Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (28): 4469-4476.doi: 10.12307/2024.340
Previous Articles Next Articles
Metformin pretreatment induces cardiac autophagy to reduce myocardial injury in septic mice
Tian Yong1, 2, Zhou Ying1, Gu Yongxiang1, Yang Guohui3
- 1Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2Intensive Care Unit, Tongren City People’s Hospital, Tongren 554300, Guizhou Province, China; 3Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2023-04-18Accepted:2023-06-01Online:2024-10-08Published:2023-11-27 -
Contact:Yang Guohui, Professor, Chief physician, Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Tian Yong, Master candidate, Guizhou Medical University, Guiyang 550004, Guizhou Province, China; Intensive Care Unit, Tongren City People’s Hospital, Tongren 554300, Guizhou Province, China -
Supported by:Culture Program for the Affiliated Hospital of Guizhou Medical University, The National Natural Science Foundation of China, No. gyfynsfc-2021-54 (to YGH); Guizhou Provincial Health Commission Science and Technology Fund Project, No. gzwjkj2020-1-026 (to YGH)
CLC Number:
Cite this article
Tian Yong, Zhou Ying, Gu Yongxiang, Yang Guohui. Metformin pretreatment induces cardiac autophagy to reduce myocardial injury in septic mice[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(28): 4469-4476.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
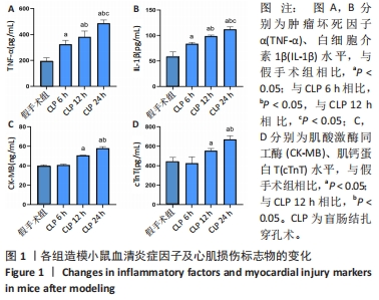
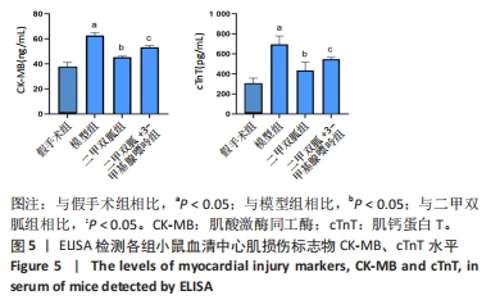
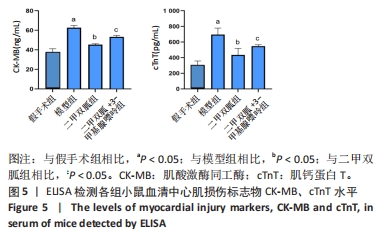
2.1 造模小鼠的一般情况分析 假手术组10 只小鼠及CLP 6 h组10 只小鼠全部存活,假手术组小鼠术后24 h精神、活动及进食正常,无腹泻、畏寒、排泄物异常增多等情况;CLP 6 h组小鼠精神稍萎靡,活动基本正常,无腹泻;CLP 12 h组在术后12 h内死亡1 只小鼠,余存活9 只小鼠出现畏寒、腹泻,进食明显减少,活动明显减少,精神萎靡;CLP 24 h组死亡4 只小鼠,存活6 只小鼠出现嗜睡、拒食、腹泻、竖毛、呼吸急促、眼部有较多分泌物、运动迟缓、不能直立。 2.2 造模小鼠炎症因子及心肌损伤标志物的变化 ELISA检测各组小鼠血清TNF-α、IL-1β、CK-MB及cTnT水平,如图1所示,与假手术组相比,CLP各组炎症因子TNF-α、IL-1β水平均升高(P < 0.05),以CLP 24 h组最为显著;与假手术组相比,CLP 12 h组和CLP 24 h组CK-MB、cTnT水平均显著升高(P < 0.05),以CLP 24 h组更显著,而CLP 6 h组与假手术组相比无统计学差异(P > 0.05),表明在CLP 12 h脓毒症小鼠心肌损伤模型构建成功,且CLP 24 h组较CLP 12 h组心肌损伤更重。"
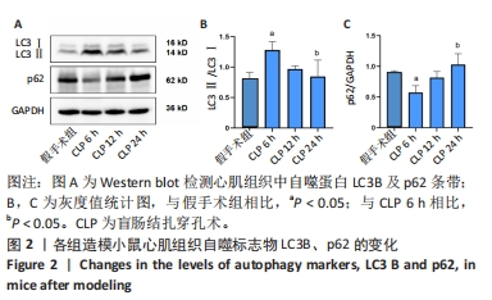
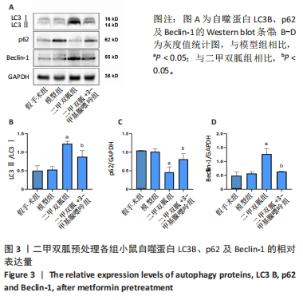
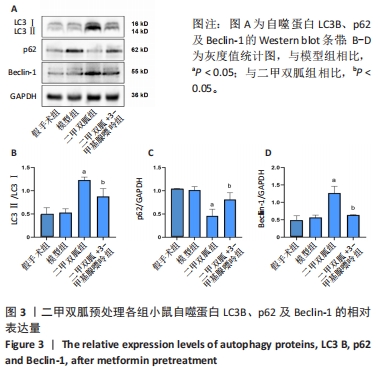
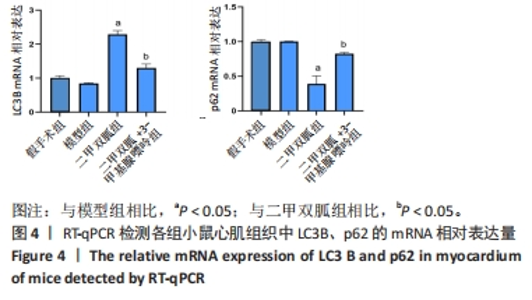
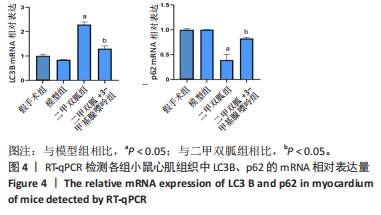
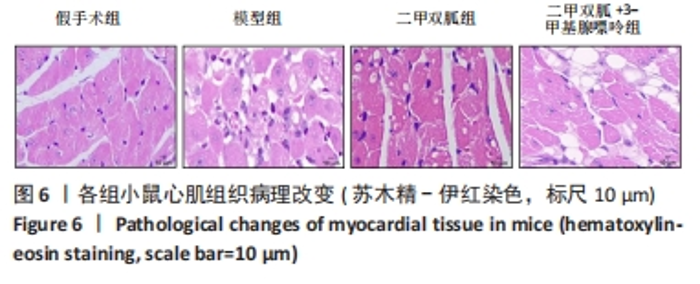
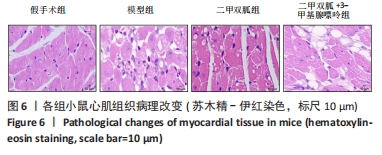
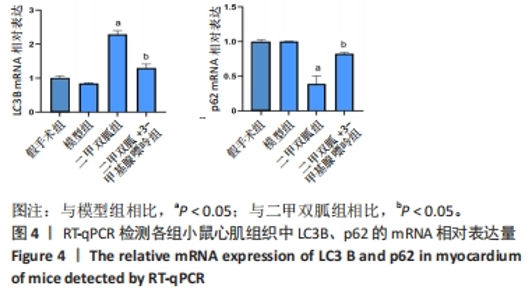
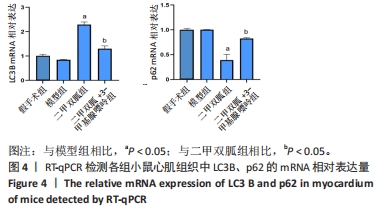
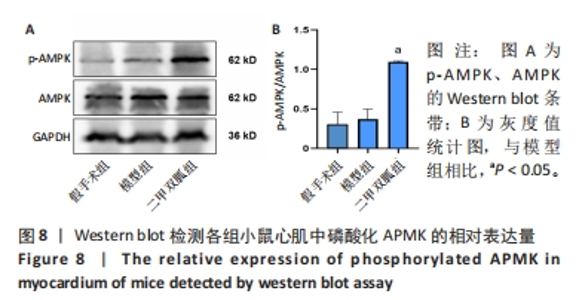
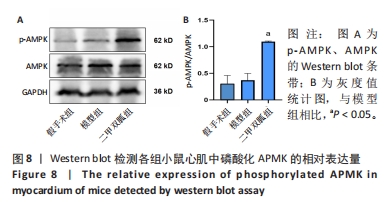
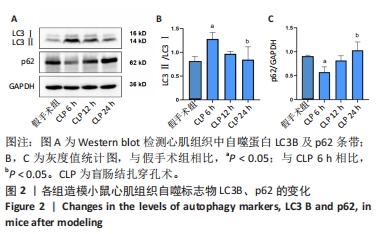
2.3 造模小鼠心肌组织中自噬水平 Western blot检测各组造模小鼠心肌组织自噬标志物(LC3B、p62)的表达水平,如图2所示,与假手术组相比,CLP 6 h组LC3Ⅱ/LC3Ⅰ显著升高(P < 0.05),之后逐渐降低,在CLP 24 h降至基础水平;与假手术组相比,CLP 6 h组p62表达水平显著降低(P < 0.05),之后逐渐恢复,在CLP 24 h已恢复至基础水平,CLP 24 h组与假手术组相比无统计学差异(P > 0.05),这提示在脓毒症早期,自噬处于高表达水平,但在脓毒症心肌损伤中自噬处于抑制状态,在CLP 24 h脓毒症小鼠中自噬处于基础水平,且心肌损伤较CLP 12 h严重,故此研究选择脓毒症心肌损伤中自噬处于显著抑制的时间点(24 h)进行后续研究。"
| [1] ZHANG YY, NING BT. Signaling pathways and intervention therapies in sepsis. Signal Transduct Target Ther. 2021;6(1):407. [2] ZHOU B, ZHANG J, CHEN Y, et al. Puerarin protects against sepsis-induced myocardial injury through AMPK-mediated ferroptosis signaling. Aging (Albany NY). 2022;14(8):3617-3632. [3] LI Y, ZHANG L, ZHANG P, et al. Dehydrocorydaline Protects Against Sepsis-Induced Myocardial Injury Through Modulating the TRAF6/NF-κB Pathway. Front Pharmacol. 2021;12:709604. [4] BI CF, LIU J, YANG LS, et al. Research Progress on the Mechanism of Sepsis Induced Myocardial Injury. J Inflamm Res. 2022;15:4275-4290. [5] SUN Y, CAI Y, ZANG QS. Cardiac Autophagy in Sepsis. Cells. 2019; 8(2):141. [6] ISMAIL HASSAN F, DIDARI T, KHAN F, et al. A Review on The Protective Effects of Metformin in Sepsis-Induced Organ Failure. Cell J. 2020;21(4): 363-370. [7] KOLAHDOUZMOHAMMADI M, PAHLAVAN S, SOTOODEHNEJADNEMATALAHI F, et al. Activation of AMPK promotes cardiac differentiation by stimulating the autophagy pathway. J Cell Commun Signal. 2023 Apr 11. doi: 10.1007/s12079-023-00744-z. [8] LI Q, WANG L, JI D, et al. Metformin attenuates cadmium-induced degeneration of spiral ganglion neuron via restoring autophagic flux in primary culture. J Inorg Biochem. 2022;234:111901. [9] LI W, HE P, HUANG Y, et al. Selective autophagy of intracellular organelles: recent research advances. Theranostics. 2021;11(1):222-256. [10] HUANG M, CAI S, SU J. The Pathogenesis of Sepsis and Potential Therapeutic Targets. Int J Mol Sci. 2019;20(21):5376. [11] GROSS AS, GRAEF M. Mechanisms of Autophagy in Metabolic Stress Response. J Mol Biol. 2020;432(1):28-52. [12] LIU C, LIU Y, CHEN H, et al. Myocardial injury: where inflammation and autophagy meet. Burns Trauma. 2023;11:tkac062. [13] 蒋大军,蒋万威,周颖,等.NLRC4炎症小体介导自噬反应在脓毒症心肌功能障碍小鼠中的作用[J].中国组织工程研究,2023,27(23): 3667-3673. [14] JINPIAO Z, ZONGZE Z, QIUYUE Y, et al. Metformin attenuates sevoflurane-induced neurocognitive impairment through AMPK-ULK1-dependent autophagy in aged mice. Brain Res Bull. 2020;157:18-25. [15] LI T, CHEN Y, LI Y, et al. FAM134B-mediated endoplasmic reticulum autophagy protects against sepsis myocardial injury in mice. Aging (Albany NY). 2021;13(10):13535-13547. [16] EVANS L, RHODES A, ALHAZZANI W, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021;47(11):1181-1247. [17] PÓVOA P, COELHO L, DAL-PIZZOL F, et al. How to use biomarkers of infection or sepsis at the bedside: guide to clinicians. Intensive Care Med. 2023;49(2):142-153. [18] LIU C, ZOU Q, TANG H, et al. Melanin nanoparticles alleviate sepsis-induced myocardial injury by suppressing ferroptosis and inflammation. Bioact Mater. 2022;24:313-321. [19] 杭成文,崔鸣.脓毒症致心肌损伤模型的研究进展[J].中国心血管杂志,2020,25(5):485-488. [20] HO J, YU J, WONG SH, et al. Autophagy in sepsis: Degradation into exhaustion? Autophagy. 2016;12(7):1073-1082. [21] ALA M, ALA M. Metformin for Cardiovascular Protection, Inflammatory Bowel Disease, Osteoporosis, Periodontitis, Polycystic Ovarian Syndrome, Neurodegeneration, Cancer, Inflammation and Senescence: What Is Next? ACS Pharmacol Transl Sci. 2021;4(6):1747-1770. [22] CEJUELA M, MARTIN-CASTILLO B, MENENDEZ JA, et al. Metformin and Breast Cancer: Where Are We Now? Int J Mol Sci. 2022;23(5):2705. [23] LIU G, WU K, ZHANG L, et al. Metformin attenuated endotoxin-induced acute myocarditis via activating AMPK. Int Immunopharmacol. 2017;47:166-172. [24] ZHANG M, SUN W, DU J, et al. Protective Effect of Metformin on Sepsis Myocarditis in Zebrafish. Dose Response. 2020;18(3): 1559325820938543. [25] LIANG H, SONG H, ZHANG X, et al. Metformin attenuated sepsis-related liver injury by modulating gut microbiota. Emerg Microbes Infect. 2022;11(1):815-828. [26] ZHAO H, LYU Y, ZHAI R, et al. Metformin Mitigates Sepsis-Related Neuroinflammation via Modulating Gut Microbiota and Metabolites. Front Immunol. 2022;13:797312. [27] GHAVIMI H, SHEIDAEI S, VAEZ H, et al. Metformin-attenuated sepsis-induced oxidative damages: a novel role for metformin. Iran J Basic Med Sci. 2018;21(5):469-475. [28] TANG G, YANG H, CHEN J, et al. Metformin ameliorates sepsis-induced brain injury by inhibiting apoptosis, oxidative stress and neuroinflammation via the PI3K/Akt signaling pathway. Oncotarget. 2017;8(58):97977-97989. [29] 高婷,陈忠.二甲双胍对脂多糖诱导的大鼠心肌细胞H9C2损伤的保护机制[J].南京医科大学学报(自然科学版),2019,39(9):1298-1303. [30] LI M, GOU Y, YU H, et al. Mechanism of Metformin on LPS-Induced Bacterial Myocarditis. Dose Response. 2019;17(2):1559325819847409. [31] WU B, SONG H, FAN M, et al. Luteolin attenuates sepsis‑induced myocardial injury by enhancing autophagy in mice. Int J Mol Med. 2020;45(5):1477-1487. [32] TANG R, JIA L, LI Y, et al. Narciclasine attenuates sepsis-induced myocardial injury by modulating autophagy. Aging (Albany NY). 2021; 13(11):15151-15163. [33] WANG X, XIE D, DAI H, et al. Clemastine protects against sepsis-induced myocardial injury in vivo and in vitro. Bioengineered. 2022;13(3):7134-7146. [34] WU S, ZHANG H, CHEN N, et al. Metformin protects cardiomyocytes against oxygen-glucose deprivation injury by promoting autophagic flux through AMPK pathway. J Drug Target. 2021;29(5):551-561. [35] WEN X, XIE B, YUAN S, et al. The “Self-Sacrifice” of ImmuneCells in Sepsis. Front Immunol. 2022;13:833479. [36] QIU P, LIU Y, ZHANG J. Review: the Role and Mechanisms of Macrophage Autophagy in Sepsis. Inflammation. 2019;42(1):6-19. [37] LU G, WU Z, SHANG J, et al. The effects of metformin on autophagy. Biomed Pharmacother. 2021;137:111286. [38] HUANG KY, QUE JQ, HU ZS, et al. Metformin suppresses inflammation and apoptosis of myocardiocytes by inhibiting autophagy in a model of ischemia-reperfusion injury. Int J Biol Sci. 2020;16(14):2559-2579. [39] WANG Y, YANG Z, ZHENG G, et al. Metformin promotes autophagy in ischemia/reperfusion myocardium via cytoplasmic AMPKα1 and nuclear AMPKα2 pathways. Life Sci. 2019;225:64-71. [40] WANG Y, AN H, LIU T, et al. Metformin Improves Mitochondrial Respiratory Activity through Activation of AMPK. Cell Rep. 2019;29(6): 1511-1523.e5. [41] BU Y, PENG M, TANG X, et al. Protective effects of metformin in various cardiovascular diseases: Clinical evidence and AMPK-dependent mechanisms. J Cell Mol Med. 2022;26(19):4886-4903. [42] TURCO E, FRACCHIOLLA D, MARTENS S. Recruitment and Activation of the ULK1/Atg1 Kinase Complex in Selective Autophagy. J Mol Biol. 2020;432(1):123-134. [43] LI SX, LI C, PANG XR, et al. Metformin Attenuates Silica-Induced Pulmonary Fibrosis by Activating Autophagy via the AMPK-mTOR Signaling Pathway. Front Pharmacol. 2021;12:719589. [44] SONG H, ZHANG X, ZHAI R, et al. Metformin attenuated sepsis-associated liver injury and inflammatory response in aged mice. Bioengineered. 2022;13(2):4598-4609. [45] LI Y, CHEN Y. AMPK and Autophagy. Adv Exp Med Biol. 2019;1206:85-108. [46] LAMOIA TE, SHULMAN GI. Cellular and Molecular Mechanisms of Metformin Action. Endocr Rev. 2021;42(1):77-96. [47] TZANAVARI T, VARELA A, THEOCHARIS S, et al. Metformin protects against infection-induced myocardial dysfunction. Metabolism. 2016; 65(10):1447-1458. [48] DI MAURO S, FILIPPELLO A, SCAMPORRINO A, et al. Metformin: When Should We Fear Lactic Acidosis? Int J Mol Sci. 2022;23(15):8320. [49] L’HEUREUX M, STERNBERG M, BRATH L, et al. Sepsis-Induced Cardiomyopathy: a Comprehensive Review. Curr Cardiol Rep. 2020; 22(5):35. [50] CHEN P, AN Q, HUANG Y, et al. Prevention of endotoxin-induced cardiomyopathy using sodium tanshinone IIA sulfonate: Involvement of augmented autophagy and NLRP3 inflammasome suppression. Eur J Pharmacol. 2021;909:174438. [51] 迪丽热巴•吐尔逊,杨春波,王毅,等.脓毒性心肌损伤机制及治疗的研究进展[J].中华危重病急救医学,2022,34(10):1112-1115. [52] NIKOUEE A, KIM M, DING X, et al. Beclin-1-Dependent Autophagy Improves Outcomes of Pneumonia-Induced Sepsis. Front Cell Infect Microbiol. 2021;11:706637. |
| [1] | Wang Ji, Zhang Min, Li Wenbo, Yang Zhongya, Zhang Long. Effect of aerobic exercise on glycolipid metabolism, skeletal muscle inflammation and autophagy in type 2 diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1200-1205. |
| [2] | Zhou Bangyu, Li Jie, Ruan Yushang, Geng Funeng, Li Shaobo. Effects of Periplaneta americana powder on motor function and autophagic protein Beclin-1 in rats undergoing spinal cord hemisection [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1223-1228. |
| [3] | Sheng Siqi, Xie Lin, Zhao Xiangyu, Jiang Yideng, Wu Kai, Xiong Jiantuan, Yang Anning, Hao Yinju, Jiao Yun. Involvement of miR-144-3p in Cbs+/- mouse hepatocyte autophagy induced by high-methionine diet [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1289-1294. |
| [4] | Pan Xiaolong, Fan Feiyan, Ying Chunmiao, Liu Feixiang, Zhang Yunke. Effect and mechanism of traditional Chinese medicine on inhibiting the aging of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1091-1098. |
| [5] | Huang Yuxin, Liang Wenzi, Chen Xiuwen, Ni Na, Zhao Yinglin, Lin Changmin. Role of autophagy in hair regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1112-1117. |
| [6] | Liu Qiwei, Zhang Junhui, Yang Yuan, Wang Jinjuan. Role and mechanism of umbilical cord mesenchymal stem cells on polycystic ovary syndrome [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1015-1020. |
| [7] | Xue Jingwen, Wang Fangfang, Zhang Xin, Pang Ruifeng, Wang Xiaoye, Ma Xiaoru. Effect of ganoderma spore on mitochondrial autophagy and apoptosis in testicular tissue of diabetic rats [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 562-568. |
| [8] | Yang Yuqing, Chen Zhiyu. Role and application of early transient presence of M1 macrophages in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 594-601. |
| [9] | Yan Binghan, Li Zhichao, Su Hui, Xue Haipeng, Xu Zhanwang, Tan Guoqing. Mechanisms of traditional Chinese medicine monomers in the treatment of osteoarthritis by targeting autophagy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 627-632. |
| [10] | Zhai Sheng, Aikeremujiang • Aerken, Zhang Zheng, Rixiati • Paerhati, Hao Feihu. CircCDR1as/miR-7-5p/RAF1 axis promotes autophagy levels in steroid-induced necrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(28): 4455-4460. |
| [11] | Chen Ying, Xia Tianqin, Hua Jianlin, Yin Jinzhu, Song Lijuan, Wang Qing, Yu Jiezhong, Huang Jianjun, Ma Cungen. The role and mechanism of TLRs/MyD88/NF-κB signaling pathway in multiple sclerosis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(28): 4578-4585. |
| [12] | Liu Ying, Liu Yalei, Liu Yu. Screening and analysis of differentially expressed long non-coding RNAs in adriamycin-induced myocardial injury antagonized with daisy leaf gentianone [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4121-4128. |
| [13] | Ma Lingju, Wang Lexin, Chi Hongyang, Zhang Jingwen, Peng Hongjian, Gao Chunlan, Jiang Yideng, Huang Hui, Yang Li, Ma Shengchao. Role of METTL3 in homocysteine-induced autophagy in mouse islet beta cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(26): 4221-4225. |
| [14] | Zhu Yongzhao, Fang Chao, Zhao Fang, Zhang Qing, Zhao Dan. Mechanism by which lycium barbarum polysaccharides inhibit keratinocyte apoptosis in burn wounds via autophagy [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(23): 3686-3691. |
| [15] | Liang Xiaolong, Zheng Kai, Geng Dechun, Xu Yaozeng. Novel programmed cell death in periprosthetic osteolysis [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(21): 3393-3399. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||