Chinese Journal of Tissue Engineering Research ›› 2023, Vol. 27 ›› Issue (18): 2928-2934.doi: 10.12307/2023.297
Previous Articles Next Articles
Regeneration mechanism and problems of tissue engineering in rotator cuff tendon-bone healing
Wang Xu1, 2, Yang Tengyun2, 3, Xiong Bohan2, 3, Zhang Yaozhang2, 3, Lu Xiaojun2, 3, Long Dan2, 3, Zhao Daohong1, 2
- 1Department of Orthopedics, The Second Affiliated Hospital of Kunming Medical University, Kunming 650000, Yunnan Province, China; 2Kunming Medical University, Kunming 650500, Yunnan Province, China; 3Department of Sports Medicine, The First Affiliated Hospital of Kunming Medical University, Kunming 650000, Yunnan Province, China
-
Received:2022-04-04Accepted:2022-05-16Online:2023-06-28Published:2022-09-19 -
Contact:Zhao Daohong, MD, Associate chief physician, Department of Orthopedics, The Second Affiliated Hospital of Kunming Medical University, Kunming 650000, Yunnan Province, China; Kunming Medical University, Kunming 650500, Yunnan Province, China -
About author:Wang Xu, Master candidate, Department of Orthopedics, The Second Affiliated Hospital of Kunming Medical University, Kunming 650000, Yunnan Province, China; Kunming Medical University, Kunming 650500, Yunnan Province, China -
Supported by:National Natural Science Foundation of China, No. 81960412 (to ZDH); Basic Research Joint Project of Yunnan Provincial Department of Science and Technology-Kunming Medical University, No. 2019FE001(-065) (to ZDH)
CLC Number:
Cite this article
Wang Xu, Yang Tengyun, Xiong Bohan, Zhang Yaozhang, Lu Xiaojun, Long Dan, Zhao Daohong. Regeneration mechanism and problems of tissue engineering in rotator cuff tendon-bone healing[J]. Chinese Journal of Tissue Engineering Research, 2023, 27(18): 2928-2934.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
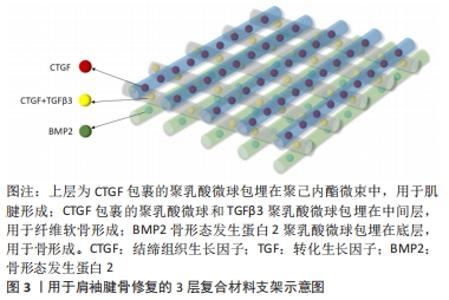
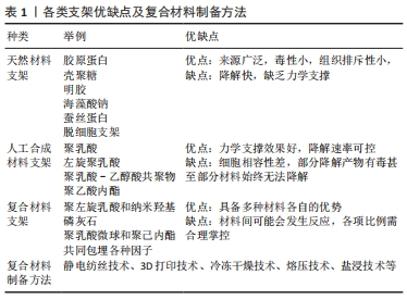
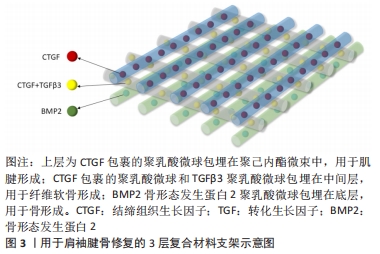
2.1 支架的材料选择 组织工程中支架的作用是传递细胞因子,刺激种子细胞黏附增殖和分化,提供良好的再生微环境并为新生组织提供结构支撑[18]。现有的支架材料可大致分为天然高分子材料、人工合成支架材料及复合支架材料等,这些材料可以被设计成水凝胶、纳米微粒、三维多孔固态支架等形式。 2.1.1 天然材料支架 天然来源的生物材料通过模拟肩袖止点处原有的细胞外基质的原理,促进细胞增殖,如胶原蛋白、壳聚糖、明胶、海藻酸钠、蚕丝蛋白等[16,19],其具有来源广泛、组织排斥性小的优势,但降解快、缺乏力学支持作用的问题有待解决。此类天然材料还包括人真皮胶原基质等自体移植材料及脱钙骨基质等异种异体移植材料[20]。近年来,脱细胞支架在腱骨修复中的应用得到关注,其具有促进细胞黏附、迁移、分化和增殖的功能,同时维持了极佳的微观结构,可增强移植后的组织细胞再生[21-22]。脱细胞支架在促进肌腱骨愈合方面发挥着独特的作用,LU等[23]于2019年创造性地用骨或纤维软骨组织制备了2种脱细胞支架,即书本型脱细胞骨支架和书本型脱细胞纤维软骨支架,2种支架都具有无细胞成分的天然细胞外基质结构,实验结果表明:书本型脱细胞骨支架具有良好的成骨诱导能力,而书本型脱细胞纤维软骨支架具有良好的软骨诱导能力;在兔部分髌骨切除模型中,2种支架均能促进肌腱-骨愈合,而书本型脱细胞纤维软骨支架比书本型脱细胞骨支架更能促进肌腱-骨愈合。 2.1.2 人工合成材料支架 在人工材料领域,多采用高分子聚合物,一类是聚碳酸酯、聚氨基甲酸乙酯和聚四氟乙烯(poly tetra fluoroethylene,PTFE)等非可降解高分子聚合物,另一类为聚乳酸(polylactic acid,PLA)、左旋聚乳酸(poly-l-lactide,PLLA)、聚乳酸-乙醇酸共聚物[poly((lactic-co-glycolic acid),PLGA]、聚乙酸内酯等新型高分子可降解支架[24]。人工合成材料的力学支撑力优于天然材料,降解速率可控,但分别存在细胞相容性差、无法降解和降解产物具有细胞毒性的弊端[25]。但其优势仍不可小觑,如CHEN等[26]实验证明3D打印PLGA支架与骨髓间充质干细胞(bone marrow mesenchymal stem cells,BMSCs)联合应用得到了很好的效果,实验结果表明BMSCs-PLGA支架植入增强了腱骨界面的胶原蛋白形成并增加了胶原蛋白的直径;生物力学分析表明,BMSCs-PLGA支架植入改善了再生肌腱的生物力学性能,同时从胶原纤维和生物力学特性的角度来看,与单独植入支架相比,植入 BMSC 种子的支架在植入后 12 周显示出更大的胶原原纤维直径和更好的机械性能,这一结论也进一步印证了组织工程技术在实际应用中的优势。 2.1.3 复合材料支架 复合生物材料具有各自材料的优势,是理想的支架选择类型。单一材料支架无法实现的仿生骨腱界面4层梯度结构和差异化设计,分层设计的仿生高分子材料支架却很好地解决了这一难题[27]。LI等[28]制备了一体化双极纤维膜,其上层和下层分别是聚左旋乳酸纤维膜和纳米羟基磷灰石纤维膜,在兔肩袖撕裂模型中,双极纤维膜增加了糖胺聚糖的形成,改善了胶原组织,诱导了骨形成和纤维形成,并产生了比单纯组更好的最终负荷。TARAFDER等[29]则设计了3层支架,支架被设计用于修复大鼠冈上肌腱和肱骨之间的损伤,采用聚乳酸微球(μS)包埋结缔组织生长因子(CTGF)、转化生长因子β3(TGFβ3)和骨形态发生蛋白2(BMP2),最终得到的支架为结缔组织生长因子包裹的聚乳酸微球包埋在聚己内酯微束中,用于肌腱形成;结缔组织生长因子包裹的聚乳酸微球和转化生长因子β3聚乳酸微球包埋在中间层,用于纤维软骨形成;骨形态发生蛋白2聚乳酸微球包埋在底层,用于骨形成。此3D打印支架成功地促进了内源性干/祖细胞的招募和区域分化,从而促进了肌腱骨界面的愈合,且由于微球缓释技术可以达到时空控制作用,是很有潜力的新型手术植入材料,复合支架具体如图3。"
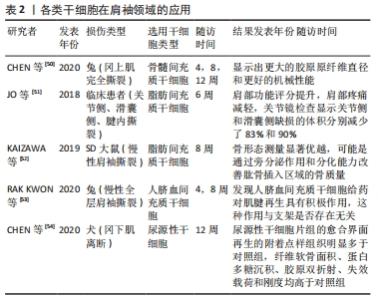
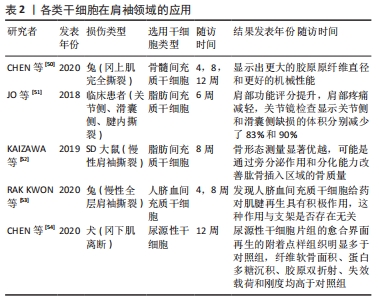
2.2 种子细胞的选择 理想的组织工程种子细胞需要易于分离培养、传代、扩增,并有分化为腱骨结构的潜能,同时种子细胞还需满足有良好的生物相容性、无免疫排斥反应的条件[30]。目前用于腱骨愈合的种子细胞主要两大类,即功能细胞和未分化细胞。功能细胞如软骨细胞、成纤维细胞、成骨细胞等[17],其为成熟体细胞,相比于未分化细胞其有更加确切的生命路线,能够直接补充损伤区域[31]。未分化细胞有不同来源的间充质干细胞、胚胎干细胞和人类诱导多能干细胞(hiPSCs)。间充质干细胞包括BMSCs、脂肪源性干细胞(adipose derived stem cells,ADSCs)、脐血间充质干细胞、肌源性间充质干细胞、腱源性间充质干细胞及尿源性干细胞。此外,胚胎干细胞也具有强大的分化能力,理论上也可以为腱骨愈合提供新的细胞来源,但由于伦理限制胚胎干细胞的应用存在一定问题,而人类诱导多能干细胞在再生领域也存在长期分化能力存疑和致畸作用的可能性,需要进一步实验[32]。因此目前最常见的组织工程种子细胞仍是各种来源的间充质干细胞。 2.2.1 BMSCs BMSCs在腱骨愈合的研究领域有着最为悠久的历史,并且已有应用于人体的研究[33]。HERNIGOU 等[34]在90例患者上进行了对照试验,45例患者在肩袖关节镜治疗时注射了BMSCs,10年随访显示BMSCs治疗组出现完整肩袖愈合的概率为87%(39例),而未治疗组只有44%(20例),并且治疗组的再撕裂率也低于未治疗组。在BMSCs治疗领域研究者不断探索,为了避免髂嵴穿刺获取BMSCs方法所导致的二次伤害,研究者创新性地在肩袖修补术的同时穿刺肱骨或肱骨大结节,通过微骨折的方法获取BMSCs,得到了很好的促进腱骨愈合表现,如KIDA 等[35]对大鼠肩袖损伤模型进行肱骨头穿刺,发现BMSCs可以穿过肌腱插入点的钻孔,爬入待修复的肩袖,此方法有望成为新型肩袖修补术式的发展方向。 2.2.2 脂肪间充质干细胞 脂肪源性干细胞具有易于分离、来源丰富,增殖率更高的优势,近年来也逐渐受到学者的关注。CHEN等[36]在大鼠模型肩袖愈合过程中将脂肪间充质干细胞注入肌腱,而对照组则注射生理盐水;在脂肪间充质干细胞注射组中,纤维排列和肌腱组织得到改善,炎症细胞数量明显减少,冈上肌腱的拉伸强度显着提高。在人类临床试验中也报道了一些有希望的结果。kIM等[37]在2017年研究了肌腱套修复后注入纤维蛋白胶装载的脂肪间充质干细胞的临床试验,基于性别、年龄、病变的大小进行匹配,35例患者接受了注射,而另35例患者仅接受手术治疗;1年随访时磁共振成像(MRI)显示,脂肪间充质干细胞注射组的再撕裂率(14.3%)显著低于对照组(28.5%)。并且有研究表明脂肪间充质干细胞可以在疾病模型中更好地维持细胞状态,发挥调节微环境的作用并且其人类白细胞抗原-ABC表达较少,免疫排斥更低,更适用于同种异体移植[38-39],同时有研究表明脂肪间充质干细胞组表现出较少的炎症,可能导致更有弹性的修复和愈合,产生更少的瘢痕[40]。因此继BMSCs后脂肪间充质干细胞似乎是肩袖腱骨愈合的又一良好种子细胞选择。 尽管脂肪间充质干细胞在许多研究中对腱骨愈合有效,但确切的机制仍需进一步研究。目前,关于脂肪间充质干细胞促进腱骨愈合的作用机制有以下几种假设,脂肪间充质干细胞不仅能够自我分化,而且能够通过刺激其他内源性干细胞沿适当的路径募集并促进它们的分化;脂肪间充质干细胞可以为损伤部位提供抗氧化化学物质或自由基清除剂,以帮助从撕裂的肌腱和肌肉骨骼组织中清除有毒物质并促进细胞恢复等[41]。 2.2.3 腱源性间充质干细胞 从健康或破裂的腱鞘分离的肌腱源性干细胞(TDSCs)已经被证明可以分化为成骨细胞、成软骨细胞和脂肪细胞[42]。SHEN等[43]研究了腱源性间充质干细胞接种在针织丝胶原海绵支架上的有效性。HUANG等[44]纳入不同年龄组及损伤情况的154例冈上肌或冈下肌肌腱全层撕裂的患者进行腱源性间充质干细胞增殖能力、基础基因表达和三系分化检测,结果表明腱源性间充质干细胞存在于大结节上的肌腱套残体中,并且具有多谱系分化能力,但其含量随着年龄增长和慢性时间延长而退化,这也限制了腱源性间充质干细胞自体移植的临床应用。虽然鉴于腱鞘中腱源性间充质干细胞含量有限,其提取也受到一定限制,在其促进肩袖腱骨愈合领域也少有相关研究,但是理论上腱源性间充质干细胞在促进腱骨愈合能力上比BMSCs更具优势,因为腱源性间充质干细胞能保证腱骨止点处生成更多的肌腱与骨,且增殖能力更强[45-46]。腱源性间充质干细胞治疗腱骨连接损伤的研究仍处于起步阶段,需要进一步的研究来测试其应用的有效性,并确定其在体内的功效。 2.2.4 脐血间充质干细胞 脐血间充质干细胞是分娩后的一种医疗废弃物,可以无创获得且成本相对较低,与其他成体间充质干细胞相比,它具有更高的自我更新潜能和增殖能力,并且具有能够在损伤组织中定位、低免疫原性、多向分化等属性[47]。PARK等[48]将脐血间充质干细胞在超声引导下注射进入肩袖全层撕裂的新西兰兔模型中,4周后肌腱撕裂大小和行走能力显著改善。此实验证明了在没有手术修复或生物支架的情况下,脐血间充质干细胞注射可在兔模型中部分治愈全厚度肩袖腱撕裂,并且结果显示再生的组织主要由Ⅰ型胶原组成。近年来研究者在兔全层肩袖肌腱撕裂模型中进行脐血间充质干细胞和聚脱氧核糖核苷酸联合注射,并同时进行微电流治疗也取得了很好的效果,实验组的组织化学和运动分析所有参数都明显优于对照组[49]。值得一提的是老年患者或有严重合并症的患者往往会出现自体间充质干细胞功能受损的情况,而由于脐血间充质干细胞具有低水平免疫原性的优势,对于老年人及有其他合并症的患者同种异体脐血间充质干细胞可能是一种有效的治疗选择[47]。 近年各种类干细胞在肩袖领域的应用,见表2。 "

2.3 生长因子 生长因子具有诱导分裂、促进细胞分化和血管形成的作用,是一类贯穿细胞分化整个过程的细胞因子或蛋白。研究发现各类生长因子在愈合过程中表现出独特的时空分布特点[55]。在以往的研究中骨诱导生长因子,包括骨形态发生蛋白(BMPs)、转化生长因子β(TGF-β)、血管内皮生长因子(VEGF)、成纤维细胞生长因子(FGF)等得到了大量实验研究,此外富血小板血浆、胰岛素样生长因子、粒细胞集落刺激因子、基质金属蛋白酶等也受到了学者们的关注,下文就骨形态发生蛋白、转化生长因子β、成纤维细胞生长因子及血管内皮生长因子的相关研究进展展开论述。 2.3.1 骨形态发生蛋白 骨形态发生蛋白属于转化生长因子β超家族的一员,它具有促进干细胞向骨、软骨转化的作用。骨形态发生蛋白有20多种亚型,其中骨形态发生蛋白2、7、12、13有许多关于腱骨愈合的研究报道,其中骨形态发生蛋白2具有最强的成骨活性,能促进成骨、软骨和脂肪三向分化并且同时促进血管形成。而 KABUTO 等[56]通过使用水凝明胶薄膜持续释放骨形态发生蛋白7,使SD大鼠肩袖腱骨界面在组织学及生物力学性能方面均得到增强,证明了骨形态发生蛋白7也具有促进腱骨愈合的作用。但骨形态发生蛋白12(又称GDF-7)的作用较为局限,不能诱导形成骨和软骨,仅能诱导干细胞向肌腱或者韧带样组织分化[57]。LAMPLOT等[58]将骨形态发生蛋白13 与富血小板血浆进行对照试验,结果显示肌腱损伤后的2周内,只有注射骨形态发生蛋白13 组促进了肌腱愈合,并且结果证明了骨形态发生蛋白13有效上调了Ⅲ型胶原的形成。总的来说,骨形态发生蛋白的应用显示出了几个挑战性问题,具体表现为不同递送时间和给药的方式可能会显著影响骨形态发生蛋白在体内的生物学功能。 2.3.2 转化生长因子β 转化生长因子β有3个亚型:转化生长因子β1、转化生长因子β2、转化生长因子β3。在损伤修复过程中,转化生长因子β通过促进成纤维细胞和巨噬细胞聚集的作用抑制了慢性炎症,同时还可以调节自身免疫、抑制排斥反应,最终达到促进腱骨愈合的目的。例如,HAN等[59]研究表明下调转化生长因子β会导致肩袖生物力学性能降低。YAMAKADO等[60]采用肝素/纤维蛋白将转化生长因子β作用于肩袖腱骨冈上肌损伤处,证实了转化生长因子β具有促进肩袖腱骨愈合、改善力学性能的作用。以上两实验通过正反两方面共同证明了转化生长因子β在肩袖腱骨愈合中的潜力这一结果。在转化生长因子β的内在作用机制方面,WANG等[61]则通过兔前交叉韧带重建模型发现了TGF-β/MAPK 信号通路,其实验结果论证了hBMSC的条件培养基通过磷酸化Smad2/3的形式激活了TGF-β/Smad信号通路,进而发挥了调控巨噬细胞极化方向的作用。但也有一些针对部分亚型的实验得出了完全不同的结果,SHAH等[62]抑制转化生长因子β1,得出了显著减少纤维化、脂肪浸润和肌肉萎缩的结果。MANNING等[63]的研究表明磷酸钙+转化生长因子β3组的愈合效果比磷酸钙组更差,提示转化生长因子β3对腱骨愈合过程可能具有干扰作用。因此,在转化生长因子β各亚型的具体作用方面仍存在一些争议,需要更多研究者进行进一步的深入研究。 2.3.3 成纤维细胞生长因子 成纤维细胞生长因子分为碱性成纤维细胞生长因子(bFGF)和酸性成纤维细胞生长因子(aFGF)。ZHAO等[64]在大鼠慢性肩袖损伤研究中发现在胶原组织再生、纤维软骨化、最大载荷及刚度方面,碱性成纤维细胞生长因子+PLGA静电纺丝膜组均优于单纯使用PLGA组。YONEMITSU等[65]的实验表明在大鼠慢性肩袖损伤中,与对照组相比,碱性成纤维细胞生长因子组在第6,12周的机械强度显著增加,存在更成熟的肌腱样组织,同时腱细胞谱系标记物Scx或Tnmd表达细胞的增加。LU等[66]则应用了酸性成纤维细胞生长因子,并证实了在兔肩袖愈合实验中酸性成纤维细胞生长因子具有促进细胞增殖、分化和增加细胞存活率的作用。在肩袖腱骨愈合领域,碱性成纤维细胞生长因子的应用较为广泛,而酸性成纤维细胞生长因子的应用并不多见,未来可以考虑将两者得出的结果进行对比研究,验证在肩袖腱骨愈合领域中两者作用的区别。 2.3.4 血管内皮生长因子 血管内皮生长因子是一种具有促进血管内皮细胞生长的因子,在肩袖愈合的局部血管新生中起重要作用。HUANG等[67]近期对于腱骨愈合的血管生成机制进行了研究,在大鼠冈上肌腱损伤手术模型中证实了vegf-vegfr激活Akt和 Erk,抑制hippo信号通路中的yap磷酸化,诱导血管生成从而促进大鼠肌腱-骨骼重建中的愈合。间接证实了血管内皮生长因子可以帮助肩袖腱骨获得更好的机械性能。而在以往的研究中显示只有在早期适当的血管内皮生长因子才能促进肌腱修复,而持续的高血管内皮生长因子表达和长期存在可能会在后期损害肌腱修复[68]。这是因为血管内皮生长因子的过度表达会产生渗漏和曲折的异常脉管系统,这些血管无法输送所需的氧气和营养物质[69]。此外,高水平血管内皮生长因子导致的血管生长已被证明可以维持炎症并增加瘢痕组织[59]。"

| [1] TEKAVEC E, JÖUD A, RITTNER R, et al. Population-based consultation patterns in patients with shoulder pain diagnoses. BMC Musculoskelet Disord. 2012;13:238. [2] YOSHIDA M, COLLIN P, JOSSEAUME T, et al. Post-operative rotator cuff integrity, based on Sugayas classification, can reflect abduction muscle strength of the shoulder. Knee Surg Sports Traumatol Arthrosc. 2018;26(1):161-168. [3] BOORMAN RS, MORE KD, HOLLINSHEAD RM, et al. What happens to patients when we do not repair their cuff tears? Five-year rotator cuff quality-of-life index outcomes following nonoperative treatment of patients with full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2018;27(3):444-448. [4] KARPINSKI KATRIN, PLACHEL FABIAN, GERHARDT CHRISTIAN, et al. Different expectations of patients and surgeons with regard to rotator cuff repair. J Shoulder Elbow Surg. 2022;31:1096-1105. [5] LE BT, WU XL, LAM PH, et al. Factors predicting rotator cuff retears: an analysis of 1000 consecutive rotator cuff repairs. Am J Sports Med. 2014;42(5):1134-1142. [6] DUQUIN TR, BUYEA C, BISSON LJ. Which method of rotator cuff repair leads to the highest rate of structural healing A systematic review. Am J Sports Med.2010;38(4):835-841. [7] TEUNIS T, LUBBERTS B, REILLY BT, et al. A systematic review and pooled analysis of the prevalence of rotator cuff disease with increasing age. J Shoulder Elbow Surg. 2014;23(12):1913-1921. [8] KIM JH, HONG IT, RYU KJ, et al. Retear rate in the late postoperative period after arthroscopic rotator cuff repair. Am J Sports Med. 2014; 42(11):2606-2613. [9] ROSSETTI L, KUNTZ LA, KUNOLD E, et al . The microstructure and micromechanics of the tendon-bone insertion. Nat Mater. 2017;16(6): 664-670. [10] CHEN C, WANG Z, ZHENG C, et al. Characterization of zinc and calcium spatial distribution at the fibrocartilage of rabbit patella-patellar tendon complex: A synchrotron radiation micro X-ray fluorescence study. J Orthopaedic Translat. 2014;2(4):242-243. [11] GENIN GM, THOMOPOULOS S. The tendon-to-bone attachment: Unification through disarray. Nat Mater. 2017;16(6):607-608. [12] BEDEIR YEHIA H, SCHUMAIER ADAM P, ABU-SHEASHA GHADA et al. Type 2 retear after arthroscopic single-row, double-row and suture bridge rotator cuff repair: a systematic review. Eur J Orthop Surg Traumatol. 2019;29:373-382. [13] LU H, LIU F, CHEN H, et al. The effect of low intensity pulsed ultrasound on bone tendon junction healing: Initiating after inflammation stage. J Orthop Res. 2016;34(10):1697-1706. [14] SCHEIBEL MARKUS, BROWN ANNA, WOERTLER KLAUS, et al. Preliminary results after rotator cuff reconstruction augmented with an autologous periosteal flap. Knee Surg Sports Traumatol Arthrosc. 2007;15(3):305-314. [15] ZHAO Y, DAI H F, LIU S S, et al. Therapeutic effect of autologous periosteum on the healing of tendon-bone interface in rabbit rotator cuff tear. Chinese Journal of Comparative Medicine. 2018;28(2):74-79. [16] WANG X, ZHAI R, YANG J, et al. Effect of hamstring tendon transfected with adenovirus-mediated transforming growth factor β 1 gene on histomorphology of tendon-bone interface healing after anterior cruciate ligament reconstruction in rabbits. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2015;29(12):1488. [17] FONT TELLADO S, BALMAYOR ER, VAN GRIENSVEN M. Strategies to engineer tendon/ligament-to-bone interface: Biomaterials,cells and growth factors. Adv Drug DelivRev. 2015;94:126-140. [18] 周勇,朱伟民,彭亮权,等.构建组织工程软骨支架材料:现状及未来[J].中国组织工程研究,2017,21(10):1604-1610. [19] BONANI WALTER,SINGHATANADGIGE WEERASAK,PORNANONG ARAMWIT, et al. Natural Origin Materials for Osteochondral Tissue Engineering. Adv Exp Med Biol. 2018;1058:3-30. [20] 刘嘉鑫,安丽萍,张广瑞,等.促进肩袖止点腱骨愈合的研究进展[J].中国骨伤,2020,33(7):684-688. [21] SABETKISH S, KAJBAFZADEH AM, SABETKISH N, et al. Whole-organ tissue engineering: decellularization and recellularization of three-dimensional matrix liver scaffolds. J Biomed Mater Res A. 2015;103(4): 1498-1508. [22] LIAO J, XU B, ZHANG R, et al. Applications of decellularized materials in tissue engineering: advantages, drawbacks and current improvements, and future perspectives. J Mater Chem B. 2020;8:10023-10049. [23] LU H, TANG Y, LIU F, et al. Comparative Evaluation of the Book-Type Acellular Bone Scaffold and Fibrocartilage Scaffold for Bone-Tendon Healing. J Orthop Res. 2019;37:1709-1722. [24] 廖昊燃,余伟林,胡庆翔,等.生物学方法促进肩袖腱骨愈合研究进展[J].中华肩肘外科电子杂志,2021,9(2):183-186. [25] ENCALADA-DIAZ I, COLE BJ, MACGILLIVRAY JD, et al . Rotator cuff repair augmentation using a novel polycarbonate polyurethane patch: preliminary results at 12 months’ follow-up. J Shoulder Elbow Surg. 2011;20(5):788-7894. [26] CHEN P, CUI L, FU S, et al. The 3D-Printed PLGA Scaffolds Loaded with Bone Marrow-Derived Mesenchymal Stem Cells Augment the Healing of Rotator Cuff Repair in the Rabbits. Cell Transplant. 2020;29: 963689720973647. [27] ATESOK K, DORAL MN, KARLSSON J, et al. Multilayer scaffolds in orthopaedic tissue engineering. Knee Surg Sports Traumatol Arthrosc. 2016;24(7):2365-2373. [28] LI X, CHENG R, SUN Z, et al. Flexible bipolar nanofibrous membranes for improving gradient microstructure in tendon-to-bone healing. Acta Biomater. 2017;61: 204-216. [29] TARAFDER S, BRITO JA, MINHAS S, et al. In situ tissue engineering of the tendon-to-bone interface by endogenous stem/progenitor cells. Biofabrication. 2019;12(1):015008. [30] BHARDWAJ N, DEVI D, MANDAL BB. Tissue-engineered cartilage: the crossroads of biomaterials, cells and stimulating factors. Macromol Biosci. 2015;15:153-182. [31] LEONG DANIEL J, SUN HUI B. Mesenchymal stem cells in tendon repair and regeneration: basic understanding and translational challenges. Ann N Y Acad Sci. 2016;1383:88-96. [32] ZEINEDDINE HA , FRUSH TJ, SALEH ZM, et al. Applications of Tissue Engineering in Joint Arthroplasty: Current Concepts Update. Orthopedic Clinics of North America. 2017;48(3):275. [33] BEITZEL K, SOLOVYOVA O, COTE MP, et al. The future role of mesenchymal stem cells in the management of shoulder disorders. Arthroscopy. 2013;29(10):1702-1711. [34] HERNIGOU P, FLOUZAT LACHANIETTE CH, DELAMBRE J, et al. Biologic augmentation of rotator cuff repair with mesenchymalstem cells during arthroscopy improves healing and prevents further tears: a case-controlled study. Int Orthop 2014;38(9):1811-1818. [35] KIDA Y, MORIHARA T, MATSUDA K, et al. Bone marrow-derived cells from the footprint infiltrate into the repaired rotator cuff. J Shoulder Elbow Surg. 2013;22(2):197-205. [36] CHEN HS, SU YT, CHAN TM, et al. Human adipose-derived stem cells accelerate the restoration of tensile strength of tendon and alleviate the progression of rotator cuff injury in a rat model. Cell Transplant. 2015;24(3):509-520. [37] KIM YS, SUNG CH, CHUNG SH, et al. Does an injection of adipose-derived mesenchymal stem cells loaded in fibringlue influence rotator cuff repair outcomes? A clinical and magnetic resonance imaging study. Am J Sports Med. 2017;45:2010-2018. [38] ZHENG CX, SUI BD, LIU N, et al. Adipose mesenchymal stem cells from osteoporotic donors preserve functionality and modulate systemic inflammatory microenvironment in osteoporotic cytotherapy. Sci Rep. 2018;8:5215. [39] LI CY, WU XY, TONG JB, et al. Comparative analysis of human mesenchymal stem cells from bone marrow and adipose tissue under xeno-free conditions for cell therapy. Stem Cell Res Ther. 2015;6:55. [40] VALENCIA MORA M, ANTUÑA ANTUÑA S, GARCÍA ARRANZ M, et al. Application of adipose tissue-derived stem cells in a rat rotator cuff repair model. Injury. 2014;45 Suppl 4:S22-S27. [41] GIMBLE JM, KATZ AJ, BUNNELL BA. Adipose-derived stem cells for regenerative medicine. Circ Res. 2007;100:1249-1260 [42] NAGURA I, KOKUBU T, MIFUNE Y, et al. Characterization of progenitor cells derived from torn human rotator cuff tendons by gene expression patterns of chondrogenesis, osteogenesis, and adipogenesis. J Orthop Surg Res. 2016;11:40. [43] SHEN W, CHEN J, YIN Z, et al. Allogenous tendon stem/progenitor cells in silk scaffold for functional shoulder repair. Cell Transplant. 2012; 21(5):943-958. [44] HUANG K, CHEN CA, DONG SK, et al. Differentiation Ability of Tendon-Derived Stem Cells and Histological Characteristics of Rotator Cuff Remnant on the Greater Tuberosity Degenerated with Age and Chronicity. Arthroscopy. 2022;38(4):1037-1048. [45] TAN Q, LUI PP, RUI YF, et al. Comparison of potentials of stem cells isolated from tendon and bone marrow for musculoskeletal tissue engineering. Tissue Eng Part A. 2012;18(7-8):840-851. [46] BI Y, EHIRCHIOU D, KILTS TM, et al. Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat Med. 2007;13(10):1219-1227. [47] BAI L, LI D, LI J, et al. Bioactive molecules derived from umbilicalcord mesenchymal stem cells. Acta Histochem. 2016;118(8):761-769. [48] PARK GY, KWON DR, LEE SC. Regeneration of Full-Thickness Rotator Cuff Tendon Tear After Ultrasound-Guided Injection With Umbilical Cord Blood-Derived Mesenchymal Stem Cells in a Rabbit Model. Stem Cells Transl Med. 2015;4:1344-1351. [49] KWON DR, KIM KL, MOON YS. Regeneration of Chronic Rotator Cuff Tear in a Rabbit Model: Synergetic Benefits of Human Umbilical Cord Blood-Derived Mesenchymal Stem Cells, Polydeoxyribonucleotides, and Microcurrent Therapy. Biomed Res Int. 2022;2022:6496773 [50] CHEN P, CUI L, FU SC, et al. The 3D-Printed PLGA Scaffolds Loaded with Bone Marrow-Derived Mesenchymal Stem Cells Augment the Healing of Rotator Cuff Repair in the Rabbits. Cell Transplant. 2020;29: 963689720973647. [51] JO CH, CHAI JW, JEONG EC, et al. Intratendinous Injection of Autologous Adipose Tissue-Derived Mesenchymal Stem Cells for the Treatment of Rotator Cuff Disease: A First-In-Human Trial. Stem Cells. 2018;36(9):1441-1450. [52] KAIZAWA Y, FRANKLIN A, LEYDEN J, et al. Augmentation of chronic rotator cuff healing using adipose-derived stem cell-seeded human tendon-derived hydrogel. J Orthop Res. 2019;37(4):877-886. [53] RAK KWON D, JUNG S, JANG J, et al. A 3-Dimensional Bioprinted Scaffold With Human Umbilical Cord Blood-Mesenchymal Stem Cells Improves Regeneration of Chronic Full-Thickness Rotator Cuff Tear in a Rabbit Model. Am J Sports Med. 2020;48(4):947-958. [54] CHEN Y, XU Y, LI M, et al. Application of Autogenous Urine-Derived Stem Cell Sheet Enhances Rotator Cuff Healing in a Canine Model. Am J Sports Med. 2020;48(14):3454-3466. [55] WÜRGLER HAURI CC, DOURTE LM, BARADET TC, et al. Temporal expression of 8 growth factors in tendon to bone healing in a rat supraspinatus model. J Shoulder Elbow Surg. 2007;16(5 Suppl): S198-S203. [56] KABUTO Y, MORIHARA T, SUKENARI T, et al. Stimulation of rotator cuff repair by sustained release of bone morphogenetic protein-7 using a gelatinhydrogel sheet. Tissue Eng Part A.2015;21(13-14):2025-2033. [57] CHEN P, CUI L, CHEN G, et al. The application of BMP-12-overexpressing mesenchymal stem cells loaded 3D-printed PLGA scaffolds in rabbit rotator cuff repair. Int J Biol Macromol. 2019;138:79-88. [58] LAMPLOT JD, ANGELINE M, ANGELES J, et al. Distinct effects of platelet-rich plasma and BMP13 on rotator cuff tendon injury healing in a rat model. Am J Sports Med. 2014;42(12):2877-2887. [59] HAN B, JONES IA, YANG Z, et al . Repair of rotator cuff tendon defects in aged rats using a growth factor injectable gel scaffold. Arthroscopy. 2020;36(3):629-637. [60] YAMAKADO K, KITAOKA K, YAMADA H, et al . The influence of mechanical stress on graft healing in a bone tunnel. Arthroscopy. 2002; 18(1):82-90. [61] WANG R, XU B, XU HG. Up-regulation of TGF-β promotes tendon- to- bone healing after anterior cruciate ligament reconstruction using bone marrow-derived mesenchymal stem cells through the TGF-β/MAPK signaling pathway in a New Zealand white rabbit model. Cell Physiol Biochem. 2017;41(1):213-226. [62] SHAH M, FOREMAN DM, FERGUSON MW. Neutralisation of TGF-beta 1 and TGF-beta 2 or exogenous addition of TGF-beta 3 to cutaneous rat wounds reduces scarring. J Cell Sci. 1995;108(Pt 3):985-1002. [63] MANNING CN, KIM HM, SAKIYAMA-ELBERT S, et al. Sustained delivery of transforming growth factor beta three enhances tendon- to- bone healing in a rat model. J Orthop Res. 2011;29(7):1099-1105. [64] ZHAO S, ZHAO J, DONG S, et al. Biological augmentation of rotator cuff repair using bFGF-loaded electrospun poly (lactide-co-glycolide) fibrous membranes. Int J Nanomedicine. 2014;9:2373-2385. [65] YONEMITSU R, TOKUNAGA T, SHUKUNAMI C, et al. Fibroblast Growth Factor 2 Enhances Tendon-to-Bone Healing in a Rat Rotator Cuff Repair of Chronic Tears. Am J Sports Med.2019;47:1701-1712. [66] LU LY, MA M, CAI JF, et al. Effects of Local Application of Adipose-Derived Stromal Vascular Fraction on Tendon-Bone Healing after Rotator Cuff Tear in Rabbits. Chin Med J (Engl). 2018;131(21):2620-2622. [67] HUANG Y, PAN M, SHU H, et al . Vascular endothelial growth factor enhances tendon-bone healing by activating Yes-associated protein for angiogenesis induction and rotator cuff reconstruction in rats. J Cell Biochem. 2020;121(3):2343-2353 [68] KORNTNER S, LEHNER C, GEHWOLF R, et al. Limiting angiogenesis to modulate scar formation. Adv Drug Deliv Rev. 2019;146:170-189 [69] JÄRVINEN TA. Neovascularisation in tendinopathy: from eradication to stabilisation? Br J Sports Med. 2020;54(1):1-2. |
| [1] | Nong Fuxiang, Jiang Zhixiong, Li Yinghao, Xu Wencong, Shi Zhilan, Luo Hui, Zhang Qinglang, Zhong Shuang, Tang Meiwen. Bone cement augmented proximal femoral nail antirotation for type A3.3 intertrochanteric femoral fracturalysis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-10. |
| [2] | Sun Kexin, Zeng Jinshi, Li Jia, Jiang Haiyue, Liu Xia. Mechanical stimulation enhances matrix formation of three-dimensional bioprinted cartilage constructs [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(在线): 1-7. |
| [3] | Dang Yi, Du Chengyan, Yao Honglin, Yuan Nenghua, Cao Jin, Xiong Shan, Zhang Dingmei, Wang Xin. Hormonal osteonecrosis and oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1469-1476. |
| [4] | Pan Zhongjie, Qin Zhihong, Zheng Tiejun, Ding Xiaofei, Liao Shijie. Targeting of non-coding RNAs in the pathogenesis of the osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1441-1447. |
| [5] | Cai Zhihao, Xie Zhaoyong. Femoral neck anteversion measurement assessment: how to establish a unified method and standard [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(9): 1448-1454. |
| [6] | Wang Ji, Zhang Min, Yang Zhongya, Zhang Long. A review of physical activity intervention in type 2 diabetes mellitus with sarcopenia [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1272-1277. |
| [7] | Nie Chenchen, Su Kaiqi, Gao Jing, Fan Yongfu, Ruan Xiaodi, Yuan Jie, Duan Zhaoyuan, Feng Xiaodong. The regulatory role of circular RNAs in cerebral ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1286-1291. |
| [8] | Gao Yu, Han Jiahui, Ge Xin. Immunoinflammatory microenvironment after spinal cord ischemia-reperfusion injury [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1300-1305. |
| [9] | Xu Xingxing, Wen Chaoju, Meng Maohua, Wang Qinying, Chen Jingqiao, Dong Qiang. Carbon nanomaterials in oral implant [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1062-1070. |
| [10] | Yang Yitian, Wang Lu, Yao Wei, Zhao Bin. Application of the interaction between biological scaffolds and macrophages in bone regeneration [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1071-1079. |
| [11] | Li Cheng, Zheng Guoshuang, Kuai Xiandong, Yu Weiting. Alginate scaffold in articular cartilage repair [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1080-1088. |
| [12] | Xu Cong, Zhao He, Sun Yan. Regeneration of facial nerve injury repaired by biomaterial nerve conduits [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1089-1095. |
| [13] | Chen Shisong, Liu Xiaohong, Xu Zhiyun. Current status and prospects of bioprosthetic heart valves [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1096-1102. |
| [14] | Lu Di, Zhang Cheng, Duan Rongquan, Liu Zongxiang. Osteoinductive properties of calcium phosphate ceramic bone repair materials [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1103-1109. |
| [15] | Shi Yehong, Wang Cheng, Chen Shijiu. Early thrombosis and prevention of small-diameter blood vessel prosthesis [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(7): 1110-1116. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||