中国组织工程研究 ›› 2015, Vol. 19 ›› Issue (12): 1924-1930.doi: 10.3969/j.issn.2095-4344.2015.12.023
• 材料力学及表面改性 material mechanics and surface modification • 上一篇 下一篇
球形多孔微载体支持下高密度培养人肝细胞L-02的定时形态变化
张 瑞,刘 明
- 内蒙古医科大学附属医院普外科,内蒙古自治区呼和浩特市 010050
-
修回日期:2015-02-17出版日期:2015-03-19发布日期:2015-03-19 -
通讯作者:刘明,主任医师。内蒙古医科大学附属医院普外科,内蒙古自治区呼和浩特市 010050 -
作者简介:张瑞,男,汉族,1972年生,内蒙古自治区包头市人,上海交通大学医学院附属瑞金医院毕业,主任医师,医学博士。 -
基金资助:国家高新技术研究发展计划“863”项目(2007AA02Z487 );国家自然科学基金项目(30672043,30772105,20074031);内蒙古科技厅面上项目(2012MS1124),内蒙古自治区高等学校科学研究项目(NJSZY11128)
Timed morphological changes of human hepatocytes L-02 cultured at high density by the support of spherical porous chitosan microcarriers
Zhang Rui, Liu Ming
- Department of General Surgery, First Affiliated Hospital, Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China
-
Revised:2015-02-17Online:2015-03-19Published:2015-03-19 -
Contact:Liu Ming, Chief physician, Department of General Surgery, First Affiliated Hospital, Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China -
About author:Zhang Rui, M.D., Chief physician, Department of General Surgery, First Affiliated Hospital, Inner Mongolia Medical University, Hohhot 010050, Inner Mongolia Autonomous Region, China -
Supported by:the National High Technology Research and Development Program of China (863 Program), No. 2007AA02Z487; the National Natural Science Foundation of China, No. 30672043, 30772105, 20074031; the General Program of the Science and Technology Department of Inner Mongolia Autonomous Region, No. 2012MS1124; the Scientific Research Program in High Education of Inner Mongolia Autonomous Region, No. NJSZY11128
摘要:
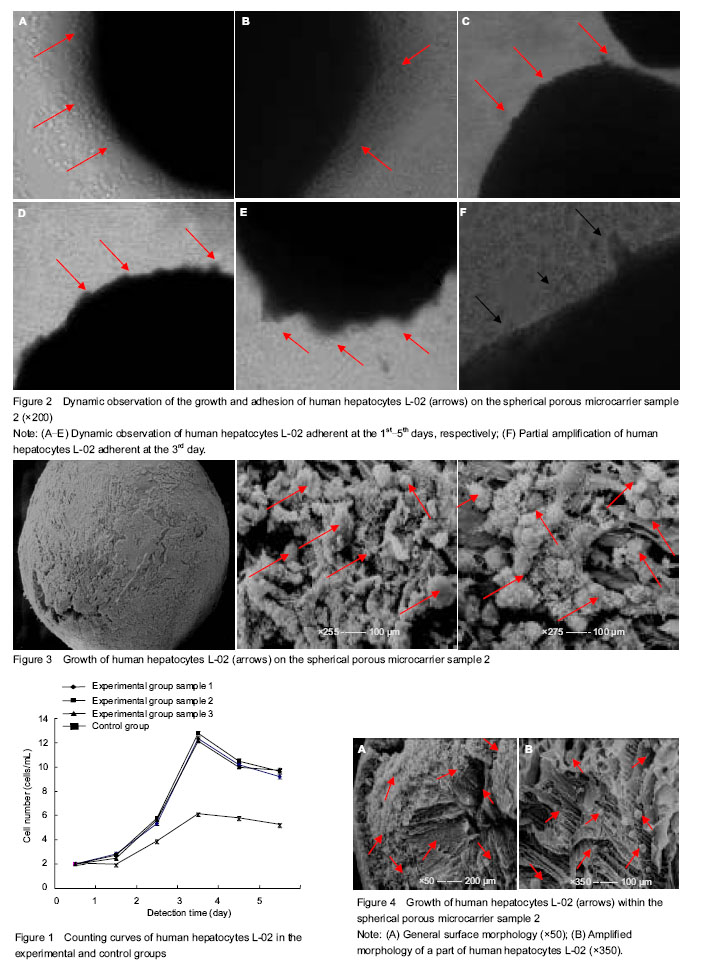
背景:微载体培养技术作为一项体外高浓度细胞培养技术,近年来已在肝细胞的体外培养中得到应用。 目的:对壳聚糖球形多孔微载体培养的人肝细胞L-02进行定时的形态学观察。 方法:以自制的壳聚糖球形多孔微载体样本为支架来培养人肝细胞L-02设为实验组;无壳聚糖球形多孔微载体支持下人肝细胞的培养设为对照组。对两组细胞进行定时的细胞计数,对实验组进行形态学观察,包括倒置相差生物显微镜观察和扫描电子显微镜观察。 结果与结论:两组培养的细胞数量均呈现前3 d增长,在培养第3天细胞数量达到最高值;实验组3个样本培养的细胞数明显高于对照组无微载体培养的细胞数量(P < 0.05),实验组各样本之间细胞数差异无显著性意义(P > 0.05)。倒置相差生物显微镜下动态观察,可见前3 d微载体表面黏附生长的肝细胞则逐渐增多,培养第3天可见大部分微载体表面有许多肝细胞黏附成团,总的存活率均在90%以上,且肝细胞保持着良好的形态学结构;扫描电子显微镜观察,微载体表面、切面和内部均可看到有许多球状肝细胞紧密黏附。结果说明,以自制的壳聚糖球形多孔微载体作为一种支架,在体外三维环境下可以进行高浓度细胞培养。
中图分类号:
引用本文
张 瑞,刘 明. 球形多孔微载体支持下高密度培养人肝细胞L-02的定时形态变化[J]. 中国组织工程研究, 2015, 19(12): 1924-1930.
Zhang Rui, Liu Ming. Timed morphological changes of human hepatocytes L-02 cultured at high density by the support of spherical porous chitosan microcarriers[J]. Chinese Journal of Tissue Engineering Research, 2015, 19(12): 1924-1930.
| [1] Kang H, Lu S, Peng J, et al. Chondrogenic differentiation of human adipose?derived stem cells using microcarrier and bioreactor combination technique. Mol Med Rep. 2015;11(2): 1195-1199. [2] Zhang Z, Zhou HC, Li ZG, et al. Microgravity culture ofhepatocytes on cellulose/gelatin macroporous microcarrier. Nan Fang Yi Ke Da Xue Xue Bao. 2010;30(4): 704-707. [3] Jin ZK, Xu CX, Tian PX, et al. Immune monitoring in kidney transplant recipients could predict acute rejection by a new method: flow cytometric microcarrier assay. Transplant Proc. 2013;45(4):1508-1510. [4] Bock A, Schulze-Horsel J, Schwarzer J, et al. High-density microcarrier cell cultures for influenza virus production. Biotechnol Prog. 2011;27(1):241-250. [5] Yu P, Huang Y, Zhang Y, et al. Production and evaluation of a chromatographically purified Vero cell rabies vaccine (PVRV) in China using microcarrier technology. Hum Vaccin Immunother. 2012;8(9):1230-1235. [6] Lam AT, Chen AK, Li J, et al. Conjoint propagation and differentiation of human embryonic stem cells to cardiomyocytes in a defined microcarrier spinner culture. Stem Cell Res Ther. 2014;5(5):110. [7] Tashiro S, Tsumoto K, Sano E. Establishment of a microcarrier culture system with serial sub-cultivation for functionally active human endothelial cells. J Biotechnol. 2012;160(3-4):202-213. [8] Lai JY. Biofunctionalization of gelatin microcarrier with oxidized hyaluronic acid for corneal keratocyte cultivation. Colloids Surf B Biointerfaces. 2014;122:277-286. [9] Ting S, Chen A, Reuveny S, et al. An intermittent rocking platform for integrated expansion and differentiation of human pluripotent stem cells to cardiomyocytes in suspended microcarrier cultures. Stem Cell Res. 2014;13(2):202-213. [10] Rodrigues CA, Diogo MM, da Silva CL, et al. Microcarrier expansion of mouse embryonic stem cell-derived neural stem cells in stirred bioreactors. Biotechnol Appl Biochem. 2011; 58(4):231-242. [11] Abeille F, Mittler F, Obeid P, et al. Continuous microcarrier-based cell culture in a benchtop microfluidic bioreactor. Lab Chip. 2014;14(18):3510-3518. [12] Kim BJ, Zhao T, Young L, et al. Batch, fed-batch, and microcarrier cultures with CHO cell lines in a pressure-cycle driven miniaturized bioreactor. Biotechnol Bioeng. 2012; 109(1):137-145. [13] Hupfeld J, Gorr IH, Schwald C, et al. Modulation of mesenchymal stromal cell characteristics by microcarrier culture in bioreactors. Biotechnol Bioeng. 2014;111(11): 2290-2302. [14] Ning B, Liu HF, Gong WM, et al. The changes of extracellular matrix in adult degenerative nucleus pulposus cells with stiring microcarrier culture system in vitro. Zhonghua Wai Ke Za Zhi. 2013;51(5):432-436. [15] Lu SJ, Kelley T, Feng Q, et al. 3D microcarrier system for efficient differentiation of human pluripotent stem cells into hematopoietic cells without feeders and serum. Regen Med. 2013;8(4):413-424. [16] Chen AK, Chen X, Choo AB, et al. Critical microcarrier properties affecting the expansion of undifferentiated human embryonic stem cells. Stem Cell Res. 2011;7(2):97-111. [17] Kurokawa M, Sato S. Growth and poliovirus production of Vero cells on a novel microcarrier with artificial cell adhesive protein under serum-free conditions. J Biosci Bioeng. 2011; 111(5): 600-604. [18] Goh TK, Zhang ZY, Chen AK, et al. Microcarrier culture for efficient expansion and osteogenic differentiation of human fetal mesenchymal stem cells. Biores Open Access. 2013;2(2):84-97. [19] Park Y, Subramanian K, Verfaillie CM, et al. Expansion and hepatic differentiation of rat multipotent adult progenitor cells in microcarrier suspension culture. J Biotechnol. 2010;150(1): 131-139. [20] Chen AK, Reuveny S, Oh SK. Application of human mesenchymal and pluripotent stem cell microcarrier cultures in cellular therapy: achievements and future direction. Biotechnol Adv. 2013;31(7):1032-1046. [21] Ning B, Liu H, Gong W, et al. Biological characteristics of adult degenerative nucleus pulposus cells in a three-dimensional microcarrier stirring culture system. J Orthop Res. 2013;31(6): 858-863. [22] Chang J, Lei H, Liu Q,et al. Optimization of culture of mesenchymal stem cells: a comparison of conventional plate and microcarrier cultures. Cell Prolif. 2012;45(5):430-437. [23] Bardy J, Chen AK, Lim YM, et al. Microcarrier suspension cultures for high-density expansion and differentiation of human pluripotent stem cells to neural progenitor cells. Tissue Eng Part C Methods. 2013;19(2):166-180. [24] Shakhbazau A, Shcharbin D, Bryszewska M, et al. Non-viral engineering of skin precursor-derived Schwann cells for enhanced NT-3 production in adherent and microcarrier culture. Curr Med Chem. 2012;19(32):5572-5579. [25] Lecina M, Ting S, Choo A, Reuveny S, et al. Scalable platform for human embryonic stem cell differentiation to cardiomyocytes in suspended microcarrier cultures. Tissue Eng Part C Methods. 2010;16(6):1609-1619. [26] Lippens E, Cornelissen M. Slow cooling cryopreservation of cell-microcarrier constructs. Cells Tissues Organs. 2010; 192(3): 177-186. [27] Justice C, Leber J, Freimark D, et al. Online- and offline- monitoring of stem cell expansion on microcarrier. Cytotechnology. 2011;63(4):325-335. [28] Surrao DC, Khan AA, McGregor AJ, et al. Can microcarrier-expanded chondrocytes synthesize cartilaginous tissue in vitro? Tissue Eng Part A. 2011;17(15-16): 1959-1967. [29] Schrobback K, Klein TJ, Schuetz M, et al. Adult human articular chondrocytes in a microcarrier-based culture system: expansion and redifferentiation. J Orthop Res. 2011;29(4): 539-546. [30] Leung HW, Chen A, Choo AB, et al. Agitation can induce differentiation of human pluripotent stem cells in microcarrier cultures. Tissue Eng Part C Methods. 2011;17(2):165-172. |
| [1] | 张同同, 王中华, 文 杰, 宋玉鑫, 刘 林. 3D打印模型在颈椎肿瘤手术切除与重建中的应用[J]. 中国组织工程研究, 2021, 25(9): 1335-1339. |
| [2] | 曾燕华, 郝延磊. 许旺细胞体外培养及纯化的系统性综述[J]. 中国组织工程研究, 2021, 25(7): 1135-1141. |
| [3] | 徐东紫, 张 婷, 欧阳昭连. 心脏组织工程领域全球专利竞争态势分析[J]. 中国组织工程研究, 2021, 25(5): 807-812. |
| [4] | 李晨杰, 吕林蔚, 宋 阳, 刘静娜, 张春秋. 预紧力作用下钛合金人工假体界面骨小梁形态参数测量与统计分析[J]. 中国组织工程研究, 2021, 25(4): 516-520. |
| [5] | 张国梅, 祝 军, 胡 杨, 焦红卫. E-Max瓷嵌体三维有限元模型粘接界面应力分析[J]. 中国组织工程研究, 2021, 25(4): 537-541. |
| [6] | 吴子健, 胡昭端, 谢有琼, 王 峰, 李 佳, 李柏村, 蔡国伟, 彭 锐. 3D打印技术与骨组织工程研究文献计量及研究热点可视化分析[J]. 中国组织工程研究, 2021, 25(4): 564-569. |
| [7] | 李 黎, 马 力. 磁性壳聚糖微球固定化乳糖酶及其酶学性质[J]. 中国组织工程研究, 2021, 25(4): 576-581. |
| [8] | 常文辽, 赵 杰, 孙晓亮, 王 锟, 吴国锋, 周 剑, 李树祥, 孙 晗. 人工骨膜的材料选择、理论设计及生物仿生功能[J]. 中国组织工程研究, 2021, 25(4): 600-606. |
| [9] | 刘 旒, 周箐竹, 龚 桌, 刘博言, 杨 斌, 赵 娴. 胶原/无机材料构建组织工程骨的特点及制造技术[J]. 中国组织工程研究, 2021, 25(4): 607-613. |
| [10] | 刘 飞, 崔宇韬, 刘 贺. 局部抗生素递送系统治疗骨髓炎的优势与问题[J]. 中国组织工程研究, 2021, 25(4): 614-620. |
| [11] | 李晓壮, 段 浩, 王伟舟, 唐志宏, 王旸昊, 何 飞. 骨组织工程材料治疗骨缺损疾病在体内实验中的应用[J]. 中国组织工程研究, 2021, 25(4): 626-631. |
| [12] | 张振坤, 李 喆, 李 亚, 王莹莹, 王亚苹, 周馨魁, 马珊珊, 关方霞. 海藻酸盐基水凝胶/敷料在创面愈合中的应用:持续、动态与顺序释放[J]. 中国组织工程研究, 2021, 25(4): 638-643. |
| [13] | 陈佳娜, 邱燕玲, 聂敏海, 刘旭倩. 组织工程支架材料修复口腔颌面部软组织缺损[J]. 中国组织工程研究, 2021, 25(4): 644-650. |
| [14] | 张 羽, 冯 硕, 杨 志, 张 野, 孙健宁, 安 伦, 陈向阳. 高位髋关节中心全髋关节置换治疗发育性髋关节发育不良后的三维步态评价[J]. 中国组织工程研究, 2021, 25(3): 350-355. |
| [15] | 邢 浩, 张永红, 王 栋. 长骨大段骨缺损修复方法的优势与不足[J]. 中国组织工程研究, 2021, 25(3): 426-430. |
Normal human hepatocytes L-02 were purchased from Shanghai Institute of Cell Biology, Chinese Academy of Sciences; spherical porous microcarriers (samples 1, 2, 3) for small-size three-dimensional culture were made in the laboratory.
.jpg)
Experimental methods
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||