中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (26): 6930-6936.doi: 10.12307/2026.737
• 生物材料综述 biomaterial review • 上一篇 下一篇
天然口服水凝胶在药物递送系统中的开发与应用
杨 淇1,向 蹊1,王 涵2,邹 圳3,张伦慈2,米热阿德力·阿布力米提2,廖 悦1,李新志2,3
- 三峡大学,1基础医学院,3健康医学院,湖北省宜昌市 443002;2三峡大学附属仁和医院,湖北省宜昌市 443099
-
接受日期:2025-09-01出版日期:2026-09-18发布日期:2026-03-16 -
通讯作者:廖悦,博士,三峡大学基础医学院,湖北省宜昌市 443002 李新志,医学博士,教授,三峡大学健康医学院,湖北省宜昌市 443002;三峡大学附属仁和医院,湖北省宜昌市 443099 -
作者简介:杨淇,女,2000年生,湖北省宜昌市人,土家族,三峡大学在读硕士,主要从事水凝胶研究。 -
基金资助:肿瘤微环境与免疫治疗湖北省重点实验室开放基金项目(2023KZL07),项目负责人:廖悦;宜昌市医疗卫生科技项目(A24-2-059),项目负责人:廖悦;三峡大学人才科研启动基金项目(2023RCKJ013),项目负责人:廖悦;国家自然科学基金面上项目(81871956),项目负责人:李新志;中国康复医学会科技发展项目(2023132-55),项目负责人:李新志
Development and application of natural oral hydrogels in drug delivery systems
Yang Qi1, Xiang Xi1, Wang Han2, Zou Zhen3, Zhang Lunci2, Mireadeli·Abulimiti2, Liao Yue1, Li Xinzhi2, 3
- 1School of Basic Medicine, 3School of Health Sciences, China Three Gorges University, Yichang 443002, Hubei Province, China; 2Renhe Hospital Affiliated to China Three Gorges University, Yichang 443099, Hubei Province, China
-
Accepted:2025-09-01Online:2026-09-18Published:2026-03-16 -
Contact:Liao Yue, MD, School of Basic Medicine, China Three Gorges University, Yichang 443002, Hubei Province, China Li Xinzhi, MD, Professor, Renhe Hospital Affiliated to China Three Gorges University, Yichang 443099, Hubei Province, China; School of Health Sciences, China Three Gorges University, Yichang 443002, Hubei Province, China -
About author:Yang Qi, MS candidate, School of Basic Medicine, China Three Gorges University, Yichang 443002, Hubei Province, China -
Supported by:Open Foundation of Hubei Province Key Laboratory of Tumor Microenvironment and Immunotherapy, No. 2023KZL07 (to LY); Yichang Medical and Health Research Project, No. A24-2-059 (to LY); Talent Research Startup Fund of China Three Gorges University, No. 2023RCKJ013 (to LY); National Natural Science Foundation of China (Top-level Project), No. 81871956 (to LXZ); Science and Technology Development Project of Chinese Association of Rehabilitation Medicine, No. 2023132-55 (to LXZ)
摘要:
文题释义:
水凝胶:是一种由三维高分子网络结构和大量水分组成的软材料,具有高吸水性、生物相容性、可调控物理化学性质等特点,广泛应用于生物医学、环境工程、柔性电子等领域。
天然材料:来源于自然界的物质,通常由生物、矿物或天然资源中提取。根据组成成分和结构的不同,天然材料可以分为多种类型,包括植物来源、动物来源和矿物来源材料。
背景:口服给药因较高的依从性一直是患者最为偏好的药物递送途径,相较于注射给药能显著提升患者的治疗体验,但消化道内的极端环境严重限制了药物的生物利用度。随着医学领域对生物相容性和生物降解性的需求日益增加,天然水凝胶作为一种理想的药物递送载体正在受到广泛关注。
目的:从材料选择到合成方法探讨不同天然材料口服水凝胶的开发与应用。
方法:检索PubMed、Web of Science数据库,以“oral hydrogels,physical crosslinking,chemical crosslinking,natural material,therapy,drug delivery,application of disease research”为英文检索词,检索2009-2024年发表的相关文献,最终筛选出83篇文献进行综述。
结果与结论:水凝胶作为一种极具潜力的新型药物递送系统,在实现药物的精准递送与控释方面展现出巨大优势。口服天然水凝胶凭借卓越的生物相容性、良好的可降解性及极低的潜在毒性,在药物递送领域脱颖而出,它不仅能够实现药物的精准投递,还能有效避免药物与胃肠道直接接触而产生的刺激,为药物递送提供了更为安全、有效的途径。随着科研工作者的不懈探索,基于天然材料的新型智能水凝胶递送系统不断涌现,如果胶基 pH 响应性水凝胶,透明质酸基活性氧响应性水凝胶等,这些新型材料为药物递送的智能化和精准化开辟了新的道路。然而,天然材料水凝胶在应用过程中也暴露出一些有待解决的问题:一方面,天然材料普遍存在机械性能和拉伸强度不足的问题,这使它们难以满足复杂的药物输送需求;另一方面,尽管天然材料源于自然,但它们仍有可能引发人体免疫反应。
https://orcid.org/0009-0007-4608-398X(杨淇)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中图分类号:
引用本文
杨 淇, 向 蹊, 王 涵, 邹 圳, 张伦慈, 米热阿德力·阿布力米提, 廖 悦, 李新志. 天然口服水凝胶在药物递送系统中的开发与应用[J]. 中国组织工程研究, 2026, 30(26): 6930-6936.
Yang Qi, Xiang Xi, Wang Han, Zou Zhen, Zhang Lunci, Mireadeli·Abulimiti, Liao Yue, Li Xinzhi. Development and application of natural oral hydrogels in drug delivery systems[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(26): 6930-6936.
2.2 天然材料 天然材料来源天然且常被用于食品添加剂,如常见的魔芋胶、果胶,因此有绝对优越的生物相容性和可降解特性,易被人体代谢吸收。天然材料来源广泛、成本低廉,基于这些优势它们在医药学以及生物工程领域呈现出极为广阔的应用前景。常用于合成口服水凝胶的天然材料往往还有着各种各样的特性,如海藻酸盐水凝胶在酸性环境下稳定、在肠道环境下易降解,透明质酸拥有特异性靶向能力,果胶和葡聚糖对肠道酶类表现出极强的敏感性,还有很多天然中药成分(如大黄酸、甘草酸)不仅具备优秀的抗炎、抗氧化、抗肿瘤效果,还能通过物理自组装,从而提升药物利用率和药物稳定性。依据分子尺寸大小,天然材料可分为天然高分子材料与天然小分子材料,天然高分子材料包含多糖类海藻酸盐、壳聚糖、透明质酸、硫酸软骨素以及蛋白质类(如明胶、胶原蛋白)等[20],天然小分子材料包含葡萄糖、甘露糖、大黄酸、甘草酸、小檗碱等[21]。
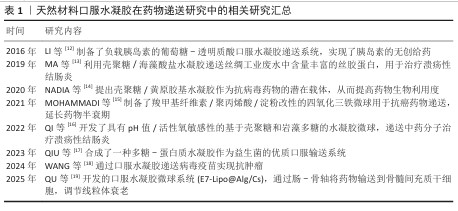
2.2.1 天然高分子材料
海藻酸盐:主要来源于褐藻和细菌,是由β-D-甘露醇α-L-古洛糖醛酸通过β-1,4糖苷键连接的长链聚合物[22]。由于海藻酸盐富含羧基基团,因此对pH值敏感:在胃酸环境下,羧基质子化从而与阳离子(如Ca2+)自发凝胶化,起到抵抗胃酸的作用;在高pH值环境下,羧基发生去质子化,离子间的静电斥力相互排斥,使得水凝胶网络变得松散,从而释放装载的药物。这种pH值智能响应性,使得海藻酸盐能够很好地实现药物定向释放。
壳聚糖:来源于虾蟹等动物外壳中的天然多糖,由N-乙酰葡萄糖胺和葡萄糖胺通过β-1,4糖苷键连接而成,分子中富含大量的氨基基团,能够黏附在胃肠道黏膜上。壳聚糖具有优异的抗炎、抗菌作用,对于细菌(如金黄色葡萄球菌、大肠杆菌)和真菌(白色念珠菌)有广谱抑制作用,可促进伤口愈合和组织修复,常被用于伤口敷料。
透明质酸:是肠壁黏膜、上皮和细胞外基质的主要成分之一,具有丰富的官能团,包括羧基和羟基,可用于化学修饰。ZHANG等[23]通过在透明质酸上接枝巯基自交联合成水凝胶,用于口服递送益生菌缓解肠道炎症。此外,透明质酸还能够与癌细胞过度表达的CD44受体特异性结合,从而实现抗癌药物的靶向递送[24]。
硫酸软骨素:是结缔组织和软骨中的重要结构成分,由β-(1,4)-D.-葡萄糖醛酸和 β-(1,3)-N-乙酰-D-半乳糖胺的交替单元组成。硫酸软骨素富含硫酸基团和糖胺基团,与炎症结肠中巨噬细胞表面高度过表达的糖蛋白CD44 表现出很强的亲和力,并通过CD44介导的内吞作用促进细胞内化[25]。因此,在很多肠道炎症部位,高表达的CD44跨膜蛋白给予了硫酸软骨素一定的靶向性,使硫酸软骨素构成的水凝胶具有靶向递送特性。OPREA等[26]制备的硫酸软骨素纳米凝胶颗粒用于递送姜黄素治疗溃疡性结肠炎,正是利用了硫酸软骨素的靶向特点。
2.2.2 天然小分子材料
大黄酸:是一种亲脂性蒽醌化合物,来源于各种传统中草药,包括大黄、芦荟和番泻叶。大黄酸具有多种有益特性,例如抗炎、抗氧化、抗肿瘤和神经保护作用[27],但药物稳定性差、水溶性低和靶向能力有限等限制了它在临床上的应用。LIANG等[28]利用大黄酸分子物理自组装特性合成大黄酸水凝胶,用于抗肿瘤,发现大黄酸可抑制糖酵解途径、诱导免疫原性细胞死亡、激活树突状细胞、向 CD8 T细胞呈递主要组织相容性复合体Ⅰ类,从而增强肿瘤杀伤效果。大黄酸水凝胶的出现使大黄酸突破了自身性质的限制,显著提升了药物稳定性和生物利用率。
甘草酸:是一种源自甘草植物根部的天然草本皂苷,具有广泛的生物活性,如抗炎、保肝、抗癌和抗病毒活性。ZHANG等[29]研究的镓/甘草酸水凝胶可用于多重耐药的铜绿假单胞菌感染。由于甘草酸具有两亲性,它能自发的直接自组装成水凝胶,并且不需要额外的化学基团修饰,方法简便[30]。
没食子酸:是来自茶叶、牡丹、中国坚果、山茱萸和其他植物的天然多酚,具有多重生物学功能,包括抗炎、抗氧化,用于癌症以及心血管和神经系统疾病的治疗中[31]。与其他天然多酚一样,没食子酸容易被氧化成醌类化合物,同时生物利用度低、亲脂性差、异味和在人体内代谢快等特点阻碍了它在临床上的进一步应用[32-33]。为了提高没食子酸的生物利用率和疗效,HUANG等[34]利用没食子酸自组装性质制备了没食子酸水凝胶,通过口服给药治疗溃疡性结肠炎,研究表明没食子酸水凝胶能够抑制炎性细胞因子的表达、调节巨噬细胞极化、改善肠黏膜屏障功能。
2.3 合成方式 由于天然材料各自独特的性质,可借助多种不同方式合成口服水凝胶。口服的天然高分子水凝胶主要通过物理交联与化学交联2种路径形成,而天然小分子水凝胶则主要依赖物理自组装作用,依靠分子间的相互作用力自行组织构建而成。表2总结了口服水凝胶不同交联方式的优缺点。

以物理交联方式合成的天然高分子水凝胶,具备成胶步骤简易、环境响应性良好、可注射性强以及自我修复能力突出等特点。例如,QI等[16]利用富含羧基(-COOH)的海藻酸盐与含有大量氨基基团(-NH3+)的壳聚糖之间的静电相互作用,成功研制出具有pH值响应性的海藻酸钠-壳聚糖基口服水凝胶,该水凝胶在胃酸环境下能够保持稳定;在肠道弱碱性环境中,-COOH解离为-(-COO-),-NH3+去质子化促使水凝胶降解,赋予水凝胶出色的黏膜黏附性能,进而实现药物在肠道的定向释放。
低溶解度和不稳定性限制了天然小分子的临床应用。有研究者报道,一些天然小分子可通过氢键、π-π堆积、范德华力和疏水作用,以物理自组装方式合成口服天然水凝胶,能够显著提高天然小分子的溶解度和生物利用率,增强天然小分子的稳定性。例如,ZHONG等[38]开发了一种大黄酸微藻复合水凝胶,大黄酸分子中的芳香环通过π-π堆积与其他大黄酸分子的芳香环相互作用,含有的多个羟基(-OH)和酮基(-C=O)基团可通过氢键与其他分子或基团形成稳定的相互作用,成功解决了大黄酸因溶解问题所受到的限制。GAO等[39]探索了小檗碱与橙皮素2种天然植物小分子的自组装特性,通过静电相互作用和氢键等非共价键作用显著提高了橙皮素的溶解度,展现出协同抗炎活性。
2.3.2 化学交联方式 以化学交联方式合成的水凝胶,通过在聚合物分子之间形成共价键而制成,通常需要使用交联剂,借助化合物之间的化学反应来促进交联。依据交联机制的差异可分为动态共价键交联、自由基聚合、传统共价交联等类型。与物理交联水凝胶相比,化学交联水凝胶呈现出更卓越的稳定性、降解能力以及机械性能,然而由于交联剂存在潜在毒性,水凝胶的生物相容性逊色于物理交联方式合成的水凝胶。
动态共价键交联:动态共价键基于可逆共价键(如酰胺键、二硫键)在特定条件下能够实现连接或断裂,这种特性使得通过该方法合成的水凝胶更能适应动态变化的生物环境,能够对外部刺激做出响应。例如,DU等[40]开发了一种基于硫基修饰的透明质酸口服水凝胶,具有灵敏的活性氧响应性,活性氧会促使巯基氧化成二硫键,从而在炎症部位特异性形成水凝胶[41],这种成胶方式对于病灶部位具有更好的特异性和定向性。除了二硫键以外,还有其他动态化学键用于构建口服水凝胶,如氧化透明质酸的醛基与Ⅰ-精氨酸的2个胺基产生的希夫碱组装[42],葡聚糖和巯基化牛血清白蛋白通过迈克尔加成反应交联水凝胶[17]。
自由基聚合:自由基聚合反应具有良好的可控性,特别是在反应条件(如温度、光照[43]、pH值等)和不同功能性基团修饰的单体选择方面,能够实现水凝胶的多种定制化功能。ZHANG等[44]通过酰胺化反应合成甲基丙烯酸酯酐改性的羧甲基壳聚糖口服水凝胶,通过甲基丙烯酸酯酐改性后水凝胶的交联度和结构稳定性得以改善,通过调节交联度和溶胀特性可调控药物的长期控释过程。还有研究将聚乙二醇和甲基丙烯酸接枝到果胶上,使得水凝胶具备更好的稳定性和抗压性能,在胃肠道的运动作用下仍能够保持完整的结构[45]。但自由基聚合常常使用一些有毒或对环境有害的引发剂(如过硫酸盐类、过氧化物等),这些化学品需要在严格的控制下使用,并且一些引发剂残留物可能对环境和人体健康构成风险。有学者利用衣康酸(一种由藻类发酵得到的有机二羧酸)作为甲基丙烯酸等石化基单体的无毒替代品[46]。
传统共价交联:共价交联通过强共价键连接高分子链,使得水凝胶具有较高的化学稳定性、热稳定性和机械强度,能够在恶劣环境下长期保持稳定。传统的共价交联一般需要使用交联剂,交联剂一般分为化学交联剂和天然交联剂。化学交联剂(例如戊二醛)的细胞毒性较大,可能引发炎症。现如今大多使用的化学交联剂有环氧类(如聚乙二醇二缩水甘油醚、1,4-丁二醇二缩水甘油醚),相较于戊二醛显著降低了毒性,并且能交联多种官能团,同时引入聚乙二醇链可增强水凝胶的亲水性。WANG等[47]利用1,4-丁二醇二缩水甘油醚与银耳多糖中的羟基(-OH)反应交联形成水凝胶,显著增强了材料的机械性能和肠道黏附性。天然交联剂的生物相容性和降解性更好,对人体无毒,用于合成可口服的水凝胶更为安全。常用的天然交联剂有单宁、京尼平[48]、衣康酸等。
物理化学双交联:通过物理化学双交联的口服水凝胶具备良好生物相容性与机械性能。例如,LIU等[49]报道了一种口服水凝胶用于血小板膜的递送恢复肠道微血管损伤,该水凝胶由甲基丙烯酸透明质酸和负载瑞巴派特的聚酰胺胺树枝状聚合物通过静电自组装加光交联制成,物理化学双交联的口服水凝胶确保了血小板膜的活性,避免了胃酸环境影响,为管理肠道微血管损伤引起的便血提供了新的方案。
2.4 天然水凝胶口服递送药物类型 不同天然材料水凝胶递送系统的研制,为药物递送领域开辟了新的途径。并且随着新型材料的发掘,口服递送药物的稳定性得到提升,有些难以实现口服途径给药的药物,在水凝胶的帮助下有了新的可能。
中药小分子:中药小分子通过口服给药时存在的溶解性差、吸收难度较大、生物利用度低的问题,在水凝胶递送过程中得到了解决。现有研究通过口服途径给药的中药小分子通常有大黄酸、姜黄素、甘草酸、小檗碱、黄芩苷等一系列抗炎药物,这些中药分子可以自组装成水凝胶,真正实现“药-胶”一体,使得水凝胶不再是传统意义上简单的载体,这为中药的现代化应用提供了新的思路和方法。
西药分子:对于西药分子,天然口服水凝胶的研究重点在于抗癌药物递送,缓解药物不良反应、延长药物半衰期以及靶向治疗[50]。例如,通过水凝胶递送伊马替尼、盐酸多柔比星治疗结肠癌[15,43]。
菌群:除了一些药物分子,天然口服水凝胶还被用于递送益生菌,或者一些功能化的细菌、病毒[51]。益生菌对机体有着各种有益影响,天然水凝胶可以实现益生菌的理想输送和按需释放,并且有效提高益生菌活性。
蛋白质:蛋白质在口服递送途径中极其容易受到胃肠道pH值影响而变性,如糖尿病患者必需的胰岛素仅止步在注射途径,而口服水凝胶装载胰岛素让无创给药途径成为可能[52]。还有痛风患者的尿酸代谢,通过水凝胶递送尿酸酶作用可经肠道代谢,从而减轻肾脏负担[53]。
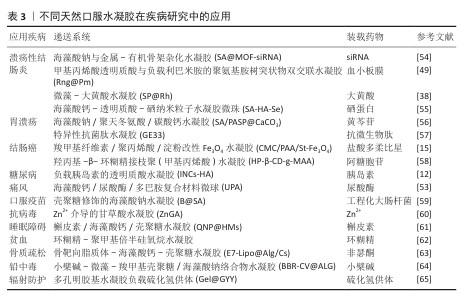
2.5 天然口服水凝胶的应用 基于天然材料合成的口服水凝胶在众多疾病领域展现出广泛的应用前景,如表3。天然口服水凝胶作为一种极具潜力的药物载体,能够精准靶向并缓慢释放药物,显著提高药物利用率,从而增强治疗效果,为疾病治疗开辟了新路径。
2.5.1 消化系统疾病
溃疡性结肠炎:口服水凝胶在消化系统疾病的治疗研究中应用广泛,如溃疡性结肠炎、胃溃疡等,尤其以溃疡性结肠炎的治疗为主。针对溃疡性结肠炎的治疗策略,临床上仍缺乏特效药物,主要治疗目标集中于缓解症状和改善患者生活质量[66]。一线常用药物(如水杨酸制剂和免疫抑制剂等)虽有一定疗效,但因对胃肠道的刺激常引发恶心、呕吐等不良反应,给患者带来身心双重困扰[67]。
研究人员利用天然高分子材料构建结肠靶向口服水凝胶递送系统,开展溃疡性结肠炎治疗的相关研究。对于治疗溃疡性结肠炎的口服水凝胶递送系统而言,需具备抵抗胃液侵蚀的能力,同时拥有肠道黏附能力以保证药物有效作用;此外,水凝胶还应具有结肠靶向性,在结肠环境中缓慢降解实现药物的缓慢释放,达到持续治疗的效果。例如,HE等[68]采用海藻酸盐-金属有机框架水凝胶递送小干扰 RNA,siRNA 药物已被广泛用作多种炎症相关疾病的潜在候选药物,但其口服利用率非常低,研究利用海藻酸盐表现出良好的抗胃酸保护作用,金属有机框架对siRNA有强吸附作用,实现口服递送,保证了siRNA的稳定性和持续缓释效果[54]。MA等[13]利用壳聚糖制备口服水凝胶,递送丝胶蛋白以加速溃疡创面愈合,丝胶蛋白可以通过抑制核因子κB 和丝裂原活化蛋白激酶通路抑制促炎细胞因子的合成和分泌,发挥抗炎活性。
胃溃疡:胃溃疡是一种慢性疾病,特征是对受阻的胃肌肉造成刺激性损害,严重影响患者的生活质量[69]。临床通常采用抗幽门螺杆菌治疗,使用质子泵抑制剂(奥美拉唑、雷贝拉唑等)或H2受体拮抗剂,然而,口服给药时,胃中的酶屏障、扩散屏障和吸收屏障阻碍了治疗胃损伤药物的有效留存,导致生物利用度降低。利用海藻酸钠、角蛋白等在胃部酸性环境中相对稳定的生物大分子可实现药物的持续递送,并形成黏附屏障,隔绝胃酸对溃疡部位的持续伤害,同时缓慢的药物释放可避免部分药物引发的不良反应。
GUO等[57]设计了胃pH值响应性凝胶系统,递送对幽门螺杆菌表现出良好治疗效果的抗微生物肽,实现抗微生物肽的持续释放作用。CHENG等[70]以头发丝中提取的角蛋白为原料,研制了口服角蛋白水凝胶,该水凝胶在伤口处诱导形成高黏度凝胶的特性可加速溃疡愈合,通过止血、分离胃液和溃疡,为组织再生提供细胞外基质支架,用于治疗胃溃疡。XU等[56]利用pH值响应性海藻酸钠/聚天冬氨酸钠/CaCO3原位水凝胶用于口服递送抗炎中药分子黄芩苷。
结肠癌:肠道和肿瘤内微生物群与肿瘤进展及对抗肿瘤治疗的反应密切相关。抗菌或肿瘤微环境调节方法已被证实可显著提高抗肿瘤疗效。天然材料口服水凝胶因具备结肠靶向、结肠保留作用及响应性降解等特性,可实现抗癌药物的精准递送。有研究报道了一种具有结肠靶向和保留作用的口服菊粉基水凝胶递送化疗药物奥沙利铂,提高了化疗效率、激活了抗肿瘤免疫应答,为治疗易受微生物侵袭的结直肠癌提供了重要的临床途径[43]。MOHAMMADi等[15]制备了羧甲基纤维素/聚丙烯酸/淀粉改性的四氧化三铁水凝胶珠,来负载抗癌药物盐酸多柔比星和5-氟尿嘧啶,以减轻药物对于消化道带来的严重不良反应,增强结直肠区域药物剂量的稳定性。BATOOL等[58]研制了一种羟基丙基β-环糊精的聚合物水凝胶,以提高低渗透性药物(例如阿糖胞苷)的功能和溶解度、延长药物半衰期、降低要给药频率,从而提高患者的依从性。
2.5.2 代谢性疾病
糖尿病:糖尿病患者通常需通过注射胰岛素维持血糖稳定,而无创的口服给药仍是未来研究的重要方向[71]。胰岛素治疗口服给药不仅可以减轻注射引起的疼痛,还可以模拟胰岛素的生理命运,可能提供更好的葡萄糖稳态。利用天然多糖的特性制备pH值响应性口服凝胶,成为实现口服胰岛素的有效途径[72],该凝胶需具备胃酸抵抗性,在肠道内通过pH值响应性、酶响应性等机制降解释放胰岛素,以促进胰岛素在肠道的吸收。CHEN等[73]使用羧甲基环糊精修饰的羧甲基壳聚糖开发了pH值敏感的水凝胶微粒,在胰岛素负载效率(70%)、胰岛素释放效率(98%)和缓释时间( > 20 h)方面取得了良好效果。LI等[12]开发了负载胰岛素的葡萄糖反应性纳米载体复合水凝胶系统,以实现胰岛素的智能控释,更好地调整体内胰岛素水平。
痛风:痛风是由高尿酸血症引起的炎症性关节炎[74],近4%的成年人表现出相关症状,并且随着生活习惯问题的凸显,痛风患者越来越年轻化。肾排泄途径是当前降尿酸药物的主要靶点,而传统的痛风药物也可能加剧肾脏负担。小肠是尿酸排泄中仅次于肾脏的第二代谢器官,大约1/3之一的尿酸通过肠道排泄代谢。TANG等[53]利用天然材料口服水凝胶递送尿酸酶,在肠道释放水解尿酸上调肠上皮细胞的多种尿酸转运蛋白,可实现尿酸的肠道排泄,该研究为痛风治疗提供了一条通过肠道原位调节实现尿酸肠道排泄的独特路径。LIU等[75]设计了基于口服微藻的水凝胶系统,可以通过消除活性氧、重编程炎症微环境以促进细胞向M2样抗炎细胞极化,抑制 NLRP3-白细胞介素1β通路和减少促炎因子的表达,抑制痛风性关节炎的进展。
2.5.3 免疫系统相关疾病
口服疫苗:口服免疫疗法作为免疫疗法药物的一种有利途径,在免疫激活和药物管理依从性方面具有巨大的发展潜力。已报道的口服疫苗水凝胶可避免胃部环境影响,将抗原物质递送至肠道,引发全身免疫反应。WANG等[18]开发的口服肿瘤疫苗,利用交联的葡聚糖水凝胶将来自B型肝炎病毒核心(HBc)抗原的病毒样颗粒递送到肠道进行黏膜免疫。CHEN等[76]
研究的pH值响应性细菌纳米纤维素/聚丙烯酸水凝胶微粒,可作为口服疫苗载体,通过口服和注射2种不同免疫接种方式证明了口服疫苗产生的血清抗体水平显著高于肌肉注射。ZHANG等[59]提出可编程的口服细菌水凝胶用于原位生产和释放肿瘤纳米疫苗,该疫苗使用壳聚糖和海藻酸钠包埋工程化的大肠杆菌并在肠道定植,为口服蛋白疫苗递送提供新思路。以上表明口服疫苗水凝胶是一种极为可行的策略,在提升患者舒适度的同时增强免疫激活效果。
抗病毒:现有的大多数抗病毒药物往往通过口服途径给药,然而半衰期短以及可能存在的胃肠道不良反应问题较突出。像婴幼儿容易感染呼吸道合胞病毒,传统片剂口服给药的方式可能存在吞咽困难或频繁给药的情况。LU
等[60]报道了新型的 Zn2+-介导的超分子自组装甘草酸水凝胶用于口服治疗感染呼吸道合胞病毒感染,口服锌补充剂可有效治疗病毒感染,该水凝胶可用于治疗婴儿和儿童的呼吸道合胞病毒感染。治疗单纯疱疹病毒感染的阿昔洛韦,市售片剂药物的平均血浆半衰期为3 h,在传统的药物输送系统中需要每天服用5次,这会导致与高剂量相关的不良反应。MALIK等[14]
提出用黄原胶/壳聚糖水凝胶口服递送阿昔洛韦,达到长效释放药物、减少给药频率的目的。SUN等[77]开发了一种pH值触发的腐植酸水凝胶递送系统,递送抑制水泡性口炎病毒和伪狂犬病病毒的复制干扰素α和干扰素γ,突出了它作为抗伪狂犬病病毒感染口服治疗剂的潜力。
类风湿性关节炎:是一种常见的慢性系统性自身免疫性疾病,可导致多个关节的软骨和骨骼损伤,最终导致残疾。青藤碱在治疗类风湿性关节炎方面具有独特的药理活性,但由于极短的半衰期和不良的消化道反应,它的临床应用受到限制。JIANG等[78]用青藤碱和甘草酸通过非共价键生产自组装纳米水凝胶,可有效延长青藤碱的半衰期、促进青藤碱的吸收利用,为中药化合物组成的成药开发新药提供了一种新的策略。
2.5.4 其他应用
贫血:贫血是一个影响成人和儿童的全球性健康问题,但治疗受到严重不良反应的阻碍,主要与口服药物的胃肠道反应有关。环糊精的羧基是Fe2+结合位点,ORLOVA等[62]研制了环糊精-聚甲基倍半硅氧烷水凝胶口服给药系统,有效提高了Fe2+负载效率并延长释放时间。
解毒:铅是最普遍和毒性最强的重金属之一 。通过与负责细胞抗氧化防御系统的蛋白质和酶结合,铅诱导氧化应激,导致对多个身体系统产生毒性。LIU等[64]发了一种多功能口服水凝胶系统,该系统由小球藻、抗炎药物分子小檗碱和稳定剂羧甲基壳聚糖/海藻酸钠复合物组成,它可以吸附和去除活体中的铅,缓解铅中毒相关疾病。
骨质疏松:肠-骨轴是骨质疏松症治疗的一个有前途靶点,但现有的输送系统缺乏精确的靶向性。Qu等[19]开发了口服水凝胶微球系统(通过使用气体微流体和离子交联技术将靶向脂质体封装在由海藻酸盐 和壳聚糖制成的水凝胶微球),通过肠骨轴将药物输送到骨髓间充质干细胞,调节线粒体衰老,该系统可实现口服给药、胃保护、肠道维持/控制释放和骨组织的主动靶向。
睡眠障碍:高海拔睡眠障碍是急性高山病的常见症状,可通过调节肠-脑轴来缓解。槲皮素可用于调节肠道微生物群,并作为调节肠-脑轴的潜在药物,但较差的溶解度和生物利用度会影响它的生物学功能。WU等[61]利用静电纺丝技术制备了负载槲皮素纳米颗粒的海藻酸钙水凝胶微球,以提高槲皮素纳米颗粒的胃肠道稳定性和肠黏附性。
胃肠道辐射防护:辐射损伤可导致一系列胃肠道疾病,安全有效的胃肠道放射防护剂的开发在临床上仍然是一个巨大挑战。SHAN
等[65]报道了一种口服胃肠道放射防护剂,利用多孔明胶基水凝胶负载硫化氢(H2S)供体,H2S可有效清除辐射产生的自由基、减少氧化应激和DNA损伤,恢复重要的超氧化去歧化酶和谷胱甘肽过氧化物酶稳态,从而起到防护作用。
| [1] XU H, CHE Y, ZHOU R, et al. Research progress of natural polysaccharide-based and natural protein-based hydrogels for bacteria-infected wound healing. Chem Eng J. 2024;496:153803. [2] HAO ZW, ZHANG ZY, WANG ZP, et al. Bioactive peptides and proteins for tissue repair: microenvironment modulation, rational delivery, and clinical potential. Mil Med Res. 2024;11(1):75. [3] LOKE YH, JAYAKRISHNAN A, MOD RAZIF MRF, et al. A Comprehensive Review of Challenges in Oral Drug Delivery Systems and Recent Advancements in Innovative Design Strategies. Curr Pharm Des. 2024; 31(5):360-376. [4] WANG ZD, ZHANG W, LIANG TX. Advancements in Oral Delivery Systems for Probiotics Based on Polysaccharides. Polymers (Basel). 2025;17(2):144. [5] WANG D, WANG W, WANG P, et al. Research progress of colon-targeted oral hydrogel system based on natural polysaccharides. Int J Pharm. 2023;643:123222. [6] CHOUDHARY S, RECK JM, CARR AJ, et al. Hydrophobically modified alginate for extended release of pharmaceuticals. Polym AdvTechnol. 2018;29(1):198-204. [7] SONG X, HUANG Q, YANG Y, et al. Efficient Therapy of Inflammatory Bowel Disease (IBD) with Highly Specific and Durable Targeted Ta(2) C Modified with Chondroitin Sulfate (TACS). Adv Mater. 2023; 35(36):e2301585. [8] ZHANG X, MA Y, MA L, et al. Oral administration of chondroitin sulfate-functionalized nanoparticles for colonic macrophage-targeted drug delivery. Carbohydr Polym. 2019;223:115126. [9] JIANG N, YU X, ZHANG J, et al. Smart stimuli-responsive hydrogels for safe oral administration of Insulin: A Review. Int J Pharm. 2025;674:125487. [10] WU YJ, WEI ZX, ZHANG FM, et al. Structure, bioactivities and applications of the polysaccharides from Tremella fuciformis mushroom: A review. Int J Biol Macromol. 2019; 121:1005-1010. [11] TIE BSH, HALLIGAN E, ZHUO S, et al. Synthesis of NVCL-NIPAM Hydrogels Using PEGDMA as a Chemical Crosslinker for Controlled Swelling Behaviours in Potential Shapeshifting Applications. Gels. 2023;9(3):248. [12] LI L, JIANG G, YU W, et al. A composite hydrogel system containing glucose-responsive nanocarriers for oral delivery of insulin. Mater Sci Eng C Mater Biol Appl. 2016;69:37-45. [13] MA Y, TONG X, HUANG Y, et al. Oral Administration of Hydrogel-Embedding Silk Sericin Alleviates Ulcerative Colitis through Wound Healing, Anti-Inflammation, and Anti-Oxidation. ACS Biomater Sci Eng. 2019; 5(11):6231-6242. [14] MALIK NS, AHMAD M, MINHAS MU, et al. Chitosan/Xanthan Gum Based Hydrogels as Potential Carrier for an Antiviral Drug: Fabrication, Characterization, and Safety Evaluation. Front Chem. 2020;8:50. [15] MOHAMMADI R, SABOURY A, JAVANBAKHT S, et al. Carboxymethylcellulose/polyacrylic acid/starch-modified Fe3O4 interpenetrating magnetic nanocomposite hydrogel beads as pH-sensitive carrier for oral anticancer drug delivery system. Eur Polym J. 2021;153:110500. [16] QI S, LUO R, HAN X, et al. pH/ROS Dual-Sensitive Natural Polysaccharide Nanoparticles Enhance “One Stone Four Birds” Effect of Rhein on Ulcerative Colitis. ACS Appl Mater Interfaces. 2022;14(45):50692-50709. [17] QIU J, XIANG S, SUN M, et al. Preparation of Polysaccharide-Protein Hydrogels with an Ultrafast Self-Healing Property as a Superior Oral Delivery System of Probiotics. J Agric Food Chem. 2023;71(48):18842-18856. [18] WANG C, TANG H, DUAN Y, et al. Oral biomimetic virus vaccine hydrogel for robust abscopal antitumour efficacy. J Colloid Interface Sci. 2024; 674:92-107. [19] QU X, XIE Z, ZHANG J, et al. Regulating Mitochondrial Aging via Targeting the Gut-Bone Axis in BMSCs With Oral Hydrogel Microspheres to Inhibit Bone Loss. Small. 2025;21(4):2409936. [20] SILVA SS, KUNDU B, LU S, et al. Chinese Oak Tasar Silkworm Antheraea pernyi Silk Proteins: Current Strategies and Future Perspectives for Biomedical Applications. Macromol Biosci. 2019;19(3):e1800252. [21] SUN Y, LEI C, QIAO R, et al. Recent advances in carrier-free natural small molecule self-assembly for drug delivery. Biomater Sci. 2024;12(24): 6237-6252. [22] NITA LE, CHIRIAC AP, GHILAN A, et al. Alginate enriched with phytic acid for hydrogels preparation. Int J Biol Macromol. 2021;181:561-571. [23] ZHANG G, SONG D, MA R, et al. Self-crosslinking hyaluronic acid hydrogel as an enteroprotective agent for the treatment of inflammatory bowel disease. Int J Biol Macromol. 2024;273(Pt 2): 132909. [24] POURSADEGH H, BAKHSHI V, AMINI-FAZL MS, et al. Incorporating mannose-functionalized hydroxyapatite/metal-organic framework into the hyaluronic acid hydrogel film: A potential dual-targeted oral anticancer delivery system. Int J Biol Macromol. 2024;274(Pt 2):133516. [25] TSAI HY, CHIU CC, LIN PC, et al. Antitumor efficacy of doxorubicin released from crosslinked nanoparticulate chondroitin sulfate/chitosan polyelectrolyte complexes. Macromol Biosci. 2011;11(5):680-688. [26] OPREA AM, NISTOR MT, POPA MI, et al. In vitro and in vivo theophylline release from cellulose/chondroitin sulfate hydrogels. Carbohydr Polym. 2012;90(1):127-133. [27] PEI R, JIANG Y, LEI G, et al. Rhein Derivatives, A Promising Pivot? Mini Rev Med Chem. 2021;21(5):554-575. [28] LIANG X, LIU H, CHEN H, et al. Rhein-based Pickering emulsion for hepatocellular carcinoma: Shaping the metabolic signaling and immunoactivation in transarterial chemoembolization. Aggregate. 2024;5(4):e552. [29] ZHANG C, CAI E, QI X, et al. Immunomodulatory gallium/glycyrrhizic acid hydrogels for treating multidrug-resistant Pseudomonas<i> aeruginosa</i>-infected pressure ulcers. Chem Eng J. 2024; 487:150756. [30] ZHENG J, SONG X, YANG Z, et al. Self-assembling glycyrrhizic acid micellar hydrogels as encapsulant carriers for delivery of curcumin. Colloid Surface A. 2023;658:130680. [31] BAI J, ZHANG Y, TANG C, et al. Gallic acid: Pharmacological activities and molecular mechanisms involved in inflammation-related diseases. Biomed Pharmacother. 2021; 133:110985. [32] YANG Z, YANG Y, YAN W, et al. Construction of polyfunctional coatings assisted by gallic acid to facilitate co-immobilization of diverse biomolecules. ACS Appl Mater Interfaces. 2013; 5(21):10495-104501. [33] HUANG H, GONG W, WANG X, et al. Self-Assembly of Naturally Small Molecules into Supramolecular Fibrillar Networks for Wound Healing. Adv Healthc Mater. 2022;11(12):e2102476. [34] HUANG HB, GONG W, HOU YY, et al. Mucoadhesive Hydrogel with Anti-gastric Acid and Sustained-Release Functions for Amelioration of DSS-Induced Ulcerative Colitis. J Agric Food Chem. 2023;71(9): 4016-4028. [35] LI D, FENG Y, TIAN M, et al. Gut microbiota-derived inosine from dietary barley leaf supplementation attenuates colitis through PPARγ signaling activation. Microbiome. 2021;9(1): 83. [36] LIN Y, LI L, LI G. Preparation and Properties of Temperature and pH Sensitive Chitosan Supramolecular Gel Based on Host-guest Interaction of Cucurbit 8 uril. Acta Chimica Sinica. 2012;70(21):2246-2250. [37] YANG J, LIANG G, XIANG T, et al. Effect of crosslinking processing on the chemical structure and biocompatibility of a chitosan-based hydrogel. Food Chem. 2021;354:129476. [38] ZHONG D, JIN K, WANG R, et al. Microalgae‐Based Hydrogel for Inflammatory Bowel Disease and Its Associated Anxiety and Depression. Adv Mater. 2024;36(24):e2312275. [39] GAO S, ZHENG H, XU S, et al. Novel Natural Carrier-Free Self-Assembled Nanoparticles for Treatment of Ulcerative Colitis by Balancing Immune Microenvironment and Intestinal Barrier. Adv Healthc Mater. 2023;12(31):e2301826. [40] DU Y, CAI M, MU J, et al. Type I Collagen-Adhesive and ROS-Scavenging Nanoreactors Enhanced Retinal Ganglion Cell Survival in an Experimental Optic Nerve Crush Model. Macromol Rapid Commun. 2023;44(23):e2300389. [41] ZHANG G, SONG D, MA R, et al. Artificial mucus layer formed in response to ROS for the oral treatment of inflammatory bowel disease. Sci Adv. 2024;10(30):eado8222. [42] FU YJ, ZHAO X, WANG LY, et al. A Gas Therapy Strategy for Intestinal Flora Regulation and Colitis Treatment by Nanogel‐Based Multistage NO Delivery Microcapsules. Adv Mater. 2024; 36(19):e2309972. [43] WANG CY, SUN M, FAN Z, et al. Intestine Enzyme-responsive Polysaccharide-based Hydrogel to Open Epithelial Tight Junctions for Oral Delivery of Imatinib against Colon Cancer. Chin J Polym Sci. 2022;40(10):1154-1164. [44] ZHANG X, LI Z, CHE X, et al. Synthesis and Characterization of Polyhydroxyalkanoate Organo/Hydrogels. Biomacromolecules. 2019;20(9): 3303-3312. [45] BAWA P, CHOONARA YE, DU TOIT LC, et al. A novel stimuli-synchronized alloy-treated matrix for space-defined gastrointestinal delivery of mesalamine in the Large White pig model. J Control Release. 2013;166(3):234-245. [46] DUQUETTE D, DUMONT MJ. Comparative studies of chemical crosslinking reactions and applications of bio-based hydrogels. Polymer Bulletin. 2018; 76(5):2683-2710. [47] WANG X, ZHANG Z, LEI H, et al. Treatment of ulcerative colitis via the in situ restoration of local immune and microbial homeostasis by oral administration of Tremella polysaccharide drug-carrying hydrogel. Int J Biol Macromol. 2025;285:138223. [48] YU Y, XU S, LI S, et al. Genipin-cross-linked hydrogels based on biomaterials for drug delivery: a review. Biomater Sci. 2021;9(5):1583-1597. [49] LIU H, CAI Z, WANG F, et al. Platelet Membrane Fragment Self-Assembled Oral Hydrogel Microspheres for Restoring Intestinal Microvascular Injury. AdvFunct Mater. 2023; 33(33):2302007. [50] WILLIG JB, VIANNA DRB, BECKENKAMP A, et al. Imatinib mesylate affects extracellular ATP catabolism and expression of NTPDases in a chronic myeloid leukemia cell line. Purinergic Signal. 2020;16(1):29-40. [51] CHEN J, ZHANG P, WU C, et al. Reductase-Labile Peptidic Supramolecular Hydrogels Aided in Oral Delivery of Probiotics. ACS Appl MaterInterfaces. 2023;15(26):31214-31223. [52] PHAN VHG, MATHIYALAGAN R, NGUYEN MT, et al. Ionically cross-linked alginate-chitosan core-shell hydrogel beads for oral delivery of insulin. Int J Biol Macromol. 2022;222: 262-271. [53] TANG Y, DU Y, YE J, et al. Intestine-Targeted Explosive Hydrogel Microsphere Promotes Uric Acid Excretion for Gout Therapy. Adv Mater. 2024;36(3):2310492. [54] CHEN Q, XU M, ZHENG W, et al. Se/Ru-Decorated Porous Metal-Organic Framework Nanoparticles for The Delivery of Pooled siRNAs to Reversing Multidrug Resistance in Taxol-Resistant Breast Cancer Cells. ACS Appl Mater Interfaces. 2017; 9(8):6712-6724. [55] OUYANG J, DENG B, ZOU B, et al. Oral Hydrogel Microbeads-Mediated In Situ Synthesis of Selenoproteins for Regulating Intestinal Immunity and Microbiota. J Am Chem Soc. 2023; 145(22):12193-205. [56] XU L, BAI E, ZHU Y, et al. pH-Responsive Hydrogel as a Potential Oral Delivery System of Baicalin for Prolonging Gastroprotective Activity. Pharmaceutics. 2023;15(1):257. [57] GUO Z, HOU Y, TIAN Y, et al. Antimicrobial Peptide Hydrogel with pH-Responsive and Controllable Drug Release Properties for the Efficient Treatment of Helicobacter pylori Infection. ACS Appl Mater Interfaces. 2024;16(39):51981-51993. [58] BATOOL N, SARFRAZ M, MAHMOOD A, et al. Orally Administered, Biodegradable and Biocompatible Hydroxypropyl-β-Cyclodextrin Grafted Poly(methacrylic acid) Hydrogel for pH Sensitive Sustained Anticancer Drug Delivery. Gels. 2022;8(3):190. [59] ZHANG Y, KANG R, ZHANG X, et al. A programmable oral bacterial hydrogel for controllable production and release of nanovaccine for tumor immunotherapy. Biomaterials. 2023;299:122147. [60] LU C, CHANG C, ZHENG Y, et al. Supramolecular Self-Assembled Hydrogel for Antiviral Therapy through Glycyrrhizic Acid-Enhanced Zinc Absorption and Intracellular Accumulation. ACS Appl Mater Interfaces. 2024;16(44):60027-60044. [61] WU Y, TANG Z, DU S, et al. Oral quercetin nanoparticles in hydrogel microspheres alleviate high-altitude sleep disturbance based on the gut-brain axis. Int J Pharm. 2024;658:124225. [62] ORLOVA P, MESHKOV I, LATIPOV E, et al. Cyclodextrin-Polymethylsilsesquioxane Combined System as a Perspective Iron Delivery System for Oral Administration. Gels. 2024;10(9):564. [63] QU X, XIE Z, ZHANG J, et al. Regulating Mitochondrial Aging via Targeting the Gut-Bone Axis in BMSCs With Oral Hydrogel Microspheres to Inhibit Bone Loss. Small. 2024;21(4):e2409936. [64] LIU C, YE Q, HUA S, et al. Microalgae-based natural oral hydrogel system for synergistic treatment of lead poisoning-related diseases. Nano Today. 2023;53:102034. [65] SHAN P, LIAO J, LI J, et al. H2S-releasing adhesive hydrogel as oral radioprotectant for gastrointestinal tract radioprotection. Chin Chem Lett. 2024;35(1):108545. [66] MOLODECKY NA, SOON IS, RABI DM, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology. 2012;142(1):46-54.e42;quiz e30. [67] THORLACIUS H, BJOERK A, NORDLE Ö, et al. P049 A new preclinical rationale for first-line therapy of ulcerative colitis. J Crohns Colitis. 2020;14(Supplement_1):S157. [68] HE C, YUE H, XU L, et al. siRNA release kinetics from polymeric nanoparticles correlate with RNAi efficiency and inflammation therapy via oral delivery. Acta Biomater. 2020;103:213-222. [69] SøREIDE K, THORSEN K, HARRISON EM, et al. Perforated peptic ulcer. Lancet. 2015; 386(10000):1288-1298. [70] CHENG Z, QING R, HAO S, et al. Fabrication of ulcer-adhesive oral keratin hydrogel for gastric ulcer healing in a rat. Regen Biomater. 2021;8(2):rbab008. [71] KRAMER CK, RETNAKARAN R, ZINMAN B. Insulin and insulin analogs as antidiabetic therapy: A perspective from clinical trials. Cell Metab. 2021;33(4):740-747. [72] JIANG N, YU X, ZHANG J, et al. Smart stimuli-responsive hydrogels for safe oral administration of Insulin: A Review. Int J Pharm. 2025;674:125487. [73] CHEN S, MIAO Q, LIU Y, et al. Construction and functional evaluation of oral long-acting insulin hydrogel microparticles based on physical and chemical double crosslinking. Int J Biol Macromol. 2023;253(Pt 3):126915. [74] DALBETH N, MERRIMAN TR, STAMP LK. Gout. Lancet. 2016;388(10055):2039-2052. [75] LIU X, DONG J, WU Z, et al. Microalgae-based hydrogel drug delivery system for treatment of gouty arthritis with alleviated colchicine side effects. Bioact Mater. 2025;52:17-35. [76] CHEN XY, BUTT AM, MOHD AMIN MCI. Enhanced paracellular delivery of vaccine by hydrogel microparticles-mediated reversible tight junction opening for effective oral immunization. J Control Release. 2019;311:50-64. [77] SUN M, SHI Y, LEI B, et al. A pH-triggered self-releasing humic acid hydrogel loaded with porcine interferon α/γ achieves anti-pseudorabies virus effects by oral administration. Vet Res. 2024;55(1):153. [78] JIANG H, LU Q, HUANG X, et al. Sinomenine-glycyrrhizic acid self-assembly enhanced the anti-inflammatory effect of sinomenine in the treatment of rheumatoid arthritis. J Control Release. 2025;382:113718. [79] SHARPE LA, DAILY AM, HORAVA SD, et al. Therapeutic applications of hydrogels in oral drug delivery. Expert Opin Drug Deliv. 2014;11(6): 901-915. [80] ABBASI M, SOHAIL M, MINHAS MU, et al. Novel biodegradable pH-sensitive hydrogels: An efficient controlled release system to manage ulcerative colitis. Int J Biol Macromol. 2019; 136:83-96. [81] SHANGGUAN J, YU F, DING B, et al. Hydrogel-forming viscous liquid in response to ROS restores the gut mucosal barrier of colitis mice via regulating oxidative redox homeostasis. Acta Biomater. 2024;184:127-143. [82] SOHAIL M, MUDASSIR, MINHAS MU, et al. Natural and synthetic polymer-based smart biomaterials for management of ulcerative colitis: a review of recent developments and future prospects. Drug Deliv Transl Res. 2018;9(2):595-614. [83] ZHU J. Bioactive modification of poly(ethylene glycol) hydrogels for tissue engineering. Biomaterials. 2010;31(17):4639-4656. |
| [1] | 郭宇超, 倪前伟, 尹 晨, 吉格尔·赛义力汗, 高 瞻. 季铵化壳聚糖紧急止血材料:合成、机制与应用[J]. 中国组织工程研究, 2026, 30(8): 2091-2100. |
| [2] | 刘宏杰, 牟秋菊, 申玉雪, 梁 飞, 祝丽丽. 金属有机框架/羧甲基壳聚糖-氧化海藻酸钠/富血小板血浆水凝胶促糖尿病感染创面愈合[J]. 中国组织工程研究, 2026, 30(8): 1929-1939. |
| [3] | 杨 光, 印治涛, 许 燕. 3D打印异烟肼脂质体光热支架及性能评价[J]. 中国组织工程研究, 2026, 30(26): 6701-6709. |
| [4] | 赵张红, 金东升, 阮世强, 黄文良, 万 喻, 田仁元, 邓 江. 淫羊藿苷缓释微球三维支架的体外促成骨与抗炎性能[J]. 中国组织工程研究, 2026, 30(26): 6710-6718. |
| [5] | 皮志龙, 李嘉源, 谭志超, 陆小梅, 张志强, 叶翔凌. 3D打印新补骨脂异黄酮涂层支架调节成骨/破骨细胞活性促进骨再生[J]. 中国组织工程研究, 2026, 30(26): 6736-6743. |
| [6] | 周云圻, 刘 旭, 肖东琴, 李兴平, 匙 峰, 张 波, 蒲 超, 罗栩伟, 张成栋. 兼具抗菌与促成骨功能水凝胶的制备与表征[J]. 中国组织工程研究, 2026, 30(26): 6768-6778. |
| [7] | 张 静, 何丽萍, 温 玉, 付 航. 负载成纤维细胞外泌体的水凝胶促进内皮细胞功能恢复和糖尿病创面修复[J]. 中国组织工程研究, 2026, 30(26): 6798-6806. |
| [8] | 陈 刚, 葛彩军, 陈建澎, 王元斌, 王前亮. 载铁抑素1水凝胶治疗腰椎间盘突出症的机制[J]. 中国组织工程研究, 2026, 30(26): 6807-6813. |
| [9] | 赵文博, 缪 鑫, 王 洋, 刘 浩, 李胜发, 陶崎峰. 锶/比拉瑞塞共载生物活性玻璃调控骨微环境治疗骨质疏松症[J]. 中国组织工程研究, 2026, 30(26): 6814-6825. |
| [10] | 周丽静, 王 双, 向谨姣, 王会超, 柴雪姣 . 体外环境下C-Root BP材料根尖封闭性及抗力强度[J]. 中国组织工程研究, 2026, 30(26): 6868-6872. |
| [11] | 余金烨, 蒋 南, 赵一浔, 黄梦静, 杨 洁, 孙 瑞, 冯所兰, 蒋 卉, 杨 军. 用于细胞三维培养的即用型海藻酸钠@纸材料[J]. 中国组织工程研究, 2026, 30(26): 6873-6879. |
| [12] | 宋沐泽, 刘楚怡, 唐庆娟, 代元坤, 宋文山, 李八方, 王园园. 聚乳酸/胶原蛋白静电纺双层引导组织再生膜的生物相容性评价[J]. 中国组织工程研究, 2026, 30(26): 6880-6891. |
| [13] | 陈伟飞, 梅远东, 巨积辉. 双离子时序释放多功能水凝胶修复感染性骨缺损[J]. 中国组织工程研究, 2026, 30(20): 5188-5200. |
| [14] | 许艺璇, 姚 俊, 刘旭璐, 李新莲, 刘志雄, 张志红. 含万古霉素的猪皮脱细胞外基质水凝胶促进皮肤感染创面愈合[J]. 中国组织工程研究, 2026, 30(20): 5214-5228. |
| [15] | 陈 颖, 孙盱衡, 刘 青, 肖 聪, 蒋虹婧, 林展翼. 促进组织工程血管移植物早期阶段形成的无血清培养基[J]. 中国组织工程研究, 2026, 30(20): 5093-5102. |
口服给药因较高的依从性一直是患者最为偏好的药物递送途径,相较于注射给药能显著提升患者的治疗体验。然而,消化道内的极端环境、药物吸收缓慢以及存在的生物屏障等问题,严重制约了口服给药的生物利用度,这是目前口服药物递送面临的主要挑战。口服天然材料水凝胶的研发克服了这一困境[3]。大多数口服天然水凝胶由天然多糖构成,这些多糖在胃肠道内的酶作用下具有较高的稳定性,难以被降解[4-5],因此,天然水凝胶不仅能够高效地将药物输送至病变部位,确保药物的充分利用,还能展现出良好的持续缓释效果[6]。更值得关注的是,部分天然分子具备疾病靶向能力,为实现精准药物释放提供了有力保障[7-8]。除此以外,随着中医热度的不断提升,许多中药小分子被应用于新型材料的研究中。大部分中药小分子难溶于水,并且在胃肠道中的稳定性极差,限制了它们的吸收与利用。中药小分子口服水凝胶的研究,为中药的现代化应用提供了新的思路。利用天然水凝胶递送益生菌、蛋白质类等,为一些常规用药的给药途径提供了新的可能,比如口服胰岛素、口服疫苗的研究给患者带来了更加便利的给药方式[9]。当前,口服天然水凝胶在多种疾病的治疗研究中已取得了显著进展,涵盖了胃肠道疾病、代谢性疾病、肿瘤治疗以及免疫治疗等多个重要领域,特别是在胃肠道疾病的药物递送方面,可口服天然水凝胶展现出独特的优势[10]。但口服水凝胶仍然面临着一系列的问题,以物理方式合成的水凝胶往往面临机械性能不足的问题,而以化学方式合成的水凝胶在弥补机械性能的同时生物相容性又受到有毒交联剂的影响[11]。
该文主要综述基于天然材料的口服水凝胶在药物递送方面的研究,首先重点介绍了不同天然材料的性质特点以及不同交联方式合成的口服水凝胶的优势,然后介绍了近几年口服水凝胶用于药物递送研究中所递送的药物类别,最后总结了天然材料口服水凝胶在疾病中的最新应用,综合讨论了天然材料口服水凝胶在未来所面临的挑战以及需要发展的方向。
1.1.1 检索人及检索时间 由第一作者在2024年11月进行检索。
1.1.2 文献检索时限 2009-2024年发表的文献。
1.1.3 检索数据库 PubMed、Web of Science数据库。
1.1.4 检索词 英文检索词为“oral hydrogels,physical crosslinking,chemical crosslinking,natural material,therapy,drug delivery,application of disease research”。
1.1.5 检索文献类型 综述及研究性论文。
1.1.6 手工检索情况 无。
1.1.7 检索策略 以 PubMed 数据库检索策略为例,见图1。
1.1.8 检索文献量 共检索到相关文献363篇。
1.2 入选标准
纳入标准:刊登在权威期刊上的文献;研究口服水凝胶装载药物并用于医学研究的文献。
排除标准:与研究目的不相符的文献;无法获取全文的文献;用于口腔治疗的水凝胶相关文献。
1.3 数据的提取 根据检索策略,在英文数据库中共检索到了363篇文献,按入选标准排除280篇,最终纳入83篇。具体文献筛选流程如图2所示。
然而,天然材料水凝胶在应用过程中也暴露出一些有待解决的问题:一方面,天然材料普遍存在机械性能和拉伸强度不足的问题,这使它们难以满足复杂的药物输送需求,限制了它们在某些特殊场景下的应用[82];另一方面,尽管天然材料源于自然,但它们仍有可能引发人体的免疫反应[83];此外,在化学交联过程中,虽然已经摒弃了细胞毒性较大的戊二醛,采用了聚乙二醇、1,4- 丁二醇二缩水甘油醚等交联剂,但这些交联剂仍存在引发过敏反应的风险,安全性仍需进一步验证。
3.2 该综述区别于他人他篇的特点 该综述选择了口服天然水凝胶作为主题,从材料特点、合成方式以及携载药物等方面系统介绍了天然分子口服水凝胶的优势和特点、研究应用的广泛性,为口服水凝胶递送药物在未来临床转化提供了依据。
3.3 该综述的重要意义 未来,天然材料口服水凝胶将集中在更多新型天然材料的发掘,开发特异性靶向水凝胶;利用更多天然交联剂代替传统交联剂,例如食物交联剂以及树脂胶类,与此同时,提高天然交联剂的交联效率和稳定性是需要思考的问题;物理化学双交联水凝胶需优化交联过程,解决物理交联的不稳定性以及化学交联的潜在毒性,放大双交联的优势;中药小分子水凝胶的开发也是一大方向,为中药分子利用提供新思路。
天然材料水凝胶在现代医学中存在巨大的应用潜力,天然水凝胶的智能化和精准递送能力将推动它在药物递送、疾病治疗和生物医学工程领域的广泛应用。天然水凝胶作为一种绿色、可持续的药物递送平台,有望为口服药物递送提供更多的创新性的解决方案,引领生物材料领域的新发展。
该文总结近5年关于天然口服水凝胶相关文献,在对材料选择、合成方式以及携载药物等方面综合讨论天然口服水凝胶在未来临床转化的优势以及未来面临的挑战。在材料选择方面,重点介绍天然材料的特征优势;在合成方式中,介绍不同合成方式合成的口服水凝胶的优势及缺陷;最后介绍了近几年利用口服水凝胶携载的药物类别以及在疾病研究中的应用情况。为未来临床转化提供了不少新的思路,也为药物提供了新的给药途径。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||