Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (15): 2426-2431.doi: 10.12307/2022.602
Previous Articles Next Articles
Effect and application of nerve growth factor in the treatment and repair of osteoarthritis
Zhang Yong1, 2, Li Feifei1, Wang Buyu1, Huang Wenliang1, Deng Jiang1
- 1Central Laboratory of Third Affiliated Hospital of Zunyi Medical University, Zunyi 563099, Guizhou Province, China; 2Zunyi Bozhou District People’s Hospital, Zunyi 563199, Guizhou Province, China
-
Received:2021-08-30Revised:2021-10-19Accepted:2021-11-19Online:2022-05-28Published:2022-01-06 -
Contact:Deng Jiang, Professor, Chief physician, Doctoral supervisor, Central Laboratory of Third Affiliated Hospital of Zunyi Medical University, Zunyi 563099, Guizhou Province, China -
About author:Zhang Yong, Attending physician, Central Laboratory of Third Affiliated Hospital of Zunyi Medical University, Zunyi 563099, Guizhou Province, China; Zunyi Bozhou District People’s Hospital, Zunyi 563199, Guizhou Province, China -
Supported by:National Natural Science Foundation of China, No. 81660367 (to DJ); Guizhou Science and Technology Fund, No. (2016) 1420 (to DJ); Zunyi Science and Technology Joint Technology Research and Development Fund Project, No. (2018) 184 (to DJ)
CLC Number:
Cite this article
Zhang Yong, Li Feifei, Wang Buyu, Huang Wenliang, Deng Jiang. Effect and application of nerve growth factor in the treatment and repair of osteoarthritis[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(15): 2426-2431.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
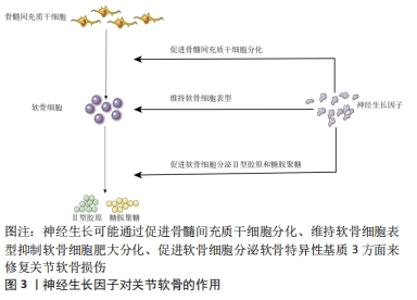
2.1 神经生长因子对关节软骨的作用 骨关节炎主要的病理表现为关节软骨不同程度的退变和磨损,这种改变也是治疗的难点。骨髓间充质干细胞是组织工程最常用的种子细胞,神经生长因子在这个过程中可能通过以下3方面发挥重要作用。 2.1.1 促进干细胞软骨分化 将骨髓间充质干细胞定向诱导分化为软骨细胞,促进软骨组织的修复是组织工程常用的方法。ZHAN等[11]报道发现,神经生长因子可以通过PI3K/AKT信号通路诱导骨髓间充质干细胞的软骨分化,并增强软骨细胞表达Ⅱ型胶原,增加透明软骨形成,促进软骨缺损组织的修复;骨关节炎软骨组织中软骨细胞明显减少,促进软骨分化后,软骨细胞增加,从而促进表达软骨基质,达到骨关节炎软骨损伤修复的目的。 2.1.2 维持软骨细胞表型、抑制肥大分化 发育学研究发现软骨细胞肥大分化发育为肥大软骨细胞,肥大的软骨细胞表达Ⅰ型胶原和钙沉积,转变成骨细胞;抑制软骨细胞肥大分化[12],一定程度上就可以保持软骨组织中软骨细胞数量的恒定,维持糖胺聚糖和Ⅱ型胶原分泌量,减少或延缓骨关节炎的进展。HUANG等[13]在体外用ATDC5软骨细胞系作为实验细胞,发现神经生长因子刺激后,可抑制软骨细胞肥大分化。GIGANTE等[14]发现神经生长因子对软骨细胞增殖、细胞活力及Ⅰ型胶原、Ⅱ型胶原和基质金属蛋白酶13的基因表达无影响,只是可降低软骨细胞基质金属蛋白酶3的表达,通过细胞染色同样也发现神经生长因子可能抑制软骨细胞的肥大分化,可能神经生长因子对软骨降解酶的促进作用不是主要的生理功能。 2.1.3 促进软骨细胞分泌软骨基质 软骨组织主要由软骨细胞及周围的软骨基质组成,软骨基质主要包括糖胺聚糖和Ⅱ型胶原,是关节软骨特异性标志物。LU等[15]同样发现神经生长因子刺激骨髓间充质干细胞可明显上调软骨特异性标志物(糖胺聚糖、Ⅱ型胶原)的表达;两人使用的神经生长因子均来源于广西眼镜蛇毒,并且发现眼镜蛇毒来源的神经生长因子和鼠来源的神经生长因子均明显促进骨髓间充质干细胞增殖并上调软骨特异性基因的表达,包括SOX9、糖胺聚糖和Ⅱ型胶原,其中SOX9是软骨分化调控的关键基因;但眼镜蛇毒来源的神经生长因子降低了Ⅰ型胶原和RUNT相关转录因子2(RUNX2)的表达水平,具体原因及机制作者未做进一步实验及分析,说明神经生长因子可一定程度上维持软骨细胞表型,抑制软骨细胞肥大分化[16]。TAN等[17]发现神经生长因子可直接上调人软骨细胞SOX9、糖胺聚糖和Ⅱ型胶原基因表达,下调Ⅰ型胶原基因表达水平,从而促进软骨基质的表达,增加软骨的修复,说明神经生长因子对软骨组织修复有积极作用。SANG等[18]在小鼠胫骨骨折中发现,神经生长因子可促进骨折后骨痂的形成,促进骨折愈合,在愈合过程中发现,骨折断端中神经生长因子表达明显升高,升高的神经生长因子通过提高Ⅱ型胶原和 SOX9 mRNA 的表达水平来促进软骨分化,骨折断端出现软骨后,进一步钙沉淀或者矿化,分化为骨组织而促进骨折愈合,表明神经生长因子首先是促进软骨形成,研究表明,这与TGF/BMP-Smad通路中Smad4因子密切相关[12]。 总之,在这些实验中证实神经生长因子可通过以上3方面促进关节软骨修复,在这个过程中,神经生长因子可能单独通过某一方面或者多方面共同促进修复,见图3。"
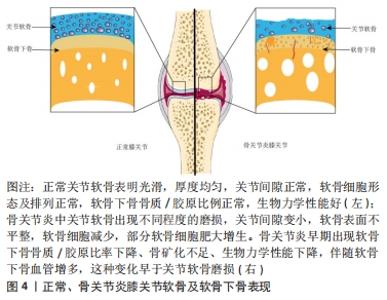
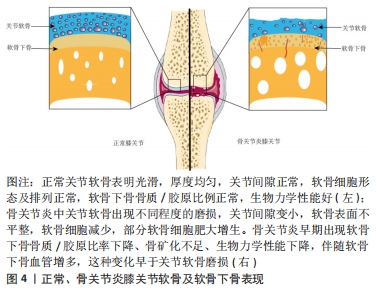
2.2 神经生长因子对软骨下骨的作用 软骨下骨是位于关节软骨下方钙化骨深面的骨组织,由软骨下骨板和底层的骨小梁组成[19]。软骨下骨板位于钙化软骨深部,是一层薄的皮质骨,是一种多孔的骨组织,通透性和渗透性佳,是关节软骨和软骨下骨骨小梁之间物质交换和信号传输的重要通道,大量的动静脉、神经通过这些管道将分枝穿入钙化软骨;下层骨小梁主要是支撑、减震及提供营养和调节新陈代谢的作用[20]。骨髓腔中动静脉、神经通过软骨下骨将营养物质运送给软骨并将代谢产物运走,为软骨下骨和软骨之间的物质转运起重要作用;同时软骨下骨与软骨组成一个动态的承重结构,吸收和转移关节受到的力学负荷,维持关节内环境的稳态和关节的形状[21]。 软骨下骨的改变是骨关节炎发生发展的关键标志之一[22],见图4。研究发现髋关节骨关节炎负重区软骨下骨的骨质/胶原比率下降、骨矿化不足,生物力学性能下降[21,23],体外实验同样发现成骨细胞的表型异常,合成活性增强,但矿化能力下降;此外,骨关节炎患者软骨下成骨细胞可释放多种蛋白酶、炎性递质与生长因子,并可促进上层软骨基质降解。"
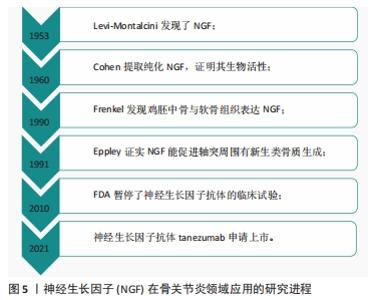
尽管长期以来人们一直认为关节软骨退变和磨损是导致骨关节炎的主要原因,并且许多疗法都旨在保护关节软骨,但是越来越多的证据表明,骨关节炎关节软骨下骨的完整性和重塑过程对维持关节的动态平衡同样重要[24-25]。最初发现神经生长因子对神经损伤修复时,在神经周围出现新生成的骨组织;同样在骨折后,神经生长因子出现在新形成的毛细血管内,包含骨髓基质细胞、成骨细胞和内皮细胞呈神经生长因子阳性,在随后的骨痂形成过程中,骨膜基质也呈神经生长因子阳性;在胫骨骨折的小鼠中,神经生长因子还通过增加成骨细胞的数量来刺激骨痂的形成;局部应用β-神经生长因子治疗大鼠颅骨缺损可诱导β-Ⅲ-微管蛋白和血管内皮生长因子的表达,提示其对神经元生长和血管生成均有调节作用;在大鼠骨关节炎模型中发现血清骨保护素及软骨下骨降钙素基因相关肽(CGRP)、神经生长因子的表达随着骨关节炎的进展发生改变,说明神经生长因子是骨关节炎的重要参与因子[26];JIANG等[27]研究发现神经生长因子或者神经生长因子抗体均会破坏人关节软骨稳态,诱导软骨细胞钙化,促进软骨下骨重塑及成骨分化,分析可能与Hedgehog通路有关;YU等[28]研究发现,神经生长因子促进人软骨细胞中FGF2的表达,增加TrkA介导的血管生成,导致软骨下骨血管生长加重骨关节炎的发展;有研究者在关节软骨下骨注射β-神经生长因子后,发现局部骨小梁数量增加,骨矿物质密度增大,表现为软骨生成减少和总骨量增加,并证明该作用是通过激活Wnt通路实现的[30]。神经和血管总是伴随生长,神经生长因子促进神经轴突生长是众所周知的,神经生长因子也能促进血管生长。软骨下骨的血管生长都会促进软骨细胞肥大分化,进而发生软骨细胞钙化,诱导骨形成。软骨下骨的血管生成和成骨分化加速骨关节炎的发生和发展[23,29];特别是成骨方面,其他一些证据进一步得到支持,如JIN等[30]用广西眼镜蛇毒分离的神经生长因子联合陶瓷支架,植入大鼠的颅骨发现神经生长因子可以促进成骨分化及诱导骨形成;LI 等[31]同样研究发现神经生长因子/TrkA信号在应力性骨折修复中起着至关重要的作用;同样在临床中发现,合并脑外伤的骨折患者,骨折愈合时间明显比单纯骨折患者短,认为这可能与神经生长因子增高有关[32];RIVERA等[33]研究发现局部注射β-神经生长因子可以通过促进软骨向骨转化加速骨折的修复。总之,这些研究均表明,神经生长因子在一定条件下可促进软骨细胞成骨分化,加重骨关节炎的发生及发展。当然也有学者发现,软骨下骨的修复同样可加速软骨组织的愈合[34-35]。 2.3 神经生长因子抗体与骨关节炎疼痛 疼痛是骨关节炎最主要的临床表现,因疼痛给全世界公共卫生造成的巨大负担,目前常用的镇痛药物虽然有一定效果,但是也存在较大的不良反应,限制了广泛的使用,所以对新的止痛药物开发迫在眉睫。最开始有研究发现局部注射神经生长因子可引起动物和人类的疼痛和痛觉过敏[36];而从骨关节炎患者中分离的关节软骨细胞可上调神经生长因子和TrkA的水平,并且动物关节中注射神经生长因子可明显增加疼痛反应,说明神经生长因子是引起骨关节炎疼痛的主要中间介质。鉴于神经生长因子在骨关节炎疼痛机制中的重要性,促使了神经生长因子抗体作为药物治疗骨关节炎的开发和临床研究[37]。虽然各种抑制剂或抗体已经在临床试验阶段取得了一些效果,但是目前只有Tanezumab和Fasinumab正在接受骨关节炎的Ⅲ期临床试验[38]。大量研究表明证实神经生长因子抗体对膝关节和髋关节骨关节炎的疼痛、功能较安慰剂组均有显著改善[39-41],与非类固醇抗炎药相比,神经生长因子抗体可显著减轻关节疼痛并改善功能,持续时间长达8周[42]。一项随机、双盲、多中心试验中发现,Tanezumab注射对骨关节炎的疼痛和功能明显改善[43];另在一项包括8 145 例骨关节炎患者的荟萃分析研究中发现,神经生长因子抗体治疗后骨关节炎患者的WOMAC指数明显改善[39];在一项对849 例患者的研究发现(欧洲人占87.5%,日本人占12.5%),Tanezumab对影像学上认为严重的骨关节炎患者有效[44]。然而较高测试剂量的神经生长因子抗体也出现一些不良反应,包括一过性的头痛、上呼吸道感染和感觉异常,以及最严重的骨坏死和关节快速破坏,从而限制了它的广泛使用及临床研究进展[29],且在研究中发现,当Tanezumab与非类固醇抗炎药联合使用时,全关节置换的发生率随着Tanezumab剂量的增加而增加,等于安慰剂组的两三倍,即使在使用较低剂量的情况下,使用Tanezumab治疗后关节恶化的风险仍然大于非类固醇抗炎药治疗。除了Tanezumab 外,Fulranumab治疗骨关节炎的研究中也发现不良反应与使用剂量有一定的相关性。对于发生关节快速破坏的原因,分析可能一方面是痛觉的丧失导致关节负荷过重并迅速恶化,另一方面在动物研究中发现神经生长因子抗体与破骨细胞的异常激活有关[45-47]。 神经生长因子在骨关节炎领域应用的研究进程,见图5。"
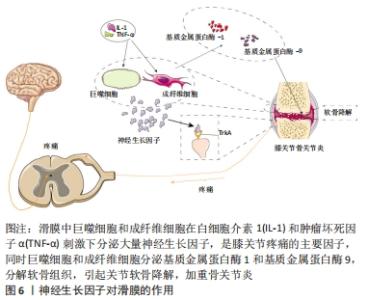
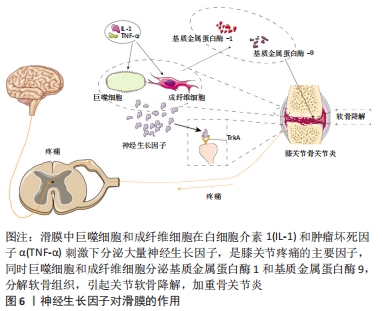
2.4 神经生长因子对滑膜的作用 关节滑膜是关节囊的内层结构,为一层疏松结缔组织,覆盖于关节囊内层表面。通过关节镜和病理学检查发现,骨关节炎滑膜炎症时一种亚急性类型的炎症,这种炎症反应早于软骨磨损[48],早期主要表现仅为轻度内膜增生、散在的炎性细胞和小的血管周围浸润[49]。滑膜细胞在正常膝关节中表达低水平的神经生长因子及其受体TrkA,当关节内环境出现炎症反应后,许多炎性因子如白细胞介素1β和肿瘤坏死因子α增加,这两种因子主要由滑膜细胞、巨噬细胞和软骨细胞产生[50],可促进滑膜细胞表达神经生长因子和TrkA,说明神经生长因子及其受体TrkA在骨关节炎中的过度表达可能参与调节而不是诱导关节炎症反应。有研究表明,神经生长因子对关节滑膜细胞有丝分裂有促进作用,所以在炎症刺激调节下,神经生长因子的失调可能导致滑膜细胞异常增殖,从而可能影响炎症性关节炎的炎症和增殖级联反应。神经生长因子主要在滑膜血管平滑肌层表达,滑膜内神经末梢分布广泛,在滑膜血管周围分布尤为明显,增加的神经生长因子刺激末梢神经可能是疼痛的主要原因。神经生长因子在膝关节骨关节炎滑膜内下方的表达明显高于它们在滑膜其他处的表达,与临床观察到的膝骨关节炎患者膝关节内下方疼痛和压痛部位一致。另一方面,在白细胞介素1β和肿瘤坏死因子α的刺激下,基质金属蛋白酶的过度产生是造成软骨丢失的主要原因,基质金属蛋白酶1和基质金属蛋白酶13在细胞外基质降解过程中起主要作用,这两种胶原酶都参与Ⅱ型胶原的降解,而基质金属蛋白酶1主要由滑膜产生,基质金属蛋白酶13是软骨细胞的产物[51]。 神经生长因子对滑膜的作用,见图6。"
| [1] ROCCO ML, SOLIGO M, MANNI L, et al. Nerve Growth Factor: Early Studies and Recent Clinical Trials. Curr Neuropharmacol. 2018;16(10): 1455-1465. [2] DENK F, BENNETT DL, MCMAHON SB. Nerve Growth Factor and Pain Mechanisms. Annu Rev Neurosci. 2017;40:307-325. [3] DO CARMO S, KANNEL B, CUELLO AC. Nerve Growth Factor Compromise in Down Syndrome. Front Aging Neurosci. 2021;13:719-507. [4] PFLUGFELDER SC, MASSARO-GIORDANO M, PEREZ VL, et al. Topical Recombinant Human Nerve Growth Factor (Cenegermin) for Neurotrophic Keratopathy: A Multicenter Randomized Vehicle-Controlled Pivotal Trial. Ophthalmology. 2020;127(1):14-26. [5] MONTAGNOLI C, TIRIBUZI R, CRISPOLTONI L, et al. β-NGF and β-NGF receptor upregulation in blood and synovial fluid in osteoarthritis. Biol Chem. 2017;398(9):1045-1054. [6] JIANG Y, HU C, YU S, et al. Cartilage stem/progenitor cells are activated in osteoarthritis via interleukin-1β/nerve growth factor signaling. Arthritis Res Ther. 2015;17:327. [7] 张滋彬,万千千,覃文聘,等. 神经-骨偶联机制促进骨关节退行性病发展的研究进展. 临床口腔医学杂志,2021,37(5):313-315. [8] CHARTIER SR, MITCHELL SA, MAJUTA LA, et al. Immunohistochemical localization of nerve growth factor, tropomyosin receptor kinase A, and p75 in the bone and articular cartilage of the mouse femur. Mol Pain. 2017;13:1744806917745465. [9] MANNI L, LUNDEBERG T, FIORITO S, et al. Nerve growth factor release by human synovial fibroblasts prior to and following exposure to tumor necrosis factor-alpha, interleukin-1 beta and cholecystokinin-8: the possible role of NGF in the inflammatory response. Clin Exp Rheumatol. 2003;21(5):617-624. [10] KOONS GL, DIBA M, MIKOS AG. Materials design for bone-tissue engineering. Nat Rev Mater. 2020;5(8):584-603. [11] ZHAN X, CAI P, LEI D, et al. Comparative profiling of chondrogenic differentiation of mesenchymal stem cells (MSCs) driven by two different growth factors. Cell Biochem Funct. 2019;37(5):359-367. [12] YAN J, LI J, HU J, et al. Smad4 deficiency impairs chondrocyte hypertrophy via the Runx2 transcription factor in mouse skeletal development. J Biol Chem. 2018;293(24):9162-9175. [13] HUANG H, SHANK G, MA L, et al. Nerve growth factor induced after temporomandibular joint inflammation decelerates chondrocyte differentiation. Oral Dis. 2013;19(6):604-610. [14] GIGANTE A, SENESI L, MANZOTTI S, et al. Effect of nerve growth factor on cultured human chondrocytes. J Biol Regul Homeost Agents. 2016;30(4 Suppl 1):1-6. [15] LU Z, LEI D, JIANG T, et al. Nerve growth factor from Chinese cobra venom stimulates chondrogenic differentiation of mesenchymal stem cells. Cell Death Dis. 2017;8(5):e2801. [16] MIAO Z, LU Z, LUO S, et al. Murine and Chinese cobra venom‑derived nerve growth factor stimulate chondrogenic differentiation of BMSCs in vitro: A comparative study. Molecular Medicine Reports. 2018;18(3): 3341-3349. [17] TAN J, LU Z, MIAO Z, et al. Effect of NGF From Venom of Chinese Cobra (Naja Atra)on Chondrocytes Proliferation and Metabolism In Vitro. J Cell Biochem. 2017;118(12):4308-4316. [18] SANG XG, WANG ZY, CHENG L, et al. Analysis of the mechanism by which nerve growth factor promotes callus formation in mice with tibial fracture. Exp Ther Med. 2017;13(4):1376-1380. [19] DONELL S. Subchondral bone remodelling in osteoarthritis. EFORT Open Rev. 2019;4(6): 221-229. [20] SALTZMAN BM, RIBOH JC. Subchondral Bone and the Osteochondral Unit: Basic Science and Clinical Implications in Sports Medicine. Sports Health. 2018;10(5):412-418. [21] HU W, CHEN Y, DOU C, et al. Microenvironment in subchondral bone: predominant regulator for the treatment of osteoarthritis. Ann Rheum Dis. 2020;80(4):413-422. [22] HOLZER LA, KRAIGER M, TALAKIC E, et al. Microstructural analysis of subchondral bone in knee osteoarthritis. Osteoporos Int. 2020;31(10): 2037-2045. [23] HU Y, CHEN X, WANG S, et al. Subchondral bone microenvironment in osteoarthritis and pain. Bone Res. 2021;9(1):20. [24] LU J, ZHANG H, CAI D, et al. Positive-Feedback Regulation of Subchondral H-Type Vessel Formation by Chondrocyte Promotes Osteoarthritis Development in Mice. J Bone Miner Res. 2018;33(5): 909-920. [25] ZHU X, CHAN YT, YUNG PSH, et al. Subchondral Bone Remodeling: A Therapeutic Target for Osteoarthritis. Front Cell Dev Biol. 2020;8:607-764. [26] 常琦,阮贞,王宪伟,等.骨关节炎模型大鼠血清骨保护素及软骨下骨降钙素基因相关肽、神经生长因子表达及Mankin评分变化[J].中国老年学杂志,2019,39(4):909-913. [27] JIANG Y, TUAN RS. Role of NGF-TrkA signaling in calcification of articular chondrocytes. FASEB J. 2019;33(9):10231-10239. [28] YU X, QI Y, ZHAO T, et al. NGF increases FGF2 expression and promotes endothelial cell migration and tube formation through PI3K/Akt and ERK/MAPK pathways in human chondrocytes. Osteoarthritis Cartilage. 2019;27(3):526-534. [29] MILLER RE, BLOCK JA, MALFAIT AM. Nerve growth factor blockade for the management of osteoarthritis pain: what can we learn from clinical trials and preclinical models? Curr Opin Rheumatol. 2017;29(1):110-118. [30] JIN P, YIN F, HUANG L, et al. Guangxi cobra venom-derived NGF promotes the osteogenic and therapeutic effects of porous BCP ceramic. Exp Mol Med. 2017;49(4):e312. [31] LI Z, MEYERS CA, CHANG L, et al. Fracture repair requires TrkA signaling by skeletal sensory nerves. J Clin Invest. 2019;129(12):5137-5150. [32] ZHANG R, LIANG Y, WEI S. The expressions of NGF and VEGF in the fracture tissues are closely associated with accelerated clavicle fracture healing in patients with traumatic brain injury. Ther Clin Risk Manag. 2018;14:2315-2322. [33] RIVERA KO, RUSSO F, BOILEAU RM, et al. Local injections of beta-NGF accelerates endochondral fracture repair by promoting cartilage to bone conversion. Sci Rep. 2020;10(1):22241. [34] YU F, LI M, YUAN Z, et al. Mechanism research on a bioactive resveratrol- PLA-gelatin porous nano-scaffold in promoting the repair of cartilage defect. Int J Nanomedicine. 2018;13:7845-7858. [35] SCHLICHTING K, SCHELL H, KLEEMANN RU, et al. Influence of scaffold stiffness on subchondral bone and subsequent cartilage regeneration in an ovine model of osteochondral defect healing. Am J Sports Med. 2008;36(12):2379-2391. [36] EPSTEIN ME. Anti-nerve growth factor monoclonal antibody: a prospective new therapy for canine and feline osteoarthritis. Vet Rec. 2019;184(1):20-22. [37] SHANG X, WANG Z, TAO H. Mechanism and therapeutic effectiveness of nerve growth factor in osteoarthritis pain. Ther Clin Risk Manag. 2017;13:951-956. [38] MONT MA, CARRINO JA, NEMETH MA, et al. Postoperative Outcome of Patients Who Underwent Total Joint Replacement During the Tanezumab Phase 3 Osteoarthritis Development Program: A 24-Week Observational Study. Surg Technol Int. 2021;38:467-477. [39] SEAH KTM, RAMMANOHAR J, SUTTON J, et al. The Effectiveness of Anti-Nerve Growth Factor Monoclonal Antibodies in the Management of Pain in Osteoarthritis of the Hip and Knee: A PRISMA Systematic Review and Meta-Analysis. Pain Med. 2021;22(5):1185-1204. [40] BERENBAUM F, BLANCO F J, GUERMAZI A, et al. Subcutaneous tanezumab for osteoarthritis of the hip or knee: efficacy and safety results from a 24-week randomised phase III study with a 24-week follow-up period. Ann Rheum Dis. 2020;79(6):800-810. [41] FAN ZR, MA JX, WANG Y, et al. Efficacy and safety of tanezumab administered as a fixed dosing regimen in patients with knee or hip osteoarthritis: a meta-analysis of randomized controlled phase III trials. Clin Rheumatol. 2021;40(6):2155-2165. [42] BANNWARTH B, KOSTINE M. Targeting nerve growth factor (NGF) for pain management: what does the future hold for NGF antagonists? Drugs. 2014;74(6):619-626. [43] SCHNITZER T J, EASTON R, PANG S, et al. Effect of Tanezumab on Joint Pain, Physical Function, and Patient Global Assessment of Osteoarthritis Among Patients With Osteoarthritis of the Hip or Knee: A Randomized Clinical Trial. JAMA. 2019;322(1):37-48. [44] BERENBAUM F, LANGFORD R, PERROT S, et al. Subcutaneous tanezumab for osteoarthritis: Is the early improvement in pain and function meaningful and sustained? Eur J Pain. 2021;25(7):1525-1539. [45] CHEN S, FU P, WU H, et al. Meniscus, articular cartilage and nucleus pulposus: a comparative review of cartilage-like tissues in anatomy, development and function. Cell Tissue Res. 2017;370(1):53-70. [46] WISE BL, SEIDEL MF, LANE NE. The evolution of nerve growth factor inhibition in clinical medicine. Nat Rev Rheumatol. 2021;17(1):34-46. [47] XU L, NWOSU L N, BURSTON J J, et al. The anti-NGF antibody muMab 911 both prevents and reverses pain behaviour and subchondral osteoclast numbers in a rat model of osteoarthritis pain. Osteoarthritis Cartilage. 2016;24(9):1587-1595. [48] INGALE D, KULKARNI P, ELECTRICWALA A, et al. Synovium-Synovial Fluid Axis in Osteoarthritis Pathology: A Key Regulator of the Cartilage Degradation Process. Genes (Basel). 2021;12(7):989. [49] PESSLER F, DAI L, DIAZ-TORNE C, et al. The synovitis of “non-inflammatory” orthopaedic arthropathies: a quantitative histological and immunohistochemical analysis. Ann Rheum Dis. 2008;67(8):1184-1187. [50] UDOMSINPRASERT W, JINAWATH A, TEERAWATTANAPONG N, et al. Interleukin-34 overexpression mediated through tumor necrosis factor-alpha reflects severity of synovitis in knee osteoarthritis. Sci Rep. 2020; 10(1):7987. [51] BENITO MJ, VEALE DJ, FITZGERALDO, et al. Synovial tissue inflammation in early and late osteoarthritis. Ann Rheum Dis. 2005;64(9):1263-1267. [52] WAN QQ, QIN WP, MA YX, et al. Crosstalk between Bone and Nerves within Bone. Adv Sci (Weinh). 2021;8(7):2003390. [53] FOERTSCH S, HAFFNER-LUNTZER M, KRONER J, et al. Chronic psychosocial stress disturbs long-bone growth in adolescent mice. Dis Model Mech. 2017;10(12):1399-1409. [54] HAFFNER-LUNTZER M, FOERTSCH S, FISCHER V, et al. Chronic psychosocial stress compromises the immune response and endochondral ossification during bone fracture healing via beta-AR signaling. Proc Natl Acad Sci U S A. 2019;116(17):8615-8622. |
| [1] | Zhang Jichao, Dong Yuefu, Mou Zhifang, Zhang Zhen, Li Bingyan, Xu Xiangjun, Li Jiayi, Ren Meng, Dong Wanpeng. Finite element analysis of biomechanical changes in the osteoarthritis knee joint in different gait flexion angles [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1357-1361. |
| [2] | Yao Xiaoling, Peng Jiancheng, Xu Yuerong, Yang Zhidong, Zhang Shuncong. Variable-angle zero-notch anterior interbody fusion system in the treatment of cervical spondylotic myelopathy: 30-month follow-up [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1377-1382. |
| [3] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [4] | Kan Houming, Fan Lijun, Chen Xuetai, Shen Wen. Application of platelet-rich plasma in neuropathic pain [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1286-1292. |
| [5] | Wang Baojuan, Zheng Shuguang, Zhang Qi, Li Tianyang. Miao medicine fumigation can delay extracellular matrix destruction in a rabbit model of knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1180-1186. |
| [6] | Xiao Yang, Gong Liqiong, Fei Jing, Li Leiji. Effect of electroacupuncture on nerve growth factor and its receptor expression in facial nerve nucleus after facial nerve injury in rabbits [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1253-1259. |
| [7] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [8] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [9] | Liu Dongcheng, Zhao Jijun, Zhou Zihong, Wu Zhaofeng, Yu Yinghao, Chen Yuhao, Feng Dehong. Comparison of different reference methods for force line correction in open wedge high tibial osteotomy [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 827-831. |
| [10] | Zhou Jianguo, Liu Shiwei, Yuan Changhong, Bi Shengrong, Yang Guoping, Hu Weiquan, Liu Hui, Qian Rui. Total knee arthroplasty with posterior cruciate ligament retaining prosthesis in the treatment of knee osteoarthritis with knee valgus deformity [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(6): 892-897. |
| [11] | He Junjun, Huang Zeling, Hong Zhenqiang. Interventional effect of Yanghe Decoction on synovial inflammation in a rabbit model of early knee osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 694-699. |
| [12] | Lin Xuchen, Zhu Hainian, Wang Zengshun, Qi Tengmin, Liu Limin, Suonan Angxiu. Effect of xanthohumol on inflammatory factors and articular cartilage in a mouse mode of osteoarthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 676-681. |
| [13] | Lu Qinxue, Xu Ning, Yang Yinglan, Han Qianqian, Duanmu Xianyu, Guo Yuwei, Han Qing. Femoroacetabular impingement: strength trainings for nerve-muscle, peripheral muscle and core muscle [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 786-791. |
| [14] | Xu Lei, Han Xiaoqiang, Zhang Jintao, Sun Haibiao. Hyaluronic acid around articular chondrocytes: production, transformation and function characteristics [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(5): 768-773. |
| [15] | He Yunying, Li Lingjie, Zhang Shuqi, Li Yuzhou, Yang Sheng, Ji Ping. Method of constructing cell spheroids based on agarose and polyacrylic molds [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(4): 553-559. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||



