Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (18): 2912-2917.doi: 10.12307/2024.058
Previous Articles Next Articles
Molecular mechanisms of autophagy-apoptosis interactions in osteoarthritis
Zhao Kui1, Pan Runsang2, Lan Fengjun1, Deng Jin3, Chen Houping2
- 1Clinical Medical College of Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2Women and Children’s Hospital Affiliated to Guizhou Medical University, Guiyang 550003, Guizhou Province, China; 3Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2023-03-01Accepted:2023-05-05Online:2024-06-28Published:2023-08-26 -
Contact:Chen Houping, Master, Chief physician, Women and Children’s Hospital Affiliated to Guizhou Medical University, Guiyang 550003, Guizhou Province, China -
About author:Zhao Kui, Master candidate, Clinical Medical College of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:National Natural Science Foundation of China, No. 82160543 (to CHP)
CLC Number:
Cite this article
Zhao Kui, Pan Runsang, Lan Fengjun, Deng Jin, Chen Houping. Molecular mechanisms of autophagy-apoptosis interactions in osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(18): 2912-2917.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
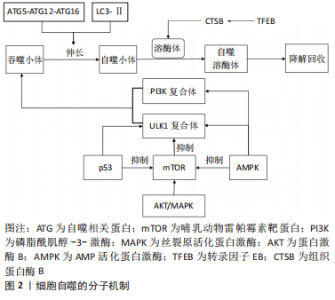
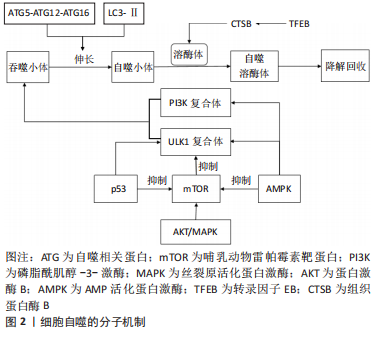
2.1 细胞自噬与骨关节炎 作为一种年龄相关的关节退行性疾病,骨关节炎已成为导致老年人肢体残疾的主要原因,它最常累及膝、髋关节。伴随着年龄的增加,骨关节也在发生退行性变,关节软骨逐渐磨损退化,关节部位骨骼摩擦加重,导致关节疼痛和活动障碍。迄今为止,骨关节炎无法被治愈,只能通过有限的治疗方法来延缓其进展,晚期行人工关节置换术来改善症状[8-9],但术后功能受到影响且关节假体寿命有限。细胞自噬促进了软骨细胞的动态平衡,广泛参与调控骨关节退变,已被证明可以保护软骨细胞免受损伤,减缓骨关节炎的发生和发展。因此,对骨关节退变中细胞自噬的分子机制进行研究,可能为骨关节炎的治疗策略带来新的见解。 2.1.1 细胞自噬的分子机制 细胞自噬是一种同时保护和杀死应激细胞的生物循环过程,通过将受损的蛋白质和细胞器隔离到溶酶体中进行降解和回收,降解后的成分被循环利用,用以构建新的细胞结构,或者加工后用作能量来源,以维持细胞的动态平衡。自噬最初由ULK1复合体介导,ULK1复合体与由Vps15、Vps34、Beclin-1组成的磷脂酰肌醇-3-激酶(phosphoinositide 3-kinase,PI3K)复合体形成一个吞噬小体,ATG5-ATG12-ATG16复合体与脂化微管相关蛋白1轻链3(LC3-Ⅱ)偶联反应刺激吞噬小体延伸形成自噬小体,然后与溶酶体融合形成自噬溶酶体,以降解内容物[10]。Beclin-1与LC3-Ⅱ是自噬的2个主要调节因子,Beclin-1可以使自噬小体成核,并与LC3-Ⅱ一起促进自噬小体的成熟[11]。哺乳动物雷帕霉素靶蛋白(mammalian target of rapamycin,mTOR)是自噬环节中的核心分子,在营养充足时,蛋白激酶B(protein kinase B,AKT)和丝裂原活化蛋白激酶(mitogen-activated protein kinase,MAPK)因子可激活mTOR信号通路,抑制ULK1复合体的形成,从而抑制自噬的启动[12]。AMP活化蛋白激酶(AMP-activated protein kinase,AMPK)和P53因子是自噬的正调控因子,可激活ULK1复合体和PI3K复合体,诱导自噬小体形成,也可负向调控mTOR,促进自噬[13]。转录因子EB(transcription factor EB,TFEB)也被证明是细胞自噬的主要调节因子之一,它可以上调组织蛋白酶B(Cathepsin B,CTSB)的表达,诱导溶酶体出现,促进溶酶体与自噬小体形成自噬溶酶体[14]。具体见图2。"
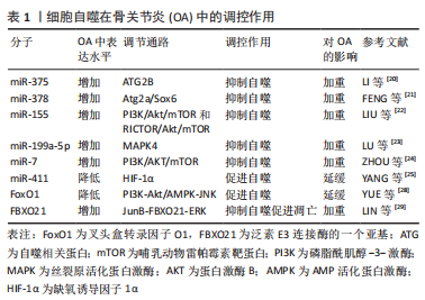
2.1.2 细胞自噬在骨关节炎中的调控作用 在骨关节炎的早期,细胞自噬作为一种适应性反应被激活,以促进软骨细胞存活,而在骨关节炎的晚期,它可能与细胞凋亡一起被激活[15]。细胞自噬功能障碍参与了骨关节的退变,而诱导自噬可以拮抗关节退变。SUN等[16]研究认为,由自噬相关蛋白(autophagy-related gene,ATG)等自噬基因控制的细胞自噬与软骨细胞的生存密切相关,细胞自噬途径对骨关节炎的防治有着十分重要的作用。线粒体自噬是细胞清除受损线粒体的过程,骨关节炎退行性变与软骨细胞线粒体自噬缺陷密切相关。目前在线粒体自噬的研究中,PINK1/Parkin经典通路被研究得最多,该通路在软骨细胞的线粒体自噬激活中起重要作用[17]。SHIN等[18]研究发现线粒体自噬与骨关节炎之间有很强的联系,线粒体自噬相关基因(LC3-Ⅱ、p62、PINK1/Parkin)在骨关节炎患者和碘乙酸单钠(MIA)诱导的骨关节炎啮齿动物模型的软骨中高度表达。大量证据表明,miRNA在软骨内环境稳态和自噬过程中有着不可或缺的作用,其中靶向自噬通路的miRNA(自噬miRNA)在软骨细胞中表达水平的增减可能影响骨关节炎的发展[19]。LI等[20]研究表明,miR-375在骨关节炎软骨组织中表达上调,其通过靶向ATG2B抑制软骨细胞自噬,进而加重骨关节炎。FENG等[21]研究证明,miR-378在骨关节炎软骨中表达增加,其表达水平与其靶基因Atg2a和软骨生成调节因子(Sox6)呈负相关,miR-378的过表达抑制骨关节炎软骨细胞的自噬,增强了白细胞介素1β诱导的软骨细胞炎症和软骨变性。LIU等[22]研究发现,2,2’,4,4’,5,5’-六氯联苯(PCB153)通过上调miR-155激活PI3K/Akt/mTOR和RICTOR/Akt/mTOR信号通路,抑制自噬,促进细胞外基质的降解,从而促进骨关节炎的发展。miR-199a-5p在骨关节炎组织中表达显著增加,MAPK4是miR-199a-5p的靶向基因,下调miR-199a-5p可抑制自噬,促进软骨细胞存活,延缓骨关节炎病情[23]。ZHOU等[24]发现,下调ciRS-7/上调miR-7轴通过激活IL-17A介导的PI3K/AKT/mTOR信号通路加重骨关节炎软骨细胞降解和自噬障碍。miR-411在骨关节炎中的表达较低,通过靶向缺氧诱导因子1α促进软骨细胞自噬,而上调miR-411则降低骨关节炎软骨细胞中ATG蛋白(ULK1、LC3、Beclin-1、P62)的表达,减弱自噬[25]。雷帕霉素(rapamycin)是公认的mTOR抑制剂之一,目前被认为是自噬的激活剂,据报道,雷帕霉素激活自噬可防止人类软骨细胞出现骨关节炎样病变,是骨关节炎治疗的有效制剂[26-27]。 FoxO-噬途径在骨关节炎的发病机制中起着重要的调节作用,叉头盒转录因子(forkhead box,Fox)O1在骨关节炎患者的软骨中表达降低,通过翻译后修饰(post‐translational modifications,PTM)如AMPK磷酸化、Sirt1/2乙酰化等调节FoxO1的活性,与ATG蛋白直接作用控制自噬小体的形成及其和溶酶小体的融合,促进自噬的启动[28]。LIN等[29]证明FBXO21(泛素E3连接酶的一个亚基)在骨关节炎中表达上调,通过JunB-FBXO21-ERK轴抑制细胞的自噬作用,FBXO21的过表达增加了软骨细胞的分解代谢和凋亡,促进了骨关节炎相关的软骨退变。软骨细胞自噬通量的阻断被认为是骨关节炎发展过程中细胞过度凋亡和衰老的主要原因,但自噬通量中断的分子机制目前尚不清楚。ZHENG等[30]研究发现TFEB的过表达能恢复被叔丁基过氧化氢(tert-Butyl hydroperoxide,TBHP)诱导损伤的自噬通量和溶酶体功能,并保护软骨细胞免受TBHP诱导的细胞凋亡和衰老。GU等[31]研究表明20-脱氧精醇(20-DOI)通过激活软骨细胞中的TFEB促进软骨细胞的自噬,抑制软骨细胞的凋亡和衰老,可能缓解骨关节炎。CHEN等[32]的研究结果表明,褪黑素可能通过AMPK/FoxO3通路维持线粒体氧化还原稳态,增强自噬通量,减轻体内软骨退变和软骨骨化。 此类关于骨关节炎中自噬的研究还有不少,这些研究扩充了人们对骨关节炎中自噬通路及分子调控机制的认识,细胞自噬在骨关节炎的病理过程中扮演着极其重要的角色,通过影响软骨细胞的自噬可以调控骨关节炎,激活并适量增强自噬可保护软骨细胞免受损害,从而使骨关节炎骨关节退变得到治疗或缓解。表1细胞自噬在骨关节炎中的调控作用研究汇总。"

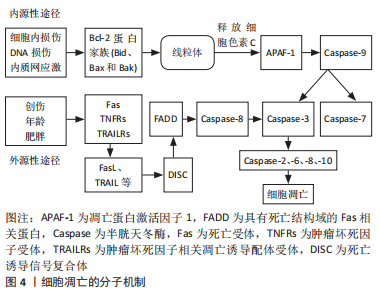
2.2.1 细胞凋亡的分子机制 细胞凋亡包含内源性途径和外源性途径两个核心途径,前者是由细胞本身响应损伤而启动的,也称为线粒体凋亡途径,后者是通过免疫系统细胞刺激的死亡受体启动的,也称死亡受体凋亡途径。在外源性途径中,机体受到创伤、年龄、肥胖等危险因素的刺激,死亡受体(Fas)、肿瘤坏死因子受体(tumor necrosis factor receptors,TNFRs)、肿瘤坏死因子相关凋亡诱导配体受体(tumor necrosis factor receptor superfamily,TRAILRs)被激活并与相应的配体(包括FasL、TRAIL)结合,形成死亡诱导信号复合体(death-inducing signaling complex,DISC)[34]。DISC激活具有死亡结构域的Fas相关蛋白(Fas-associated protein with death domain,FADD),FADD介导半胱天冬酶8(Caspase-8)的激活,Caspase-8进一步激活Caspase-3来传递凋亡信号。在内源性途径中,Bcl-2蛋白家族成员(包括Bid、Bax和Bak)被细胞内损伤、DNA损伤和内质网应激所激活[34]。Bax-BaK寡聚体促进线粒体释放细胞色素C,细胞色素C进而激活凋亡蛋白激活因子1(APAF-1)和Caspase-9,Caspase-9进一步激活Caspase-3和Caspase-7,进而激活Caspase-2、-6、-8、-10,两种凋亡途径一起形成一个正反馈环路,放大细胞凋亡信号,形成级联反应诱导细胞凋亡。Caspase-3在细胞凋亡过程中处于Caspase级联反应的核心,是最常见的也是不可或缺的凋亡执行者,其可被外源性和内源性两种途径激活,通过一系列信号通路诱导细胞凋亡[35]。具体见图4。"
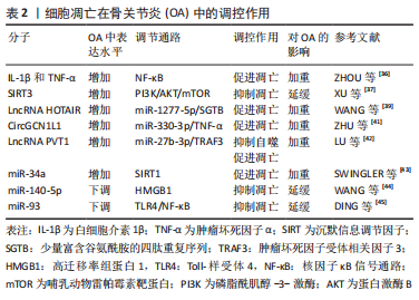
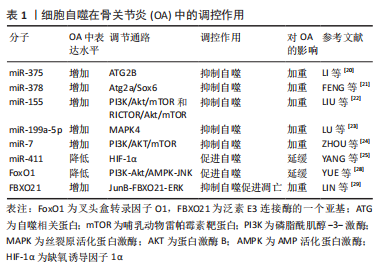
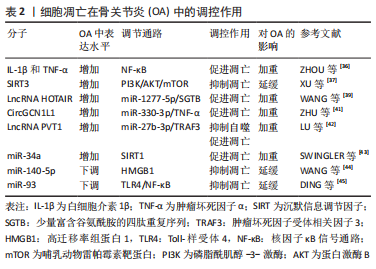
2.2.2 细胞凋亡在骨关节炎中的调控作用 越来越多的研究表明,骨关节退行性变与软骨细胞的凋亡密切相关。近年来对骨关节炎软骨细胞凋亡的研究主要集中在炎症反应、信号通路和靶点调控等方面。在骨关节炎的急性发作期,促炎性细胞因子白细胞介素1β和肿瘤坏死因子α高表达,二者通过激活核因子κB(nuclear factor-kappa B,NF-κB)信号通路上调关节软骨细胞中酸敏离子通道1a(ASIC1a)的表达,降低酸刺激下软骨细胞的存活率,促进Caspase-3、-9的表达和软骨细胞的凋亡,加重骨关节炎[36]。XU等[37]研究发现,沉默信息调节因子3(sirtuin3,SIRT3)的过表达抑制了白细胞介素1β诱导的软骨细胞炎症、凋亡及关节退变,SIRT3通过抑制PI3K/AKT/mTOR信号通路的激活,缓解骨关节炎的发生发展。白细胞介素18诱导软骨细胞的炎症和凋亡,促进骨关节炎的发生和发展,雷帕霉素能通过PI3K/AKT/mTOR通路保护软骨细胞免受白细胞介素18诱导的细胞凋亡并改善骨关节炎[38]。相比较以往局部、单一的研究方法,长链非编码RNA(long noncoding RNA,LncRNA)结合微小RNA(miRNA)的研究已成为目前研究这些分子在骨关节炎中作用的主要途径。WANG等[39]研究表明LncRNA HOTAIR在骨关节炎软骨细胞中上调,其通过海绵miR-1277-5p增强了富含谷氨酰胺的小四肽重复序列(SGTB)表达,加重骨关节炎中的软骨细胞凋亡和炎症。LU等[40]发现氢(H2)通过c-jun氨基末端激酶(c-Jun N-terminal kinases,JNK)信号通路抑制软骨细胞的凋亡和细胞外基质的降解,进而缓解骨关节炎。ZHU等[41]阐明了CircGCN1L1在颞下颌关节骨关节炎滑膜组织和细胞中丰富表达,通过海绵miR-330-3p上调滑膜细胞中肿瘤坏死因子α的表达来促进软骨细胞凋亡,加重颞下颌关节骨关节炎的进展。据LU等[42]报道,LncRNA 浆细胞瘤变异易位体1(plasmacytoma variant translocation 1,PVT1)在骨关节炎细胞中表达增强,敲低PVT1增加了细胞活力和自噬,但PVT1通过海绵miR-27b-3p正调控肿瘤坏死因子受体相关因子3(tumor necrosis factor receptor-associated factor 3,TRAF3)的表达,抑制了白细胞介素1β处理的软骨细胞的凋亡和炎症反应。SWINGLER等[43]研究表明miR-34a在人骨关节炎软骨中的表达水平增加,沉默信息调节因子1(SIRT1)是它的直接靶点,其过表达增加了促凋亡蛋白Bax的表达,降低抗凋亡蛋白Bcl-2的表达,促进软骨细胞凋亡,而miR-34a的抑制剂可以减轻骨关节炎中的软骨破坏。WANG等[44]研究发现,骨关节炎中miR-140-5p表达下调,高迁移率组蛋白1(high mobility group box 1,HMGB1)表达上调,miR-140-5p可通过靶向HMGB1抑制PI3K/AKT信号通路,抑制白细胞介素1β诱导的软骨细胞炎症、基质金属蛋白酶表达和凋亡,进而抑制骨关节炎进展。DING等[45]研究表明miR-93在骨关节炎关节软骨组织中显著降低,miR-93直接靶向NF-κB信号通路的重要调节因子Toll样受体4(Toll-like receptor 4,TLR4),又进一步证实过表达miR-93可抑制TLR4/NF-κB信号通路,进而抑制骨关节炎中的软骨细胞凋亡和炎症。根据上述研究,不难发现,软骨细胞凋亡参与了骨关节炎发生发展过程且在其中起着不可或缺的作用,通过调控软骨细胞凋亡,可以抑制关节退变,进而防治骨关节炎。表2为细胞凋亡在骨关节炎中的调控作用研究汇总。"
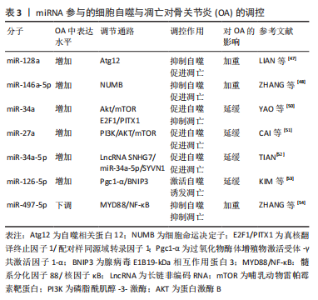
2.3 细胞自噬与凋亡在骨关节炎中的共同调控作用 2.3.1 miRNA参与了细胞自噬与凋亡对骨关节炎的调控 越来越多的证据表明,软骨细胞自噬和凋亡之间的平衡在骨关节炎的软骨退变中起关键作用。miRNA在骨关节炎组织中呈差异性表达,导致相应靶基因表达异常,能调节细胞自噬和凋亡[46]。LIAN等[47]研究发现miR-128a在骨关节炎软骨中表达升高,直接与Atg12的3′-UTR结合,显著降低了Atg12、LC3-Ⅱ表达浓度,阻碍软骨细胞自噬,他们进一步研究证明,高水平的miR-128a增加了细胞凋亡调节因子Bax、Bcl-2和Caspase-3的表达并加速软骨细胞的凋亡,驱动骨关节炎向终末期发展。ZHANG等[48]研究证明,miR-146a-5p在骨关节炎中表达显著上调,miR-146a-5p的下调可通过靶向细胞命运决定子NUMB来抑制骨关节炎软骨细胞凋亡并促进自噬,改善骨关节炎。在骨关节炎组织中环状miR let-7e(circulating miRNA let 7e)的表达显著降低,上调miR let-7e可能增强软骨细胞自噬活性并抑制凋亡[49]。YAO等[50]研究发现,miR-34a在高脂肪饮食的大鼠关节软骨中表达增加,姜黄素下调miR-34a可能通过Akt/mTOR信号通路促进细胞自噬并通过上调真核翻译终止因子1(eukaryotic translation termination factor 1,ETF1)/成对同源异型结构域转录因子1(paired-like homeodomain transcription factor 1,PITX1)抑制细胞凋亡来保护软骨,进而改善骨关节炎发展。CAI等[51]研究表明,miR-27a在骨关节炎组织中表达上调,并通过PI3K/AKT/mTOR信号通路参与骨关节炎的发生发展,抑制miR-27a的表达后,骨关节炎中LC3-Ⅰ、LC3-Ⅱ水平降低,自噬减弱;此外,Bcl-2水平升高和Bax水平降低,凋亡也受到抑制,从而加重骨关节炎。TIAN等[52]首次研究证明,miR-34a-5p在骨关节炎组织和白细胞介素1β处理的细胞中表达上调,LncRNA SNHG7通过miR-34a-5p/SYVN1信号通路影响细胞增殖、凋亡和自噬,过表达SNHG7促进细胞增殖并抑制细胞自噬和凋亡,而上调miR-34a-5p和敲低SYVN 1挽救了SNHG7过表达对细胞自噬和凋亡的抑制,保护软骨。KIM等[53]研究发现,miR-126-5p与过氧化物酶体增殖物激活受体γ共激活因子1α(peroxisome proliferator-activated receptor-gamma co-activator 1-alpha,Pgc1-α)的3’UTR区域直接结合,miR-126-5p通过靶向Pgc1-α调控软骨细胞的自噬和凋亡,过表达的miR-126-5p抑制软骨细胞中Pgc1-α的表达,最终激活腺病毒E1B19-kDa相互作用蛋白3(BNIP3)依赖性线粒体自噬并诱发软骨降解和软骨细胞凋亡。最近有研究证明,miR-497-5p在骨关节炎软骨组织和白细胞介素1β刺激的软骨细胞中表达下调,其通过髓样分化因子88(myeloid differentiation factor 88,MYD88)/NF-κB信号通路抑制骨关节炎软骨细胞凋亡和自噬,从而促进骨关节炎的进展[54]。综上可知,miRNAs可与相应的分子靶点结合,通过其不同的表达水平,实现对软骨细胞中自噬与凋亡的调控,进而影响骨关节炎的发展。表3为miRNA参与的细胞自噬与凋亡对骨关节炎的调控作用研究汇总。"


2.3.2 Beclin-1参与了细胞自噬与凋亡对骨关节炎的调控 Beclin-1是一种含有BH3结构域的蛋白,能与Vps15、Vps34结合形成PI3K复合体,是启动自噬的关键调节因子[55]。Beclin-1通过BH3结构域与Bcl-2蛋白家族成员(Bcl-2、Bcl-xl)结合形成Bcl-2/Bcl-xl/Beclin-1复合体,它们之间的结合解离作用调控Beclin-1诱导的自噬功能[56]。死亡相关蛋白激酶1(DRAK1)可以磷酸化Beclin-1的BH3结构域,降低其与Bcl-2、Bcl-xl的亲和力,使其从Bcl-2/Bcl-xl/Beclin-1复合体中解离,进而诱导自噬[57]。此外,Caspase对Beclin-1的切割,消除了Beclin-1与Bcl-2、Bcl-xl的结合解离作用,且Beclin-1的蛋白剪切片段能诱导细胞色素C释放,抑制自噬、促进凋亡[58]。HE等[59]的研究结果表明,在肿瘤坏死因子α诱导的软骨细胞中,Bcl-2蛋白与Beclin-1蛋白相互作用,Mettl3通过m6A/Ythdf1/Bcl-2信号轴抑制炎症状态下软骨细胞的自噬和凋亡,缓解骨关节炎的软骨破坏和软骨下骨退变。综上所述,Beclin-1的BH3结构域可以调控Bcl-2、Bcl-xl、Beclin-1复合体的结合与解离,从而影响细胞自噬和凋亡之间的平衡关系。 2.3.3 氧化应激参与了细胞自噬与凋亡对骨关节炎的调控 活性氧是维持软骨内环境稳定的重要介质,对软骨细胞的自噬与凋亡均有促进作用。线粒体功能障碍、氧化应激和软骨细胞死亡是骨关节炎发生发展的重要因素,线粒体功能障碍通过增加细胞的活性氧水平来引起氧化应激,诱导软骨细胞衰老,并驱动骨关节炎[60]。细胞的衰老、氧化应激等会影响细胞外基质蛋白质的合成和分泌,导致蛋白质在内质网中蓄积,这一过程即为内质网应激。内质网应激引起的凋亡被称为内质网应激凋亡通路,即内质网应激会干扰软骨细胞的稳态并启动未折叠蛋白反应(unfolded protein reaction,UPR),持续不可逆的内质网应激会触发UPR启动的软骨细胞凋亡[61]。活性氧诱导的线粒体损伤和内质网应激在骨关节炎中发挥关键作用,并最终可能触发软骨细胞凋亡的级联反应[62]。细胞内活性氧水平过高时,氧自由基对线粒体造成损伤,氧化应激激活线粒体自噬以修复细胞,维持细胞稳态[63]。ANSARI等[64]研究表明,PINK1/Parkin过表达可以促进自噬,清除功能失调的线粒体,减少线粒体中的活性氧和氧化应激,抑制软骨细胞的凋亡,从而延缓骨关节炎的发生发展。活性氧诱导氧化应激发生的同时,也激活了自噬,受损的线粒体被清除,细胞内活性氧减少,软骨细胞内环境稳态得以维持,但过度的氧化应激状态反而会抑制自噬,刺激软骨细胞过量生成活性氧,体内氧化与抗氧化系统失衡,导致细胞凋亡和衰老,损害关节软骨,促进骨关节炎的发生和发展[65]。"

| [1] HARRELL CR, MARKOVIC BS, FELLABAUM C, et al. Mesenchymal stem cell-based therapy of osteoarthritis: Current knowledge and future perspectives. Biomed Pharmacother. 2019;109:2318-2326. [2] KLOPPENBURG M, BERENBAUM F. Osteoarthritis year in review 2019: epidemiology and therapy. Osteoarthritis Cartilage. 2020;28(3):242-248. [3] VINA ER, KWOH CK. Epidemiology of osteoarthritis: literature update. Curr Opin Rheumatol. 2018;30(2):160-167. [4] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019;393(10182): 1745-1759. [5] CHENG NT, GUO A, MENG H. The protective role of autophagy in experimental osteoarthritis, and the therapeutic effects of Torin 1 on osteoarthritis by activating autophagy. BMC Musculoskelet Disord. 2016;17:150. [6] WONG SQ, KUMAR AV, MILLS J, et al. Autophagy in aging and longevity. Hum Genet. 2020;139(3):277-290. [7] KLIONSKY DJ, PETRONI G, AMARAVADI RK, et al. Autophagy in major human diseases. EMBO J. 2021;40(19):e108863. [8] MESSINA OD, VIDAL WM, VIDAL NL. Nutrition, osteoarthritis and cartilage metabolism. Aging Clin Exp Res. 2019;31(6):807-813. [9] NASIRI N, HOSSEINI S, ALINI M, et al. Targeted cell delivery for articular cartilage regeneration and osteoarthritis treatment. Drug Discovery Today. 2019;24(11): 2212-2224. [10] D’ARCY MS. Cell death: a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol Int. 2019;43(6):582-592. [11] JEON H, IM GI. Autophagy in osteoarthritis. Connect Tissue Res. 2017;58(6): 497-508. [12] SUN K, LUO J, GUO J, et al. The PI3K/AKT/mTOR signaling pathway in osteoarthritis: a narrative review. Osteoarthritis Cartilage. 2020;28(4):400-409. [13] CHOI AM, RYTER SW, LEVINE B. Autophagy in human health and disease. N Engl J Med. 2013;368(19):1845-1846. [14] DIKIC I, ELAZAR Z. Mechanism and medical implications of mammalian autophagy. Nat Rev Mol Cell Biol. 2018;19(6):349-364. [15] CASTROGIOVANNI P, RAVALLI S, MUSUMECI G. Apoptosis and Autophagy in the Pathogenesis of Osteoarthritis. J Invest Surg. 2020;33(9):874-875. [16] SUN K, JING X, GUO J, et al. Mitophagy in degenerative joint diseases. Autophagy. 2021;17(9):2082-2092. [17] WANG S, DENG Z, MA Y, et al. The Role of Autophagy and Mitophagy in Bone Metabolic Disorders. Int J Biol Sci. 2020;16(14):2675-2691. [18] SHIN HJ, PARK H, SHIN N, et al. Pink1-Mediated Chondrocytic Mitophagy Contributes to Cartilage Degeneration in Osteoarthritis. J Clin Med. 2019;8(11):1849. [19] YU Y, ZHAO J. Modulated Autophagy by MicroRNAs in Osteoarthritis Chondrocytes. Biomed Res Int. 2019;2019:1484152. [20] LI H, LI Z, PI Y, et al. MicroRNA-375 exacerbates knee osteoarthritis through repressing chondrocyte autophagy by targeting ATG2B. Aging (Albany NY). 2020;12(8):7248-7261. [21] FENG L, YANG Z, LI Y, et al. MicroRNA-378 contributes to osteoarthritis by regulating chondrocyte autophagy and bone marrow mesenchymal stem cell chondrogenesis. Mol Ther Nucleic Acids. 2022;28:328-341. [22] LIU Z, HUANG Y, JIN X, et al. PCB153 suppressed autophagy via PI3K/Akt/mTOR and RICTOR/Akt/mTOR signaling by the upregulation of microRNA-155 in rat primary chondrocytes. Toxicol Appl Pharmacol. 2022;449:116135. [23] LU H, YANG Y, OU S, et al. The silencing of miR-199a-5p protects the articular cartilage through MAPK4 in osteoarthritis. Ann Transl Med. 2022;10(10):601. [24] ZHOU X, LI J, ZHOU Y, et al. Down-regulated ciRS-7/up-regulated miR-7 axis aggravated cartilage degradation and autophagy defection by PI3K/AKT/mTOR activation mediated by IL-17A in osteoarthritis. Aging (Albany NY). 2020;12(20): 20163-20183. [25] YANG F, HUANG R, MA H, et al. miRNA-411 Regulates Chondrocyte Autophagy in Osteoarthritis by Targeting Hypoxia-Inducible Factor 1 alpha (HIF-1alpha). Med Sci Monit. 2020;26:e921155. [26] DUARTE JH. Osteoarthritis: Autophagy prevents age-related OA. Nat Rev Rheumatol. 2015;11(12):683. [27] ITO M, YURUBE T, KAKUTANI K, et al. Selective interference of mTORC1/RAPTOR protects against human disc cellular apoptosis, senescence, and extracellular matrix catabolism with Akt and autophagy induction. Osteoarthritis Cartilage. 2017;25(12):2134-2146. [28] YUE J, AOBULIKASIMU A, SUN W, et al. Targeted regulation of FoxO1 in chondrocytes prevents age-related osteoarthritis via autophagy mechanism. J Cell Mol Med. 2022;26(11):3075-3082. [29] LIN Z, MIAO J, ZHANG T, et al. JUNB-FBXO21-ERK axis promotes cartilage degeneration in osteoarthritis by inhibiting autophagy. Aging Cell. 2021;20(2):e13306. [30] ZHENG G, ZHAN Y, LI X, et al. TFEB, a potential therapeutic target for osteoarthritis via autophagy regulation. Cell Death Dis. 2018;9(9):858. [31] GU M, JIN J, REN C, et al. 20-Deoxyingenol alleviates osteoarthritis by activating TFEB in chondrocytes. Pharmacol Res. 2021;165:105361. [32] CHEN Z, ZHAO C, LIU P, et al. Anti-Apoptosis and Autophagy Effects of Melatonin Protect Rat Chondrocytes against Oxidative Stress via Regulation of AMPK/Foxo3 Pathways. Cartilage. 2021;13(2_suppl):1041S-1053S. [33] SIVAGNANAM U, PALANIRAJAN SK, GUMMADI SN. The role of human phospholipid scramblases in apoptosis: An overview. Biochim Biophys Acta Mol Cell Res. 2017;1864(12):2261-2271. [34] YANG J, HU S, BIAN Y, et al. Targeting Cell Death: Pyroptosis, Ferroptosis, Apoptosis and Necroptosis in Osteoarthritis. Front Cell Dev Biol. 2021;9:789948. [35] MEDINA CB, MEHROTRA P, ARANDJELOVIC S, et al. Metabolites released from apoptotic cells act as tissue messengers. Nature. 2020;580(7801):130-135. [36] ZHOU RP, DAI BB, XIE YY, et al. Interleukin-1beta and tumor necrosis factor-alpha augment acidosis-induced rat articular chondrocyte apoptosis via nuclear factor-kappaB-dependent upregulation of ASIC1a channel. Biochim Biophys Acta Mol Basis Dis. 2018;1864(1):162-177. [37] XU K, HE Y, MOQBEL SAA, et al. SIRT3 ameliorates osteoarthritis via regulating chondrocyte autophagy and apoptosis through the PI3K/Akt/mTOR pathway. Int J Biol Macromol. 2021;175:351-360. [38] BAO J, CHEN Z, XU L, et al. Rapamycin protects chondrocytes against IL-18-induced apoptosis and ameliorates rat osteoarthritis. Aging (Albany NY). 2020; 12(6):5152-5167. [39] WANG B, SUN Y, LIU N, et al. LncRNA HOTAIR modulates chondrocyte apoptosis and inflammation in osteoarthritis via regulating miR-1277-5p/SGTB axis. Wound Repair Regen. 2021;29(3):495-504. [40] LU H, WANG W, KANG X, et al. Hydrogen (H2) Alleviates Osteoarthritis by Inhibiting Apoptosis and Inflammation via the JNK Signaling Pathway. J Inflamm Res. 2021;14:1387-1402. [41] ZHU H, HU Y, WANG C, et al. CircGCN1L1 promotes synoviocyte proliferation and chondrocyte apoptosis by targeting miR-330-3p and TNF-alpha in TMJ osteoarthritis. Cell Death Dis. 2020;11(4):284. [42] LU X, YU Y, YIN F, et al. Knockdown of PVT1 inhibits IL-1beta-induced injury in chondrocytes by regulating miR-27b-3p/TRAF3 axis. Int Immunopharmacol. 2020;79:106052. [43] SWINGLER TE, NIU L, SMITH P, et al. The function of microRNAs in cartilage and osteoarthritis. Clin Exp Rheumatol. 2019;37 Suppl 120(5):40-47. [44] WANG Y, SHEN S, LI Z, et al. MIR-140-5p affects chondrocyte proliferation, apoptosis, and inflammation by targeting HMGB1 in osteoarthritis. Inflamm Res. 2020;69(1):63-73. [45] DING Y, WANG L, ZHAO Q, et al. MicroRNA93 inhibits chondrocyte apoptosis and inflammation in osteoarthritis by targeting the TLR4/NFkappaB signaling pathway. Int J Mol Med. 2019;43(2):779-790. [46] YU Q, ZHAO B, HE Q, et al. microRNA-206 is required for osteoarthritis development through its effect on apoptosis and autophagy of articular chondrocytes via modulating the phosphoinositide 3-kinase/protein kinase B-mTOR pathway by targeting insulin-like growth factor-1. J Cell Biochem. 2019; 120(4):5287-5303. [47] LIAN WS, KO JY, WU RW, et al. MicroRNA-128a represses chondrocyte autophagy and exacerbates knee osteoarthritis by disrupting Atg12. Cell Death Dis. 2018; 9(9):919. [48] ZHANG H, ZHENG W, LI D, et al. miR-146a-5p Promotes Chondrocyte Apoptosis and Inhibits Autophagy of Osteoarthritis by Targeting NUMB. Cartilage. 2021; 13(2_suppl):1467S-1477S. [49] FENG L, FENG C, WANG CX, et al. Circulating microRNA let‑7e is decreased in knee osteoarthritis, accompanied by elevated apoptosis and reduced autophagy. Int J Mol Med. 2020;45(5):1464-1476. [50] YAO J, LIU X, SUN Y, et al. Curcumin-Alleviated Osteoarthritic Progression in Rats Fed a High-Fat Diet by Inhibiting Apoptosis and Activating Autophagy via Modulation of MicroRNA-34a. J Inflamm Res. 2021;14:2317-2331. [51] CAI C, MIN S, YAN B, et al. MiR-27a promotes the autophagy and apoptosis of IL-1beta treated-articular chondrocytes in osteoarthritis through PI3K/AKT/mTOR signaling. Aging (Albany NY). 2019;11(16):6371-6384. [52] TIAN F, WANG J, ZHANG Z, et al. LncRNA SNHG7/miR-34a-5p/SYVN1 axis plays a vital role in proliferation, apoptosis and autophagy in osteoarthritis. Biol Res. 2020;53(1):9. [53] KIM D, SONG J, JIN EJ. BNIP3-Dependent Mitophagy via PGC1alpha Promotes Cartilage Degradation. Cells. 2021;10(7):1839. [54] ZHANG Y, LU R, HUANG X, et al. Circular RNA MELK Promotes Chondrocyte Apoptosis and Inhibits Autophagy in Osteoarthritis by Regulating MYD88/NF-kappaB Signaling Axis through MicroRNA-497-5p. Contrast Media Mol Imaging. 2022;2022:7614497. [55] LUO P, GAO F, NIU D, et al. The Role of Autophagy in Chondrocyte Metabolism and Osteoarthritis: A Comprehensive Research Review. Biomed Res Int. 2019; 2019:5171602. [56] FUCHS Y, STELLER H. Live to die another way: modes of programmed cell death and the signals emanating from dying cells. Nat Rev Mol Cell Biol. 2015;16(6):329-344. [57] MANIVANNAN P, REDDY V, MUKHERJEE S, et al. RNase L Induces Expression of A Novel Serine/Threonine Protein Kinase, DRAK1, to Promote Apoptosis. Int J Mol Sci. 2019;20(14):3535. [58] SIDDIQUI MA, MUKHERJEE S, MANIVANNAN P, et al. RNase L Cleavage Products Promote Switch from Autophagy to Apoptosis by Caspase-Mediated Cleavage of Beclin-1. Int J Mol Sci. 2015;16(8):17611-17636. [59] HE Y, WANG W, XU X, et al. Mettl3 inhibits the apoptosis and autophagy of chondrocytes in inflammation through mediating Bcl2 stability via Ythdf1-mediated m6A modification. Bone. 2022;154:116182. [60] CORYELL PR, DIEKMAN BO, LOESER RF. Mechanisms and therapeutic implications of cellular senescence in osteoarthritis. Nat Rev Rheumatol. 2021;17(1):47-57. [61] RELLMANN Y, EIDHOF E, DREIER R. Review: ER stress-induced cell death in osteoarthritic cartilage. Cell Signal. 2021;78:109880. [62] DING DF, XUE Y, WU XC, et al. Recent Advances in Reactive Oxygen Species (ROS)-Responsive Polyfunctional Nanosystems 3.0 for the Treatment of Osteoarthritis. J Inflamm Res. 2022;15:5009-5026. [63] LU J, PENG Y, ZOU J, et al. Hypoxia Inducible Factor-1alpha Is a Regulator of Autophagy in Osteoarthritic Chondrocytes. Cartilage. 2021;13(2_suppl):1030S-1040S. [64] ANSARI MY, KHAN NM, AHMAD I, et al. Parkin clearance of dysfunctional mitochondria regulates ROS levels and increases survival of human chondrocytes. Osteoarthritis Cartilage. 2018;26(8):1087-1097. [65] ROCA-AGUJETAS V, DE DIOS C, LESTON L, et al. Recent Insights into the Mitochondrial Role in Autophagy and Its Regulation by Oxidative Stress. Oxid Med Cell Longev. 2019;2019:3809308. [66] CELIA AI, COLAFRANCESCO S, BARBATI C, et al. Autophagy in Rheumatic Diseases: Role in the Pathogenesis and Therapeutic Approaches. Cells. 2022;11(8):1359. [67] VALENTI MT, DALLE CL, ZIPETO D, et al. Control of the Autophagy Pathway in Osteoarthritis: Key Regulators, Therapeutic Targets and Therapeutic Strategies. Int J Mol Sci. 2021;22(5):2700. [68] 孙明帅,范重山,李凯杰,等.骨关节炎中软骨细胞自噬的作用及其靶向治疗[J].中国组织工程研究,2021,25(35):5688-5693. |
| [1] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [2] | Li Yongjie, Fu Shenyu, Xia Yuan, Zhang Dakuan, Liu Hongju. Correlation of knee extensor muscle strength and spatiotemporal gait parameters with peak knee flexion/adduction moment in female patients with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1354-1358. |
| [3] | Qi Haodong, Lu Chao, Xu Hanbo, Wang Mengfei, Hao Yangquan. Effect of diabetes mellitus on perioperative blood loss and pain after primary total knee arthroplasty [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1383-1387. |
| [4] | Du Changling, Shi Hui, Zhang Shoutao, Meng Tao, Liu Dong, Li Jian, Cao Heng, Xu Chuang. Efficacy and safety of different applications of tranexamic acid in high tibial osteotomy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1409-1413. |
| [5] | Wei Juan, Li Ting, Huan Mengting, Xie Ying, Xie Zhouyu, Wei Qingbo, Wu Yunchuan. Mechanism by which static exercise improves insulin resistance in skeletal muscle of type 2 diabetes [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1271-1276. |
| [6] | Sheng Siqi, Xie Lin, Zhao Xiangyu, Jiang Yideng, Wu Kai, Xiong Jiantuan, Yang Anning, Hao Yinju, Jiao Yun. Involvement of miR-144-3p in Cbs+/- mouse hepatocyte autophagy induced by high-methionine diet [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1289-1294. |
| [7] | Huang Xiarong, Hu Lizhi, Sun Guanghua, Peng Xinke, Liao Ying, Liao Yuan, Liu Jing, Yin Linwei, Zhong Peirui, Peng Ting, Zhou Jun, Qu Mengjian. Effect of electroacupuncture on the expression of P53 and P21 in articular cartilage and subchondral bone of aged rats with knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1174-1179. |
| [8] | Yue Yun, Wang Peipei, Yuan Zhaohe, He Shengcun, Jia Xusheng, Liu Qian, Li Zhantao, Fu Huiling, Song Fei, Jia Menghui. Effects of croton cream on JNK/p38 MAPK signaling pathway and neuronal apoptosis in cerebral ischemia-reperfusion injury rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1186-1192. |
| [9] | Zhao Garida, Ren Yizhong, Han Changxu, Kong Lingyue, Jia Yanbo. Mechanism of Mongolian Medicine Erden-uril on osteoarthritis in rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1193-1199. |
| [10] | Wang Ji, Zhang Min, Li Wenbo, Yang Zhongya, Zhang Long. Effect of aerobic exercise on glycolipid metabolism, skeletal muscle inflammation and autophagy in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1200-1205. |
| [11] | Liu Xin, Hu Man, Zhao Wenjie, Zhang Yu, Meng Bo, Yang Sheng, Peng Qing, Zhang Liang, Wang Jingcheng. Cadmium promotes senescence of annulus fibrosus cells via activation of PI3K/Akt signaling pathway [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1217-1222. |
| [12] | Zhou Bangyu, Li Jie, Ruan Yushang, Geng Funeng, Li Shaobo. Effects of Periplaneta americana powder on motor function and autophagic protein Beclin-1 in rats undergoing spinal cord hemisection [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1223-1228. |
| [13] | Li Rui, Zhang Guihong, Wang Tao, Fan Ping. Effect of ginseng polysaccharide on the expression of prostaglandin E2/6-keto-prostaglandin 1alpha in traumatic osteoarthritis model rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1235-1240. |
| [14] | Pan Xiaolong, Fan Feiyan, Ying Chunmiao, Liu Feixiang, Zhang Yunke. Effect and mechanism of traditional Chinese medicine on inhibiting the aging of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1091-1098. |
| [15] | Huang Yuxin, Liang Wenzi, Chen Xiuwen, Ni Na, Zhao Yinglin, Lin Changmin. Role of autophagy in hair regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1112-1117. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||