Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (29): 4729-4735.doi: 10.12307/2023.489
Previous Articles Next Articles
Long non-coding RNAs regulate osteoarthritis by mediating chondrocyte-related mechanisms
Chen Cai1, Zeng Ping2, Liu Jinfu2
- 1Graduate School of Guangxi University of Chinese Medicine, Nanning 530299, Guangxi Zhuang Autonomous Region, China; 2The First Affiliated Hospital of Guangxi University of Chinese Medicine, Nanning 530023, Guangxi Zhuang Autonomous Region, China
-
Received:2022-07-13Accepted:2022-08-19Online:2023-10-18Published:2022-12-02 -
Contact:Zeng Ping, MD, Professor, Doctoral supervisor, Chief physician, The First Affiliated Hospital of Guangxi University of Chinese Medicine, Nanning 530023, Guangxi Zhuang Autonomous Region, China -
About author:Chen Cai, Master candidate, Graduate School of Guangxi University of Chinese Medicine, Nanning 530299, Guangxi Zhuang Autonomous Region, China -
Supported by:the National Natural Science Foundation of China, Nos. 81960876 and 82160913 (both to ZP); Guangxi Traditional Chinese Medicine Appropriate Technology Development and Promotion Project, No. GZSY21-14 (to ZP)
CLC Number:
Cite this article
Chen Cai, Zeng Ping, Liu Jinfu. Long non-coding RNAs regulate osteoarthritis by mediating chondrocyte-related mechanisms[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(29): 4729-4735.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
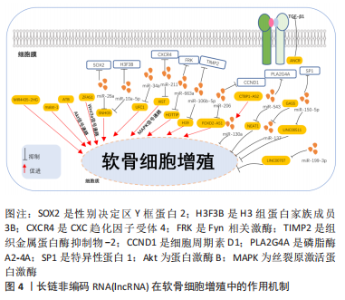
2.1 lncRNA在骨性关节炎软骨细胞增殖中的作用 软骨细胞合成胶原纤维和蛋白多糖等基质成分, 是形成和维持软骨的重要细胞[11]。正常成人关节软骨中的软骨细胞是稳定的、分化的细胞,通过合成极低水平的细胞外基质来替换受损的基质分子,从而维持组织稳态,保持软骨基质的结构完整性。当发生骨性关节炎时,软骨细胞合成与分解代谢之间的不平衡以及通过增殖和细胞死亡等过程改变细胞数量将会加速关节软骨破坏[12]。软骨细胞的异常增殖将会加重骨性关节炎的进展[13]。因此,抑制软骨细胞的过度增殖可以作为防止骨性关节炎中软骨降解的潜在治疗方法。下一部分将讨论lncRNA对骨性关节炎中软骨细胞增殖的影响,相关机制见图4。"
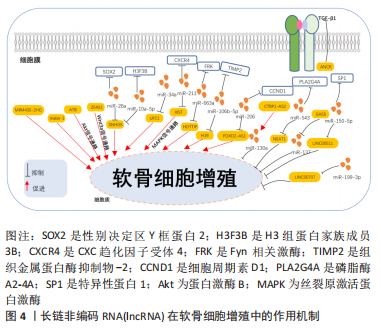
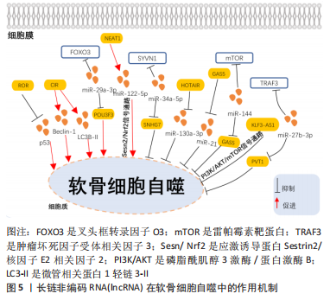
2.1.1 促进骨性关节炎软骨细胞增殖的LncRNA XIAO等[14]研究发现,骨性关节炎患者的血浆中lncRNA MIR4435-2HG 的表达水平显著低于健康对照组,通过敲低lncRNA MIR4435-2HG能够抑制软骨细胞的增殖,相反过表达lncRNA MIR4435-2HG能够促进软骨细胞增殖。DANG等[15]在细胞实验中发现lncRNA-ATB能够通过激活蛋白激酶B(protein kinase B,AKT)信号传导来促进软骨细胞的增殖和活力。YE等[16]报道,与正常软骨细胞相比,lncRNA ZFAS1在骨性关节炎软骨细胞中的表达下调,LncRNA ZFAS1可能通过靶向Wnt3a信号通路促进骨性关节炎软骨细胞的增殖和迁移,减少细胞凋亡和基质合成。LI等[17]在体内实验中通过Pearson相关系数分析观察到lncRNA ANCR 和转化生长因子β1表达在骨关节炎患者血浆水平中呈负相关,而在正常对照组中则无相关性,lncRNA ANCR可能通过下调转化生长因子β1表达促进软骨细胞增殖。PAN等[18]通过动物和细胞实验证明,在骨性关节炎环境中人间充质干细胞外囊泡来源的lncRNA malat-1能够促进软骨细胞增殖。 lncRNA还可以作为竞争性内源RNA(competing endogenous,ceRNA)促进骨性关节炎软骨细胞增殖。lncRNA SNHG5可以作为ceRNA 竞争性海绵化miR-26a,从而下调性别决定区Y框蛋白2(sex determining region Y box protein 2,SOX2)的表达,促进软骨细胞增殖[19]。此外,JIANG等[20]报道在骨性关节炎软骨组织中lncRNA SNHG5和H3组蛋白家族成员3B(H3F3B)表达下调,而miR-10a-5p表达上调。LncRNA SNHG5能够通过海绵化miR-10a-5p促进H3F3B的表达从而促进软骨细胞增殖。lncRNA UFC1能作为ceRNA海绵化miR-34a促进骨关节炎中软骨细胞的增殖[21]。LncRNA XIST可以充当miR-211的ceRNA以竞争性结合miR-211减轻其对于CXC趋化因子受体4(CXCR4)的抑制,从而通过下游丝裂原激活蛋白激酶(mitogen-activated protein kinases,MAPK)信号促进软骨细胞增殖[22]。HE等[23]在实验中发现,骨性关节炎软骨组织中lncRNA HOTTIP的表达明显高于正常软骨组织,lncRNA HOTTIP可以作为ceRNA通过竞争性结合miR-663a上调Fyn 相关激酶(FRK)的表达从而促进骨性关节炎软骨细胞的增殖。TAN等[24]发现,lncRNA H19作为ceRNA靶向竞争结合miR-106b-5p上调组织金属蛋白酶抑制物2(TIMP2)的表达来促进骨性关节炎中的软骨细胞增殖和迁移。CAO等[25]证实lncRNA FOXD2-AS1能够下调miR-206上调细胞周期素D1(CCND1)的表达促进软骨细胞增殖。 2.1.2 抑制骨性关节炎软骨细胞增殖的LncRNA ZHANG等[26]在体内实验中发现,与健康对照组相比,lncRNA CTBP1-AS2在骨性关节炎软骨组织中的表达上调,且与miR-130a表达呈负相关,lncRNA CTBP1-AS2的过表达能够导致miR-130a基因的甲基化增加,从而证实lncRNA CTBP1-AS2能够通过增加miR-130a基因甲基化修饰进而抑制软骨细胞增殖。XIAO等[27]研究表明,lncRNA NEAT1在骨性关节炎中表达量下调,并且证实了lncRNA NEAT1/miR-543/磷脂酶A2-4A(phospholipase A2-4A,PLA2G4A)调控信号通路的存在,lncRNA NEAT1能够竞争性结合miR-543诱导PLA2G4A的表达从而抑制软骨细胞增殖。GAO等[28]研究发现,lncRNA GAS5在膝关节骨性关节炎患者血清和软骨组织中表达上调,lncRNA GAS5能够通过靶向下调miR-137的表达抑制软骨细胞增殖。ZHANG等[9]通过双荧光素酶实验中发现lncRNA LINC00511是miR-150-5p的上游靶标,证实lncRNA LINC00511可以作为ceRNA海绵化miR-150-5p从而上调特异性蛋白1(SP1)的表达抑制软骨细胞增殖。以往的研究证实了lncRNA LINC00707能够促进脂多糖诱导的骨性关节炎软骨细胞损伤,而miR-199-3p能调节软骨细胞增殖,XU等[29]在细胞实验中的研究结果表明,lncRNA LINC00707能够直接与细胞质中的miR-199-3p相互作用,验证了lncRNA LINC00707可以海绵化miR-199-3p抑制骨性关节炎软骨细胞的增殖。 2.2 lncRNA在骨性关节炎软骨细胞自噬中的作用 自噬是细胞对化学和物理因素的代谢反应,例如营养或生长因子剥夺、促炎状态、缺氧、活性氧的积累、感染因子的存在和DNA损伤等。自噬主要通过降解细胞器和关键蛋白质来维持细胞内代谢的稳态[30]。研究表明,在骨性关节炎中发现软骨中的自噬水平降低,自噬可以保护软骨细胞免于凋亡[31]。软骨细胞的凋亡是造成软骨退变的主要机制,细胞自噬是真核细胞重要的分解代谢系统之一,能够维持细胞内环境稳态,提高细胞生存能力,自噬的激活能够调控软骨细胞凋亡并保护关节软骨[32]。已有的研究表明多种lncRNA能够通过软骨细胞自噬调控骨性关节炎的进展,在下面的部分中,将综述有关lncRNA在骨性关节炎期间对于软骨细胞自噬作用的最新发现,相关机制见图5。"
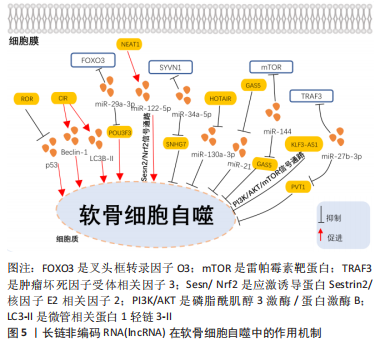
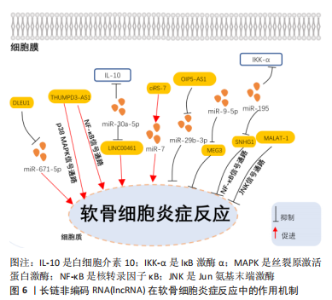
2.2.1 促进骨性关节炎软骨细胞自噬的lncRNA YANG等[33]在骨性关节炎软骨组织样本中发现,与正常软骨细胞相比,骨性关节炎中lncRNA-ROR的水平降低,并且lncRNA-ROR的过表达显著促进了骨性关节炎软骨细胞的活力;蛋白免疫印迹分析显示lncRNA-ROR 过表达抑制了p53的蛋白水平,在骨性关节炎软骨细胞中过表达p53能够通过抑制自噬特异性蛋白微管相关蛋白轻链3-Ⅱ(LC3-II)和Beclin-1的表达水平,因此发现lncRNA-ROR通过抑制p53的表达促进软骨细胞自噬。WANG等[34]研究表明,在骨性关节炎中lncRNA-CIR可以直接促进自噬特异性蛋白Beclin-1和微管相关蛋白1轻链3B-Ⅱ(LC3B-II)的表达,从而诱导软骨细胞自噬以调节关节软骨退化。SHI等[35]在体外实验中发现,miR-29a-3p可以直接与lncRNA POU3F3结合,而叉头框转录因子O3 (forkhead box O3,FOXO3)是miR-29a-3p的靶基因,lncRNA POU3F3可以海绵性吸附miR-29a-3p上调FOXO3的表达促进软骨细胞自噬抑制骨性关节炎的发展。ZHANG等[36]的实验结果显示,骨性关节炎患者骨髓间充质干细胞的细胞外囊泡提供的lncRNA NEAT1能够通过与miR-122-5p 结合来激活应激诱导蛋白Sestrin2(Sesn2)/核因子E2相关因子2(Nrf2)信号通路,从而促进软骨细胞自噬以防止骨性关节炎的发生。 2.2.2 抑制骨性关节炎软骨细胞自噬的LncRNA TIAN等[37]研究发现,与正常软骨细胞相比,经白细胞介素1β处理的软骨细胞中lncRNA SNHG7和SYVN1的表达下调,但miR-34a-5p上调;通过RNA结合蛋白免疫沉淀、RNA pull down和双荧光素酶基因实验确定miR-34a-5p是lncRNA SNHG7的目标miRNA,而SYVN1是miR-34-5p的目标mRNA,在骨性关节炎中lncRNA SNHG7能够作为ceRNA海绵性吸附miR-34a-5p上调SYVN1的表达进而抑制软骨细胞自噬。HE等[38]发现与无骨性关节炎软骨对照区域相比,骨性关节炎切除区域的股骨髁或胫骨平台软骨组织中lncRNA HOTAIR表达显著上调。在细胞实验中,与白细胞介素1β诱导的软骨细胞组相比,沉默lncRNA HOTAIR后发现LC3-II与微管相关蛋白轻链3-Ⅰ(LC3-I)的表达显著上调,荧光素酶测定表明miR-130a-3p是lncRNA HOTAIR的直接靶标,在骨性关节炎中通过促进lncRNA HOTAIR可以抑制miR-130a-3p的表达降低软骨细胞自噬,诱导骨性关节炎的发展。SONG等[39]在体内外实验中发现,用lncRNA GAS5编码的慢病毒感染软骨细胞,与无骨性关节炎患者的软骨细胞相比,骨性关节炎患者软骨细胞中细胞死亡显著增加,并且自噬相关蛋白Beclin-1,ATG7和微管相关蛋白1轻链3-B(LC3-B)显著降低;在动物实验中将接触pre-miR-21或针对miR-21的特异性si-miR-21的慢病毒注射到内侧半月板失稳手术小鼠的膝关节中发现,miR-21的引入显著降低了lncRNA GAS5水平,而si-miR-21的引入显著诱导了lncRNA GAS5的表达,miR-21的过表达刺激了软骨细胞中自噬相关蛋白ATG3,ATG5,ATG12和LC3-B的表达水平,lncRNA GAS5能够充当 miR-21 的负调节剂,抑制软骨细胞自噬,从而促进了骨性关节炎的发病。此外,JI等[40]同样发现,lncRNA GAS5在骨性关节炎大鼠软骨组织中的表达量上调,lncRNA GAS5能够与miR-144结合并调节雷帕霉素靶蛋白(mammalian target of rapamycin,mTOR)的表达从而抑制软骨细胞自噬。WEN等[41]在细胞实验中发现,间充质干细胞衍生中外泌体介导的lncRNA KLF3-AS1可能是骨性关节炎的潜在治疗靶点,在白细胞介素1β处理的软骨细胞中LncRNA KLF3-AS1可以通过磷脂酰肌醇3激酶(phosphatidylinositol-3-kinases,PI3K)/AKT/mTOR信号通路抑制软骨细胞的自噬。LU等[42]研究发现,在蛋白质印迹实验中与白细胞介素1β处理的人软骨细胞相比,miR-27b-3p模拟剂组能够促进自噬特异性蛋白Beclin-1,LC3-I,LC3-II的表达水平;通过双荧光素酶、RNA结合蛋白免疫沉淀和RNA pull down实验验证了miR-27b-3p与lncRNA PVT1,miR-27b-3p与肿瘤坏死因子受体相关因子3(tumor necrosis factor receptor-associated factor 3,TRAF3)之间的靶标关联,并且lncRNA PVT1与miR-27b-3p负相关,miR-27b-3p能够抑制TRAF3的表达。因此证实lncRNA PVT1作为ceRNA负向调控miR-27b-3p从而上调TRAF3的表达来抑制软骨细胞自噬。 2.3 lncRNA在骨性关节炎软骨细胞炎症反应中的作用 以往的研究表明,许多炎性细胞因子,尤其是白细胞介素2、白细胞介素6、白细胞介素10、白细胞介素12、白细胞介素1β、肿瘤坏死因子α和干扰素γ被认为可以介导骨性关节炎的进展[43]。软骨细胞引起的滑膜炎症在骨性关节炎中起着重要作用,滑膜病变和滑膜炎症相关因素将会引起关节软骨的降解和破坏,促进骨性关节炎的发展[44]。lncRNA在许多炎症相关疾病中发挥调节作用,发挥着广泛的生物学功能[8]。在以下部分中,总结了有关lncRNA在骨性关节炎软骨细胞相关滑膜炎中作用的最新发现,相关机制见图6。"
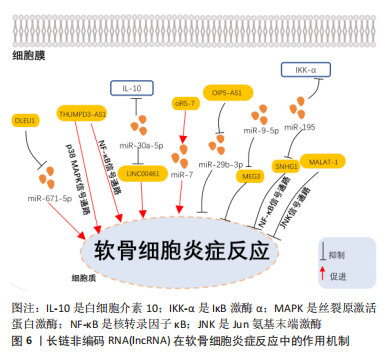
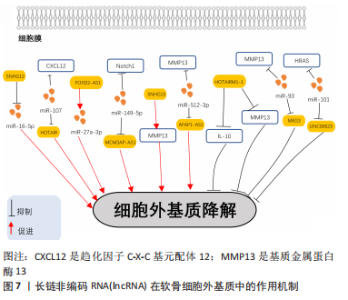
2.3.1 促进骨性关节炎软骨细胞炎症反应的LncRNA WU等[45]在骨性关节炎软骨组织标本中发现miR-671-5p的表达与lncRNA DLEU1呈负相关,炎症递质白细胞介素1β、肿瘤坏死因子α和白细胞介素6能够抑制骨性关节炎中miR-671-5p的表达;miR-671-5p的表达升高抑制软骨细胞增殖、细胞外基质降解和炎症递质分泌;过表达LncRNA DLEU1能够通过靶向下调miR-671-5p的表达促进软骨细胞炎症递质的分泌。WANG等[43]研究证实,lncRNA THUMPD3-AS1在骨性关节炎软骨组织和白细胞介素1β刺激的软骨细胞系中下调;在白细胞介素1β诱导的软骨细胞中,lncRNA THUMPD3-AS1能够通过激活p38 MAPK和核转录因子κB(nuclear factor kappa-B,NF-κB)信号通路促进白细胞介素6、一氧化氮和肿瘤坏死因子α的表达水平加重软骨细胞炎症。ZHANG等[46]报道,在骨性关节炎组织中lncRNA LINC00461的表达上调,通过TarBase数据库和双荧光酶素实验证实在软骨细胞中lncRNA LINC00461是miR-30a-5p的上游靶标,在骨性关节炎组织中miR-30a-5p的表达水平与lncRNA LINC00461表达呈负相关;miR-30a-5p过表达抑制软骨细胞中白细胞介素10的表达,过表达LINC00461的可以下调miR-30a-5p的表达水平促进白细胞介素10的表达增强了软骨细胞炎症;lncRNA ciRS-7已被证明能够充当miR-7的ceRNA并且能抑制其表达。ZHOU等[47]在体外实验中分别用lncRNA ciRS-7和miR-7模拟物或抑制剂转染软骨细胞,结果发现lncRNA ciRS-7表达的下调促进了炎性细胞因子白细胞介素6、白细胞介素8、白细胞介素10和肿瘤坏死因子α的释放,miR-7则会抑制炎性细胞因子白细胞介素6、白细胞介素8、白细胞介素10和肿瘤坏死因子α的释放。因此证实在骨性关节炎软骨细胞中lncRNA ciRS-7能够海绵化miR-7上调相关炎症因子的表达从而促进软骨细胞炎症。 2.3.2 抑制骨性关节炎软骨细胞炎症反应的LncRNA ZHI等[48]研究发现,在骨性关节炎软骨组织中LncRNA OIP5-AS1和颗粒蛋白前体(PGRN)表达明显降低,而miR-29b-3p表达则明显上调,miR-29b-3p是lncRNA OIP5-AS1的靶标且表达呈负相关,在骨性关节炎软骨细胞中,miR-29b-3p抑制剂组中炎性细胞因子白细胞介素6、白细胞介素8和肿瘤坏死因子α的表达明显降低;lncRNA OIP5-AS1能够通过抑制miR-29b-3p的表达抑制软骨细胞炎症。HUANG等[49]发现lncRNA MEG3表达在用白细胞介素1β处理的人软骨细胞系中显著下调,敲低lncRNA MEG3的表达发现软骨细胞白细胞介素6、白细胞介素8和肿瘤坏死因子α的水平升高,通过寻找其下游机制发现lncRNA MEG3可能是miR-9-5p的ceRNA,lncRNA MEG3能够竞争性结合miR-9-5p从而下调相关炎症因子的表达抑制软骨细胞炎症反应。NF-κB是介导 lncRNA对骨性关节炎软骨细胞炎症影响的主要途径,WANG等[50]在体内外实验中的研究结果表明,在骨性关节炎软骨细胞中lncRNA SNHG1直接靶向miR-195,IκB激酶α (IKK-α)与miR-195存在直接结合位点,IKK-α是NF-κB信号通路的抑制剂,证实lncRNA SNHG1能够作为ceRNA结合miR-195上调IKK-α的表达从而调节NF-κB信号通路以减轻软骨细胞炎症。此外,GAO等[51]在大鼠关节软骨细胞中发现LncRNA MALAT-1可以通过抑制Jun氨基末端激酶(Jun N-terminal kinase,JNK)信号通路从而抑制软骨细胞炎症。 2.4 lncRNA在骨性关节炎软骨细胞外基质降解中的作用 软骨细胞外基质降解与骨性关节炎发病密切相关,关节软骨细胞外基质的分解代谢增加是骨性关节炎发展的关键因素[52]。相关研究表明多种lncRNA在骨性关节炎期间参与了软骨细胞外基质降解的过程[53],在下面的部分中,将综述有关lncRNA在骨性关节炎期间软骨细胞外基质降解作用的最新发现,相关机制见图7。"
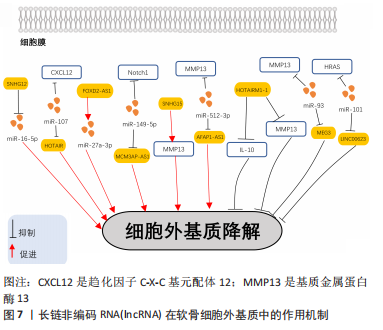

2.4.1 促进软骨细胞外基质降解的lncRNA YANG等[54]研究发现在骨性关节炎组织中lncRNA SNHG12表达上调,骨性关节炎患者组织中miR-16-5p的表达水平与lncRNA SNHG12的表达呈负相关,miR-16-5p是lncRNA SNHG12的靶点。lncRNA SNHG12通过抑制软骨细胞中miR-16-5p的表达抑制软骨细胞增殖,促进细胞外基质降解。LU等[55]在体内实验中发现,与正常对照组相比,骨性关节炎软骨中lncRNA HOTAIR的表达上调,miR-107表达下调,miR-107的表达与lncRNA HOTAIR 水平呈负相关且miR-107是lncRNA HOTAIR的下游靶标,lncRNA HOTAIR能够海绵性结合miR-107减轻其对趋化因子C-X-C基元配体12(CXCL12)的抑制从而促进软骨细胞增殖和细胞外基质降解加重骨性关节炎的发展。WANG等[56]研究发现lncRNA FOXD2-AS1可以通过靶向调节miR-27a-3p促进软骨细胞外基质降解。XU等[57]研究表明,性别决定基因-区域转录因子4(SOX4)激活的lncRNA MCM3AP-AS1能够作为ceRNA吸附miR-149-5p调节Notch1促进软骨细胞外基质降解来加重骨性关节炎的进展。基质金属蛋白酶13是一种在骨骼和关节部位产生的细胞外基质降解相关酶。ZHANG等[58]通过lncRNA SNHG15转染软骨细胞后并通过白细胞介素1β刺激软骨细胞,发现基质金属蛋白酶13水平增加以及胶原蛋白Ⅲ和聚集蛋白聚糖蛋白水平降低,上调lncRNA SNHG15的表达能够抑制白细胞介素1β诱导的软骨细胞外基质的降解。此外,ZHAO等[59]研究表明LncRNA AFAP1-AS1的表达在骨关节炎软骨及软骨细胞中上调;miR-512-3p的表达在骨关节炎软骨中下调,lncRNA AFAP1-AS1能够作为ceRNA海绵化结合miR-512-3p上调基质金属蛋白酶13的表达从而促进软骨细胞外基质降解。 2.4.2 抑制软骨细胞外基质降解的lncRNA LIU等[60]研究表明,与健康对照样本相比,骨性关节炎样本组织中lncRNA HOTAIRM1-1的表达显著降低,敲低lncRNA HOTAIRM1-1能够上调基质金属蛋白酶13和白细胞介素10的表达促进细胞外基质降解,相反过表达lncRNA HOTAIRM1-1可以使基质金属蛋白酶13和白细胞介素10表达量下调抑制细胞外基质降解。CHEN等[61]在细胞实验中发现,在用白细胞介素1β处理的软骨细胞中lncRNA MEG3表达显著下调,而miR-93上调,miR-93能够直接与lncRNA MEG3结合,在白细胞介素1β处理的软骨细胞中,lncRNA MEG3过表达抑制基质金属蛋白酶13的表达,通过转染miR-93模拟物可以显著降低lncRNA MEG3对于基质金属蛋白酶13表达的抑制作用,lncRNA MEG3能够作为ceRNA 海绵化miR-93抑制基质金属蛋白酶13的表达从而抑制细胞外基质降解。LV等[62]的细胞实验报道HRAS过表达显著减弱了白细胞介素1β诱导的软骨细胞外基质降解,lncRNA LINC00623能够作为ceRNA与HRAS竞争结合miR-101,从而降低miR-101对其下游靶标HRAS表达的抑制作用,该实验结果证明lncRNA LINC00623能够通过靶向于miR-101上调HRAS的表达从而抑制软骨细胞外基质降解。"

| [1] XU B, LI YY, MA J, et al. Roles of microRNA and signaling pathway in osteoarthritis pathogenesis. J Zhejiang Univ Sci B. 2016;17(3):200-208. [2] TAO XM, LIU PF, GU HY, et al. Cordycepin Alleviates Anterior Cruciate Ligament Transection (ACLT)-Induced Knee Osteoarthritis Through Regulating TGF-β Activity and Autophagy. Drug Des Devel Ther. 2020;14:2809-2817. [3] XIONG W, ZHAO J, MA X, et al. Mechanisms and Molecular Targets of BuShenHuoXue Formula for Osteoarthritis. ACS Omega. 2022;7(5):4703-4713. [4] 朱芳晓,王晓桃.长非编码RNA在骨关节炎中的作用研究进展[J].中国老年学杂志,2022,42(10):2557-2559. [5] QIAN X, ZHAO J, YEUNG PY, et al. Revealing lncRNA Structures and Interactions by Sequencing-Based Approaches. Trends Biochem Sci. 2019;44(1):33-52. [6] CHEN J, YU X, ZHANG X. Advances on biological functions of exosomal non-coding RNAs in osteoarthritis. Cell Biochem Funct. 2022;40(1):49-59. [7] SILVA AM, MOURA SR, TEIXEIRA JH, et al. Long noncoding RNAs: a missing link in osteoporosis. Bone Res. 2019;7:10. [8] HE L, LI Y, WANG G, et al. Regulation of long non-coding RNA in cartilage injury of osteoarthritis. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2020;34(11): 1486-1491. [9] ZHANG Y, DONG Q, SUN X. Positive Feedback Loop LINC00511/miR-150-5p/SP1 Modulates Chondrocyte Apoptosis and Proliferation in Osteoarthritis. DNA Cell Biol. 2020;39(9):1506-1512. [10] WANG J, SUN Y, LIU J, et al. Roles of long non‑coding RNA in osteoarthritis (Review). Int J Mol Med. 2021;48(1):133. [11] 张兴宇,向萌.力学改变激活软骨细胞介导创伤性骨关节炎的研究进展[J].复旦学报(医学版),2018,45(2):233-239. [12] AIGNER T, SODER S, GEBHARD PM, et al. Mechanisms of disease:role of chondrocytes in the pathogenesis of osteoarthritis--structure, chaos and senescence. Nat Clin Pract Rheumatol. 2007;3(7):391-399. [13] ZHANG W, ZHONG B, ZHANG C, et al. miR-373 regulates inflammatory cytokine-mediated chondrocyte proliferation in osteoarthritis by targeting the P2X7 receptor. FEBS Open Bio. 2018;8(3):325-331. [14] XIAO Y, BAO Y, TANG L, et al. LncRNA MIR4435-2HG is downregulated in osteoarthritis and regulates chondrocyte cell proliferation and apoptosis. J Orthop Surg Res. 2019;14(1):247. [15] DANG X, LIAN L, WU D. The diagnostic value and pathogenetic role of lncRNA-ATB in patients with osteoarthritis. Cell Mol Biol Lett. 2018;23:55. [16] YE D, JIAN W, FENG J, et al. Role of long noncoding RNA ZFAS1 in proliferation, apoptosis and migration of chondrocytes in osteoarthritis. Biomed Pharmacother. 2018;104:825-831. [17] LI Q, ZHANG Z, GUO S, et al. LncRNA ANCR is positively correlated with transforming growth factor-β1 in patients with osteoarthritis. J Cell Biochem. 2019;120(9):14226-14232. [18] PAN C, HUANG W, CHEN Q, et al. LncRNA Malat-1 From MSCs-Derived Extracellular Vesicles Suppresses Inflammation and Cartilage Degradation in Osteoarthritis. Front Bioeng Biotechnol. 2021;9:772002. [19] SHEN H, WANG Y, SHI W, et al. LncRNA SNHG5/miR-26a/SOX2 signal axis enhances proliferation of chondrocyte in osteoarthritis. Acta Biochim Biophys Sin (Shanghai). 2018;50(2):191-198. [20] JIANG H, PANG H, WU P, et al. LncRNA SNHG5 promotes chondrocyte proliferation and inhibits apoptosis in osteoarthritis by regulating miR-10a-5p/H3F3B axis. Connect Tissue Res. 2021;62(6):605-614. [21] ZHANG G, WU Y, XU D, et al. Long Noncoding RNA UFC1 Promotes Proliferation of Chondrocyte in Osteoarthritis by Acting as a Sponge for miR-34a. DNA Cell Biol. 2016;35(11):691-695. [22] LI L, LV G, WANG B, et al. The role of lncRNA XIST/miR-211 axis in modulating the proliferation and apoptosis of osteoarthritis chondrocytes through CXCR4 and MAPK signaling. Biochem Biophys Res Commun. 2018;503(4):2555-2562. [23] HE X, GAO K, LU S, et al. LncRNA HOTTIP leads to osteoarthritis progression via regulating miR-663a/ Fyn-related kinase axis. BMC Musculoskelet Disord. 2021; 22(1):67. [24] TAN F, WANG D, YUAN Z. The Fibroblast-Like Synoviocyte Derived Exosomal Long Non-coding RNA H19 Alleviates Osteoarthritis Progression Through the miR-106b-5p/TIMP2 Axis. Inflammation. 2020;43(4):1498-1509. [25] CAO L, WANG Y, WANG Q, et al. LncRNA FOXD2-AS1 regulates chondrocyte proliferation in osteoarthritis by acting as a sponge of miR-206 to modulate CCND1 expression. Biomed Pharmacother. 2018;106:1220-1226. [26] ZHANG H, LI J, SHAO W, et al. LncRNA CTBP1-AS2 is upregulated in osteoarthritis and increases the methylation of miR-130a gene to inhibit chondrocyte proliferation. Clin Rheumatol. 2020;39(11):3473-3478. [27] XIAO P, ZHU X, SUN J, et al. LncRNA NEAT1 regulates chondrocyte proliferation and apoptosis via targeting miR-543/PLA2G4A axis. Hum Cell. 2021;34(1):60-75. [28] GAO ST, YU YM, WAN LP, et al. LncRNA GAS5 induces chondrocyte apoptosis by down-regulating miR-137. Eur Rev Med Pharmacol Sci. 2020;24(21):10984-10991. [29] XU Y, DUAN L, LIU S, et al. Long intergenic non-protein coding RNA 00707 regulates chondrocyte apoptosis and proliferation in osteoarthritis by serving as a sponge for microRNA-199-3p. Bioengineered. 2022;13(4):11137-11145. [30] XIN Y, JIANG F, YANG C, et al. Role of autophagy in regulating the radiosensitivity of tumor cells. J Cancer Res Clin Oncol. 2017;143(11):2147-2157. [31] DUAN R, XIE H, LIU ZZ. The Role of Autophagy in Osteoarthritis. Front Cell Dev Biol. 2020;8:608388. [32] 曾凡,陈柏屹,王康,等.加味独活寄生合剂对膝骨关节炎模型兔膝关节软骨组织细胞自噬及凋亡相关蛋白的影响[J].中医杂志,2022,63(8):773-780. [33] YANG Z, TANG Y, LU H, et al. Long non-coding RNA reprogramming (lncRNA-ROR) regulates cell apoptosis and autophagy in chondrocytes. J Cell Biochem. 2018;119(10):8432-8440. [34] WANG CL, PENG JP, CHEN XD. LncRNA-CIR promotes articular cartilage degeneration in osteoarthritis by regulating autophagy. Biochem Biophys Res Commun. 2018;505(3):692-698. [35] SHI M, SUN M, WANG C, et al. Therapeutic potential of POU3F3, a novel long non-coding RNA alleviates the pathogenesis of osteoarthritis by regulating miR-29a-3p/FOXO3 axis. Curr Gene Ther. 2022 Mar 9. doi: 10.2174/1566523222666220309150722. [36] ZHANG S, JIN Z. Bone Mesenchymal Stem Cell-Derived Extracellular Vesicles Containing Long Noncoding RNA NEAT1 Relieve Osteoarthritis. Oxid Med Cell Longev. 2022;2022:5517648. [37] TIAN F, WANG J, ZHANGh Z, et al. LncRNA SNHG7/miR-34a-5p/SYVN1 axis plays a vital role in proliferation, apoptosis and autophagy in osteoarthritis. Biol Res. 2020;53(1):9. [38] HE B, JIANG D. HOTAIR-induced apoptosis is mediated by sponging miR-130a-3p to repress chondrocyte autophagy in knee osteoarthritis. Cell Biol Int. 2020; 44(2):524-535. [39] SONG J, AHN C, CHUN CH, et al. A long non-coding RNA, GAS5, plays a critical role in the regulation of miR-21 during osteoarthritis. J Orthop Res. 2014;32(12): 1628-1635. [40] JI Q, QIAO X, LIU Y, et al. Expression of long-chain noncoding RNA GAS5 in osteoarthritis and its effect on apoptosis and autophagy of osteoarthritis chondrocytes. Histol Histopathol. 2021;36(4):475-484. [41] WEN C, LIN L, ZOU R, et al. Mesenchymal stem cell-derived exosome mediated long non-coding RNA KLF3-AS1 represses autophagy and apoptosis of chondrocytes in osteoarthritis. Cell Cycle. 2022;21(3):289-303. [42] LU X, YU Y, YIN F, et al. Knockdown of PVT1 inhibits IL-1β-induced injury in chondrocytes by regulating miR-27b-3p/TRAF3 axis. Int Immunopharmacol. 2020;79:106052. [43] WANG Y, LI T, YANG Q, et al. LncRNA THUMPD3-AS1 enhances the proliferation and inflammatory response of chondrocytes in osteoarthritis. Int Immunopharmacol. 2021;100:108138. [44] SONG PF, KAN WB, YUAN Q, et al. Manifestation of synovial lesions in osteoarthritis. Zhongguo Gu Shang. 2012;25(5):442-444. [45] WU X, YIN S, YAN L, et al. lncRNA DLEU1 Modulates Proliferation, Inflammation, and Extracellular Matrix Degradation of Chondrocytes through Regulating miR-671-5p. J Immunol Res. 2022;2022:1816217. [46] ZHANG Y, MA L, WANG C, et al. Long noncoding RNA LINC00461 induced osteoarthritis progression by inhibiting miR-30a-5p. Aging (Albany NY). 2020; 12(5):4111-4123. [47] ZHOU X, JIANG L, FAN G, et al. Role of the ciRS-7/miR-7 axis in the regulation of proliferation, apoptosis and inflammation of chondrocytes induced by IL-1β. Int Immunopharmacol. 2019;71:233-240. [48] ZHI L, ZHAO J, ZHAO H, et al. Downregulation of LncRNA OIP5-AS1 Induced by IL-1β Aggravates Osteoarthritis via Regulating miR-29b-3p/PGRN. Cartilage. 2021;13(2_suppl):1345S-1355S. [49] HUANG Y, CHEN D, YAN Z, et al. LncRNA MEG3 Protects Chondrocytes From IL-1β-Induced Inflammation via Regulating miR-9-5p/KLF4 Axis. Front Physiol. 2021;12:617654. [50] WANG Q, DENG F, LI J, et al. The long non-coding RNA SNHG1 attenuates chondrocyte apoptosis and inflammation via the miR-195/IKK-α axis. Cell Tissue Bank. 2022 Jul 7. doi: 10.1007/s10561-022-10019-3. [51] GAO GC, CHENG XG, WEI QQ, et al. Long noncoding RNA MALAT-1 inhibits apoptosis and matrix metabolism disorder in interleukin-1β-induced inflammation in articular chondrocytes via the JNK signaling pathway. J Cell Biochem. 2019;120(10):17167-17179. [52] RAHMATI M, NALESSO G, MOBASHERI A, et al. Aging and osteoarthritis: Central role of the extracellular matrix. Ageing Res Rev. 2017;40:20-30. [53] CHEN WK, YU XH, YANG W, et al. lncRNAs: novel players in intervertebral disc degeneration and osteoarthritis. Cell Prolif. 2017;50(1):e12313. [54] YANG X, CHEN H, ZHENG H, et al. LncRNA SNHG12 Promotes Osteoarthritis Progression Through Targeted Down-Regulation of miR-16-5p. Clin Lab. 2022; 68(1). doi: 10.7754/Clin.Lab.2021.210402. [55] LU J, WU Z, XIONG Y. Knockdown of long noncoding RNA HOTAIR inhibits osteoarthritis chondrocyte injury by miR-107/CXCL12 axis. J Orthop Surg Res. 2021;16(1):410. [56] WANG Y, CAO L, WANG Q, et al. LncRNA FOXD2-AS1 induces chondrocyte proliferation through sponging miR-27a-3p in osteoarthritis. Artif Cells Nanomed Biotechnol. 2019;47(1):1241-1247. [57] XU F, HU QF, LI J, et al. SOX4-activated lncRNA MCM3AP-AS1 aggravates osteoarthritis progression by modulating miR-149-5p/Notch1 signaling. Cytokine. 2022;152:155805. [58] ZHANG X, HUANG CR, PAN S, et al. Long non-coding RNA SNHG15 is a competing endogenous RNA of miR-141-3p that prevents osteoarthritis progression by upregulating BCL2L13 expression. Int Immunopharmacol. 2020;83:106425. [59] ZHAO Z, WANG Z, PEI L, et al. Long non-coding ribonucleic acid AFAP1-AS1 promotes chondrocyte proliferation via the miR-512-3p/matrix metallopeptidase 13 (MMP-13) axis. Bioengineered. 2022;13(3):5386-5395. [60] LIU WB, LI GS, SHEN P, et al. Long non-coding RNA HOTAIRM1-1 silencing in cartilage tissue induces osteoarthritis through microRNA-125b. Exp Ther Med. 2021;22(3):933. [61] CHEN K, ZHU H, ZHENG MQ, et al. LncRNA MEG3 Inhibits the Degradation of the Extracellular Matrix of Chondrocytes in Osteoarthritis via Targeting miR-93/TGFBR2 Axis. Cartilage. 2021;13(2_suppl):1274S-1284S. [62] LV G, LI L, WANG B, et al. LINC00623/miR-101/HRAS axis modulates IL-1β-mediated ECM degradation, apoptosis and senescence of osteoarthritis chondrocytes. Aging (Albany NY). 2020;12(4):3218-3237. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [3] | Li Xiaomin, Tian Xiangdong, Tan Yetong, Zhu Guangyu, Wang Rongtian, Wang Jian, Xue Zhipeng, Ma Sheng, Hu Yuanyi, Huang Ye, Ding Tiansong. Changes of lower limb force line and knee function after high tibial osteotomy in osteoporotic medial ventricular knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1325-1329. |
| [4] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [5] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [6] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [7] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [8] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [9] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [10] | Huang Linke, Wei Linhua, Jiang Jie, Liu Qian, Chen Weiwei. Effects of estrogen combined with treadmill exercise on bone mass and articular cartilage in ovariectomized mice [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1166-1171. |
| [11] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [12] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [13] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [14] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| [15] | Tang Haotian, Liao Rongdong, Tian Jing. Application and design of piezoelectric materials for bone defect repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1117-1125. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||