Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (27): 4408-4413.doi: 10.12307/2023.604
Previous Articles Next Articles
Risk and prevention of intervertebral implant subsidence after anterior cervical decompression and fusion
Peng Jiancheng1, Yao Xiaoling1, Ling Haiqian1, Lai Ying2, Li Jie1
- 1Shenzhen Longgang District Orthopedic Hospital, Shenzhen 518116, Guangdong Province, China; 2School of Clinical Medicine, Guangdong Pharmaceutical University, Guangzhou 510006, Guangdong Province, China
-
Received:2022-05-12Accepted:2022-07-18Online:2023-09-28Published:2022-11-08 -
Contact:Li Jie, Associate chief physician, Shenzhen Longgang District Orthopedic Hospital, Shenzhen 518116, Guangdong Province, China -
About author:Peng Jiancheng, Master, Physician, Shenzhen Longgang District Orthopedic Hospital, Shenzhen 518116, Guangdong Province, China
CLC Number:
Cite this article
Peng Jiancheng, Yao Xiaoling, Ling Haiqian, Lai Ying, Li Jie. Risk and prevention of intervertebral implant subsidence after anterior cervical decompression and fusion[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(27): 4408-4413.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
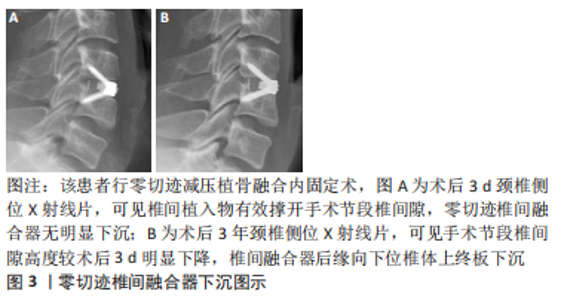
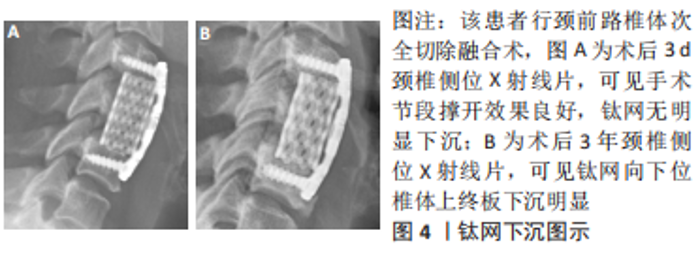
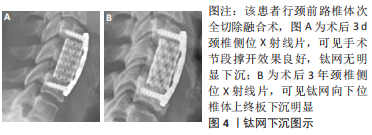
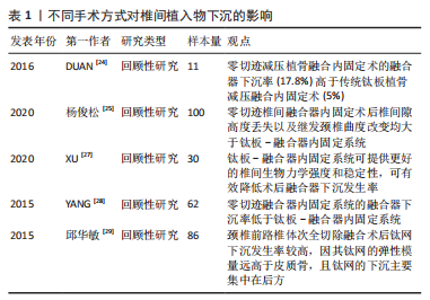
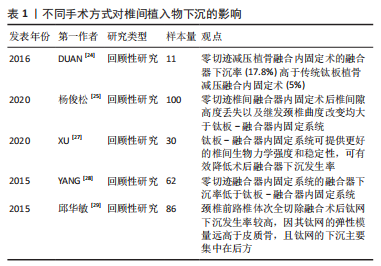
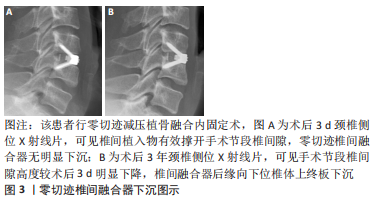
2.1 椎间植入物下沉的临床特点 椎间植入物下沉是指术后椎间盘高度的丢失,其诊断标准为颈椎间盘高度缺失≥3 mm[2]。 CORNIOLA等[3]对椎间植入物下沉有了更具体的定义,即末次随访颈椎间盘高度较术后即刻椎间盘高度下降≥3 mm。PINTER等[4] 提出了融合器下沉分度的观点,将手术节段的融合器下沉≤2 mm定义为轻度,下沉在2-4 mm之间为中度,下沉≥4 mm为重度。FUJIBAYASHI等[5]将融合器下沉分为2种类型,短暂下沉型表现为1-3 mm下沉而没有进一步变化,而渐进下沉型则导致骨不连。目前,对于颈前路减压融合术后椎间植入物下沉的标准和类型尚未形成完全统一。 一项多中心研究报道颈前路减压融合术后椎间植入物下沉的发生率为17.7%,大多数的下沉在术后3个月时即可通过影像资料发现[6-7]。唐福波等[8]对行颈前路椎体次全切减压融合术的81例患者随访发现术后钛网下沉的发生率为29.6%。椎间植入物下沉可能导致颈椎曲度改变、轴性症状、节段不稳甚至颈椎后凸畸形,影响长期疗效,并会造成椎间孔狭窄,初期无临床症状,术后6个月左右则可因下沉产生的椎间孔狭窄引起神经根性疼痛,影响术后神经根减压的效果[9-10]。但是,近年来有越来越多的文献提出了不同观点,尽管术后出现了椎间植入物下沉,但与临床疗效、融合率并无明显相关性[7,11]。对于该观点的解释,有学者认为融合器下沉是骨终板与椎间融合器融合过程中发生的一个固有过程,包括骨的吸收和重塑[12]。MACCORMICK等[13]研究显示,颈前路减压融合术后有91%的手术节段出现融合器轻微下沉(< 2 mm),没有患者需要进行翻修手术,作者认为该影像学结果可能是椎间融合的自然进程。LEE等[14]研究认为,颈前路减压融合术后椎间融合器下沉不会影响临床疗效,而当严重下沉继发的节段性颈椎后凸则会影响长期疗效。 2.2 椎间植入物下沉的影响原因 2.2.1 术前因素 在性别方面,不同研究得出了不同的结论。韩俊等[15]研究认为,椎间植入物下沉与患者性别无明显相关性。部分学者认为,绝经后女性为患骨质疏松症的高危人群,低骨密度可导致终板刚度降低,椎间植入物刚度高于终板刚度时下沉风险增加[16]。JI等[17]研究认为,对于女性患者,术前6个月开始抗骨质疏松治疗可有效预防术后椎间植入物下沉。 但也有学者观察到男性的发生率高于女性,认为可能与男性术后的颈椎活动范围大于女性相关[18]。体质量指数被认为是椎间植入物下沉的危险因素之一。YAMAGATA等[19]报道体质量指数低的患者椎间植入物下沉发生率明显增高,认为体质量较大患者骨的力学性能较强,因此能承受更大的负荷。蔡忠香等[20]通过术后1年随访发现,颈椎外伤患者的椎间植入物下沉率显著高于颈椎退行性变患者,其认为骨折椎体、毗邻椎体及后方韧带复合体的生物学强度可能受损,从而导致椎间植入物的负荷相对增加,术后出现下沉的风险增大。术前颈椎矢状位参数对颈前路术后椎间植入物下沉的影响主要表现在T1倾斜率和颈椎角,更高的T1倾斜率和倒置的颈椎角可增加术后椎间植入物下沉风险。 2.2.2 手术方式、节段 不同的手术节段被认为是影响椎间植入物下沉的因素之一。VAN JONBERGEN等[21]报道颈前路减压融合术后发现C6/7节段是融合器下沉的好发节段。椎间植入物下沉更可能发生在2个以上手术节段的患者中,并且手术节段C5-7比C2-5更可能发生椎间植入物下沉[22-23]。JIN等[18]研究认为,从生物力学的角度来看,直接传递到终板-椎间植入物界面的负载压力可以由植入物前方的金属镀层分担,相比单节段颈前路减压融合术,该金属镀层在多节段手术中承载更大的压力,导致椎间植入物下沉的发生率更高。 在颈前路减压融合手术中,不同手术方式对椎间植入物下沉也产生一定的影响。DUAN等[24]研究报道零切迹减压植骨融合内固定术的融合器下沉率(17.8%)高于传统钛板植骨减压融合内固定术(5%)。国内学者通过随访发现,颈前路术中采用零切迹椎间融合器虽能获得与钛板-融合器内固定系统相似的疗效,但术后椎间隙高度丢失以及继发颈椎曲度改变均大于钛板-融合器内固定系统[25-26],见图3。零切迹减压植骨融合内固定术发生较高的融合器下沉发生率可能与术后节段性微运动相关,造成颈椎应力分布改变,这种“微动”在可变角度零切迹前路椎间融合内固定系统(Zero-P VA)中更加明显,而传统钛板-融合器内固定系统则提供了更好的稳定性。XU等[27]通过对比分析钛板-融合器内固定系统(CP)、零切迹融合器内固定系统(Zero-P)、带钢板的髂骨移植物(IP)、单独的聚醚醚酮融合器(PCA)、髂骨自体移植物(ICAG)和单独的钛融合器(TCA),发现钛板-融合器内固定系统组下沉率最低,钛板-融合器内固定系统可提供更好的椎间生物力学强度和稳定性,而带钢板的髂骨移植物组下沉率显著低于聚醚醚酮融合器组、髂骨自体移植物组和钛融合器组。JIN等[18]研究报道,零切迹自稳型颈椎融合器(ROI-C)的下沉率高于传统钛板-融合器内固定系统,在多节段手术中有显著差异,但在单节段手术中差异无统计学意义。但也有学者研究发现,零切迹融合器内固定系统的融合器下沉率低于钛板-融合器内固定系统[28],因此尚需更深入的研究,探讨零切迹融合器内固定系统与传统钛板-融合器内固定系统对术后融合器下沉的影响是否存在差异。颈前路椎体次全切除融合术后钛网下沉具有较高的发生率,见图4,因其钛网的弹性模量远高于皮质骨,且钛网的下沉主要集中在后方,有观点认为,钛网下沉有利于恢复颈椎前凸,但对远期临床结果的影响还需进一步研究[29-30]。见表1。"
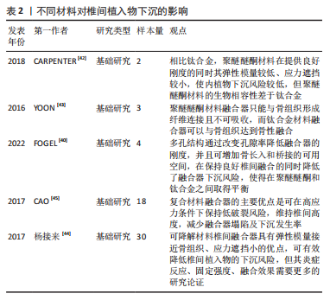
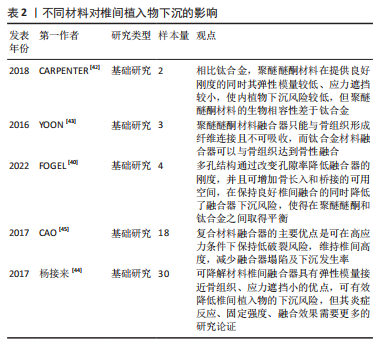
2.2.3 植入物的大小、类型、位置、材质 椎间植入物的大小、位置是引起植入物下沉的危险因素。国内外主流观点认为,椎间植入物的直径和宽度越大,与相邻椎体终板接触的面积就越大,根据压强的原理,在承受轴向负荷时椎间植入物下沉的风险就越小[10,31-32]。YANG等[32]研究报道,与使用12 mm直径的融合器相比,使用14 mm直径的融合器下沉的风险显著降低。更大椎间植入物的高度被认为可有效恢复颈椎曲度,但过高的融合器意味着对椎间过度撑开,YAMAGATA等[19]对47例行颈前路减压融合术的患者进行回顾性研究,1年随访后发现,与4.5 mm或5.5 mm相比,6.5 mm或7.5 mm的融合器有更高的下沉风险。但具体多少直径、宽度、高度的椎间植入物最合适,如何做到每例患者的个体化设计,还需生物力学方面及临床方面的进一步研究,融合器并非越大越好,更大的融合器会增加骨组织损伤,进而导致融合器下沉。椎体前方的皮质骨较中央区域多,因此能承受更大的负荷,术中处理终板后椎体中央以松质骨为主,因此,颈前路椎体次全切除融合术中钛网前缘与椎体前缘的距离可影响术后钛网下沉,钛网放置位置越趋近于椎体中部时,越容易发生下沉[15]。马永刚等[33]对104例行颈前路椎体次全切除融合术的患者进行随访研究,发现钛网前缘与椎体前缘距离< 1 mm组的钛网下沉发生率显著低于> 1 mm组,作者推测可能与皮质骨、松质骨的骨质强度差异相关。 不同形状的椎间植入物可引起椎间力学性能的差异,导致下沉风险出现差异。肾形融合器的前凸设计利于恢复颈椎曲度,但其放置位置靠近终板中央松质骨,而矩形融合器跨越终板周围皮质骨,对终板的应力集中减少,故矩形融合器的下沉发生率低于肾形融合器[34]。有学者设计出一种中间为圆柱状两侧带扁平翼的椎间融合器(WING Cage)[35],认为“侧翼”设计可以降低融合器下沉风险。研究表明,四叶形和肾形的融合器能更好地促进椎间融合,但在降低融合器下沉发生率方面,十二叶形的融合器更具有优势[36]。铰链式融合器的设计可减少植入过程中对周围组织的损伤,但融合器与终板的贴合度较低,导致融合器下沉风险增加,而解剖形融合器通过增大终板-融合器接触面积,达到了减少终板-融合器界面压力的目的,从而降低融合器下沉风险[37-38]。仿生学应用到椎间融合器的设计中,为研究提供了新的思考。国内学者通过研究马腿第三掌骨滋养小孔,将其力学特性应用在了椎间融合器上,设计了仿生凹坑表面形态,将凹坑表面形态应用在融合器侧面,模拟出了兼顾预防下沉和促进椎间融合的最优凹坑数据,同时,通过生物力学分析得出,该学者设计的二元耦合仿生颈椎融合器较结构单元仿生融合器能更好地预防下沉[36]。 近年,3D打印技术也逐渐运用到颈椎的生物力学研究中。舒启航等[39]研究设计了新型颈椎3D打印多孔椎间融合器,通过有限元分析发现,该设计相对于传统融合器能减少终板的应力集中,一定程度上可降低内植物下沉风险。FOGEL等[40]研究认为,多孔结构通过改变孔隙率降低融合器的刚度,使椎间融合器的刚度降低16.7%,使其弹性模量更接近椎体的弹性模量,多孔结构的钛融合器可增加骨长入和桥接的可用空间,在保持良好椎间融合的同时降低了融合器下沉风险。 目前,临床中主流椎间融合器材质类型有聚醚醚酮(PEEK)和钛合金。相比钛合金,聚醚醚酮材料在提供良好刚度的同时其弹性模量较低、应力遮挡较小,使内植物下沉风险较低,且X射线可透性更好,利于术后进行影像学评估,但聚醚醚酮材料的生物相容性差于钛合金,聚醚醚酮材料融合器只能与骨组织形成纤维连接且不可吸收,而钛合金材料融合器可以与骨组织达到骨性融合[41-43]。3D打印技术的发展则在二者之间取得了平衡,通过多孔结构改变孔隙率从而降低弹性模量,使3D打印钛合金多孔融合器兼备了力学优势与生物学优势。随着材料学的发展,可降解材料椎间融合器的研究越来越多,聚乳酸(PLA)和聚乙醇酸(PGA)是研究的主要两大类,它们具有弹性模量接近骨组织、应力遮挡小的优点,可有效降低椎间植入物的下沉风险,但是否能减少降解中的炎症反应、提供良好的固定强度和融合效果,需要更多的研究论证,长期降解后导致的骨溶解亦会引起植入物的进一步下沉[30,44]。复合材料融合器的研发改善了单一材料的不足,在提供高韧性及高抗形变强度的同时,也提供了高抗压缩强度,在维持椎间高度方面具有优势,减少融合器下沉发生率,但在生物力学和促成骨方面的远期效果仍需进一步研究[45-47]。见表2。"
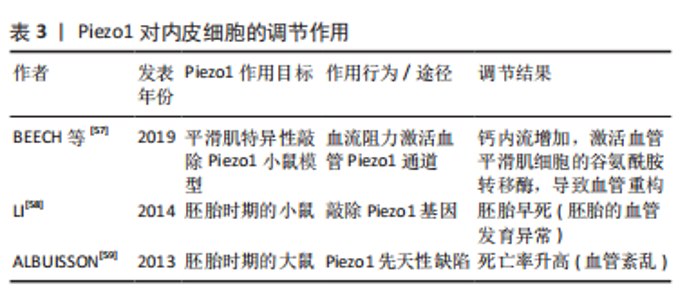
2.2.4 术中因素 终板处理方式、椎间撑开程度、颈椎牵开器的使用、螺钉的置入位置是颈前路减压融合术中影响椎间植入物下沉的因素。术中过度刮除终板是融合器下沉的重要因素,LI等[48-49]通过多变量logistic分析证实了颈前路手术后终板切除厚度与融合器下沉之间的关联,同时,超过3.7 mm的终板切除厚度与融合器下沉显著相关。术中对椎体前缘过度撑开或选择高度过大的融合器时,会导致终板后部应力集中,导致终板-融合器接触面积减小,同时椎间高度的过度增加会导致周围的韧带和肌肉牵引力增加,从而压缩内植物,增加内植物下沉风险[18,50]。WANG等[51]报道的改良颈前路减压融合术(mACDF)通过在放置融合器前释放Caspar颈椎牵开器来避免过度撑开,研究结果显示通过这种处理可恢复颈椎的“自高度”,缓解术后即刻疼痛,防止椎间融合器快速下沉。既往生物力学研究已证实,工作长度的缩短可增加结构刚度,而在颈前路减压融合术中,螺钉的置入高度可影响螺钉的工作长度,从而对椎间植入物的刚度产生影响,PINTER等[4]研究报道近端螺钉尖端高度与椎体高度比降低是发生融合器下沉> 2 mm的危险因素。在颈前路椎体次全切除融合术中,钛笼倾斜角度与术后钛笼下沉相关,研究报道钛笼倾斜角增加1°,下沉风险增加约1.209倍,当钛笼倾斜角度取最佳临界值为8.6°时,对预测下沉的灵敏度为85.2%,> 8.6°将增加钛笼出现下沉的风险[52]。见表3。"
| [1] ZHANG AS, MYERS C, MCDONALD CL, et al. Cervical Myelopathy: Diagnosis, Contemporary Treatment, and Outcomes. Am J Med. 2022; 135(4):435-443. [2] NOORDHOEK I, KONING MT, JACOBS WCH, et al. Incidence and clinical relevance of cage subsidence in anterior cervical discectomy and fusion: a systematic review. Acta Neurochir (Wien). 2018;160(4):873-880. [3] CORNIOLA MV, JAGERSEERG M, STIENEN MN, et al. Complete cage migration/subsidence into the adjacent vertebral body after posterior lumbar interbody fusion. J Clin Neurosci. 2015;22(3):597-598. [4] PINTER ZW, REED R, TOWNSLEY SE, et al. Titanium Cervical Cage Subsidence: Postoperative Computed Tomography Analysis Defining Incidence and Associated Risk Factors. Global Spine J. 2021: 21925682211046897. [5] FUJIBAYASHI S, NEO M, NAKAMURA T. Stand-alone interbody cage versus anterior cervical plate for treatment of cervical disc herniation: sequential changes in cage subsidence. J Clin Neurosci. 2008;15(9):1017-1022. [6] NAKANISHI Y, NAITO K, YAMAGATA T, et al. Safety of anterior cervical discectomy and fusion using titanium-coated polyetheretherketone stand-alone cages: Multicenter prospective study of incidence of cage subsidence. J Clin Neurosci. 2020;74:47-54. [7] CHEN Y, LV G, WANG B, et al. A comparison of anterior cervical discectomy and fusion (ACDF) using self-locking stand-alone polyetheretherketone (PEEK) cage with ACDF using cage and plate in the treatment of three-level cervical degenerative spondylopathy: a retrospective study with 2-year follow-up. Eur Spine J. 2016;25(7): 2255-2262. [8] 唐福波,周劲衍,张翼升,等.颈椎前路单椎体次全切钛笼植骨融合后钛笼下沉的相关危险因素[J].中国组织工程研究,2019,23(12): 1818-1823. [9] KARIKARI IO, JAIN D, OWENS TR, et al. Impact of subsidence on clinical outcomes and radiographic fusion rates in anterior cervical discectomy and fusion: a systematic review. J Spinal Disord Tech. 2014;27(1):1-10. [10] ALIMI M, LANG G, NAVARRO-RAMIREZ R, et al. The Impact of Cage Dimensions, Positioning, and Side of Approach in Extreme Lateral Interbody Fusion. Clin Spine Surg. 2018;31(1):E42-E49. [11] WU WJ, JIANG LS, LIANG Y, et al. Cage subsidence does not, but cervical lordosis improvement does affect the long-term results of anterior cervical fusion with stand-alone cage for degenerative cervical disc disease: a retrospective study. Eur Spine J. 2012;21(7):1374-1382. [12] ZHANG JD, POFFYN B, SYS G, et al. Are stand-alone cages sufficient for anterior lumbar interbody fusion. Orthop Surg. 2012;4(1):11-14. [13] MACCORMICK AP, SHARMA H. Analysis of the Variables Affecting the Incidence, Location, and Severity of Cage Subsidence Following Anterior Cervical Discectomy and Fusion Operation. Int J Spine Surg. 2020;14(6):896-900. [14] LEE CH, KIM KJ, HYUN SJ, et al. Subsidence as of 12 months after single-level anterior cervical inter-body fusion. Is it related to clinical outcomes. Acta Neurochir. 2015;157(6):1063-1068. [15] 韩俊,黄勇.颈椎ACCF术后出现钛笼下沉的危险因素Logistic回归分析[J].颈腰痛杂志,2019,40(4):442-444. [16] 邱华敏.颈椎前路椎体次全切除减压融合术后钛笼下沉的影响因素分析[D].南宁:广西医科大学,2015. [17] JI C, YU S, YAN N, et al. Risk factors for subsidence of titanium mesh cage following single-level anterior cervical corpectomy and fusion. BMC Musculoskelet Disord. 2020;21(1):32. [18] JIN ZY, TENG Y, WANG HZ, et al. Comparative Analysis of Cage Subsidence in Anterior Cervical Decompression and Fusion: Zero Profile Anchored Spacer (ROI-C) vs. Conventional Cage and Plate Construct. Front Surg. 2021;8:736680. [19] YAMAGATA T, TAKAMI T, UDA T, et al. Outcomes of contemporary use of rectangular titanium stand-alone cages in anterior cervical discectomy and fusion: cage subsidence and cervical alignment. J Clin Neurosci. 2012;19(12):1673-1678. [20] 蔡忠香,马永刚.颈椎前路融合术后12例钛笼下沉临床分析[J].生物骨科材料与临床研究,2011,8(1):52-53. [21] VAN JONBERGEN HP, SPRUIT M, ANDERSON PG, et al. Anterior cervical interbody fusion with a titanium box cage: early radiological assessment of fusion and subsidence. Spine J. 2005;5(6):645-649. [22] KAO TH, WU CH, CHOU YC, et al. Risk factors for subsidence in anterior cervical fusion with stand-alone polyetheretherketone (PEEK) cages: a review of 82 cases and 182 levels. Arch Orthop Trauma Surg. 2014; 134(10):1343-1351. [23] LU Y, FANG Y, SHEN X, et al. Does zero-profile anchored cage accompanied by a higher postoperative subsidence compared with cage-plate construct? A meta-analysis. J Orthop Surg Res. 2020;15(1): 189. [24] DUAN Y, YANG Y, WANG Y, et al. Comparison of anterior cervical discectomy and fusion with the zero-profile device versus plate and cage in treating cervical degenerative disc disease: A meta-analysis. J Clin Neurosci. 2016;33:11-18. [25] 杨俊松,刘鹏,刘团江,等.零切迹椎间融合器与钛板-椎间融合器系统对颈前路术后颈椎曲度维持的临床研究[J].中国修复重建外科杂志,2020,34(2):151-156. [26] 姚晓玲,彭建城,许岳荣,等.可变角度零切迹前路椎间融合内固定系统治疗脊髓型颈椎病:30个月随访[J].中国组织工程研究, 2022,26(9):1377-1382. [27] XU J, HE Y, LI Y, et al. Incidence of Subsidence of Seven Intervertebral Devices in Anterior Cervical Discectomy and Fusion: A Network Meta-Analysis. World Neurosurg. 2020;141:479-489. [28] YANG H, CHEN D, WANG X, et al. Zero-profile integrated plate and spacer device reduces rate of adjacent-level ossification development and dysphagia compared to ACDF with plating and cage system. Arch Orthop Trauma Surg. 2015;135(6):781-787. [29] 邱华敏,詹新立.双节段颈椎前路椎体次全切除融合术后钛笼下沉的影响因素分析[J].骨科,2015,6(3):130-134. [30] 杨毅,刘浩,孟阳.颈椎融合器的研究历史与现状[J].生物骨科材料与临床研究,2020,17(3):62-65. [31] 程招军,彭兵,范俊超,等.脊髓型颈椎病患者颈椎 MRI与X线片矢状位参数的对比分析[J].中国脊柱脊髓杂志,2016,26(7):591-595. [32] YANG JJ, YU CH, CHANG BS, et al. Subsidence and nonunion after anterior cervical interbody fusion using a stand-alone polyetheretherketone (PEEK) cage. Clin Orthop Surg. 2011;3(1): 16-23. [33] 马永刚,刘世清,李亚明,等.颈椎前路减压融合术后钛笼下沉临床分析[J].中国脊柱脊髓杂志,2011,21(1):21-23. [34] CHOI WS, KIM JS, HUR JW, et al. Minimally Invasive Transforaminal Lumbar Interbody Fusion Using Banana-Shaped and Straight Cages: Radiological and Clinical Results from a Prospective Randomized Clinical Trial. Neurosurgery. 2018;82(3):289-298. [35] WILKE HJ, KETTLER A, GOETZ C, et al. Subsidence resulting from simulated postoperative neck movements: an in vitro investigation with a new cervical fusion cage. Spine (Phila Pa 1976). 2000;25(21): 2762-2770. [36] 王甲甲.人体颈椎有限元建模及仿生颈椎椎间融合器研究[D].长春:吉林大学,2014. [37] WANG Y, LU T, HE X, et al. Effect of Dome-Shaped Titanium Mesh Cages on Cervical Endplate Under Cyclic Loading: An In Vitro Biomechanics Study. Med Sci Monit. 2019;25:142-149. [38] ZHANG H, JIANG Y, WANG B, et al. Direction-changeable lumbar cage versus traditional lumbar cage for treating lumbar spondylolisthesis: A retrospective study. Medicine (Baltimore). 2018;97(7):e9984. [39] 舒启航,廖亦佳,薛静波,等.新型颈椎3D打印多孔椎间融合器的三维有限元分析[J].中国组织工程研究,2021,25(24):3810-3815. [40] FOGEL G, MARTIN N, LYNCH K, et al. Subsidence and fusion performance of a 3D-printed porous interbody cage with stress-optimized body lattice and microporous endplates - a comprehensive mechanical and biological analysis. Spine J. 2022;22(6):1028-1037. [41] 王林楠,杨曦,宋跃明. 3D打印技术在椎间融合临床应用的研究进展[J].华西医学,2018,33(9):1061-1067. [42] CARPENTER RD, KLOSTERHOFF BS, TORSTRICK FB, et al. Effect of porous orthopaedic implant material and structure on load sharing with simulated bone ingrowth: A finite element analysis comparing titanium and PEEK. J Mech Behav Biomed Mater. 2018;80:68-76. [43] YOON BJ, XAVIER F, WALKER BR, et al. Optimizing surface characteristics for cell adhesion and proliferation on titanium plasma spray coatings on polyetheretherketone. Spine J. 2016;16(10):1238-1243. [44] 杨接来,徐俊,谷辉杰,等. 3D打印聚乳酸/纳米级 -磷酸钙可吸收山羊颈椎融合器的生物相容性及生物力学评价[J].中国临床医学,2017,24(4):525-530. [45] CAO L, CHEN Q, JIANG LB, et al. Bioabsorbable self-retaining PLA/nano-sized beta-TCP cervical spine interbody fusion cage in goat models: an in vivo study. Int J Nanomedicine. 2017;12:7197-7205. [46] BRENKE C, KINDLING S, SCHARF J, et al. Short-term experience with a new absorbable composite cage (β-tricalcium phosphate-polylactic acid) in patients after stand-alone anterior cervical discectomy and fusion. Spine (Phila Pa 1976). 2013;38(11):E635-E640. [47] YANG X, SONG Y, LIU L, et al. Mid-term effectiveness of anterior decompression and fusion with nano-hydroxyapatite/polyamide 66 cage in treatment of cervical spondylotic myelopathy. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2013;27(9):1085-1089. [48] LI S, ZHANG H, SHEN Y, et al. Risk factors of cage subsidence after removal of localized heterotopic ossification by anterior cervical discectomy and fusion: A retrospective multivariable analysis. Medicine (Baltimore). 2019;98(42):e17505. [49] KIM MC, CHUNG HT, CHO JL, et al. Subsidence of polyetheretherketone cage after minimally invasive transforaminal lumbar interbody fusion. J Spinal Disord Tech. 2013;26(2):87-92. [50] CHOI MK, KIM SB, LEE JH. Rare Intractable Cervicalgia Related to Exaggerated Disc Height Distraction : Report of Two Cases and Literature Review. J Korean Neurosurg Soc. 2018;61(4):530-536. [51] WANG S, ZHU J, SUN K, et al. Clinical impact and imaging results after a modified procedure of ACDF: a prospective case-controlled study based on ninety cases with two-year follow-up. BMC Musculoskelet Disord. 2021;22(1):605. [52] 宋萌,朱灏宇,朱庆三,等.单节段颈椎椎体次全切除钛笼植骨融合术后钛笼下沉相关危险因素分析[J].中国脊柱脊髓杂志,2017, 27(8):681-685. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Wu Tianliang, Tao Xiuxia, Xu Hongguang. Influence of different bone mineral densities on cage subsidence after stand-alone oblique lateral interbody fusion: three-dimensional finite element analysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1352-1358. |
| [3] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [4] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [5] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [6] | Wang Yanjin, Zhou Yingjie, Chai Xubin, Zhuo Hanjie. Meta-analysis of the efficacy and safety of 3D printed porous titanium alloy fusion cage in anterior cervical discectomy and fusion [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1434-1440. |
| [7] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [8] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [9] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [10] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [11] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [12] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [13] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [14] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [15] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||