Chinese Journal of Tissue Engineering Research ›› 2021, Vol. 25 ›› Issue (32): 5222-5226.doi: 10.12307/2021.228
Previous Articles Next Articles
Peroxisome proliferator-activated receptor signaling pathway is involved in the metabolic regulation of obesity by intestinal flora and brown adipose tissue
Ye Erbai1, Xiao Xiaoling1, He Ziyu1, Yang Chun1, Du Yikuan2#br#
- 1Key Laboratory of Stem Cell and Regenerative Tissue Engineering, Guangdong Medical University, Dongguan 523808, Guangdong Province, China; 2Dongguan People’s Hospital, Dongguan 523059, Guangdong Province, China
-
Received:2020-10-21Revised:2020-10-24Accepted:2020-12-07Online:2021-11-18Published:2021-07-26 -
Contact:Yang Chun, MD, Associate professor, Key Laboratory of Stem Cell and Regenerative Tissue Engineering, Guangdong Medical University, Dongguan 523808, Guangdong Province, China Du Yikuan, MD, Associate researcher, Dongguan People’s Hospital, Dongguan 523059, Guangdong Province, China -
About author:Ye Erbai, Key Laboratory of Stem Cell and Regenerative Tissue Engineering, Guangdong Medical University, Dongguan 523808, Guangdong Province, China -
Supported by:the National Natural Science Foundation of China (Youth Program), No. 81501242 (to YC); Guangdong Provincial Natural Science Foundation (General Project), No. 2020A1515010011 (to YC); General Project of Guangdong Provincial Traditional Chinese Medicine Bureau, No. 20202101 (to YC); Guangdong Provincial Medical Science Foundation, No. A2020096 (to YC); Guangdong Provincial Science and Technology Innovation Strategy Special Fund (Special Fund for Climbing Plan), No. pdjh2020a0255 (to YEB) and pdjh2019b0216; College Student Innovation Project of Guangdong Medical University, No. 2019ZZDS007 (to YEB) and 2019ZZDS003; College Student Innovation and Entrepreneurship Training Project of Guangdong Medical University, No. GDMU2019003 (to YEB), GDMU2018063 and GDMU2019041; 2020 Guangdong Provincial and National College Students Innovation and Entrepreneurship Training Program, No. 202010571003 (to YEB) and S202010571041
CLC Number:
Cite this article
Ye Erbai, Xiao Xiaoling, He Ziyu, Yang Chun, Du Yikuan. Peroxisome proliferator-activated receptor signaling pathway is involved in the metabolic regulation of obesity by intestinal flora and brown adipose tissue[J]. Chinese Journal of Tissue Engineering Research, 2021, 25(32): 5222-5226.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
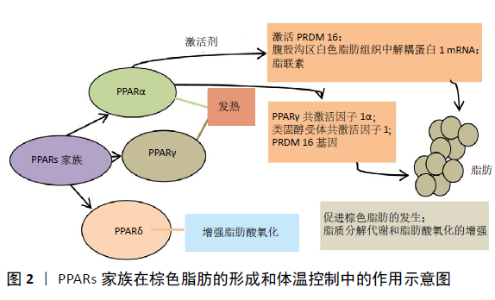
2.1 PPARs-BAT-肥胖轴在小鼠代谢调节中的作用 2.1.1 BAT与肥胖 肥胖是导致代谢紊乱的危险因素之一,可导致高血压、胆固醇和三酰甘油升高、胰岛素抵抗和糖尿病。肥胖的特征是通过脂肪细胞增生(脂肪细胞数量增加)和脂肪细胞肥大(脂肪细胞体积增大)以及脂肪组织功能障碍而发生的脂肪量膨胀[1]。长期摄入超过维持需求所需的能量会导致体内脂肪堆积,最终导致肥胖[2]。脂肪组织是控制能量消耗、食欲、胰岛素敏感性、内分泌、生殖功能、炎症和免疫等重要代谢途径的复杂而活跃的器官[3]。其中BAT是一种通过解偶联热形成来燃烧能量的器官[4],它的生热能力与线粒体解偶联蛋白1(uncoupling protein 1,UCP1)有关[5]。棕色脂肪细胞内含有大量线粒体和分散的脂质,通过UCP1产生热量[1]。BAT同时也是一种有效的三酰甘油血症调节剂,被认为是啮齿类动物血浆中主要的脂类清除器官[3]。研究人员估计,每天只有56.698 g(两盎司)的BAT可以燃烧高达2.093 kJ的热量,这意味着在治疗肥胖和相关疾病方面有巨大的治疗潜力[6]。 2.1.2 BAT与PPARs 研究发现,PPARs可通过调节合成代谢和氧化过程之间的平衡来调节脂肪组织的动态平衡,在肥胖的调节中发挥着重要的调节作用[7]。PPARs家族在棕色脂肪的形成和体温控制中起着核心的作用,迄今已鉴定出3种PPARs亚型,分别是PPARα、PPARγ和PPARδ。PPARα和PPARγ与热发生直接相关,PPARδ具有增加脂肪酸氧化的能力[8]。PPARα与PPARγ共激活因子1α(PGC-1α)、类固醇受体共激活因子1(SRC-1)和PRDM 16基因(激活BAT关键特征的基因)共同促进棕色脂肪的发生:当PPARα被相应激动剂激活时,直接激活PRDM 16、腹股沟区白色脂肪组织中UCP-1 mRNA及脂联素的表达,促进脂肪的褐变,同时伴随着脂质分解代谢和脂肪酸氧化的增强[9](图2)。当PPARα基因缺失时,β3肾上腺素能受体激动剂诱导的脂肪组织褐变减弱[10]。 "
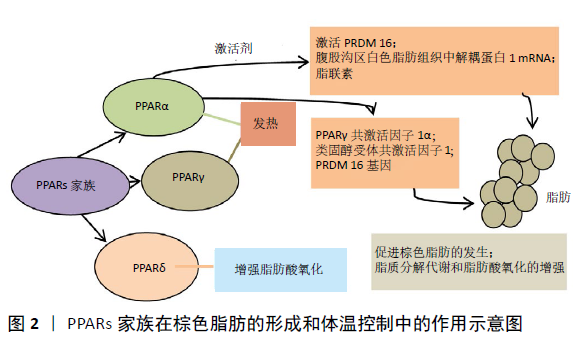

2.1.3 BAT与PPARγ PPARγ激活时,可通过促进BAT的褐变以及对支链氨基酸的代谢,双向影响体质量。PPARγ通过调节AK 079912(一种棕色脂肪细胞富含的长链非编码RNA)基因的表达,促使棕色脂肪细胞的分化以及相关热原基因的表达,从而达到减轻体质量的作用[11]。多数研究都表明PPARγ抑制肥胖,也有部分文献报道PPARγ也参与体质量的增加。研究表明当PPARγ被慢性激活时,可促进BAT对外周支链氨基酸的分解代谢,导致血液中的支链氨基酸浓度下降,相反地起到增加体质量的作用[12]。 BAT的线粒体呼吸中还存在着一种逆行性信号,反过来调节BAT的遗传和代谢,它是依靠PPARγ来实现的。当BAT中的线粒体呼吸能力受损时,激活逆行信号通路,影响PPARγ启动子的异性募集,导致产热、氧化基因的表达的下降,减少组织中的氧化达到能量储存的效果[13]。细菌代谢产物和细菌副产物如丁酸酯和脂多糖也能够激活PPARγ。在热量限制的条件下,小鼠肠上皮PPARγ增加交感神经系统活性并促进储存脂肪的使用[14]。 2.1.4 BAT与PPARδ 作为BAT与肥胖的调节剂,PPARδ主要通过调节BAT中热源基因的表达以及增强脂肪酸氧化,间接影响肥胖。因此,从BAT、PPARδ两者中研究抵抗肥胖的相关药物前景可观。如有研究显示在小鼠体内补充绿茶儿茶素可明显减轻体质量,绿茶儿茶素处理提高了BAT中PPARδ、脂肪酸氧化相关基因的表达,部分绿茶儿茶素通过调节PPARs信号通路发挥其抗肥胖作用[15]。同时,在脂肪组织中能表达一种被激活的PPARδ转基因的瘦小鼠能够抵抗遗传或高脂饮食引起的肥胖、高脂血症和组织脂肪变性。受体被激活后,脂肪酸分解代谢和适应性产热所需的基因表达上调。同时,高脂饮食的PPARδ缺陷型小鼠表现出能量解耦性降低,并且容易引起肥胖[16]。实验结果共同确定PPARδ通过引起脂肪燃烧,从而达到降低脂肪含量的作用[17]。 PPARs家族的转录活性受到细胞信号网络的严格控制,β-arrestins是G蛋白偶联受体的主要次级信使,通过与多种蛋白质的功能相互作用发挥作用。实验发现β-arrestin-1通过LXXXLXXXL基序直接与PPARα和PPARγ相互作用,对BAT的生成有双重调节的作用:它可以增加PPARα的活性促进白色脂肪组织的褐变,同时抑制PPARγ的转录,从而影响棕色脂肪细胞的稳定[18]。但该文献只局限于对β-arrestins通过双向调节BAT增加小鼠产热从而维持体温的讨论,对体质量的影响尚未进行探讨。 以上可见,BAT、PPARs与肥胖三者之间构成PPARs-BAT-肥胖这一轴线来影响小鼠的代谢调节。BAT主要通过UCP1产热从而达到能量的高消耗,起到直接抵抗肥胖的作用。 2.2 肠道菌群与肥胖 肠道菌群是机体肠道的正常微生物,其构成主要为细菌、古细菌、真菌、病毒等。肠道菌群中含有数以百计的外源和内在宿主因子调控的大量和多样的微生物和基因。微生物区系产生的代谢产物(包括短链脂肪酸)在PPARs家族中发挥了重要作用,如PPARs家族中γ主要表达于脂肪组织和胃肠道,参与脂质代谢调节、葡萄糖稳态、细胞增殖分化和局部炎症反应[19]。 短链脂肪酸来自于膳食纤维的微生物发酵,是肠道微生物发酵活性的主要产物,包括乙酸、丙酸和丁酸[20],其中丁酸盐和丙酸盐可以抵抗肥胖,而乙酸盐被大量吸收后,可以表现出很强的致肥胖作用[21]。一项研究中短链脂肪酸可以降低脂肪组织中的PPARγ,PPARγ表达或活性的降低刺激UCP2的表达和脂肪酸氧化,并降低脂肪生成和肝三酰甘油水平,这表明短链脂肪酸可作为廉价和选择性的PPARγ调节剂用于治疗肥胖[22]。 短链脂肪酸是重要的能量和信号分子。人们逐渐意识到产生丁酸的细菌和丁酸本身可能对人体健康有益。高脂饲料喂养的小鼠和低脂对照组小鼠的肠道微生物多样性有显著差异,与高脂饮食的小鼠相比,喂养低热量脂肪限制饮食(21.3%脂肪和60%碳水化合物)小鼠的柔膜细菌相对丰度显著降低,直肠中拟杆菌数量增加,体质量增加减少,其体质量的减少的原因与丁酸盐的产生有关[23]。宿主可以通过结肠上皮高表达的PPARγ识别丁酸[24],在喂食富含丁酸的高脂饮食的小鼠生热和能量消耗增加,并对肥胖具有抵抗力[20]。 有趣的是,肠道微生物也能够从食物和脂肪酸衍生物(包括PPARγ诱导)中合成多不饱和脂肪酸的代谢产物[25]。饮食中添加n-3多不饱和脂肪酸往往与肥胖啮齿动物的脂肪组织改变有关。据报道,食用含有n-3多不饱和脂肪酸的油可以改善肥胖患者的葡萄糖和脂肪代谢[26]。此外,n-3多不饱和脂肪酸被鉴定为PPARs家族的潜在内源性配体,在脂肪细胞分化和脂质代谢中起着至关重要的作用。n-3多不饱和脂肪酸以不受体质量增加或脂肪积累限制的方式协调调节脂肪组织基因表达程序,对脂肪生成有重要影响。此外该研究证明保护蛋白dx存在于脂肪组织中,并且保护蛋白dx和保护蛋白d1促进类似的PPARγ转录活性,表明保护蛋白可能通过其对PPARγ的影响脂肪细胞分化和脂质代谢[27]。有研究证明,多不饱和脂肪酸还可以促进不同时期的3T3-L1脂肪细胞棕色化、线粒体生成和葡萄糖消耗,这些作用与多不饱和脂肪酸激活PPARγ有关[28]。同时有研究结果表明,用白藜芦醇喂养的小鼠中,类杆菌属、类杆菌科和类杆菌纲的相对丰度较高,脂肪细胞因子信号和PPAR信号通路显著上调,该研究还表明,用白藜芦醇治疗可以重塑小鼠的肠道微生物群,从而改善肥胖导致的生物失调和葡萄糖稳态[29]。综上,不难看出肠道菌群的代谢产物通过激活PPARs信号通路,调节脂肪组织,从而对肥胖症产生影响。 2.3 PPARs在BAT与肠道菌群的联系作用 越来越多的证据表明,肠道微生物区系通过与脂肪组织的交通轴参与宿主的代谢,从而影响与肥胖相关的代谢变化的发展。有数据表明,肠道微生物群调节脂质代谢的BAT,肠道微生物群的缺乏刺激肝和BAT脂解作用而抑制脂肪生成[30]。然而,鲜有文献集中讨论PPARs信号通路在两者之间的联系作用。 研究发现桑叶可以通过激活小鼠BAT和调节肠道微生物群来改善肥胖,PGC-1α、PGC-1β和PPARγ等几种关键基因参与增强BAT的活性来减轻肥胖;此外,桑叶显著提高了与肥胖发展和进展密切相关的拟杆菌/硬度比和Akkermansia水平,并降低了粪便中潜在的促炎性蛋白杆菌;桑叶富含黄酮类化合物和膳食纤维,这可能对调节肠道微生物群有益[31]。 一种亚马逊河水果卡木卡木(CC)处理小鼠BAT和腹股沟脂肪组织中UCP1和脱碘酶2的mRNA水平显著上调;与较高的BAT活性一致,CC小鼠的BAT中PPARg2的mRNA表达增加,而CC小鼠的BAT中PPARγ协同激活子1α(PGC-1α)的基因表达趋势更高;CC显著增加了饮食诱导肥胖小鼠肠道微生物群中的黏蛋白A。研究表明,口服黏蛋白原A饮食诱导的肥胖小鼠中和了肥胖以及代谢综合征的其他特征,肠道微生物群的变化与CC治疗的主要代谢益处有关[32]。 褪黑激素是一种天然激素,主要由松果体合成和分泌。褪黑素能调节肠道微生物群,促进类杆菌与硬壁菌的比例降低,增加艾克曼菌在高脂饮食喂养的小鼠中的相对丰度;同时该研究观察到褪黑激素在基因和蛋白质水平上增加UCP1的表达,增强脂肪酸运输和分解代谢,并促进产热[33]。另有研究发现,在高脂饮食小鼠中,短链脂肪酸减少,而褪黑素处理改善了乙酸的产生;褪黑素可逆转高脂饮食诱导的脂质积累和肠道微生物区系改变。在小鼠肝脏中,PPARγ和LXR的mRNA表达随高脂饮食的增加而降低,而褪黑素显著增加PPARγ、LXR和ACC的mRNA表达[34]。 补充谷子全谷类和麸皮可以使小鼠从肥胖状态转变为非肥胖状态。补充Kodo小米(KM)全谷物(KM-WG)和麸皮(KM-BR)可增加小鼠乳酸杆菌、双歧杆菌、玫瑰指孢囊菌和粘杆菌的丰度。在内脏白色脂肪组织中,PPARγ和acox1(一种脂肪酸氧化基因)在较低剂量的Kodo小米全谷物和麸皮补充下下调,提示内脏白色脂肪组织中脂肪生成减少[35]。 也有论文最近提出,内源性大麻素(Endocannabinoid,ECB)系统可能是肠道和脂肪组织之间沟通的媒介之一。内源性大麻素系统由生物活性脂质组成,这些脂质与大麻素受体结合并引起细胞信号传导[36]。在生理条件下,大麻素受体激动剂(Hu-210)激活内源性大麻素系统会增加瘦小鼠的脂肪生成。事实上,结果发现大麻素受体的激活显著增加了参与脂肪细胞分化(PPAR-g,AP2,C/EBP-a)和脂肪生成(SREBP-1c,ACC,FAS)的基因的表达[37]。同时研究发现丹参素冰片脂也可以同时影响BAT与肠道菌群来调节肥胖。丹参素冰片脂又称丹参醇龙脑酯,来源于丹参,是一种有效的天然PPARγ激动剂,丹参素冰片脂可能是一种有益的益生菌制剂,可改善高脂饮食引起的肥胖相关代谢综合征。丹参素冰片脂不仅能刺激脂肪组织褐变,维持肠屏障完整性,而且能逆转高脂饮食引起的肠道菌群失调[38]。摄入桑叶、水果卡木卡木、褪黑素、丹参素冰片脂后BAT和肠道菌群的代谢变化都伴随着PPARγ基因表达水平的改变,这表明了PPARs信号通路可能在肠道菌群和BAT中的联系作用,提示可以通过食疗和药物摄入来改变BAT与肠道菌群的代谢,改善肥胖症状。 "

| [1] LIU J, WANG Y, LIN L. Small molecules for fat combustion: targeting obesity. Acta Pharmaceutica Sinica B. 2019;9(2):220-236. [2] PRPIC V, WATSON PM, FRAMPTON IC, et al. Adaptive changes in adipocyte gene expression differ in AKR/J and SWR/J mice during diet-induced obesity. J Nutr. 2002;132(11):3325-3332. [3] CALDERON-DOMINGUEZ M, MIR JF, FUCHO R, et al. Fatty acid metabolism and the basis of brown adipose tissue function. Adipocyte. 2016;5(2):98-118. [4] YIN N, ZHANG H, YE R, et al. Fluvastatin Sodium Ameliorates Obesity through Brown Fat Activation. Int J Mol Sci. 2019;20(7):1622. [5] THARP KM, STAHL A. Bioengineering Beige Adipose Tissue Therapeutics. Front Endocrinol (Lausanne). 2015;6:164. [6] YANG JP, ANDERSON AE, MCCARTNEY A, et al. Metabolically Active Three-Dimensional Brown Adipose Tissue Engineered from White Adipose-Derived Stem Cells. Tissue Engineering Part A. 2017;23(7-8): 253-262. [7] CORRALES P, VIDAL-PUIG A, MEDINA-GOMEZ G. PPARs and Metabolic Disorders Associated with Challenged Adipose Tissue Plasticity. Int J Mol Sci. 2018;19(7):2124. [8] LIU J, WANG Y, LIN L. Small molecules for fat combustion: targeting obesity. Acta Pharmaceutica Sinica B. 2019;9(2):220-236. [9] ARAKI M, NAKAGAWA Y, OISHI A, et al.The Peroxisome Proliferator-Activated Receptor alpha (PPARalpha) Agonist Pemafibrate Protects against Diet-Induced Obesity in Mice. Int J Mol Sci. 2018;19(7):2148. [10] NAKATSU Y, MATSUNAGA Y, YAMAMOTOYA T, et al. Prolyl Isomerase Pin1 Suppresses Thermogenic Programs in Adipocytes by Promoting Degradation of Transcriptional Co-activator PRDM16. Cell Reports. 2019;26(12):3221-3230. [11] XIONG Y, YUE F, JIA Z, et al. A novel brown adipocyte-enriched long non-coding RNA that is required for brown adipocyte differentiation and sufficient to drive thermogenic gene program in white adipocytes. Biochim Biophys Acta Mol Cell Biol Lipids. 2018;1863(4):409-419. [12] BLANCHARD PG, MOREIRA RJ, CASTRO E, et al. PPARgamma is a major regulator of branched-chain amino acid blood levels and catabolism in white and brown adipose tissues. Metabolism. 2018;89:27-38. [13] NAM M, AKIE TE, SANOSAKA M, et al. Mitochondrial retrograde signaling connects respiratory capacity to thermogenic gene expression. Scientific Reports. 2017;7(1):2012-2013. [14] DUSZKA K, PICARD A, ELLERO-SIMATOS S, et al. Intestinal PPARγ signalling is required for sympathetic nervous system activation in response to caloric restriction. Scientific Reports. 2016;6(1):36937. [15] MAKI KC, REEVES MS, FARMER M, et al. Green tea catechin consumption enhances exercise-induced abdominal fat loss in overweight and obese adults. J Nutr. 2009;139(2):264-270. [16] WANG YX, LEE CH, TIEP S, et al. Peroxisome-proliferator-activated receptor delta activates fat metabolism to prevent obesity. Cell. 2003;113(2):159-170. [17] EVANS RM, BARISH GD, WANG YX. PPARs and the complex journey to obesity. Nat Med. 2004;10(4):355-361. [18] WANG C, ZENG X, ZHOU Z, et al. beta-arrestin-1 contributes to brown fat function and directly interacts with PPARalpha and PPARgamma. Sci Rep. 2016;6:26999. [19] COUVIGNY B, DE WOUTERS T, KACI G, et al. Commensal Streptococcus salivarius Modulates PPARgamma Transcriptional Activity in Human Intestinal Epithelial Cells. PLoS One. 2015;10(5):e125371. [20] KOH A, DE VADDER F, KOVATCHEVA-DATCHARY P, et al. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell. 2016;165(6):1332-1345. [21] SANNA S, VAN ZUYDAM NR, MAHAJAN A, et al. Causal relationships among the gut microbiome, short-chain fatty acids and metabolic diseases. Nat Genet. 2019;51(4):600-605. [22] HASAN AU, RAHMAN A, KOBORI H. Interactions between Host PPARs and Gut Microbiota in Health and Disease. Int J Mol Sci. 2019;20(2):387. [23] SHANG Y, KHAFIPOUR E, DERAKHSHANI H, et al. Short Term High Fat Diet Induces Obesity‐Enhancing Changes in Mouse Gut Microbiota That are Partially Reversed by Cessation of the High Fat Diet. Lipids. 2017;52(6):499-511. [24] BYNDLOSS MX, OLSAN EE, RIVERA-CHÁVEZ F, et al. Microbiota-activated PPAR-γ signaling inhibits dysbiotic Enterobacteriaceae expansion. Science (New York, N.Y.). 2017;357(6351):570-575. [25] MARION-LETELLIER R, SAVOYE G, GHOSH S. Fatty acids, eicosanoids and PPAR gamma. Eur J Pharmacol. 2016;785:44-49. [26] LU J, BORTHWICK F, HASSANALI Z, et al.Chronic dietary n-3 PUFA intervention improves dyslipidaemia and subsequent cardiovascular complications in the JCR:LA- cp rat model of the metabolic syndrome. Br J Nutr. 2011;105(11):1572-1582. [27] WHITE PJ, MITCHELL PL, SCHWAB M, et al. Transgenic omega-3 PUFA enrichment alters morphology and gene expression profile in adipose tissue of obese mice: Potential role for protectins. Metabolism. 2015; 64(6):666-676. [28] 裘洁妮. 多不饱和脂肪酸对3T3-L1脂肪细胞白色脂肪棕色化的作用及机制研究[D].杭州:浙江大学,2016. [29] HUI S, LIU Y, HUANG L, et al. Resveratrol enhances brown adipose tissue activity and white adipose tissue browning in part by regulating bile acid metabolism via gut microbiota remodeling. Int J Obes (Lond). 2020;44(8):1678-1690. [30] MESTDAGH R, DUMAS ME, REZZI S, et al. Gut microbiota modulate the metabolism of brown adipose tissue in mice. J Proteome Res. 2012;11(2):620-630. [31] SHENG Y, LIU J, ZHENG S, et al. Mulberry leaves ameliorate obesity through enhancing brown adipose tissue activity and modulating gut microbiota. Food Funct. 2019;10(8):4771-4781. [32] ANHE FF, NACHBAR RT, VARIN TV, et al. Treatment with camu camu (Myrciaria dubia) prevents obesity by altering the gut microbiota and increasing energy expenditure in diet-induced obese mice. Gut. 2019;68(3):453-464. [33] XU P, WANG J, HONG F, et al. Melatonin prevents obesity through modulation of gut microbiota in mice. J Pineal Res. 2017;62(4). doi: 10.1111/jpi.12399. [34] YIN J, LI Y, HAN H, et al. Melatonin reprogramming of gut microbiota improves lipid dysmetabolism in high‐fat diet‐fed mice. J Pineal Res. 2018;65(4):e12524. [35] SARMA SM, KHARE P, JAGTAP S, et al. Kodo millet whole grain and bran supplementation prevents high-fat diet induced derangements in a lipid profile, inflammatory status and gut bacteria in mice. Food Funct. 2017;8(3):1174-1183. [36] GEURTS L, NEYRINCK AM, DELZENNE NM, et al. Gut microbiota controls adipose tissue expansion, gut barrier and glucose metabolism: novel insights into molecular targets and interventions using prebiotics. Benef Microbes. 2014;5(1):3-17. [37] MUCCIOLI GG,NASLAIN D,BACKHED F,et al.The endocannabinoid system links gut microbiota to adipogenesis. Mol Syst Biol. 2010;6:392. [38] XU J, XU P, HONG F, et al. DBZ is a putative PPARγ agonist that prevents high fat diet-induced obesity, insulin resistance and gut dysbiosis. Biochim Biophys Acta Gen Subj. 2017;1861(11):2690-2701. |
| [1] | Ruan Ling, Wang Guanghua, Wu Rongping, Jin Zhan, Lyu Zhenqing, Zhang Nan, Li Shoubang. Correlation between exercise intensity and lipid metabolism disorder and oxidative stress in a high-diet rat model [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1149-1155. |
| [2] | Zhang Yan, He Ruibo, Wang Qingbo, Pi Yihua, Lu Chunmin, Xu Chuanyi, Ma Gang, Peng Peng. Effects of aerobic exercises with different load volumes on inflammatory response and insulin signaling pathway of skeletal muscle in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1237-1244. |
| [3] | Liang Jiaqi, Liu Hengxu, Yang Jinxin, Yang Yi, Deng Xuhui, Tan Mingjian, Luo Jiong. Health benefit relationship between exercise and intestinal bacteria [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(8): 1292-1299. |
| [4] | Li Yujiao, Su Kunxia. High-intensity endurance exercise influences browning of white adipose tissue in a mouse model of high-fat diet induced obesity [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(5): 707-713. |
| [5] | Cui Zhenhua, Lin Xiafei, Chen Yongmin, Lin Ye, Li Guanyu, Song Zhenhua. Acupuncture and moxibustion combined with rehabilitation therapy improve neurological function and intestinal flora following cerebral ischemia in rats [J]. Chinese Journal of Tissue Engineering Research, 2023, 27(17): 2692-2698. |
| [6] | Lin Jiayu, Huang Huibin, Liang Bo, Chen Lijun. Changes in irisin, leptin, adiponectin and visceral fat in obese rats undergoing high-intensity intermittent exercise [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(35): 5583-5588. |
| [7] | Wu Juhua, Yang Yanan, Weng Xiquan, Zhao Fangfang, Xu Guoqin, Lin Wenta. Effects of hypoxic exercise on skeletal muscle energy metabolism of rats with alimentary obesity [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(29): 4598-4604. |
| [8] | Chen Chichi, Zhang Yu, He Jiachen, Shi Qin. Osteogenic differentiation of bone marrow mesenchymal stem cells in obese mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3846-3851. |
| [9] | Wu Siyi, Wu Qiong. Effect of adipose-derived stem cell transplantation on the immune microenvironment of adipose tissue in aged mice [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(24): 3773-3778. |
| [10] | Chen Ziyang, Pu Rui, Deng Shuang, Liu Min. N6-methyladenosine methylation and its regulation in metabolic diseases [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(20): 3250-3255. |
| [11] | Shang Wandi, Wang Xingze, Wei Xiaoyan. Effects of different types of abdominal support on lumbar-back muscle surface electromyography signals in people with abdominal obesity [J]. Chinese Journal of Tissue Engineering Research, 2022, 26(11): 1656-1661. |
| [12] | Bu Jianhua, Wang Zhen, Liu Ziming, Li Lin, Yu Liang. Effect of calorie restriction and its combined exercise on fat reduction and irisin expression in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(35): 5638-5643. |
| [13] | Liu Jianguo, Pan Yong, Li Hanyu, Liu Yan, Zhang Zhentian, Lin Xiuping, Zhang Yuan, Liu Yajin, Zhang Fan, Zhang Leijun, Xiao Liehui, Xu Aimin, Zhu Cuifeng. Effect of microecological preparation combined with an improved low-carbon diet on fat metabolism and intestinal barrier function in obese patients [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(29): 4672-4679. |
| [14] | Chen Ziyang, Pu Rui, Deng Shuang, Yuan Lingyan. Regulatory effect of exosomes on exercise-mediated insulin resistance diseases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(25): 4089-4094. |
| [15] | Wang Chaoge, Weng Xiquan, Lin Baoxuan, Chen Lina, Xu Guoqin. Exercises under cold exposure change fat type and function in obese rats [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(20): 3162-3167. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||