):1337-1350. [49] KIMURA T, HORIKOSHI Y, KURIYAGAWA C, et al. Rho/ROCK pathway and noncoding RNAs: implications in ischemic stroke and spinal cord injury. Int J Mol Sci. 2021;22(21):11573. [50] GU H, YU SP, GUTEKUNST CA, et al. Inhibition of the Rho signaling pathway improves neurite outgrowth and neuronal differentiation of mouse neural stem cells. Int J Physiol Pathophysiol Pharmacol. 2013;5(1):11-20. [51] WANG QM, LIAO JK. ROCKs as immunomodulators of stroke. Expert Opin Ther Targets. 2012;16(10):1013-1025. [52] WANG J, FU Z, WANG M, et al. Knockdown of XIST attenuates cerebral ischemia/reperfusion injury through regulation of miR-362/ROCK2 axis. Neurochem Res. 2021; 46(8):2167-2180. [53] ZHONG Y, YU C, QIN W. LncRNA SNHG14 promotes inflammatory response induced by cerebral ischemia/reperfusion injury through regulating miR-136-5p /ROCK1. Cancer Gene Ther. 2019;26(7-8):234-247. [54] XU Q, ZHAO B, YE Y, et al. Relevant mediators involved in and therapies targeting the inflammatory response induced by activation of the NLRP3 inflammasome in ischemic stroke. Neuroinflammation. 2021;18(1):123. [55] ZHANG H, LI M, LIANG J, et al. Long Non-coding RNA PVT1 inhibits miR-30c-5p to upregulate Rock2 to modulate cerebral ischemia/reperfusion injury through MAPK signaling pathway activation. Mol Neurobiol. 2021;58(11):6032-6048. [56] XIANG Y, ZHANG Y, XIA Y, et al. LncRNA MEG3 targeting miR-424-5p via MAPK signaling pathway mediates neuronal apoptosis in ischemic stroke. Aging (Albany NY). 2020;12(4):3156-3174. [57] SHI YL, WANG Q, WEI JC. Influence of lncRNA-MALAT1 on neuronal apoptosis in rats with cerebral infarction through regulating the ERK/MAPK signaling pathway. Eur Rev Med Pharmacol Sci. 2019;23(18):8039-8048. [58] ZHANG S, SUN Y, XIAO Q, et al. Lnc_000048 promotes histone H3K4 methylation of MAP2K2 to reduce plaque stability by recruiting KDM1A in carotid atherosclerosis. Mol Neurobiol. 2023;60(5):2572-2586. [59] GAO M, FU J, WANG Y. The lncRNA FAL1 protects against hypoxia-reoxygenation- induced brain endothelial damages through regulating PAK1. J Bioenerg Biomembr. 2020;52(1):17-25. [60] ZANG L, YANG H, LI WJ, et al. LncRNA MALAT1 Promotes OGD-Induced apoptosis of brain microvascular endothelial cells by sponging miR-126 to repress PI3K/Akt signaling pathway. Neurochem Res. 2020;45(9):2091-2099. [61] NI X, SU Q, XIA W, et al. Knockdown lncRNA NEAT1 regulates the activation of microglia and reduces AKT signaling and neuronal apoptosis after cerebral ischemic reperfusion. Sci Rep. 2020;10(1):19658. [62] FASOLO F, JIN H, WINSKI G, et al. Long Noncoding RNA MIAT controls advanced atherosclerotic lesion formation and plaque destabilization. Circulation. 2021;144(19): 1567-1583. [63] GUO T, LIU Y, REN X, et al. Errate: promoting role of long non-coding RNA small nucleolar RNA host gene 15 (SNHG15) in neuronal injury following ischemic stroke via the MicroRNA-18a/CXC chemokine ligand 13 (CXCL13)/ERK/MEK axis. Med Sci Monit. 2022;28:e938473. |
| [1] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [2] | Yue Yun, Wang Peipei, Yuan Zhaohe, He Shengcun, Jia Xusheng, Liu Qian, Li Zhantao, Fu Huiling, Song Fei, Jia Menghui. Effects of croton cream on JNK/p38 MAPK signaling pathway and neuronal apoptosis in cerebral ischemia-reperfusion injury rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1186-1192. |
| [3] | Liu Xin, Hu Man, Zhao Wenjie, Zhang Yu, Meng Bo, Yang Sheng, Peng Qing, Zhang Liang, Wang Jingcheng. Cadmium promotes senescence of annulus fibrosus cells via activation of PI3K/Akt signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1217-1222. |
| [4] | Wei Juan, Li Ting, Huan Mengting, Xie Ying, Xie Zhouyu, Wei Qingbo, Wu Yunchuan. Mechanism by which static exercise improves insulin resistance in skeletal muscle of type 2 diabetes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1271-1276. |
| [5] | Wang Weiqing, Zhou Yue. Chronic inflammation regulates adipose tissue fibrosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1307-1312. |
| [6] | Yang Yifeng, Huang Jian, Ye Nan, Wang Lin. Ischemia-reperfusion injury in total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 955-960. |
| [7] | Zhang Kefan, Shi Hui. Research status and application prospect of cytokine therapy for osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(6): 961-967. |
| [8] | Liu Anhong, Cai Mengmeng, Han Xiao, Wang Zhanhui. Research status on element selection of medical magnesium alloy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 777-782. |
| [9] | Wei Yuanxun, Chen Feng, Lin Zonghan, Zhang Chi, Pan Chengzhen, Wei Zongbo. The mechanism of Notch signaling pathway in osteoporosis and its prevention and treatment with traditional Chinese medicine [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 587-593. |
| [10] | Liu Luxing, Di Mingyuan, Yang Qiang. Signaling pathways in the mechanism underlying active ingredients of Chinese medicine in the treatment of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 609-614. |
| [11] | Ye Haiming, Zeng Hui, Yang Qi, Zhang Geng, Weng Jian, Yu Fei. Exploration of signaling pathways with unclear action status and possible effects on related diseases or functions after knockdown of silencing information regulator 1 gene in chondrocytes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3123-3129. |
| [12] | Ji Weixiu, Bai Yi, Wang Shuo, Zhao Yungang. Protective effect and mechanism of 3-nitro-N-methyl salicylamide on the skeletal muscle of rats with limb ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(20): 3164-3169. |
| [13] | Liu Baofang, Xu Bin, Chen Lei. Pueraria decoction in the treatment of osteoarthritis: network pharmacology analysis and animal model validation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 193-199. |
| [14] | Zhao Kui, Pan Runsang, Lan Fengjun, Deng Jin, Chen Houping. Molecular mechanisms of autophagy-apoptosis interactions in osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(18): 2912-2917. |
| [15] | Yang Yunyun, Chen Qiqing, Zhao Jirong, Zhu Bao, Ma Dong, Huang Junkai, An Dehao, Zou Jipeng, Liu Weihang. Research status of traditional Chinese medicine monomer mediating related signaling pathways in treatment of intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(18): 2918-2924. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||

