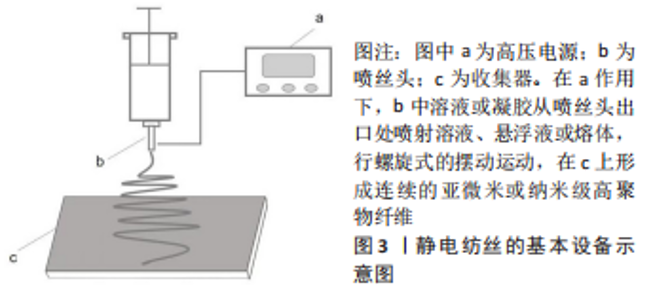
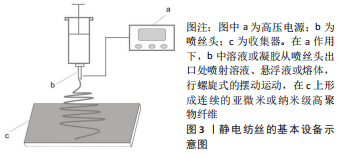
Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (34): 5554-5560.doi: 10.12307/2023.502
Previous Articles Next Articles
Application of platelet-rich plasma combined with electrospun nanoscaffolds in bone and soft tissue
Zhao Mingyue1, Yang Shun1, Tu Xiling1, Gao Li2, Yang Kun2, Liu Qi2
- 1Zunyi Medical University, Zunyi 563000, Guizhou Province, China; 2Department of Periodontics, Affiliated Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China
-
Received:2022-06-28Accepted:2022-08-22Online:2023-12-08Published:2023-04-23 -
Contact:Liu Qi, PhD, Professor, Department of Periodontics, Affiliated Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China ang Kun, PhD, Master’s supervisor, Associate professor, Department of Periodontics, Affiliated Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China Gao Li, Master, Master’s supervisor, Associate professor, Department of Periodontics, Affiliated Stomatological Hospital of Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
About author:Zhao Mingyue, Master candidate, Zunyi Medical University, Zunyi 563000, Guizhou Province, China -
Supported by:National Natural Science Foundation of China, No. 82060204 (to GL); Municipal Key Project, No. 15851 (to LQ); Guizhou Provincial Health Commission Science and Technology Fund Project, No. gzwkj2022-169 (to YK); Science and Technology Fund Program of Guizhou Province, No. [2020]1Y328 (to YK)
CLC Number:
Cite this article
Zhao Mingyue, Yang Shun, Tu Xiling, Gao Li, Yang Kun, Liu Qi. Application of platelet-rich plasma combined with electrospun nanoscaffolds in bone and soft tissue[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(34): 5554-5560.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks

近年来,通过静电纺丝工艺制备出的纳米纤维支架由平均直径50-500 nm的纤维组成。静电纺丝在组织工程支架的优势主要有:①直径与天然细胞外基质(extracellular matrixc,ECM)相近,能最大程度地模拟人体内天然细胞外基质环境;②电纺丝材料可以是多种聚合物的复合体,甚至可以引入生长因子、细胞调控因子、药物及活细胞等;③制备的支架具有较高的孔隙率和极大的比表面积,为细胞的黏附、增殖和分化等提供了良好的条件;④通过材料的选择和各项参数的调节,可以很好地控制支架的直径、三维结构和力学性能,并通过对材料表面的修饰从而提高支架的生物相容性[14]。 2.3 静电纺丝纳米纤维支架的分类 静电纺丝纳米支架主要分为天然聚合物和合成聚合物[15]: 2.3.1 天然聚合物 由机体组织天然产生,具有良好的生物相容性;缺点则是降解速率较快、机械强度较差,以及不同天然聚合物之间不同的浓度、溶解体系难以控制[16-19]。目前,临床上应用比较常见的有: 水凝胶(hydrogel):为类似于细胞外基质的三维结构,具有对水、氧、生物液及代谢物高渗透的能力,并可维持生长因子释放等[20-22],其缺点是强度低、韧性差和吸水速度慢。 明胶(gelatin,Gel):是一种从动物结缔组织中提取的胶原蛋白(collagen,COL)类似物,可以促进软组织和骨骼的愈合。具有生物可降解性、良好的生物相容性、凝胶性和低成本等优点[23-25];缺点为机械性能、防潮、抗湿及抗菌方面性能的较低。 透明质酸(hyaluronic acid):具有独特的黏弹性、良好的生物相容性和生物降解性,以及强亲水性,可提供适合组织生长的湿润环境。此外,透明质酸还可以通过促进间充质干细胞的增殖使伤口内胶原蛋白的含量不断增加 [26-29],缺点为容易降解。 壳聚糖(chitosan):是广泛存在于甲壳类动物体内的一种动物性纤维素,因具有生物降解性、生物相容性、无毒性、抑菌、抗癌、降脂、增强免疫等多种生理功能,可以用作临时支架来修饰和刺激新组织的生长[30],但不溶于水。 丝素蛋白(silk fibroin,SF):又名蚕丝蛋白,是从蚕丝中提取的天然高分子纤维蛋白,具有优良的机械性能和生物降解性能,以及无毒、无免疫原性[31],但不溶于水。 锶(Sr2+):可通过涂覆或掺杂Sr2+对生物材料进行改性,以提高材料的成骨潜力,促进成骨细胞分化和降低破骨细胞活性,从而促进骨形成[32];目前相关研究较为局限,有待进一步研究锶在静电纺丝作用下产生的效果。 2.3.2 合成无毒聚合物 此类材料可以设计纤维支架的特性、超细直径、孔径和机械性能,常见的合成聚合物有: 聚己内酯(polycaprolactone,PCL):无毒,不溶于水,易溶于多种极性有机溶剂。具有良好的生物相容性、生物降解性、有机高聚物相容性和形状记忆温控性[33-35],但力学性能不佳。 羟基磷灰石(Hydroxyapatite):是人体和动物骨骼的主要无机成分。在体内有一定的溶解度,能释放对机体无害的离子,能参与体内代谢,对骨质增生有刺激或诱导作用,促进缺损组织的修复[36],缺点为缺乏结构的完整性和力学稳定性。 聚偏氟乙烯(polyvinylidene fluoride,PVDF):具有良好的耐化学腐蚀性、耐高温性、耐氧化性、耐射线辐射性能、压电性、介电性及热电性等特殊性能[37]。 聚(乙烯醇)(polyvinyl alcohol,vinylalcohol polymer,PVA):对人体无毒,无副反应,具有良好的生物相容性[38],不过耐水性相对较差。 β-磷酸三钙(β-tricalcium phosphate,β-TCP):具有良好的生物降解性、生物相容性和生物无毒性[39]。 天然聚合物和合成聚合物各具优缺点,许多关于骨、软骨组织及软组织再生的研究都致力于开发将天然聚合物的细胞相容性与合成聚合物的机械和物理化学性相结合的支架;却存在着降解速度与骨成长不相匹配的缺点。 作者总结了静电纺丝纳米纤维支架的分类情况,见表2。"

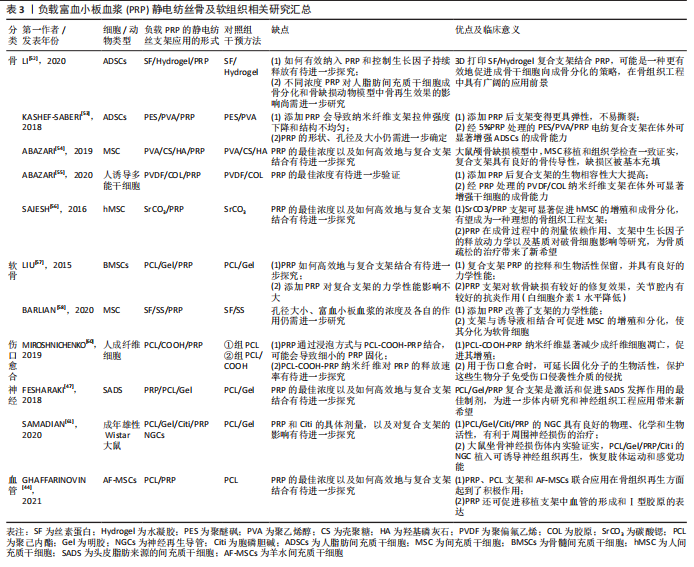
2.4 负载富血小板血浆的静电纺丝纳米支架的性能 静电纺丝纳米复合纤维作为组织工程材料具有广泛而重要的生物学特性,通过改变纳米纤维孔径及材料组分可获得良好的机械可塑性、骨诱导性、生物相容性、抗菌性和降解速度的可控性等。富血小板血浆富含多种生长因子,可促进组织再生、骨折愈合、瘢痕修复等,因而在临床上得到了广泛的应用[40-43]。单独使用富血小板血浆支架或静电纺丝纳米纤维支架均存在或多或少的不足,其中单独使用静电纺丝纳米支架存在骨诱导性不强、促细胞生长分化能力弱等缺点;而单独使用富血小板血浆又存在降解性慢、机械性可塑性较差、无法调控合适的孔径和孔隙率等问题,所以将富血小板血浆负载于静电纺丝纳米复合支架上应用于组织工程,彼此弥补自身缺点,同时更具临床应用前景。 通过对负载富血小板血浆的聚己内酯纤维纳米支架体外培养羊水间充质干细胞的研究证实,羊水间充质干细胞能够很好地黏附于复合支架表面,并且随着时间的推移,细胞黏附率增加,同时对骨组织再生具有积极推动作用,表明负载富血小板血浆静电纺丝纳米支架具有良好的骨诱导性,有利于细胞的黏附和生长[44]。此外,负载富血小板血浆的静电纺丝纳米支架具有良好细胞相容性,能为细胞的生长提供良好的微环境。PRABHAKARAN等[45]通过把人骨髓间充质干细胞培养于负载富血小板血浆的电纺聚(乳酸)-聚(?-己内酯)/胶原纳米纤维支架上,研究表明负载富血小板血浆的复合纳米纤维支架对神经元细胞增殖、分化的促进作用均优于单纯聚(乳酸)-聚(?-己内酯)/胶原纳米纤维组。YIN等[46]通过对负载血小板血浆的丝素蛋白/聚乳酸-聚己内酯的纳米纤维支架材料的研究发现,它在拉伸强度、抗压力等方面具有与天然组织相当的力学性能,这充分证明了负载富血小板血浆静电纺丝纳米纤维良好的机械性能。同时负载富血小板血浆的静电纺丝纳米纤维支架也具有优良的的降解性,且可以对细胞提供良好的微环境,此外,FESHARAKI等 [47]通过静电纺丝机制备出负载富血小板血浆的PCL/Gel纳米复合支架,他们的研究进一步证实,复合支架在力学性能、生物降解率、细胞黏附和代谢产物运输方面均具备更优异的性能。 综上所述,负载富血小板血浆的静电纺丝纳米支架兼顾两者的优点,为细胞的生长提供良好的微环境,具有良好机械可塑性、骨诱导性,利于细胞的黏附和生长,促进新骨向材料内部的长入,以及营养成分的运输和代谢产物的排出。 2.5 负载富血小板血浆的静电纺丝纳米复合支架在骨及软骨再生医学领域的应用 2.5.1 骨组织的修复 骨是人体的重要组成部分,主要起支撑作用,当骨组织受到较大损害时,就会产生不可逆性损伤。目前自体或同种异体骨移植被认为是骨骼重建的“金标准”[48-49]。然而,这些治疗方法存在一定的缺点,如自体骨移植受制于取骨量的限制,可能会导致供骨部位的严重并发症;而同种异体骨移植存在多种并发症如感染、移植物与宿主组织难以融合等。因此,亟需更可靠、稳定的治疗方法来克服骨组织再生的难题。而负载富血小板血浆的静电纺丝纳米复合支架中的富血小板血浆可在静电纺丝复合支架上形成交联的网状结构,因而能更好地模仿细胞外基质结构,更好地促进细胞的生长、增殖和分化[50- 51],已经成为近年来骨组织再生修复方面的研究热点。 单独使用天然聚合物或合成聚合物制备支架均存在或多或少的不足,而按照一定比例共混并通过电纺技术加工后形成一种复合支架,不仅具有良好的相容性和可生物降解性的,还具有与目标组织相似的机械性能[47]。并且有时为了进一步增加支架的促进组织愈合力,常常在负载富血小板血浆的静电纺丝纳米支架基础上制作成3D复合支架。LI等[52]将富血小板血浆与丝素蛋白-水凝胶(Silk Fibroin/Hydrogel,SF/Hydrogel)电纺成一种新型的复合3D纳米支架,研究发现SF/Hydrogel/富血小板血浆与单纯的SF/Hydrogel相比,得益于富血小板血浆在SF/Hydrogel支架中形成的血小板网络交联结构能持续释放生长因子,SF/Hydrogel/富血小板血浆复合3D纳米支架对细胞的生长、增殖和分化产生更明显的作用;并且通过聚合酶链式反应进一步证实其成骨基因骨钙素和骨保护素的表达明显高于对照组的单纯3D复合支架,表明负载富血小板血浆的3D复合支架对细胞的成骨性、生物相容性均产生积极的影响。有研究则将聚醚砜/聚乙烯醇/富血小板血浆(polyethersulfone/polyvinyl alcohol/platelet-rich plasma,PES/PVA/PRP)复合支架用于体外诱导人脂肪间充质干细胞成骨分化,研究证实该复合支架在促进细胞增殖的同时,也会产生相对合适的网架,不仅对细胞渗透产生积极影响,也对成骨的诱导具有积极的作用,促进了细胞的黏附。由此可见,负载富血小板血浆的静电纺丝纳米支架也具有优良的骨诱导性和生物相容性[53]。 ABAZARI等[54]将富血小板血浆与聚乙烯醇/壳聚糖/羟基磷灰石(polyethersulfone/Chitosan/Hydroxyapatite/platelet-rich plasma,PVA/CS/HA)复合支架植入大鼠颅骨缺损模型中,第8周以后,通过多层螺旋CT和苏木精-伊红染色实验显示,虽然单纯植入的PVA/CS/HA复合支架缺损部位可以检测到骨再生,但是植入PVA/CS/HA/富血小板血浆复合支架后骨缺损几乎完全再生,这表明负载富血小板血浆的静电纺丝纳米支架可代替骨移植用于临床,促进骨组织再生。还有研究通过比较单一的负载富血小板血浆的静电纺丝支架对羊水间充质干细胞分化的影响,发现聚己内酯/富血小板血浆(Polycaprolactone/Platelet Rich Plasma,PCL/PRP)纳米纤维膜支架能够增强羊水间充质干细胞成骨分化和增殖的能力,表明该负载富血小板血浆的静电纺丝支架适合骨组织工程[44]。在骨修复治疗中负载富血小板血浆的静电纺丝支架不总是单一的,静电纺丝常以不同的形式及复合多种生物材料与富血小板血浆联合应用于实验研究。ABAZARI等[55]将聚偏氟乙烯/胶原(polyvinylidene fluoride/collagen,PVDF/COL)与富血小板血浆联合作为实验组,结果表明,与PVDF/COL相比,PVDF/COL/富血小板血浆具有更强的骨诱导活性,而且由于富血小板血浆富含多种生长因子,提高了支架的生物相容性,并支持干细胞的黏附、生长和增殖,因此可作为一种很有前途的骨组织工程骨植入材料。此外,SAJESH等[56]采用逐层静电纺丝技术制备出负载富血小板血浆的SrCO3 (nano strontium carbonate)/聚(3-羟基丁酸-co-3-羟基戊酸盐)(3-hydroxybutyrate-co-3-hydroxyvalerate,PHBV) 纤维支架,通过测量负载富血小板血浆的SrCO3/PHBV纤维支架上的碱性磷酸酶浓度和矿物质沉积证实了人骨髓间充质干细胞显著增强的成骨分化,并推测静电纺丝复合支架可以通过调控富血小板血浆生长因子浓度及成分来提高生物活性,以上发现为该复合支架在临床骨缺损修复应用提供了可靠的依据与方向,不过对于如何有效纳入富血小板血浆和控制生长因子持续释放以及对不同富血小板血浆浓度对干细胞的成骨分化和骨缺损动物模型植入后再生功效的影响有待进一步研究。 2.5.2 软骨组织的修复 由于负载富血小板血浆的静电纺丝纳米支架能够为细胞黏附和增殖提供良好的环境,因此该复合支架在修复软骨方面同样显示出了良好的疗效。LIU等[57]将富血小板血浆负载于聚己内酯/明胶(Polycaprolactone/gelatin,PCL/Gel)支架纳米纤维上,发现兔骨髓间充质干细胞能增殖、黏附在PCL/Gel/富血小板血浆复合支架上,且通过计算得到的PCL/Gel/富血小板血浆纳米纤维支架的杨氏模数为(8.11±1.05) Mpa,与人关节软骨的杨氏模数(1-10 Mpa)和牛关节软骨的平衡拉伸模数(2-7 Mpa)相当,表明PCL/ Gel/富血小板血浆纳米纤维支架可作为代替合成软骨工程的细胞支架。此外,作者使用36只兔的膝关节建立兔膝关节软骨缺损模型,在术后6周,大体观可以明显看到富血小板血浆复合支架覆盖的缺损区呈凹陷状;1个月后,再生组织表面较粗糙,边缘与正常软骨组织间有缝隙,在术后第12周时,再生组织则几乎填满了整个缺损区,且如正常关节表面平坦。由此可得出,PCL/Gel/富血小板血浆纳米纤维支架适合作为软骨修复的组织工程支架。同样,BARLIAN等[58]通过将间充质干细胞种植在丝素蛋白-蚕丝蛋白-富小血版血浆(silk fibroin -silk spidroin-platelet rich plasma,SF-SS-PRP)支架上的研究发现,与单纯的SF-SS纳米支架相比,SF-SS-PRP更能促进间充质干细胞的软骨分化,骨髓间充质干细胞能很好地在支架上增殖分化,软骨缺损几乎被修复,这充分表明SF-SS-PRP支架能够作为骨髓间充质干细胞的载体用于治疗软骨损伤。 2.6 软组织的修复 2.6.1 伤口愈合 创伤的愈合可有2种方式:一种是一期愈合或直接愈合,另一种是二期愈合或间接愈合,去除感染和坏死物质,较大的伤口可以行自体皮肤移植。富血小板血浆中的血小板被激活后可释放出多种协同作用的生长因子,促进局部修复细胞的增殖分化及细胞外基质的合成,从而增强了组织再生和修复能力。而静电纺丝材料作为一种新型的功能性敷料,可调控材料的孔隙率、表面积,具有良好的生物学功能,既有益于细胞呼吸,又可以抑制伤口感染,因此负载富血小板血浆的静电纺丝纳米支架是一种适用于伤口愈合的良好生物医学材料[59]。MIROSHNICHENKO等[60]通过将成纤维细胞种植在负载富血小板血浆的PCL-COOH支架上,在干预7 d后,支架上的成纤维细胞凋亡水平降至(6.0±0.9)%,同时保持(7.2±1.3)%的细胞增殖活性,说明PCL-COOH-富血小板血浆对于维持成纤维细胞的功能活力是十分有效的,可促进细胞增殖,从而进一步提高伤口愈合率。 2.6.2 神经损伤修复 由于脑外伤、脑血管硬化(脑出血和脑血栓)及脑炎等原因容易导致神经损伤,其轴突的不可再生性使神经的再生修复成为一大难题。因此通过利用生物组织工程支架治疗脊髓神经损伤成为近年来的研究热点。FESHARAKI团队[47]通过研究人头皮脂肪来源的间充质干细胞对富血小板血浆/PCL/Gel纳米纤维支架的黏附特性、增殖情况及形态,发现与PCL/Gel纳米支架相比富血小板血浆/PCL/Gel纳米纤维支架效果最好,从而为利用组织工程方法修复神经组织工程中的应用提供了基础。此外,SAMADIAN等[61]发现PCL/Gel/Citi/富血小板血浆纳米纤维神经导管对人头皮脂肪来源的间充质干细胞具有良好的力学和生物学特性,可以促进神经细胞的生长,并可促进受损的神经部分功能恢复,再次说明了负载富血小板血浆的静电纺丝纳米支架在神经组织工程中的应用价值。 2.6.3 血管组织损伤 目前治疗性血管生成的方法包括使用血管生成生长因子、细胞或两者的组合,其广阔的生物医学应用价值已在许多体外实验和临床缺血环境中得到证实[62]。然而,新生微血管的不稳定性和组织易损伤性已成为其临床推广使用的主要障碍。因此,治疗性血管生成的许多策略都集中在综合方法上,其中包括血管生成生长因子、细胞与模拟细胞外基质的三维支架的结合。负载富血小板血浆的静电纺丝纳米支架已被证实可修复血管组织损伤,GHAFFARINOVIN等[44]在大鼠的顶骨的双侧形成圆形全层骨缺损,并将羊水间充质干细胞附着在PCL/富血小板血浆的支架植入右侧病灶中,在第8周,研究结果发现,与没有富血小板血浆支架组相比,所有PCL/富血小板血浆支架组不但骨缺损修复明显,且都形成了血管,表明负载富血小板血浆的静电纺丝纳米支架能够促进骨组织和血管修复。 作者对负载富血小板血浆静电纺丝骨及软组织相关研究作一汇总,见表3。"
| [1] PIGEOT S, KLEIN T, GULLOTTA F, et al. Manufacturing of human tissues as off-the-shelf grafts programmed to induce regeneration. Adv Mater. 2021;33(43):e2103737. [2] MAIA FR, BASTOS AR, OLIVEIRA JM, et al. Recent approaches towards bone tissue engineering. Bone. 2022;154:116256. [3] ATALA A. Bioengineered tissues for urogenital repair in children. Pediatr Res. 2008;63(5):569-575. [4] ELGHBLAWI E. Platelet-rich plasma, the ultimate secret for youthful skin elixir and hair growth triggering. J Cosmet Dermatol. 2018;17(3):423-430. [5] SARKAR R, GUPTA M. Platelet-rich plasma in melasma-a systematic review. Dermatol Surg. 2022;48(1):131-134. [6] WU J, PIAO Y, LIU Q, et al. Platelet-rich plasma-derived extracellular vesicles: a superior alternative in regenerative medicine? Cell Prolif. 2021;54(12):e13123. [7] BELK JW, KRAEUTLER MJ, HOUCK DA, et al. Platelet-rich plasma versus hyaluronic acid for knee osteoarthritis: a systematic review and meta-analysis of randomized controlled trials. Am J Sports Med. 2021;49(1):249-260. [8] KONWARH R, KARAK N, MISRA M. Electrospun cellulose acetate nanofibers: the present status and gamut of biotechnological applications. Biotechnol Adv. 2013;31(4):421-437. [9] FOSTER TE, PUSKAS BL, MANDELBAUM BR, et al. Platelet-rich plasma: from basic science to clinical applications. Am J Sports Med. 2009;37(11):2259-2272. [10] ZHANG X, WANG J, REN M, et al. A novel collagen/platelet-rich plasma (COL/PRP) scaffold: preparation and growth factor release analysis. Cell Tissue Bank. 2016;17(2):327-334. [11] OUDELAAR BW, PEERBOOMS JC, HUIS IN’T VELD R, et al. Concentrations of blood components in commercial platelet-rich plasma separation systems: a review of the literature. Am J Sports Med. 2019;47(2):479-487. [12] KIM JI, BAE HC, PARK HJ, et al. Effect of Storage conditions and activation on growth factor concentration in platelet-rich plasma. J Orthop Res. 2020;38(4):777-784. [13] SHAHRIAR SMS, MONDAL J, HASAN MN, et al. Electrospinning nanofibers for therapeutics delivery. Nanomaterials (Basel). 2019;9(4):532. [14] CHAHAL S, KUMAR A, HUSSIAN FSJ. Development of biomimetic electrospun polymeric biomaterials for bone tissue engineering: a review. J Biomater Sci Polym Ed. 2019;30(14):1308-1355. [15] BHATTARAI DP, AGUILAR LE, PARK CH, et al. A review on properties of natural and synthetic based electrospun fibrous materials for bone tissue engineering. Membranes (Basel). 2018;8(3):62. [16] ZHANG CL, YU SH. Nanoparticles meet electrospinning: recent advances and future prospects. Chem Soc Rev. 2014;43(13):4423-4448. [17] ASTI A, GIOGLIO L. Natural and synthetic biodegradable polymers: different scaffolds for cell expansion and tissue formation. Int J Artif Organs. 2014;37(3):187-205. [18] LI WJ, COOPER JA JR, MAUCK RL, et al. Fabrication and characterization of six electrospun poly(alpha-hydroxy ester)-based fibrous scaffolds for tissue engineering applications. Acta Biomater. 2006;2(4):377-385. [19] YILDIRIMER L, THANH NT, SEIFALIAN AM. Skin regeneration scaffolds: a multimodal bottom-up approach. Trends Biotechnol. 2012;30(12):638-648. [20] ASKARZADEH N, NAZARPAK MH, MANSOORI K, et al. Bilayer cylindrical conduit consisting of electrospun polycaprolactone nanofibers and DSC cross-linked sodium alginate hydrogel to bridge peripheral nerve gaps. Macromol Biosci. 2020;20(9):e2000149. [21] XI-XUN Y, FEI L, YUAN-TING X, et al. In vitro study in the endothelial cell compatibility and endothelialization of genipin-crosslinked biological tissues for tissue-engineered vascular scaffolds. J Mater Sci Mater Med. 2010;21(2):777-785. [22] SONG JY, RYU HI, LEE JM, et al. Conformal fabrication of an electrospun nanofiber mat on a 3D ear cartilage-shaped hydrogel collector based on hydrogel-assisted electrospinning. Nanoscale Res Lett. 2021;16(1):116. [23] 高婷婷,常丹,刘凤岐,等.有机复合水凝胶的研究进展[J].功能材料,2016, 47(S1):72-77. [24] LIU X, ZU E, CHANG X, et al. Bi-phasic effect of gelatin in myogenesis and skeletal muscle regeneration. Dis Model Mech. 2021;14(12):dmm049290. [25] 卢伟鹏,张兵,郭燕川.明胶静电纺丝的研究进展[J].明胶科学与技术,2013, 33(1):8-14. [26] BARDONOVA L, KOTZIANOVA A, SKUHROVCOVA K, et al. Effects of emulsion, dispersion, and blend electrospinning on hyaluronic acid nanofibers with incorporated antiseptics. Int J Biol Macromol. 2022;194:726-735. [27] CASTRO KC, CAMPOS MGN, MEI LHI. Hyaluronic acid electrospinning: challenges, applications in wound dressings and new perspectives. Int J Biol Macromol. 2021;173:251-266. [28] NIU Y, STADLER FJ, FANG J, et al. Hyaluronic acid-functionalized poly-lactic acid (PLA) microfibers regulate vascular endothelial cell proliferation and phenotypic shape expression. Colloids Surf B Biointerfaces. 2021;206:111970. [29] SNETKOV P, MOROZKINA S, OLEKHNOVICH R, et al. Curcumin/usnic acid-loaded electrospun nanofibers based on hyaluronic acid. Materials (Basel). 2020;13(16):3476. [30] KERSANI D, MOUGIN J, LOPEZ M, et al. Stent coating by electrospinning with chitosan/poly-cyclodextrin based nanofibers loaded with simvastatin for restenosis prevention. Eur J Pharm Biopharm. 2020;150:156-167. [31] PINA S, CANADAS RF, JIMENEZ G, et al. Biofunctional ionic-doped calcium phosphates: silk fibroin composites for bone tissue engineering scaffolding. Cells Tissues Organs. 2017;204(3-4):150-163. [32] TSAI SW, YU WX, HWANG PA, et al. Fabrication and characteristics of pcl membranes containing strontium-substituted hydroxyapatite nanofibers for guided bone regeneration. Polymers (Basel). 2019;11(11):1761. [33] DAELEMANS L, STEYAERT I, SCHOOLAERT E, et al. Nanostructured hydrogels by blend electrospinning of polycaprolactone/gelatin nanofibers. Nanomaterials (Basel). 2018;8(7):551. [34] QIAN Y, CHEN H, XU Y, et al. The preosteoblast response of electrospinning PLGA/PCL nanofibers:effects of biomimetic architecture and collagen I. Int J Nanomedicine. 2016;11:4157-4171. [35] EOM J, KWAK Y, NAM C. Electrospinning fabrication of magnetic nanoparticles-embedded polycaprolactone (PCL) sorbent with enhanced sorption capacity and recovery speed for spilled oil removal. Chemosphere. 2022;303(Pt 3):135063. [36] YOSHIKAWA H, TAMAI N, MURASE T, et al. Interconnected porous hydroxyapatite ceramics for bone tissue engineering. J R Soc Interface. 2009;6 Suppl 3:S341-S348. [37] KALIMULDINA G, TURDAKYN N, ABAY I, et al. A review of piezoelectric pvdf film by electrospinning and its applications. Sensors (Basel). 2020;20(18):5214. [38] WU JY, WANG CY, CHEN KH, et al. Electrospinning of quaternized chitosan-poly(vinyl alcohol) composite nanofiber membrane: processing optimization and antibacterial efficacy. Membranes (Basel). 2022;12(3):332. [39] DOS SANTOS VI, MERLINI C, ARAGONES Á, et al. In vitro evaluation of bilayer membranes of PLGA/hydroxyapatite/β-tricalcium phosphate for guided bone regeneration. Mater Sci Eng C Mater Biol Appl. 2020;112:110849. [40] MIJIRITSKY E, ASSAF HD, PELEG O, et al. Use of PRP, PRF and CGF in periodontal regeneration and facial rejuvenation-a narrative review. Biology (Basel). 2021;10(4):317. [41] CHENG G, MA X, LI J, et al. Incorporating platelet-rich plasma into coaxial electrospun nanofibers for bone tissue engineering. Int J Pharm. 2018;547(1-2):656-666. [42] VOCETKOVA K, SOVKOVA V, BUZGO M, et al. A simple drug delivery system for platelet-derived bioactive molecules, to improve melanocyte stimulation in vitiligo treatment. Nanomaterials (Basel). 2020;10(9):1801. [43] LANA JFSD, DA FONSECA LF, MACEDO RDR, et al. Platelet-rich plasma vs bone marrow aspirate concentrate:An overview of mechanisms of action and orthobiologic synergistic effects. World J Stem Cells. 2021;13(2):155-167. [44] GHAFFARINOVIN Z, SOLTANINIA O, MORTAZAVI Y, et al. Repair of rat cranial bone defect by using amniotic fluid-derived mesenchymal stem cells in polycaprolactone fibrous scaffolds and platelet-rich plasma. Bioimpacts. 2021;11(3):209-217. [45] PRABHAKARAN MP, VENUGOPAL JR, RAMAKRISHNA S. Mesenchymal stem cell differentiation to neuronal cells on electrospun nanofibrous substrates for nerve tissue engineering. Biomaterials. 2009;30(28):4996-5003. [46] YIN A, BOWLIN GL, LUO R, et al. Electrospun silk fibroin/poly (L-lactide-ε-caplacton) graft with platelet-rich growth factor for inducing smooth muscle cell growth and infiltration. Regen Biomater. 2016;3(4):239-245. [47] FESHARAKI M, RAZAVI S, GHASEMI-MOBARAKEH L, et al. Differentiation of human scalp adipose-derived mesenchymal stem cells into mature neural cells on electrospun nanofibrous scaffolds for nerve tissue engineering applications. Cell J. 2018;20(2):168-176. [48] BHUMIRATANA S, BERNHARD JC, ALFI DM, et al. Tissue-engineered autologous grafts for facial bone reconstruction. Sci Transl Med. 2016;8(343):343ra83. [49] LEE JW, HAN HS, HAN KJ, et al. Long-term clinical study and multiscale analysis of in vivo biodegradation mechanism of Mg alloy. Proc Natl Acad Sci U S A. 2016; 113(3):716-721. [50] Koç S, Çakmak S, Gümüşderelioğlu M, et al. Three dimensional nanofibrous and compressible poly(L-lactic acid) bone grafts loaded with platelet-rich plasma. Biomed Mater. 2021;16(4):045012. [51] ZHAO C, TAN A, PASTORIN G, et al. Nanomaterial scaffolds for stem cell proliferation and differentiation in tissue engineering. Biotechnol Adv. 2013; 31(5):654-668. [52] LI Z, ZHANG X, YUAN T, et al. Addition of platelet-rich plasma to silk fibroin hydrogel bioprinting for cartilage regeneration. Tissue Eng Part A. 2020;26(15-16):886-895. [53] KASHEF-SABERI MS, HAYATI RN, PARIVAR K, et al. Enhanced osteogenic differentiation of mesenchymal stem cells on electrospun polyethersulfone/poly (vinyl) alcohol/platelet rich plasma nanofibrous scaffolds. ASAIO J. 2018;64(5):e115-e122. [54] ABAZARI MF, NEJATI F, NASIRI N, et al. Platelet-rich plasma incorporated electrospun PVA-chitosan-HA nanofibers accelerates osteogenic differentiation and bone reconstruction. Gene. 2019;720:144096. [55] ABAZARI MF, SOLEIMANIFAR F, AMINI F, et al. Improved osteogenic differentiation of human induced pluripotent stem cells cultured on polyvinylidene fluoride/collagen/platelet-rich plasma composite nanofibers. J Cell Physiol. 2020;235(2):1155-1164. [56] SAJESH KM, KIRAN K, NAIR SV, et al. Sequential layer-by-layer electrospinning of nano SrCO3/PRP loaded PHBV fibrous scaffold for bone tissue engineering. Compos B Eng. 2016;99:445-452. [57] LIU J, NIE H, XU Z, et al. Construction of PRP-containing nanofibrous scaffolds for controlled release and their application to cartilage regeneration. J Mater Chem B. 2015;3(4):581-591. [58] BARLIAN A, JUDAWISASTRA H, ALFARAFISA NM, et al. Chondrogenic differentiation of adipose-derived mesenchymal stem cells induced by L-ascorbic acid and platelet rich plasma on silk fibroin scaffold. Sci Rep. 2020;10(1):19449. [59] QIU M, CHEN D, SHEN C, et al. Platelet-rich plasma-loaded poly (d,l-lactide)-poly (ethylene glycol)-poly (d, l-lactide) hydrogel dressing promotes full-thickness skin wound healing in a rodent model. Int J Mol Sci. 2016;17(7):1001. [60] MIROSHNICHENKO S, TIMOFEEVA V, PERMYKOVA E, et al. Plasma-coated polycaprolactone nanofibers with covalently bonded platelet-rich plasma enhance adhesion and growth of human fibroblasts. Nanomaterials (Basel). 2019;9(4):637. [61] SAMADIAN H, EHTERAMI A, SARRAFZADEH A, et al. Sophisticated polycaprolactone/gelatin nanofibrous nerve guided conduit containing platelet-rich plasma and citicoline for peripheral nerve regeneration:In vitro and in vivo study. Int J Biol Macromol. 2020;150:380-388. [62] WU D, LIU J, PANG X, et al. Palmitic acid exerts pro-inflammatory effects on vascular smooth muscle cells by inducing the expression of C-reactive protein, inducible nitric oxide synthase and tumor necrosis factor-α. Int J Mol Med. 2014;34(6):1706-1712. [63] WANG K, LIU L, XIE J, et al. Facile strategy to generate aligned polymer nanofibers:effects on cell adhesion. ACS Appl Mater Interfaces. 2018;10(2):1566-1574. [64] JIN G, HE R, SHA B, et al. Electrospun three-dimensional aligned nanofibrous scaffolds for tissue engineering. Mater Sci Eng C Mater Biol Appl. 2018;92:995-1005. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [3] | Zheng Hongrui, Zhang Wenjie, Wang Yunhua, He Bin, Shen Yajun, Fan Lei. Femoral neck system combined with platelet-rich plasma in the treatment of femoral neck fracture [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1390-1395. |
| [4] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [5] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [6] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [7] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [8] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [9] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [10] | Lian Shilin, Zhang Yan, Jiang Qiang, Zhang Hanshuo, Li Tusheng, Ding Yu. Interventional effects of whole blood and platelet-rich plasma with different preparation methods on nucleus pulposus cells [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1199-1204. |
| [11] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [12] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [13] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [14] | Xu Cong, Zhao He, Sun Yan. Regeneration of facial nerve injury repaired by biomaterial nerve conduits [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1089-1095. |
| [15] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||