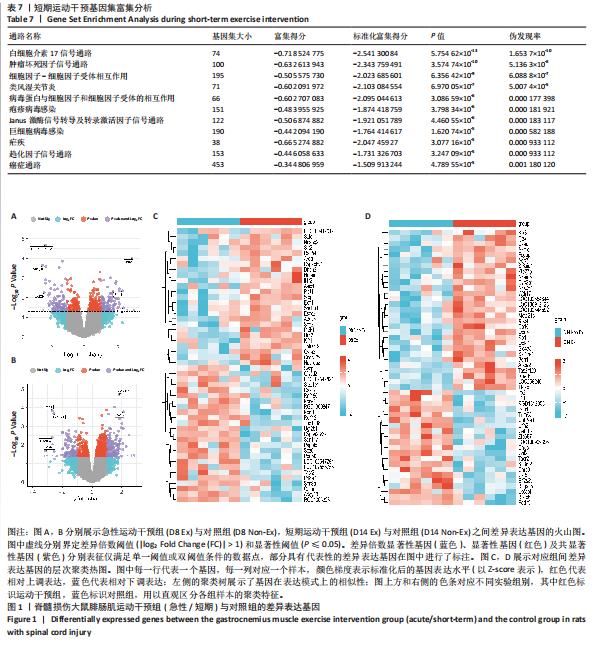
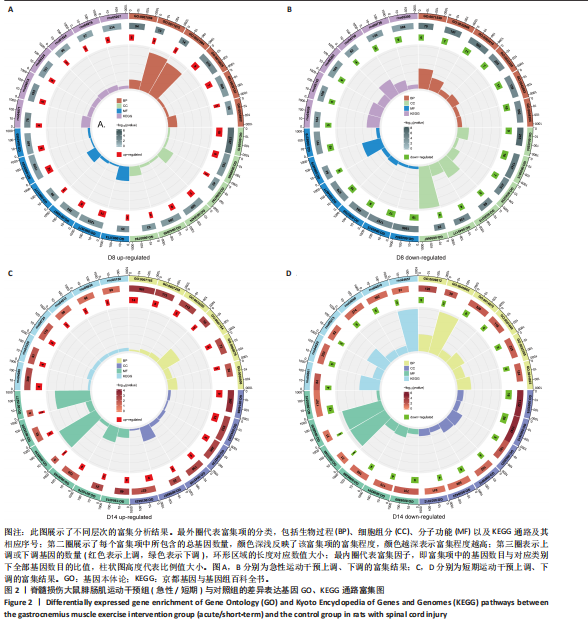
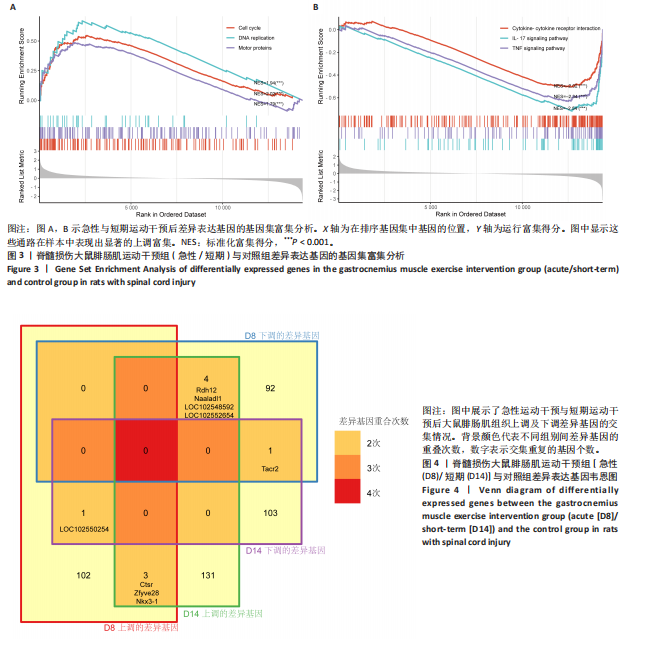
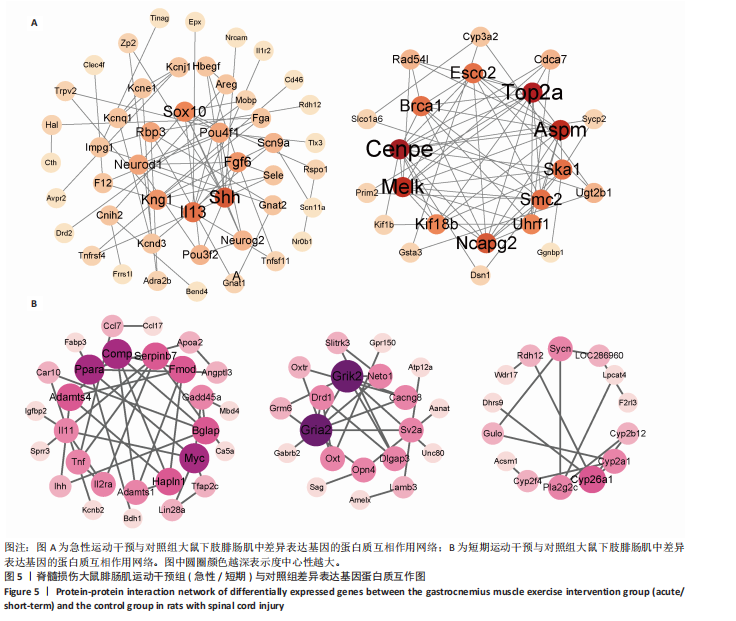
[1] 刘帅祎,赵晓璇,李奇,等.中央模式发生器在脊髓损伤后下肢运动恢复中作用的研究进展[J].中国脊柱脊髓杂志,2024,34(10):1092-1098.
[2] RYBAK IA, SHEVTSOVA NA, MARKIN SN, et al. Operation regimes of spinal circuits controlling locomotion and the role of supraspinal drives and sensory feedback. Elife. 2024;13:RP98841.
[3] MINASSIAN K, BAYART A, LACKNER P, et al. Rare phenomena of central rhythm and pattern generation in a case of complete spinal cord injury. Nat Commun. 2023;14(1):3276.
[4] YU P, ZHANG W, LIU Y, et al. The effects and potential mechanisms of locomotor training on improvements of functional recovery after spinal cord injury. Int Rev Neurobiol. 2019;147:199-217.
[5] LOY K, BAREYRE FM. Rehabilitation following spinal cord injury: how animal models can help our understanding of exercise-induced neuroplasticity. Neural Regen Res. 2019;14(3):405-412.
[6] ZENG CW, ZHANG CL. Neuronal regeneration after injury: a new perspective on gene therapy. Front Neurosci. 2023;17:1181816.
[7] HU X, XU W, REN Y, et al. Spinal cord injury: molecular mechanisms and therapeutic interventions. Signal Transduct Target Ther. 2023;8(1):245.
[8] 潘璐,谭波涛,虞乐华,等.运动训练影响脊髓损伤后功能恢复机制的研究进展[J].中国康复医学杂志,2020,35(12):1537-1541.
[9] KAUR S, ARUMUGAM N, CHHABRA H. A Systematic Review: Exercise based approaches to activate central pattern generator in spinal cord injury survivors. Int J Neurol Phys Ther. 2024;10:8-15.
[10] HORNBY TG, REISMAN DS, WARD IG, et al. Clinical Practice Guideline to Improve Locomotor Function Following Chronic Stroke, Incomplete Spinal Cord Injury, and Brain Injury. J Neurol Phys Ther. 2020;44(1):49-100.
[11] BILCHAK JN, CARON G, CÔTÉ MP. Exercise-Induced Plasticity in Signaling Pathways Involved in Motor Recovery after Spinal Cord Injury. Int J Mol Sci. 2021;22(9):4858.
[12] GOLDSHMIT Y, LYTHGO N, GALEA MP, et al. Treadmill training after spinal cord hemisection in mice promotes axonal sprouting and synapse formation and improves motor recovery. J Neurotrauma. 2008;25(5):449-465.
[13] ZHANG H, LIU Y, ZHOU K, et al. Restoring Sensorimotor Function Through Neuromodulation After Spinal Cord Injury: Progress and Remaining Challenges. Front Neurosci. 2021;15:749465.
[14] CHANG W, PEDRONI A, BERTUZZI M, et al. Locomotion dependent neuron-glia interactions control neurogenesis and regeneration in the adult zebrafish spinal cord. Nat Commun. 2021;12(1):4857.
[15] KAZIM SF, BOWERS CA, COLE CD, et al. Corticospinal Motor Circuit Plasticity After Spinal Cord Injury: Harnessing Neuroplasticity to Improve Functional Outcomes. Mol Neurobiol. 2021;58(11):5494-5516.
[16] WANG H, LIU NK, ZHANG YP, et al. Treadmill training induced lumbar motoneuron dendritic plasticity and behavior recovery in adult rats after a thoracic contusive spinal cord injury. Exp Neurol. 2015;271:368-378.
[17] YU JI, SEO TB. The effects of weight- and non-weight-bearing exercise on corticospinal axon sprouting, regeneration-related proteins and functional recovery after spinal cord contusion. J Exerc Rehabil. 2024;20(6):213-219.
[18] ERSCHBAMER MK, PHAM TM, ZWART MC, et al. Neither environmental enrichment nor voluntary wheel running enhances recovery from incomplete spinal cord injury in rats. Exp Neurol. 2006;201(1):154-164.
[19] LEWIS NE, TABARESTANI TQ, CELLINI BR, et al. Effect of Acute Physical Interventions on Pathophysiology and Recovery After Spinal Cord Injury: A Comprehensive Review of the Literature. Neurospine. 2022;19(3):671-686.
[20] MIGLIORINI F, COCCONI F, SCHÄFER L, et al. Pharmacological management of secondary chronic spinal cord injury: a systematic review. Br Med Bull. 2024; 151(1):49-68.
[21] ANJUM A, YAZID MD, FAUZI DAUD M, et al. Spinal Cord Injury: Pathophysiology, Multimolecular Interactions, and Underlying Recovery Mechanisms. Int J Mol Sci. 2020;21(20):7533.
[22] ALIZADEH A, DYCK SM, KARIMI-ABDOLREZAEE S. Traumatic Spinal Cord Injury: An Overview of Pathophysiology, Models and Acute Injury Mechanisms. Front Neurol. 2019;10:282.
[23] HOU J, NELSON R, MOHAMMAD N, et al. Effect of Simultaneous Combined Treadmill Training and Magnetic Stimulation on Spasticity and Gait Impairments after Cervical Spinal Cord Injury. J Neurotrauma. 2020;37(18):1999-2013.
[24] LI X, LI Q, LI C, et al. Effect of high-intensity exercise training on functional recovery after spinal cord injury. Front Neurol. 2025;16:1442004.
[25] HE LW, GUO XJ, ZHAO C, et al. Rehabilitation Training after Spinal Cord Injury Affects Brain Structure and Function: From Mechanisms to Methods. Biomedicines. 2023;12(1):41.
[26] BALIGAND C, CHEN YW, YE F, et al. Transcriptional Pathways Associated with Skeletal Muscle Changes after Spinal Cord Injury and Treadmill Locomotor Training. Biomed Res Int. 2015;2015:387090.
[27] 郭小军,常佳琪,何乐玮,等.硬膜外脊髓电刺激在脊髓损伤后下肢运动恢复的研究进展[J].中国康复医学杂志,2024,39(6):898-904.
[28] LYAKHOVETSKII V, MERKULYEVA N, GORSKII O, et al. Simultaneous bidirectional hindlimb locomotion in decerebrate cats. Sci Rep. 2021;11(1):3252.
[29] TOMÀS J, CILLEROS-MAÑÉ V, JUST-BORRÀS L, et al. Brain-derived neurotrophic factor signaling in the neuromuscular junction during developmental axonal competition and synapse elimination. Neural Regen Res. 2025;20(2):394-401.
[30] CHAN WS, NG CF, PANG BPS, et al. Exercise-induced BDNF promotes PPARδ-dependent reprogramming of lipid metabolism in skeletal muscle during exercise recovery. Sci Signal. 2024;17(828):eadh2783.
[31] SANDROW-FEINBERG HR, IZZI J, SHUMSKY JS, et al. Forced exercise as a rehabilitation strategy after unilateral cervical spinal cord contusion injury. J Neurotrauma. 2009;26(5):721-731.
[32] ASANO K, NAKAMURA T, FUNAKOSHI K. Early mobilization in spinal cord injury promotes changes in microglial dynamics and recovery of motor function. IBRO Neurosci Rep. 2022;12:366-376.
[33] 王子恒,吴霜.脊髓损伤后氧化应激相关基因及分子机制:基于GEO数据库的数据分析及验证[J].中国组织工程研究,2025,29(32):6893-6904.
[34] 于浩洋,祝英文,李亚楠,等.跑台运动小鼠骨组织外泌体微小RNA表达谱及其生物信息学分析[J].医用生物力学,2024,39(S1):612.
[35] 章森,刘文彬,夏杰,等.运动改善ASMT基因敲除小鼠抑郁行为的海马蛋白质组学机制[J].上海体育大学学报,2024,48(3):36-48.
[36] HENRY M, COXE RC, BARRY A, et al. A research protocol to study the critical time window for rehabilitation after incomplete spinal cord injury: early vs. late locomotor training. BMC Neurol. 2024;24(1):482.
[37] MUN S, HAN K, HYUN JK. The Time Sequence of Gene Expression Changes after Spinal Cord Injury. Cells. 2022;11(14):2236.
[38] MATSON KJE, RUSS DE, KATHE C, et al. Single cell atlas of spinal cord injury in mice reveals a pro-regenerative signature in spinocerebellar neurons. Nat Commun. 2022;13(1):5628.
[39] LI C, WU Z, ZHOU L, et al. Temporal and spatial cellular and molecular pathological alterations with single-cell resolution in the adult spinal cord after injury. Signal Transduct Target Ther. 2022;7(1):65.
[40] CHIO JCT, WANG J, SURENDRAN V, et al. Delayed administration of high dose human immunoglobulin G enhances recovery after traumatic cervical spinal cord injury by modulation of neuroinflammation and protection of the blood spinal cord barrier. Neurobiol Dis. 2021;148:105187.
[41] DAVIS JQ, LAMBERT S, BENNETT V. Molecular composition of the node of Ranvier: identification of ankyrin-binding cell adhesion molecules neurofascin (mucin+/third FNIII domain-) and NrCAM at nodal axon segments. J Cell Biol. 1996;135(5):1355-1367.
[42] LAMBERT S, DAVIS JQ, BENNETT V. Morphogenesis of the node of Ranvier: co-clusters of ankyrin and ankyrin-binding integral proteins define early developmental intermediates. J Neurosci. 1997;17(18):7025-7036.
[43] ESHED-EISENBACH Y, DEVAUX J, VAINSHTEIN A, et al. Precise Spatiotemporal Control of Nodal Na+ Channel Clustering by Bone Morphogenetic Protein-1/Tolloid-like Proteinases. Neuron. 2020;106(5):806-815.e6.
[44] RASBAND MN, PELES E. Mechanisms of node of Ranvier assembly. Nat Rev Neurosci. 2021;22(1):7-20.
[45] TSAYTLER P, BLAESS G, SCHOLZE-WITTLER M, et al. Early neural specification of stem cells is mediated by a set of SOX2-dependent neural-associated enhancers. Stem Cell Reports. 2024;19(5):618-628.
[46] LIU JA, TAI A, HONG J, et al. Fbxo9 functions downstream of Sox10 to determine neuron-glial fate choice in the dorsal root ganglia through Neurog2 destabilization. Proc Natl Acad Sci U S A. 2020;117(8):4199-4210.
[47] FARIOLI-VECCHIOLI S, MATTERA A, MICHELI L, et al. Running rescues defective adult neurogenesis by shortening the length of the cell cycle of neural stem and progenitor cells. Stem Cells. 2014;32(7):1968-1982.
[48] KRUPPA AJ, BUSS F. Motor proteins at the mitochondria-cytoskeleton interface. J Cell Sci. 2021;134(7):jcs226084.
[49] ONG KH, LAI HY, SUN DP, et al. Prognostic Significance of DNA Topoisomerase II Alpha (TOP2A) in Cholangiocarcinoma. Front Biosci (Landmark Ed). 2023; 28(4):75.
[50] TCHAKARSKA G, SOLA B. The double dealing of cyclin D1. Cell Cycle. 2020;19(2): 163-178.
[51] CRASKE B, WELBURN JPI. Leaving no-one behind: how CENP-E facilitates chromosome alignment. Essays Biochem. 2020;64(2):313-324.
[52] GRAVES LY, KEANE KF, TAYLOR JY, et al. Subacute and Chronic Spinal Cord Injury: A Scoping Review of Epigenetics and Secondary Health Conditions. Epigenet Insights. 2023;16:25168657231205679.
[53] ZHAO YN, LI JM, CHEN CX, et al. Effect on intensity of treadmill running on learning, memory and expressions of cell cycle-related proteins in rats with cerebral ischemia. Oncotarget. 2017;8(25):40633-40642.
[54] PEHAR M, HEWITT M, WAGNER A, et al. Histamine stimulates human microglia to alter cellular prion protein expression via the HRH2 histamine receptor. Sci Rep. 2024;14(1):25519.
[55] CARTHY E, ELLENDER T. Histamine, Neuroinflammation and Neurodevelopment: A Review. Front Neurosci. 2021;15:680214.
[56] 於来康,吕媛媛,顾博雅,等.有氧运动对APP/PS1小鼠海马突触超微结构和功能的可塑性作用[J].中国体育科技,2023,59(2):18-24.
[57] 戴雯,金晖,彼末一之.目标设置在不同肌肉强度的短期运动训练中对大脑运动皮层突触可塑性影响研究[J].中国体育科技,2021,57(9):46-54.
[58] HELLENBRAND DJ, QUINN CM, PIPER ZJ, et al. Inflammation after spinal cord injury: a review of the critical timeline of signaling cues and cellular infiltration. J Neuroinflammation. 2021;18(1):284.
[59] XU X, TALIFU Z, ZHANG CJ, et al. Mechanism of skeletal muscle atrophy after spinal cord injury: A narrative review. Front Nutr. 2023;10:1099143.
[60] DING Y, CHEN Q. Recent advances on signaling pathways and their inhibitors in spinal cord injury. Biomed Pharmacother. 2024;176:116938.
[61] SUN D, LI W, DING D, et al. IL-17a promotes hepatocellular carcinoma by increasing FAP expression in hepatic stellate cells via activation of the STAT3 signaling pathway. Cell Death Discov. 2024;10(1):230.
[62] ZHAO Y, URBONAVICIUTE V, XU B, et al. Cartilage Oligomeric Matrix Protein Induced Arthritis-A New Model for Rheumatoid Arthritis in the C57BL/6 Mouse. Front Immunol. 2021;12:631249.
[63] GRABACKA M, PIERZCHALSKA M, REISS K. Peroxisome proliferator activated receptor α ligands as anticancer drugs targeting mitochondrial metabolism. Curr Pharm Biotechnol. 2013;14(3):342-356.
[64] STRAND E, LYSNE V, GRINNA ML, et al. Short-Term Activation of Peroxisome Proliferator-Activated Receptors α and γ Induces Tissue-Specific Effects on Lipid Metabolism and Fatty Acid Composition in Male Wistar Rats. PPAR Res. 2019;2019:8047627.
[65] TAYLOR DF, BISHOP DJ. Transcription Factor Movement and Exercise-Induced Mitochondrial Biogenesis in Human Skeletal Muscle: Current Knowledge and Future Perspectives. Int J Mol Sci. 2022;23(3):1517.
[66] TANG Y, WANG X, HUANG M, et al. Sports training improves motor function after spinal cord injury by regulating microtubule dynamics. Biochim Biophys Acta Mol Basis Dis. 2025;1871(3):167587.
|