Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (11): 1737-1742.doi: 10.12307/2024.295
Previous Articles Next Articles
The role of silent information regulator in periodontitis
Sun Jinyi, Wang Qinying, Li Ying, Meng Maohua, Chen Helin, Zeng Xiao, Shu Jiayu, Li Wenjie, Luo Yuncai, Dong Qiang
- School of Stomatology, Guizhou Medical University/Affiliated Stomatological Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2023-02-22Accepted:2023-04-28Online:2024-04-18Published:2023-07-27 -
Contact:Dong Qiang, MD, Chief physician, Doctoral supervisor, School of Stomatology, Guizhou Medical University/Affiliated Stomatological Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Sun Jinyi, Master candidate, School of Stomatology, Guizhou Medical University/Affiliated Stomatological Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
CLC Number:
Cite this article
Sun Jinyi, Wang Qinying, Li Ying, Meng Maohua, Chen Helin, Zeng Xiao, Shu Jiayu, Li Wenjie, Luo Yuncai, Dong Qiang. The role of silent information regulator in periodontitis[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(11): 1737-1742.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

2.1 Sirtuin1在牙周炎中的研究进展 目前,Sirtuins家族中最受关注的、研究报道最多、最深入的是Sirtuin1(SIRT1)。周丰等[17]研究发现,SIRT1通过调节炎症因子及炎症可以起到缓解牙周炎症状的作用,但具体机制研究不多。CARIBé等[18]在一项前瞻性病例对照研究中,通过对比伴有或不伴有冠心病患者牙周病治疗前后血清 SIRT1和甘露糖结合凝集素的浓度,发现牙周治疗后甘露糖结合凝集素和C-反应蛋白浓度降低,SIRT1水平升高。此外,该研究团队又对40例牙周炎患者进行非手术牙周治疗后分析,发现与治疗前相比,所有患者血清甘露糖结合凝集素浓度降低,SIRT1升高,两者呈负相关,同时,C-反应蛋白水平也降低[19]。牙周治疗后能使血清SIRT1水平升高,这有希望为牙周炎的预防和治疗提供一个新思路,即增加血清中SIRT1浓度。再一次佐证了SIRT1在牙周炎中对组织起保护作用的观点。 氧化应激是调节牙周炎相关全身病理生理效应的关键因素。SIRT1基因的激动剂白藜芦醇具有抗氧化、消炎的作用,能减缓氧化应激,减轻炎症反应。TAMAKI等[20]构建大鼠牙周炎模型中,发现白藜芦醇的摄入可缓解牙周炎引起的骨质破坏吸收,这主要是通过激活 SIRT1/AMPK和Nrf2/抗氧化防御通路改善氧化应激,进而阻碍牙周炎症的发展。在CORRêA等[21]构建吸入香烟烟雾的实验性牙周炎的大鼠模型中,发现白藜芦醇通过降低NADPH氧化酶水平和上调SIRT1、超氧化物歧化酶水平,减轻了香烟烟雾吸入组大鼠牙周组织的破坏程度,氧化应激水平降低。CIRANO等[22]在糖尿病合并牙周炎大鼠模型的研究中发现,牙周组织内超氧化物歧化酶和SIRT1的水平降低会引起组织抗氧化能力下降,出现牙周炎症和骨质破坏等炎症表现。但将白藜芦醇和胰岛素联合使用,或者单独使用均可降低大鼠牙周组织炎症水平,减缓牙槽骨吸收速度。上述的几项研究都支持白藜芦醇作为SIRT1激动剂、因抗氧化抑炎的特性可用于治疗牙周炎的观点,对于伴有糖尿病以及长期吸烟史的牙周炎患者也适用。 在LI等[23]的研究中,发现当SIRT1的表达下调时,牙周炎大鼠肾组织损伤加重。该团队在更深入的研究中发现,牙周炎会导致大鼠肾脏功能紊乱、氧化应激水平升高、线粒体结构破坏、活性氧积累、三磷酸腺苷减少以及SIRT1和氧化物酶体增殖物激活受体γ共激活因子1α水平降低[24];但在经过白藜芦醇治疗的牙周炎组中,未出现上述情况。这提示白藜芦醇通过预防线粒体功能障碍对牙周炎引起的肾损伤发挥预防作用,进一步说明SIRT1不仅能减轻牙周组织的炎症,而且能减轻牙周炎引起的全身组织病变。 PARK等[25]的研究发现脂多糖会抑制人牙周膜细胞中碱性磷酸酶、骨桥蛋白和骨唾液酸蛋白等成骨标志物的表达,促进破骨标志物的表达,首次证明牙龈卟啉单胞菌脂多糖与牙周炎的骨质破坏有关。在脂多糖刺激的人牙周膜细胞中,SIRT1的蛋白质和mRNA水平增加、白细胞介素17(interleukin17,IL-17)和IL-23水平升高。当抑制SIRT1表达时,脂多糖刺激的IL-17和IL-23表达减少,该过程依赖于SIRT1-PI3K/Akt-MAPK-NF-κB信号传导。但在研究人员的进一步研究中却得出了接近于相反的结论[26],在受脂多糖或尼古丁刺激的人牙龈纤维组织中发现,白藜芦醇和重组SIRT1腺病毒激活SIRT1,能降低细胞毒性、活性氧生成和促炎性细胞因子产生,这说明SIRT1的过表达对牙周炎症有抑制作用。LI等[27]的研究发现也支持上述观点,SIRT1的过表达会抑制脂多糖诱导的牙周膜成纤维细胞的凋亡,抑制促炎细胞因子释放。同时,SIRT1过表达会通过降低Toll样受体4(Toll-like receptor 4,TLR4)的水平和抑制JNK/NF-κB通路改善脂多糖引发的牙周组织炎性反应,为牙周炎治疗提供了理论基础和新的治疗策略。 牙周炎导致的牙槽骨吸收,主要是由于成骨细胞介导的骨形成和破骨细胞介导的骨吸收之间的骨稳态平衡被打破[28],核因子κB受体活化因子配体(receptor activator of nuclear factor kappa-B ligand,RANKL)/核因子κB 受体活化因子(receptor activator of nuclear factor-κB,RANK)/骨保护素(osteoprotegerin,OPG)是参与骨改建的主要信号通路,成骨细胞内表达的RANKL能与破骨细胞前体膜上的功能性受体RANK结合,进而使破骨细胞前体发育为成熟破骨细胞,在牙槽骨吸收中发挥重要作用[29]。高迁移率族蛋白B1已被证实参与了脓毒症、缺血再灌注损失、糖尿病、自身免疫性疾病等疾病的发生发展[30-31]。MORIMOTO等[32]发现高迁移率族蛋白B1只存在于牙周炎患者的龈沟液中。此外,有研究发现,分泌的高迁移率族蛋白B1诱导促炎细胞因子如IL-1β、IL-6和肿瘤坏死因子α的表达,延长牙周炎的病程[33]。以上研究表明在牙周炎的进展过程中,高迁移率族蛋白B1蛋白扮演着某种角色。KIM等[34]发现,高迁移率族蛋白B1在人牙周膜细胞中通过激活p38 MAPK、核因子κB、TLR2和TLR4诱导破骨细胞因子表达。但抑制SIRT1会阻断高迁移率族蛋白B1引起人牙周膜细胞破骨细胞因子表达。基质金属蛋白酶13(matrix metalloproteinase 13,MMP-13)与慢性牙周炎密切相关,其胶原酶活性参与调控软组织和骨组织的破坏,是牙周炎的潜在治疗靶点[35]。QU等[36]在研究中发现敲除SIRT1会增强脂多糖诱导的MMP-13 mRNA表达,但激活SIRT1情况则相反。脂多糖诱导的MMP-13表达依赖于核因子κB p65直接与MMP-13启动子结合,但白藜芦醇会抑制了核因子κB p65的乙酰化和转录活性。这提示SIRT1通过靶向抑制核因子κB p65的活性进而抑制MMP-13的表达,充分说明SIRT1参与了抗炎以及抗骨吸收的过程。 microRNAs是一类进化上高度保守的非编码小分子RNA,通过靶向特定的 mRNA 参与调节许多生物学或病理过程,包括发育、分化、凋亡、细胞周期、细胞骨架组织和自噬[37]。最近的几项研究也揭示了microRNAs可通过调控SIRT1的表达,间接参与牙周炎症的调控。ZHENG等[38]研究证实,在牙周膜干细胞中的miR-22-3p可通过SIRT1途径上调炎性细胞因子的表达水平,下调TLR-2和TLR-4的表达。此外,首次发现miR-22-3P通过SIRT1沉默调控牙周膜干细胞的增殖和分化,为治疗牙周炎或减轻正畸引起的炎症提供了一个潜在的新方案。有研究人员发现在牙周炎环境中,从牙周膜干细胞提取的外泌体中microRNA-155-5P表达降低,摄入外泌体的CD4+ T细胞中SIRT1表达升高,打破了Th17/Treg的平衡,参与免疫调节,故microRNA-155-5P有希望成为治疗牙周炎症导致宿主免疫功能失衡的一个新的位点[39]。 SIRT1能参与调控牙周膜细胞的成骨分化,进而起维持骨稳态的作用。LEE等[40]研究发现SIRT1过表达可促进人牙周膜细胞向成骨样细胞分化,矿化结节形成增加,成骨相关基因表达上调,当SIRT1沉默会阻断人牙周膜细胞分化并成骨细胞相关基因的表达。富含血小板血浆活化后释放大量生长因子,具有促进骨再生的潜能。自噬分解代谢对维持骨骼稳态至关重要,它可以调节成骨细胞、骨细胞和破骨细胞的存活和功能。XU等[41]研究结果提示,凝血酶激活的富含血小板血浆是通过SIRT1介导的自噬诱导进一步提高牙周膜干细胞的细胞存活率、碱性磷酸酶活性以及成骨相关基因表达。 综合上述的许多研究发现,SIRT1既能通过PI3K/AKT-MAPK-NF-κB、高迁移率族蛋白B1、microRNAs等促进炎症因子的释放以及破骨细胞因子的表达,进而加速牙周炎的发生发展,也能通过抑制JNK/NF-κB通路、下调TLR4等抑制牙周炎的进展,然而,目前关于SIRT1调控牙周炎的具体机制还尚未完全阐明。 SIRT1在牙周炎中的研究进展总结见表1。"

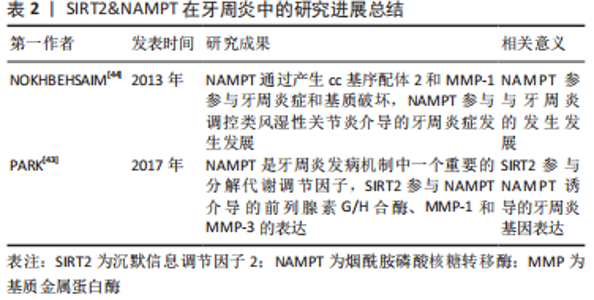
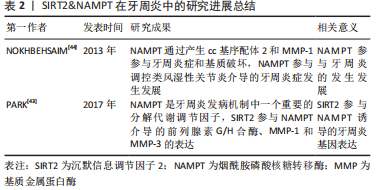
2.2 Sirtuin2&NAMPT在牙周炎中的研究进展 NAMPT具有促炎作用,能控制NAD依赖性酶(包括去乙酰化酶、PARP和NAD 酶)的活性[42]。PARK等[43]已经发现,NAMPT是牙周炎发病机制中一个重要的分解代谢调节因子。NAMPT上调环氧合酶2、MMP-1和MMP-3,这些都是牙周炎症、破骨细胞激活和牙齿周围骨质破坏的关键效应因子。此外还发现,Sirtuin2(SIRT2)参与NAMPT介导的前列腺素G/H合酶、MMP-1和MMP-3的表达,这提示SIRT2参与NAMPT诱导的牙周炎基因表达。 NOKHBEHSAIM等[44]发现,NAMPT刺激人牙周膜细胞产生cc基序配体2(cc motif ligand 2,CCL2)和MMP-1,NAMPT通过产生CCL2和MMP-1参与牙周炎症和基质破坏。除此之外,NAMPT参与调控类风湿性关节炎介导的牙周炎症发生发展。 以上这些结果,提示NAMPT可以作为治疗或预防牙周疾病的新思路,同时,SIRT2也参与NAMPT介导的牙周炎症,SIRT2也在牙周疾病的治疗和转归中发挥一定的作用。SIRT2&NAMPT在牙周炎中的研究进展总结见表2。"

2.3 Sirtuin3在牙周炎中的研究进展 自噬在维持细胞稳态方面具有重要意义,研究表明,Sirtuin3(SIRT3)通过调节AMPK-mTOR通路激活自噬来保护氧糖剥夺诱导的神经元损伤[45]。YU等[46]发现,高糖环境抑制了SIRT3-FOXO3A信号转导,下调Parkin介导的线粒体自噬,从而加重糖尿病患者的心功能障碍;激活SIRT3时,心肌细胞Parkin升高,恢复心肌细胞自噬功能。PI等[47]的研究中发现,褪黑激素通过增强体内SIRT3活性来抑制镉诱导的人肝癌细胞系自噬,使用SIRT3抑制剂后,SIRT3-SOD2信号传导受到抑制,从而阻断褪黑激素介导的自噬抑制作用。综上,SIRT3可通过AMPK-mTOR、SIRT3-FOXO3A、SIRT3-SOD2等途径调节细胞自噬。 自噬可通过抵抗病原菌的入侵、调节炎症反应、减少活性氧的产生以及调控细胞凋亡等参与牙周炎和慢性肾脏疾病的发生、发展与演变[48]。牙周炎症下,牙周组织自噬活性增强,体内和体外研究中自噬相关基因的表达增加[49]。牙周炎大鼠会出现肾脏组织损伤,SIRT3的表达和自噬水平在牙周组织和肾组织中均受到抑制。经白藜芦醇(SIRT3激活剂)干预后可激活牙周及肾组织的自噬水平,使牙周组织破坏程度降低和由牙周炎引起的肾脏损害得到改善[50]。 CHEN等[51]研究发现,SIRT3 在与年龄相关的牙周病中发挥一定的作用,衰老过程中,由于NAD+/NADH 比率下调导致牙槽骨中 SIRT3表达降低,SIRT3可调节牙槽骨吸收和破骨细胞形成,破骨细胞数量在衰老的SIRT3-KO小鼠中达到最高峰。SIRT3-KO小鼠会出现牙槽骨吸收加快和牙龈组织氧化损伤加重,这与牙周组织中过氧化物酶体增殖物激活受体γ辅激活因子1α的表达降低和线粒体呼吸水平降低有关。FENG等[52]研究指出,天麻素能阻止脂多糖诱导的人牙周膜干细胞凋亡,恢复人牙周膜干细胞中抗凋亡和促凋亡蛋白的失衡,能使人牙周膜干细胞免受脂多糖引起的炎症、凋亡和氧化应激的影响,并促进成骨分化。此外,研究发现,脂多糖可抑制SIRT3的表达,但天麻素促进SIRT3的表达。沉默SIRT3后发现,有效抑制天麻素对脂多糖诱导的人牙周膜干细胞活性的抑制作用,抑制炎症、氧化应激和成骨分化,增加细胞凋亡水平。因此,天麻素对人牙周膜干细胞成骨分化的影响依赖于SIRT3的上调表达。 关于SIRT3与牙周炎的关系还需进一步深入研究。但总体上SIRT3能抑制氧化应激,调节细胞自噬水平,缓解牙周炎导致的牙周组织及其他脏器(如肾脏)的损伤。SIRT3在牙周炎中的研究进展总结见表3。"

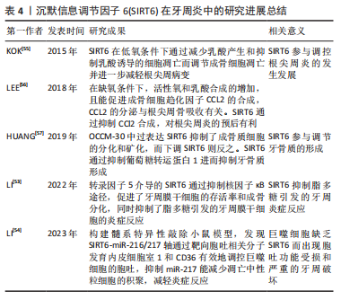
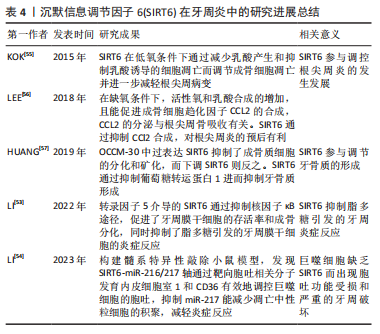
2.4 Sirtuin6在牙周炎中的研究进展 Sirtuin6(SIRT6)能通过抑制糖酵解和减少氧化应激来保护慢性炎症引起的组织损伤。LI等[53]研究发现转录因子5可调控SIRT6,干扰转录因子5会逆转SIRT6对脂多糖诱导的牙周膜干细胞炎症反应的抑制作用。转录因子5介导的SIRT6通过抑制核因子κB途径,促进了牙周膜干细胞的存活率和成骨分化,同时抑制了脂多糖引发的牙周膜干细胞的炎症反应。 糖尿病相关性牙周炎患者由于巨噬细胞缺乏SIRT6而出现胞吐功能受损和严重的牙周破坏。研究人员通过构建髓系特异性敲除小鼠模型[54],发现SIRT6-miR-216/217轴通过靶向胞吐相关分子发育内皮细胞室1和CD36有效地调控巨噬细胞的胞吐,抑制miR-217能减少凋亡中性粒细胞的积聚,减轻炎症反应,可作为促进牙周组织修复的一种新策略。 研究发现,SIRT6也参与调控根尖周炎的发生发展[55],低氧环境会增强成骨细胞的糖酵解活性,同时糖酵解的终末产物乳酸也能诱导成骨细胞凋亡,说明细胞对缺氧的凋亡反应可能依赖于糖酵解活性。在体外实验中,缺氧和炎症递质使成骨细胞SIRT6表达水平下调。此外,在细菌诱导的根尖周病变中,SIRT6在成骨细胞中的表达减少。SIRT6在低氧条件下通过减少乳酸产生和抑制乳酸诱导的细胞凋亡而调节成骨细胞凋亡并进一步减轻根尖周病变。LEE等[56]发现在缺氧条件下,活性氧和乳酸合成增加,另外,活性氧和乳酸都能促进成骨细胞趋化因子CCL2的合成,CCL2的分泌与根尖周骨吸收有关。SIRT6通过抑制CCL2合成,对根尖周炎的预后有利。 牙骨质是维持牙周组织稳态的关键,HUANG等[57]研究中发现,小鼠成牙骨质细胞系OCCM-30中过表达SIRT6抑制了成骨质细胞的分化和矿化,而下调SIRT6则反之。葡萄糖转运蛋白1参与了牙骨质的形成过程,SIRT6通过抑制葡萄糖转运蛋白1进而抑制牙骨质形成。当葡萄糖转运蛋白1过度表达时,SIRT6对成牙骨质细胞分化和矿化的抑制作用就会被消除。另外,AMPK还参与了SIRT6调控的牙骨质形成过程,并对成骨质细胞的分化和矿化起抑制作用。然而,SIRT6是否参与调控牙周炎过程中的牙骨质吸收过程,目前还尚未有研究报道。 综合上述研究,不难发现SIRT6可以抑制牙周组织炎症反应,对牙周炎和根尖周炎的预后有较好的影响。但有研究表明SIRT6会抑制牙骨质的形成,这可能会加剧牙周炎引起的牙槽骨吸收。故仍需更多的研究去探索SIRT6与牙周炎深层次的关系。SIRT6在牙周炎中的研究进展总结见表4。"
| [1] HAJISHENGALLIS G. Immunomicrobial pathogenesis of periodontitis: keystones, pathobionts, and host response. Trends Immunol. 2014;35(1):3-11. [2] SANZ M, MARCO DEL CASTILLO A, JEPSEN S, et al. Periodontitis and cardiovascular diseases: Consensus report. J Clin Periodontol. 2020;47(3):268-288. [3] SALHI L, RENERS M. Update on the Bidirectional Link Between Diabetes and Periodontitis. Adv Exp Med Biol. 2022;1373:231-240. [4] KANEKO C, KOBAYASHI T, ITO S, et al. Association among periodontitis severity, anti-agalactosyl immunoglobulin G titer, and the disease activity of rheumatoid arthritis. J Periodontal Res. 2021;56(4):702-709. [5] SADRAMELI M, BATHINI P, ALBERI L. Linking mechanisms of periodontitis to Alzheimer’s disease. Curr Opin Neurol. 2020;33(2):230-238. [6] BOBETSIS YA, GRAZIANI F, GÜRSOY M, et al. Periodontal disease and adverse pregnancy outcomes. Periodontol 2000. 2020;83(1):154-174. [7] 王兴.第四次全国口腔健康流行病学调查报告[M].北京:人民卫生出版社,2018 [8] BONKOWSKI MS, SINCLAIR DA. Slowing ageing by design: the rise of NAD+ and sirtuin-activating compounds. Nat Rev Mol Cell Biol. 2016;17(11):679-690. [9] PALOMER X, ROMÁN-AZCONA MS, PIZARRO-DELGADO J, et al. SIRT3-mediated inhibition of FOS through histone H3 deacetylation prevents cardiac fibrosis and inflammation. Signal Transduct Target Ther. 2020;5(1):14. [10] LUNAR SILVA I, CASCALES E. Molecular Strategies Underlying Porphyromonas gingivalis Virulence. J Mol Biol. 2021;433(7):166836. [11] WADDINGTON RJ, MOSELEY R, EMBERY G. Reactive oxygen species: a potential role in the pathogenesis of periodontal diseases. Oral Dis. 2000; 6(3):138-151. [12] SCZEPANIK FSC, GROSSI ML, CASATI M, et al. Periodontitis is an inflammatory disease of oxidative stress: We should treat it that way. Periodontol 2000. 2020;84(1):45-68. [13] SINGH CK, CHHABRA G, NDIAYE MA, et al. The Role of Sirtuins in Antioxidant and Redox Signaling. Antioxid Redox Signal. 2018;28(8):643-661. [14] PARK S, SHIN J, BAE J, et al. SIRT1 Alleviates LPS-Induced IL-1β Production by Suppressing NLRP3 Inflammasome Activation and ROS Production in Trophoblasts. Cells. 2020;9(3):728. [15] YE Q, XU H, LIU S, et al. Apoptotic extracellular vesicles alleviate Pg-LPS induced inflammatory responses of macrophages via AMPK/SIRT1/NF-κB pathway and inhibit osteoclast formation. J Periodontol. 2022;93(11): 1738-1751. [16] 丁旭. Sirt3诱导的自噬在大鼠牙周炎相关肾损伤中的作用研究[D].长春:吉林大学,2022. [17] 周丰,陈野,陈晨,等.沉默信息调节因子1调控牙周炎发生发展的机制[J].国际口腔医学杂志,2021,48(3):341-346. [18] CARIBÉ PMV, VILLAR CC, ROMITO GA, et al. Prospective, case-controlled study evaluating serum concentration of sirtuin-1 and mannose-binding lectin in patients with and without periodontal and coronary artery disease. Ther Adv Chronic Dis. 2020;11:2040622320919621. [19] CARIBÉ PMV, VILLAR CC, ROMITO GA, et al. Influence of the treatment of periodontal disease in serum concentration of sirtuin 1 and mannose-binding lectin. J Periodontol. 2020;91(7):900-905. [20] TAMAKI N, CRISTINA ORIHUELA-CAMPOS R, INAGAKI Y, et al. Resveratrol improves oxidative stress and prevents the progression of periodontitis via the activation of the Sirt1/AMPK and the Nrf2/antioxidant defense pathways in a rat periodontitis model. Free Radic Biol Med. 2014;75:222-229. [21] CORRÊA MG, ABSY S, TENENBAUM H, et al. Resveratrol attenuates oxidative stress during experimental periodontitis in rats exposed to cigarette smoke inhalation. J Periodontal Res. 2019;54(3):225-232. [22] CIRANO FR, MOLEZ AM, RIBEIRO FV, et al. Resveratrol and insulin association reduced alveolar bone loss and produced an antioxidant effect in diabetic rats. J Periodontol. 2021;92(5):748-759. [23] LI X, LIU XC, DING X, et al. Resveratrol protects renal damages induced by periodontitis via preventing mitochondrial dysfunction in rats [published online ahead of print, 2022 Feb 10]. Oral Dis. 2022;10.1111/odi.14148. [24] 李鑫,丁旭,刘笑梦,等.沉默信息调节因子1对慢性牙周炎模型大鼠肾损伤的影响[J].吉林大学学报(医学版),2022,48(5):1200-1208. [25] PARK YD, KIM YS, JUNG YM, et al. Porphyromonas gingivalis lipopolysaccharide regulates interleukin (IL)-17 and IL-23 expression via SIRT1 modulation in human periodontal ligament cells. Cytokine. 2012; 60(1):284-293. [26] PARK GJ, KIM YS, KANG KL, et al. Effects of sirtuin 1 activation on nicotine and lipopolysaccharide-induced cytotoxicity and inflammatory cytokine production in human gingival fibroblasts. J Periodontal Res. 2013;48(4): 483-492. [27] LI K, LV G, PAN L. Sirt1 alleviates LPS induced inflammation of periodontal ligament fibroblasts via downregulation of TLR4. Int J Biol Macromol. 2018; 119:249-254. [28] INTINI G, KATSURAGI Y, KIRKWOOD KL, et al. Alveolar bone loss: mechanisms, potential therapeutic targets, and interventions. Adv Dent Res. 2014;26(1):38-46. [29] LACEY DL, TIMMS E, TAN HL, et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 1998;93(2):165-176. [30] WANG H, BLOOM O, ZHANG M, et al. HMG-1 as a late mediator of endotoxin lethality in mice. Science. 1999;285(5425):248-251. [31] TANIGUCHI N, KAWAHARA K, YONE K, et al. High mobility group box chromosomal protein 1 plays a role in the pathogenesis of rheumatoid arthritis as a novel cytokine. Arthritis Rheum. 2003;48(4):971-981. [32] MORIMOTO Y, KAWAHARA KI, TANCHAROEN S, et al. Tumor necrosis factor-alpha stimulates gingival epithelial cells to release high mobility-group box 1. J Periodontal Res. 2008;43(1):76-83. [33] YAMASHIRO K, IDEGUCHI H, AOYAGI H, et al. High Mobility Group Box 1 Expression in Oral Inflammation and Regeneration. Front Immunol. 2020; 11:1461. [34] KIM YS, LEE YM, PARK JS, et al. SIRT1 modulates high-mobility group box 1-induced osteoclastogenic cytokines in human periodontal ligament cells. J Cell Biochem. 2010;111(5):1310-1320. [35] 王亚敏,张瑞敏.基质金属蛋白酶作为牙周炎调节因子的研究进展[J].口腔医学,2019,39(3):271-274. [36] QU L, YU Y, QIU L, et al. Sirtuin 1 regulates matrix metalloproteinase-13 expression induced by Porphyromonas endodontalis lipopolysaccharide via targeting nuclear factor-κB in osteoblasts. J Oral Microbiol. 2017;9(1):1317578. [37] RIAHI RAD Z, RIAHI RAD Z, GOUDARZI H, et al. MicroRNAs in the interaction between host-bacterial pathogens: A new perspective. J Cell Physiol. 2021; 236(9):6249-6270. [38] Zheng M, Guo J. NICOTINAMIDE-INDUCED SILENCING OF SIRT1 BY MIR-22-3P INCREASES periodontal ligament stem cell proliferation and differentiation. Cell Biol Int. 2020;44(3):764-772. [39] ZHENG Y, DONG C, YANG J, et al. Exosomal microRNA-155-5p from PDLSCs regulated Th17/Treg balance by targeting sirtuin-1 in chronic periodontitis. J Cell Physiol. 2019;234(11):20662-20674. [40] LEE YM, SHIN SI, SHIN KS, et al. The role of sirtuin 1 in osteoblastic differentiation in human periodontal ligament cells. J Periodontal Res. 2011;46(6):712-721. [41] XU Y, WANG X, LIU W, et al. Thrombin-activated platelet-rich plasma enhances osteogenic differentiation of human periodontal ligament stem cells by activating SIRT1-mediated autophagy. Eur J Med Res. 2021;26(1):105. [42] AUDRITO V, MESSANA VG, DEAGLIO S. NAMPT and NAPRT: Two Metabolic Enzymes With Key Roles in Inflammation. Front Oncol. 2020;10:358. [43] PARK KH, KIM DK, HUH YH, et al. NAMPT enzyme activity regulates catabolic gene expression in gingival fibroblasts during periodontitis. Exp Mol Med. 2017;49(8):e368. [44] NOKHBEHSAIM M, EICK S, NOGUEIRA AV, et al. Stimulation of MMP-1 and CCL2 by NAMPT in PDL cells. Mediators Inflamm. 2013;2013:437123. [45] DAI SH, CHEN T, LI X, et al. Sirt3 confers protection against neuronal ischemia by inducing autophagy: Involvement of the AMPK-mTOR pathway. Free Radic Biol Med. 2017;108:345-353. [46] YU W, GAO B, LI N, et al. Sirt3 deficiency exacerbates diabetic cardiac dysfunction: Role of Foxo3A-Parkin-mediated mitophagy. Biochim Biophys Acta Mol Basis Dis. 2017;1863(8):1973-1983. [47] PI H, XU S, REITER RJ, et al. SIRT3-SOD2-mROS-dependent autophagy in cadmium-induced hepatotoxicity and salvage by melatonin. Autophagy. 2015;11(7):1037-1051. [48] GREABU M, GIAMPIERI F, IMRE MM, et al. Autophagy, One of the Main Steps in Periodontitis Pathogenesis and Evolution. Molecules. 2020;25(18):4338. [49] HE S, ZHOU Q, LUO B, et al. Chloroquine and 3-Methyladenine Attenuates Periodontal Inflammation and Bone Loss in Experimental Periodontitis. Inflammation. 2020;43(1):220-230. [50] DING X, HOU Y, LIU X, et al. The role of Sirt3-induced autophagy in renal structural damage caused by periodontitis in rats. J Periodontal Res. 2023; 58(1):97-108. [51] CHEN J, ZHANG Y, GAO J, et al. Sirtuin 3 deficiency exacerbates age-related periodontal disease. J Periodontal Res. 2021;56(6):1163-1173. [52] FENG Q. Gastrodin attenuates lipopolysaccharide-induced inflammation and oxidative stress, and promotes the osteogenic differentiation of human periodontal ligament stem cells through enhancing sirtuin3 expression. Exp Ther Med. 2022;23(4):296. [53] LI C, XIAO F, WEN Y, et al. Krüppel-like factor 5 -mediated Sirtuin6 promotes osteogenic differentiation and inhibits inflammatory injury of lipopolysaccharide-induced periodontal membrane stem cells by inhibiting nuclear factor kappa-B pathway. Bioengineered. 2022;13(3):6966-6977. [54] LI B, XIN Z, GAO S, et al. SIRT6-regulated macrophage efferocytosis epigenetically controls inflammation resolution of diabetic periodontitis. Theranostics. 2023;13(1):231-249. [55] KOK SH, HOU KL, HONG CY, et al. Sirtuin 6 Modulates Hypoxia-induced Apoptosis in Osteoblasts via Inhibition of Glycolysis: Implication for Pathogenesis of Periapical Lesions. J Endod. 2015;41(10):1631-1637. [56] LEE YL, LIN SK, HOU KL, et al. Sirtuin 6 attenuates periapical lesion propagation by modulating hypoxia-induced chemokine (C-C motif) ligand 2 production in osteoblasts. Int Endod J. 2018;51 Suppl 2:e74-e86. [57] HUANG L, SUN H, SONG F, et al. SIRT6 overexpression inhibits cementogenesis by suppressing glucose transporter 1. J Cell Physiol. 2019; 234(4):4005-4014. |
| [1] | Yang Yifeng, Ye Nan, Wang Lin, Guo Shuaicheng, Huang Jian. Signaling pathway of dexmedetomidine against ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1464-1469. |
| [2] | Lou Guo, Zhang Yan, Fu Changxi. Role of endothelial nitric oxide synthase in exercise preconditioning-induced improvement of myocardial ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(8): 1283-1288. |
| [3] | Pan Xiaolong, Fan Feiyan, Ying Chunmiao, Liu Feixiang, Zhang Yunke. Effect and mechanism of traditional Chinese medicine on inhibiting the aging of mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1091-1098. |
| [4] | Cao Sheng, Kong Lingwei, Xu Kun, Sun Zhijie. Effect of gelatin methacryloyl hydrogel loaded with salvianolic acid B on intervertebral disc degeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 380-386. |
| [5] | Sun Yuan, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Zhang Yan. Effects of early and late aerobic exercise on right heart failure induced by monocrotaline in rats with pulmonary hypertension [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 177-185. |
| [6] | Chen Simin, Hu Yingjun, Yan Wenrui, Ji Le, Shao Mengli, Sun Ze, Zheng Hongxing, Qi Shanshan. Establishment and evaluation of a streptozotocin-induced diabetic encephalopathy rat model [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(2): 237-241. |
| [7] | Ma Suilu, He Zhijun, Liu Tao, Li Yan, He Yuanxu, He Bo, Wang Weiwei, Wei Xiaotao. Traditional Chinese medicine monomer in the prevention and treatment of flap necrosis by regulating “autophagy” [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(1): 153-158. |
| [8] | Fang Xingyan, Tian Zhenli, Zhao Zheyi, Wen Ping, Xie Tingting. Effects of sodium arsenite on human umbilical vein endothelial cell injury and sphingosine kinases 1/sphingosine 1-phosphate signaling axis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [9] | Guo Shuhui, Yang Ye, Jiang Yangyang, Xu Jianwen. Screening and validation of neurogenic bladder miRNA-mRNA regulatory network [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-8. |
| [10] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [11] | Li Mengfei, Zhang Hong, Zhao Shaojian, Yin Guanghao, Wang Qibao. Expression of forkhead box protein 3 in refractory periapical periodontitis in rats with Enterococcus faecalis infection [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1187-1192. |
| [12] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [13] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [14] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [15] | Tian Qinyu, Tian Xinggui, Tian Zhuang, Sui Xiang, Liu Shuyun, Lu Xiaobo, Guo Quanyi. Protection of manganese oxide nanoparticles for bone marrow mesenchymal stem cell spreading against oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(6): 821-826. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||