Chinese Journal of Tissue Engineering Research ›› 2022, Vol. 26 ›› Issue (19): 3084-3089.doi: 10.12307/2022.390
Previous Articles Next Articles
Effect of mesenchymal stem cells on negative regulation of immune response of myeloid-derived suppressor cells
Min Keting1, 2, Zhang Yiguo1, 2, Yang Hao2, Lü Xin1, 2
- 1Wannan Medical College, Wuhu 241002, Anhui Province, China; 2Shanghai Pulmonary Hospital of Tongji University, Shanghai 200433, China
-
Received:2021-06-04Revised:2021-07-16Accepted:2021-08-03Online:2022-07-08Published:2021-12-29 -
Contact:Lü Xin, PhD, Professor, Wannan Medical College, Wuhu 241002, Anhui Province, China; Shanghai Pulmonary Hospital of Tongji University, Shanghai 200433, China Yang Hao, PhD, Attending physician, Shanghai Pulmonary Hospital of Tongji University, Shanghai 200433, China -
About author:Min Keting, Master candidate, Wannan Medical College, Wuhu 241002, Anhui Province, China; Shanghai Pulmonary Hospital of Tongji University, Shanghai 200433, China Zhang Yiguo, Master candidate, Wannan Medical College, Wuhu 241002, Anhui Province, China; Shanghai Pulmonary Hospital of Tongji University, Shanghai 200433, China Min Keting and Zhang Yiguo contributed equally to this article. -
Supported by:National Natural Science Foundation of China, No. 82000085 (to YH), 81871601 (to LX)
CLC Number:
Cite this article
Min Keting, Zhang Yiguo, Yang Hao, Lü Xin. Effect of mesenchymal stem cells on negative regulation of immune response of myeloid-derived suppressor cells[J]. Chinese Journal of Tissue Engineering Research, 2022, 26(19): 3084-3089.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
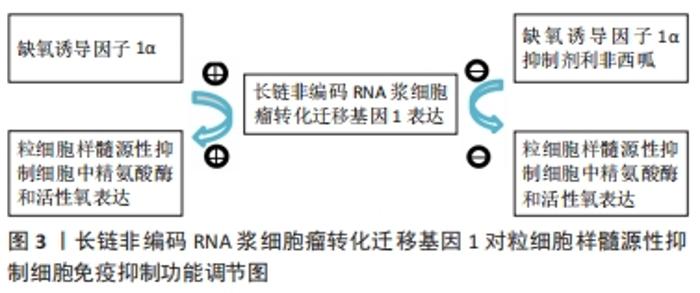
2.2.1 精氨酸酶 精氨酸酶能将精氨酸水解为鸟氨酸和尿素,提高精氨酸酶表达可导致组织微环境中精氨酸含量降低。精氨酸酶通过降低T细胞CD3δ表达或阻止T细胞周期调节因子和细胞周期调节蛋白依赖激酶4表达来抑制T细胞增殖[4]。研究表明髓源性抑制细胞通过高表达精氨酸酶来抑制T细胞功能。环氧合酶2抑制剂能够抑制精氨酸酶合成,从而增强机体的免疫细胞功能发挥抗肿瘤效应。有效的免疫抑制诱导后,髓源性抑制细胞中精氨酸酶表达增加,髓源性抑制细胞自身的免疫抑制作用显著增强,从而减轻免疫排斥反应、延长移植物存活时间[5]。 2.2.2 调节性T细胞 髓源性抑制细胞与调节性T细胞都具有诱导免疫耐受的功能。调节性T细胞是CD4+T细胞的一个特定亚群,可通过抑制T细胞增殖和细胞因子(如转化生长因子)等生成而发挥免疫抑制作用[6-7]。研究表明,在心脏移植术后,髓源性抑制细胞在心脏组织内聚集,通过分泌一系列细胞因子如转化生长因子β、白细胞介素10、程序性死亡配体1(programmed death ligand 1,PD-L1)等诱导调节性T细胞聚集,从而减轻心脏免疫排斥反应,此外,髓源性抑制细胞在诱导调节性T细胞分化中也发挥重要作用[8-9]。虽然目前髓源性抑制细胞诱导调节性T细胞分化的机制尚未完全清楚,但可以确定髓源性抑制细胞与调节性T细胞在移植物内含量呈正相关。髓源性抑制细胞发挥免疫调节作用的时间较调节性T细胞短,其可能主要在移植早期发挥免疫调节作用,而调节性T细胞则主要在移植晚期起关键作用,故髓源性抑制细胞与调节性T细胞在诱导移植免疫耐受中具有协同作用[10]。 2.2.3 吲哚胺2,3-双加氧酶 吲哚胺2,3-双加氧酶主要来源于树突状细胞或树突状细胞诱导的调节性T细胞。QU 等[11]发现在髓源性抑制细胞中存在一条胱天蛋白酶募集域蛋白9-核转录因子κB-吲哚胺2,3-双加氧酶(CARD9-NF-κB-IDO)通路,该通路可以抑制髓源性抑制细胞免疫调节功能,从而阻止肺癌的发生,胱天蛋白酶募集域蛋白9缺失可通过非典型的核转录因子κB途径增强髓源性抑制细胞中吲哚胺2,3-双加氧酶的表达。髓源性抑制细胞通过分泌吲哚胺2,3-双加氧酶刺激L-色氨酸降解为N-甲酰基尿嘧啶,导致T细胞无能[12-13]。在慢性乙型肝炎病毒感染中,乙肝抗原诱导髓源性抑制细胞扩增,并通过吲哚胺2,3-双加氧酶途径损害T细胞功能,从而有利于慢性乙型肝炎的形成[14]。 2.2.4 白细胞介素6 白细胞介素6可通过信号传导与转录激活因子3(signal transducer and activator of transcription hepatocyte growth factor,STAT3)依赖机制上调髓源性抑制细胞中精氨酸酶的表达。由于精氨酸酶表达上调和活性氧产生增加,与未经白细胞介素6诱导的髓源性抑制细胞相比,白细胞介素6诱导的髓源性抑制细胞对CD8+T细胞的功能有更强的抑制作用[15]。体内白细胞介素6分泌增加可导致信号传导与转录激活因子3介导的程序性死亡配体1在抗原提呈细胞中表达上调,而由白细胞介素6诱导产生的髓源性抑制细胞抗原提呈基因则显著减少[16]。 在转基因黑素瘤小鼠模型中,白细胞介素6能够上调髓源性抑制细胞上的趋化因子受体CCR5,并诱导髓源性抑制细胞进入黑色素瘤组织中,白细胞介素6过度表达的肿瘤在CD8+T细胞激活的情况下生长明显缓慢。然而,当使用白细胞介素6信号阻断疗法治疗转基因黑素瘤小鼠时肿瘤发展速度显著加快[17]。 2.2.5 程序性死亡配体1 程序性死亡配体1在肿瘤微环境中高表达,能够抑制效应T细胞的免疫应答[18-19]。在器官移植模型中,应用程序性死亡配体1抗原CD40抗体治疗能够显著延长移植物的存活时间[20]。程序性死亡配体1/程序性死亡受体1(PD-L1/PD-1)轴在延长移植物存活时间中也起重要作用。研究表明,在小鼠心脏移植模型中,髓源性抑制细胞可通过高表达程序性死亡配体1来诱导调节性T细胞在心脏内聚集,进而诱导移植物免疫耐受[21]。KIM等[22-23]在小鼠胃癌实验中发现,5-氟尿嘧啶和奥沙利铂等化疗药物可减少小鼠体内髓源性抑制细胞的数量,增强抗程序性死亡配体1的作用,从而促进CD8+T细胞对肿瘤的浸润,延缓肿瘤病情进展,而胃上皮细胞表达程序性死亡配体1则增加了髓源性抑制细胞在肿瘤组织内的聚集,促进肿瘤进展。 2.2.6 诱导型一氧化氮合酶 诱导型一氧化氮合酶能够将L-精氨酸转化为一氧化氮,一氧化氮可与超氧化物反应生成对硝基甲苯,然后通过硝化T细胞抗原受体直接抑制T细胞,从而提高T细胞抗原受体对抗原-主要组织相容性复合体的亲和力,并通过硝化T细胞特异性趋化因子阻断T细胞迁移[24-25]。研究表明,在髓源性抑制细胞诱导移植免疫耐受过程中诱导型一氧化氮合酶发挥着重要作用。体外研究发现在淋巴细胞混合液中,髓源性抑制细胞能够抑制CD3、CD28抗原、T细胞增殖,加入诱导型一氧化氮合酶阻滞剂后,髓源性抑制细胞的抑制作用消失[26]。 2.2.7 长链非编码RNA(long non-coding RNA,lncRNA) 长链非编码RNA浆细胞瘤转化迁移基因1(long non-coding RNA Plasmacytoma variant translocation 1,lncRNA Pvt1)是一种电位辅助基因,在肺癌小鼠实验模型中被首次发现。在低氧条件下,缺氧诱导因子1α可明显上调lncRNA Pvt1表达,应用缺氧诱导因子1α抑制剂利非西呱,可明显降低髓源性抑制细胞中lncRNA Pvt1的表达,故lncRNA Pvt1可能是缺氧诱导因子1α的作用靶点。体外敲除lncRNA Pvt1后,粒细胞样的髓源性抑制细胞中精氨酸酶和活性氧的水平降低,从而显著减弱粒细胞样的髓源性抑制细胞的免疫抑制功能[27],见图3。"
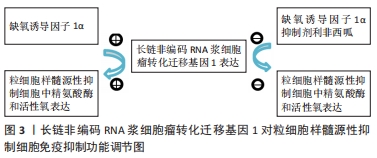
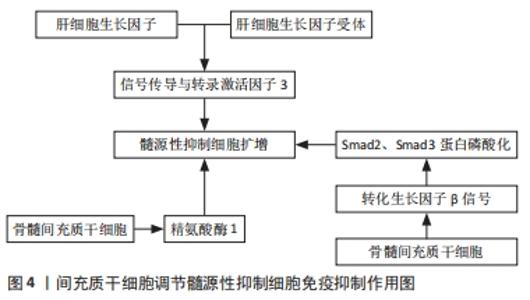
TIAN等[28]在荷瘤小鼠模型中敲除长链非编码RNAAK036396,Fcnb蛋白的稳定性降低,粒细胞样的髓源性抑制细胞抑制功能下调,Fcnb蛋白代表粒细胞样的髓源性抑制细胞中长链非编码RNA表达的产物。因此,长链非编码RNA可以通过增强Fcnb蛋白的稳定性来抑制粒细胞样的髓源性抑制细胞的成熟并加强其免疫抑制功能。 2.3 间充质干细胞对髓源性抑制细胞免疫抑制作用的影响 2.3.1 间充质干细胞调节髓源性抑制细胞免疫抑制作用的机制 间充质干细胞是一类起源于中胚层的成体干细胞,具有自我复制、多向分化潜能和免疫调节特性。作者前期多项研究发现间充质干细胞可有效缓解急性呼吸窘迫综合征、神经病理性疼痛、椎间盘退变等疾病[29-31]。间充质干细胞表达的程序性死亡配体1与T淋巴细胞表达的程序性死亡受体1结合后,可导致T细胞活化和增殖受到抑制及其免疫抑制功能减弱[32]。间充质干细胞除了通过与免疫效应细胞直接接触及分泌多种可溶性因子调控固有免疫及适应性免疫外,还可通过诱导调节性免疫细胞如髓源性抑制细胞等来发挥免疫调控作用[33]。 研究报道,在小鼠体内高水平分泌肝细胞生长因子的肝脏中含有大量髓源性抑制细胞,间充质干细胞分泌的肝细胞生长因子对髓源性抑制细胞的扩增作用与肝细胞生长因子受体和转录激活因子3的磷酸化有关。研究发现抑制肝细胞生长因子受体表达时,肝细胞生长因子对髓源性抑制细胞的扩增作用消失。外源性添加肝细胞生长因子可导致信号传导与转录激活因子3磷酸化水平升高,在外源性肝细胞生长因子中添加信号传导与转录激活因子3抑制剂,髓源性抑制细胞的扩增作用消失。因此,肝细胞生长因子通过结合肝细胞生长因子受体导致信号传导与转录激活因子3磷酸化增加,从而促进髓源性抑制细胞增殖,由此可见信号传导与转录激活因子3是诱导髓源性抑制细胞扩增的关键因素[34]。此外有研究发现在使用脐带间充质干细胞治疗脓毒症小鼠的实验中,脐带间充质干细胞显著提高小鼠体内精氨酸酶1的mRNA水平,且小鼠脾脏中髓源性抑制细胞含量明显增加,髓源性抑制细胞可通过精氨酸酶1发挥免疫抑制功能。当使用精氨酸酶1抑制剂后,可下调髓源性抑制细胞中抗炎细胞因子白细胞介素10的产生。这些结果表明脐带间充质干细胞可通过上调精氨酸酶1表达而增强髓源性抑制细胞的免疫抑制活性,这有助于促进脓毒症小鼠体内促炎和抗炎因子重新达到平衡[35]。骨髓间充质干细胞可以在机体内外直接调节髓源性抑制细胞的免疫抑制功能。在实验性干燥综合征小鼠模型中,骨髓间充质干细胞与髓源性抑制细胞共培养在高浓度精氨酸酶和一氧化氮中,转化生长因子β信号被激活,表达上调,Smad2蛋白和Smad3蛋白磷酸化增强,髓源性抑制细胞的免疫抑制功能明显增强。当髓源性抑制细胞的转化生长因子β通路被阻断时,骨髓间充质干细胞的诱导作用消失,髓源性抑制细胞的免疫抑制功能恢复到诱导前状态[35]。综上所述,骨髓间充质干细胞通过转化生长因子β/Smad途径增强髓源性抑制细胞的免疫抑制功能来缓解干燥综合征的进展,为间充质干细胞治疗干燥综合征提供了一个新的机制[36],见图4。"
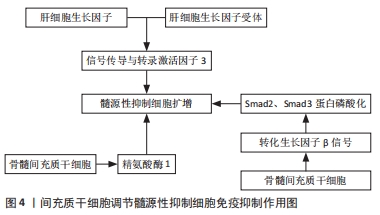

2.3.2 间充质干细胞对髓源性抑制细胞的调节作用与移植物免疫耐受 目前认为间充质干细胞通过影响髓样细胞分化发挥免疫抑制作用[37]。YANG等[38]在移植物抗宿主病动物实验中发现:脐血来源的间充质干细胞经粒细胞集落刺激因子诱导后可分泌人白细胞抗原G蛋白,促进并维持外周血髓源性抑制细胞扩增,减少髓源性抑制细胞的凋亡,促进精氨酸酶、吲哚胺2,3-双加氧酶分泌从而抑制T细胞功能,延长移植物术后存活时间,减轻移植物抗宿主病的严重程度。进一步研究证实,使用人白细胞抗原G蛋白刺激间充质干细胞后,与对照组相比,孕妇外周血中髓源性抑制细胞数量显著增加,而人白细胞抗原G蛋白中和抗体组孕妇外周血中的髓源性抑制细胞则明显减少,这说明间充质干细胞可通过分泌人白细胞抗原G蛋白来促进髓源性抑制细胞扩增[39-40]。由此可见,间充质干细胞可通过促进髓源性抑制细胞增殖而缓解移植物抗宿主病,提示间充质干细胞在防治移植物抗宿主病中具有重要的临床应用价值。 2.3.3 间充质干细胞对髓源性抑制细胞的调节作用与自身免疫性疾病 间充质干细胞中应用最广泛的是人脐带间充质干细胞,先前研究证实人脐带间充质干细胞可通过减少巨噬细胞浸润并将巨噬细胞极化为抗炎的M2表型以减轻肾脏足细胞损伤,最终改善狼疮性肾炎[40]。此外在探讨人脐带间充质干细胞对原发性系统性红斑狼疮患者体内滤泡辅助T细胞的影响中发现,人脐带间充质干细胞还可通过分泌吲哚胺2,3-双加氧酶抑制滤泡辅助性T细胞分化[41-42],研究发现人脐带间充质干细胞可通过不同机制调节髓源性抑制细胞的两个亚群分化,如人脐带间充质干细胞可通过环氧化酶2/前列腺素E2(COX2/PGE2)轴抑制粒细胞样的髓源性抑制细胞分化,而经由干扰素β抑制单核细胞样的髓源性抑制细胞分化,最终提高髓源性抑制细胞的免疫抑制能力[43]。QI等[44]研究患有涎腺炎的非肥胖性糖尿病小鼠时发现人脐带间充质干细胞可通过抑制体内单核细胞样的髓源性抑制细胞分化、减少其含量以及提高其免疫抑制功能来改善干燥综合征样症状。 2.3.4 间充质干细胞对髓源性抑制细胞的调节作用与炎症的关系 间充质干细胞可通过诱导髓源性抑制细胞进入炎症部位,抑制CD4+T细胞增殖并诱导其死亡。动物实验表明,在小鼠体内输注间充质干细胞,大多数被肺巨噬细胞吞噬,随后巨噬细胞通过产生白细胞介素10发挥抗炎作用[45-46]。LEE等[43,47]在实验性自身免疫性葡萄膜炎小鼠模型中首次发现,在炎症微环境中间充质干细胞可通过产生趋化因子CCl2募集髓源性抑制细胞进入炎症部位来发挥作用,且伴随着小鼠体内髓源性抑制细胞的增加,诱导型一氧化氮合酶和精氨酸酶表达也显著升高,髓源性抑制细胞进一步抑制CD4+T细胞的增殖和分化,并诱导T细胞凋亡,从而减轻炎症。 2.3.5 间充质干细胞对髓源性抑制细胞的调节作用与肿瘤的关系 在人和小鼠肿瘤组织中,间充质干细胞分泌的外泌体通过诱导髓源性抑制细胞分化为具有更强免疫抑制活性的M2型巨噬细胞,从而加速乳腺癌进展。肿瘤细胞免疫逃逸可能直接依赖于间充质干细胞,也可能通过髓源性抑制细胞在肿瘤微环境中介导的免疫抑制特性发挥作用[48]。GARCIA-GOMEZ等[49]在研究单克隆免疫球蛋白病、多发性骨髓瘤患者体内的间充质干细胞与来自健康人体内的间充质干细胞对粒细胞样的髓源性抑制细胞的扩张和激活的作用中发现,浆细胞促进间充质干细胞的增殖并影响其基因表达谱,同时间充质干细胞通过释放多种细胞因子和生长因子促进髓源性抑制细胞活化,进而促进多发性骨髓瘤细胞生长[7]。"

| [1] DOLEN Y, GUNAYDIN G, ESENDAGLI G, et al. Granulocytic subset of myeloid derived suppressor cells in rats with mammary carcinoma. Cell Immunol. 2015;295(1):29-35. [2] CONDAMINE T, GABRILOVICH DI. Molecular mechanisms regulating myeloid-derived suppressor cell differentiation and function. Trends Immunol. 2011; 32(1):19-25. [3] SAFARI E, GHORGHANLU S, AHMADI-KHIAVI H, et al. Myeloid-derived suppressor cells and tumor: Current knowledge and future perspectives. J Cell Physiol. 2019;234(7):9966-9981. [4] RODRIGUEZ PC, QUICENO DG, OCHOA AC. L-arginine availability regulates T-lymphocyte cell-cycle progression. Blood. 2007;109(4):1568-1573. [5] NAKAMURA T, NAKAO T, YOSHIMURA N, et al. Rapamycin Prolongs Cardiac Allograft Survival in a Mouse Model by Inducing Myeloid-Derived Suppressor Cells. Am J Transplant. 2015;15(9):2364-2377. [6] SHEVYREV D, TERESHCHENKO V. Treg Heterogeneity, Function, and Homeostasis. Front Immunol. 2020;10:3100. [7] GIALLONGO C, TIBULLO D, PARRINELLO NL, et al. Granulocyte-like myeloid derived suppressor cells (G-MDSC) are increased in multiple myeloma and are driven by dysfunctional mesenchymal stem cells (MSC). Oncotarget. 2016;7(52):85764-85775. [8] MEI Y, LI Z, ZHANG Y, et al. Low miR-210 and CASP8AP2 expression is associated with a poor outcome in pediatric acute lymphoblastic leukemia. Oncol Lett. 2017;14(6):8072-8077. [9] TOMIĆ S, JOKSIMOVIĆ B, BEKIĆ M, et al. Prostaglanin-E2 Potentiates the Suppressive Functions of Human Mononuclear Myeloid-Derived Suppressor Cells and Increases Their Capacity to Expand IL-10-Producing Regulatory T Cell Subsets. Front Immunol. 2019;10:475. [10] OKANO S, ABU-ELMAGD K, KISH DD, et al. Myeloid-derived suppressor cells increase and inhibit donor-reactive T cell responses to graft intestinal epithelium in intestinal transplant patients. Am J Transplant. 2018;18(10): 2544-2558. [11] QU J, LIU L, XU Q, et al. CARD9 prevents lung cancer development by suppressing the expansion of myeloid-derived suppressor cells and IDO production. Int J Cancer. 2019;145(8):2225-2237. [12] YU J, DU W, YAN F, et al. Myeloid-derived suppressor cells suppress antitumor immune responses through IDO expression and correlate with lymph node metastasis in patients with breast cancer. J Immunol. 2013;190(7):3783-3797. [13] LI F, ZHAO Y, WEI L, et al. Tumor-infiltrating Treg, MDSC, and IDO expression associated with outcomes of neoadjuvant chemotherapy of breast cancer. Cancer Biol Ther. 2018;19(8):695-705. [14] YANG F, YU X, ZHOU C, et al. Hepatitis B e antigen induces the expansion of monocytic myeloid-derived suppressor cells to dampen T-cell function in chronic hepatitis B virus infection. PLoS Pathog. 2019;15(4):e1007690. [15] WEBER R, GROTH C, LASSER S, et al. IL-6 as a major regulator of MDSC activity and possible target for cancer immunotherapy. Cell Immunol. 2021; 359:104254. [16] TARTOUR E, PERE H, MAILLERE B, et al. Angiogenesis and immunity: a bidirectional link potentially relevant for the monitoring of antiangiogenic therapy and the development of novel therapeutic combination with immunotherapy. Cancer Metastasis Rev. 2011;30(1):83-95. [17] WEBER R, RIESTER Z, HÜSER L, et al. IL-6 regulates CCR5 expression and immunosuppressive capacity of MDSC in murine melanoma. J Immunother Cancer. 2020;8(2):e000949. [18] SUN C, MEZZADRA R, SCHUMACHER TN. Regulation and Function of the PD-L1 Checkpoint. Immunity. 2018;48(3):434-452. [19] RUAN WS, FENG MX, XU J, et al. Early Activation of Myeloid-Derived Suppressor Cells Participate in Sepsis-Induced Immune Suppression via PD-L1/PD-1 Axis. Front Immunol. 2020;11:1299. [20] GAO W, DEMIRCI G, STROM TB, et al. Stimulating PD-1-negative signals concurrent with blocking CD154 co-stimulation induces long-term islet allograft survival. Transplantation. 2003;76(6):994-999. [21] NAKAMURA T, NAKAO T, ASHIHARA E, et al. Myeloid-derived Suppressor Cells Recruit CD4(+)/Foxp3(+) Regulatory T Cells in a Murine Cardiac Allograft. Transplant Proc. 2016;48(4):1275-1278. [22] KiM W, CHU TH, NIENHÜSER H, et al. PD-1 Signaling Promotes Tumor-Infiltrating Myeloid-Derived Suppressor Cells and Gastric Tumorigenesis in Mice. Gastroenterology. 2021;160(3):781-796. [23] WANG Z, ZHU F, WANG J, et al. Increased CD14(+)HLA-DR(-/low) Myeloid-Derived Suppressor Cells Correlate With Disease Severity in Systemic Lupus Erythematosus Patients in an iNOS-Dependent Manner. Front Immunol. 2019;10:1202. [24] GARCÍA-ORTIZ A, SERRADOR JM. Nitric Oxide Signaling in T Cell-Mediated Immunity. Trends Mol Med. 2018;24(4):412-427. [25] XUE Q, YAN Y, ZHANG R, et al. Regulation of iNOS on Immune Cells and Its Role in Diseases. Int J Mol Sci. 2018;19(12):3805. [26] BRONTE V, BRANDAU S, CHEN SH, et al. Recommendations for myeloid-derived suppressor cell nomenclature and characterization standards. Nat Commun. 2016;7:12150. [27] ZHENG Y, TIAN X, WANG T, et al. Long noncoding RNA Pvt1 regulates the immunosuppression activity of granulocytic myeloid-derived suppressor cells in tumor-bearing mice. Mol Cancer. 2019;18(1):61. [28] TIAN X, ZHENG Y, YIN K, et al. LncRNA AK036396 Inhibits Maturation and Accelerates Immunosuppression of Polymorphonuclear Myeloid-Derived Suppressor Cells by Enhancing the Stability of Ficolin B. Cancer Immunol Res. 2020;8(4):565-577. [29] DENG H, WU L, LIU M, et al. Bone Marrow Mesenchymal Stem Cell-Derived Exosomes Attenuate LPS-Induced ARDS by Modulating Macrophage Polarization Through Inhibiting Glycolysis in Macrophages. Shock. 2020; 54(6):828-843. [30] Yang H, Wu L, Deng H, et al. Anti-inflammatory protein TSG-6 secreted by bone marrow mesenchymal stem cells attenuates neuropathic pain by inhibiting the TLR2/MyD88/NF-kappaB signaling pathway in spinal microglia. J Neuroinflammation. 2020;17(1):154. [31] YANG H, TIAN W, WANG S, et al. TSG-6 secreted by bone marrow mesenchymal stem cells attenuates intervertebral disc degeneration by inhibiting the TLR2/NF-κB signaling pathway. Lab Invest. 2018;98(6):755-772. [32] WANG G, ZHANG S, WANG F, et al. Expression and biological function of programmed death ligands in human placenta mesenchymal stem cells. Cell Biol Int. 2013;37(2):137-148. [33] PAPAEMMANUIL E, GERSTUNG M, MALCOVATI L, et al. Clinical and biological implications of driver mutations in myelodysplastic syndromes. Blood. 2013; 122(22):3616-3627. [34] NOLL JE, WILLIAMS SA, TONG CM, et al.Myeloma plasma cells alter the bone marrow microenvironment by stimulating the proliferation of mesenchymal stromal cells. Haematologica. 2014;99(1):163-171. [35] LONG X, LI X, LI T, et al. Umbilical cord mesenchymal stem cells enhance the therapeutic effect of imipenem by regulating myeloid-derived suppressor cells in septic mice. Ann Transl Med. 2021;9(5):404. [36] TIAN J, HONG Y, ZHU Q, et al. Mesenchymal Stem Cell Enhances the Function of MDSCs in Experimental Sjogren Syndrome. Front Immunol. 2020;11:604607. [37] CHEUNG TS, DAZZI F. Mesenchymal-myeloid interaction in the regulation of immunity. Semin Immunol. 2018;35:59-68. [38] YANG S, WEI Y, SUN R, et al. Umbilical cord blood-derived mesenchymal stromal cells promote myeloid-derived suppressor cell proliferation by secreting HLA-G to reduce acute graft-versus-host disease after hematopoietic stem cell transplantation. Cytotherapy. 2020;22(12):718-733. [39] KÖSTLIN N, OSTERMEIR AL, SPRING B, et al. HLA-G promotes myeloid-derived suppressor cell accumulation and suppressive activity during human pregnancy through engagement of the receptor ILT4. Eur J Immunol. 2017;47(2):374-384. [40] ZHANG Z, NIU L, TANG X, et al. Mesenchymal stem cells prevent podocyte injury in lupus-prone B6.MRL-Faslpr mice via polarizing macrophage into an anti-inflammatory phenotype. Nephrol Dial Transplant. 2018;33(11):2069. [41] LIU R, SU D, ZHOU M, et al. Umbilical cord mesenchymal stem cells inhibit the differentiation of circulating T follicular helper cells in patients with primary Sjogren’s syndrome through the secretion of indoleamine 2,3-dioxygenase. Rheumatology (Oxford). 2015;54(2):332-342. [42] LI X, XU Z, BAI J, et al. Umbilical Cord Tissue-Derived Mesenchymal Stem Cells Induce T Lymphocyte Apoptosis and Cell Cycle Arrest by Expression of Indoleamine 2, 3-Dioxygenase. Stem Cells Int. 2016;2016:7495135. [43] LEE HJ, KO JH, JEONG HJ, et al. Mesenchymal stem/stromal cells protect against autoimmunity via CCL2-dependent recruitment of myeloid-derived suppressor cells. J Immunol. 2015;194(8):3634-3645. [44] QI J, TANG X, LI W, et al. Mesenchymal stem cells inhibited the differentiation of MDSCs via COX2/PGE2 in experimental sialadenitis. Stem Cell Res Ther. 2020;11(1):325. [45] NÉMETH K, LEELAHAVANICHKUL A, YUEN PS, et al. Bone marrow stromal cells attenuate sepsis via prostaglandin E(2)-dependent reprogramming of host macrophages to increase their interleukin-10 production. Nat Med. 2009;15(1):42-49. [46] LUK F, DE WITTE SF, KOREVAAR SS, et al. Inactivated Mesenchymal Stem Cells Maintain Immunomodulatory Capacity. Stem Cells Dev. 2016;25(18): 1342-1354. [47] WANG T, WEN Y, FAN X. Myeloid-derived suppressor cells suppress CD4+ T cell activity and prevent the development of type 2 diabetes. Acta Biochim Biophys Sin (Shanghai). 2018;50(4):362-369. [48] ALTANEROVA U, JAKUBECHOVA J, BENEJOVA K, et al. Prodrug suicide gene therapy for cancer targeted intracellular by mesenchymal stem cell exosomes. Int J Cancer. 2019;144(4):897-908. [49] GARCIA-GOMEZ A, DE LAS RIVAS J, OCIO EM, et al. Transcriptomic profile induced in bone marrow mesenchymal stromal cells after interaction with multiple myeloma cells: implications in myeloma progression and myeloma bone disease. Oncotarget. 2014;5(18):8284-8305. |
| [1] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhou Qian, Zhang Qiang, Chen Qiu. Human salivary components and osteoporosis/osteopenia [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1439-1444. |
| [2] | Jin Tao, Liu Lin, Zhu Xiaoyan, Shi Yucong, Niu Jianxiong, Zhang Tongtong, Wu Shujin, Yang Qingshan. Osteoarthritis and mitochondrial abnormalities [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1452-1458. |
| [3] | Zhang Lichuang, Xu Hao, Ma Yinghui, Xiong Mengting, Han Haihui, Bao Jiamin, Zhai Weitao, Liang Qianqian. Mechanism and prospects of regulating lymphatic reflux function in the treatment of rheumatoid arthritis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(9): 1459-1466. |
| [4] | Xiao Hao, Liu Jing, Zhou Jun. Research progress of pulsed electromagnetic field in the treatment of postmenopausal osteoporosis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1266-1271. |
| [5] | Zhu Chan, Han Xuke, Yao Chengjiao, Zhang Qiang, Liu Jing, Shao Ming. Acupuncture for Parkinson’s disease: an insight into the action mechanism in animal experiments [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1272-1277. |
| [6] | Xiang Xinjian, Liu Fang, Wu Liangliang, Jia Daping, Tao Yue, Zhao Zhengnan, Zhao Yu. High-dose vitamin C promotes the survival of autologous fat transplantation in rats [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1242-1246. |
| [7] | Wang Jing, Xiong Shan, Cao Jin, Feng Linwei, Wang Xin. Role and mechanism of interleukin-3 in bone metabolism [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(8): 1260-1265. |
| [8] | Wu Weiyue, Guo Xiaodong, Bao Chongyun. Application of engineered exosomes in bone repair and regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1102-1106. |
| [9] | Zhou Hongqin, Wu Dandan, Yang Kun, Liu Qi. Exosomes that deliver specific miRNAs can regulate osteogenesis and promote angiogenesis [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1107-1112. |
| [10] | Zhang Jinglin, Leng Min, Zhu Boheng, Wang Hong. Mechanism and application of stem cell-derived exosomes in promoting diabetic wound healing [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1113-1118. |
| [11] | Huang Chenwei, Fei Yankang, Zhu Mengmei, Li Penghao, Yu Bing. Important role of glutathione in stemness and regulation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1119-1124. |
| [12] | Hui Xiaoshan, Bai Jing, Zhou Siyuan, Wang Jie, Zhang Jinsheng, He Qingyong, Meng Peipei. Theoretical mechanism of traditional Chinese medicine theory on stem cell induced differentiation [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1125-1129. |
| [13] | An Weizheng, He Xiao, Ren Shuai, Liu Jianyu. Potential of muscle-derived stem cells in peripheral nerve regeneration [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1130-1136. |
| [14] | Fan Yiming, Liu Fangyu, Zhang Hongyu, Li Shuai, Wang Yansong. Serial questions about endogenous neural stem cell response in the ependymal zone after spinal cord injury [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1137-1142. |
| [15] | Xuan Juanjuan, Bai Hongtai, Zhang Jixiang, Wang Yaoquan, Chen Guoyong, Wei Sidong. Role of regulatory T cell subsets in liver transplantation and progress in clinical application [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(7): 1143-1148. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||