中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (1): 78-92.doi: 10.12307/2026.510
• 脐带脐血干细胞 umbilical cord blood stem cells • 上一篇 下一篇
人脐血富血小板血浆、单个核细胞及间充质干细胞修复大鼠薄型子宫内膜
牟彦郦1,胡安春1,须文驰1,陈盼盼1,陈 浩2,赵淑云2,黄官友2,陈小娟3
- 1贵州医科大学,贵州省贵阳市 550004;贵州医科大学附属医院,2生殖中心,3高压氧科,贵州省贵阳市 550004
-
收稿日期:2024-10-31接受日期:2024-12-31出版日期:2026-01-08发布日期:2025-06-19 -
通讯作者:赵淑云,博士,主任医师,贵州医科大学附属医院生殖中心,贵州省贵阳市 550004 -
作者简介:牟彦郦,女,1998年生,四川省泸州市人,汉族,贵州医科大学在读硕士,主要从事生殖内分泌的研究。 -
基金资助:贵州省卫生健康委科学技术基金项目(gzwkj2013-1-101),项目负责人:黄官友;贵州省卫生健康委科学技术基金项目(gzwkj2022-415),项目负责人:陈小娟;贵州省卫生健康委科学技术基金项目(g2wkj2024-439),项目负责人:胡安春
Effects of human umbilical cord blood platelet-rich plasma, mononuclear cells, and mesenchymal stem cells in repairing thin endometrium in rats
Mu Yanli1, Hu Anchun1, Xu Wenchi1, Chen Panpan1, Chen hao2, Zhao Shuyun2, Huang Guanyou2, Chen Xiaojuan3
- 1Guizhou Medical University, Guiyang 550004, Guizhou Province, China; 2Department of Reproductive Center, 3Department of Hyperbaric Oxygen, Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China
-
Received:2024-10-31Accepted:2024-12-31Online:2026-01-08Published:2025-06-19 -
Contact:Zhao Shuyun, MD, Chief physician, Department of Reproductive Center, Affiliated Hospital of Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
About author:Mu Yanli, Master candidate, Guizhou Medical University, Guiyang 550004, Guizhou Province, China -
Supported by:Guizhou Provincial Health Commission Science and Technology Fund, No. gzwkj2013-1-101 (to HGY); Guizhou Provincial Health Commission Science and Technology Fund, No. gzwkj2022-415 (to CXJ); Guizhou Provincial Health Commission Science and Technology Fund, No. g2wkj2024-439 (to HAC)
摘要:
文题释义:
人脐血衍生物:指从人脐血中提取的多种生物活性物质和细胞成分,主要包括单个核细胞、间充质干细胞和造血细胞,以及各种生长因子和抗炎/免疫调节因子,还有血液衍生因子富血小板血浆。这些衍生物展现出多样化且重要的生物学功能,如促进细胞增殖、分化、迁移和免疫调节等,在血液疾病治疗、再生医学、组织修复和免疫调节等领域具有广泛的应用前景。薄型子宫内膜:是指在特定生理或病理情况下,子宫内膜厚度相对较薄,无法满足正常妊娠的必要条件,主要表现为细胞增殖不足和功能失衡,是导致女性月经异常、不孕症和反复流产的重要因素,能够影响子宫内膜的耐受性,进而降低胚胎的着床率和临床妊娠率,同时与辅助生殖治疗的预后质量下降相关。
摘要
背景:研究发现人脐血富血小板血浆与人脐血间充质干细胞对薄型子宫内膜有一定治疗效果,但目前针对人脐血单个核细胞对薄型子宫内膜的研究尚未报道,且目前尚缺乏三者比较的相关研究。
目的:探讨人脐血富血小板血浆、单个核细胞及间充质干细胞修复大鼠薄型子宫内膜的效果与机制。
方法:60只雌性SPF级SD大鼠随机分为假手术组、模型组、人脐血富血小板血浆组、人脐血单个核细胞组和人脐血间充质干细胞组,每组12只。假手术组宫角注射0.5 mL生理盐水,5 min后再灌注0.5 mL PBS;模型组、人脐血富血小板血浆组、人脐血单个核细胞组和人脐血间充质干细胞组宫角注射0.5 mL体积分数95%乙醇,5 min后吸出剩余乙醇,经生理盐水冲洗2次后再分别灌注0.5 mL PBS、人脐血富血小板血浆、人脐血单个核细胞悬液(1×107个)和人脐血间充质干细胞悬液(1×107个),于再灌注后第3个正常动情周期的动情间期收集脏器组织与血清,检测相关指标。
结果与结论:①子宫组织大体观、苏木精-伊红染色和Masson染色结果:假手术组结构完整,内膜厚度适中,血管结构清晰。与假手术组相比,模型组子宫萎缩,结构不完整,内膜厚度、腺体数量明显减少,血管结构紊乱,纤维化增加;与模型组相比,经人脐血衍生物治疗后,子宫大小、结构、内膜厚度均有所恢复(均P < 0.01),并且纤维化均减轻,其中以人脐血单个核细胞组恢复最为明显。在腺体数量增加方面,以人脐血富血小板血浆组效果最为显著(P < 0.000 1)。②子宫组织免疫组化与免疫荧光结果显示,与假手术组相比,模型组细胞增殖相关指标角蛋白9、波形蛋白,内膜容受性相关指标白血病抑制因子、整合素αγβ3,血小板内皮细胞黏附分子、碱性成纤维细胞生长因子、血管内皮生长因子表达均降低(均P < 0.05)。与模型组相比,经人脐血衍生物治疗后以上指标均有显著性升高,人脐血衍生物各组比较:角蛋白9、血管内皮生长因子蛋白:人脐血单个核细胞组>人脐血间充质干细胞组>人脐血富血小板血浆组;波形蛋白、白血病抑制因子蛋白:人脐血间充质干细胞组>人脐血单个核细胞组>人脐血富血小板血浆组;整合素αγβ3蛋白:人脐血富血小板血浆组>人脐血间充质干细胞组>人脐血单个核细胞组;血小板内皮细胞黏附分子蛋白:人脐血富血小板血浆组>人脐血单个核细胞组>人脐血间充质干细胞组;碱性成纤维细胞生长因子蛋白:人脐血单个核细胞组>人脐血富血小板血浆组>人脐血间充质干细胞组。③Western blot检测显示,与假手术组相比,模型组白细胞介素6、白细胞介素1β和肿瘤坏死因子α蛋白表达水平显著升高(均P < 0.001),经人脐血衍生物治疗后上述指标均降低(均P < 0.05)。④ELISA检测显示,与假手术组相比,模型组抗缪勒氏管激素、雌二醇、孕酮水平降低,促卵泡生成素、促黄体生成素水平增加(除促黄体生成素外,均P < 0.05)。经人脐血衍生物治疗后,性激素和抗缪勒氏管激素水平有一定程度恢复。⑤生育力实验显示,与假手术组相比,模型组受孕时间有所增加,产仔数明显降低(均P < 0.05)。经人脐血衍生物治疗后,3组产仔数均有所增加(P < 0.05),组间比较尚未发现明显差异。此研究初步表明人脐血单个核细胞对薄型子宫内膜有一定治疗效果,且人脐血富血小板血浆、人脐血单个核细胞和人脐血间充质干细胞分别在改善薄型子宫内膜再生功能、内膜容受性、血管生成、炎症调控及妊娠结局方面具有不同的优势与差异。
https://orcid.org/0009-0003-3871-6506(牟彦郦)
中图分类号:
引用本文
牟彦郦, 胡安春, 须文驰, 陈盼盼, 陈 浩, 赵淑云, 黄官友, 陈小娟. 人脐血富血小板血浆、单个核细胞及间充质干细胞修复大鼠薄型子宫内膜[J]. 中国组织工程研究, 2026, 30(1): 78-92.
Mu Yanli, Hu Anchun, Xu Wenchi, Chen Panpan, Chen hao, Zhao Shuyun, Huang Guanyou, Chen Xiaojuan. Effects of human umbilical cord blood platelet-rich plasma, mononuclear cells, and mesenchymal stem cells in repairing thin endometrium in rats [J]. Chinese Journal of Tissue Engineering Research, 2026, 30(1): 78-92.
2.2 各组大鼠基本参数对比 阴道脱落细胞涂片检查确认SD大鼠的动情周期,主要表现为动情前期(P):全部是有核上皮细胞,偶有少量角化细胞;动情期(E):全部是无核角化细胞或间有少量上皮细胞;动情间期(M):白细胞、角化细胞、有核上皮细胞均有;动情后期(DI):大量白细胞及少量上皮细胞和黏液,见图1A。
动情周期正常大鼠入组,再灌注后第3个正常动情周期的动情间期取大鼠子宫组织。假手术组子宫组织大小正常,形态规则,呈粉红色,组织质地均匀、富有弹性,切面观内膜厚度适中,血管分布丰富且均匀,无明显肿块与淤血坏死。模型组子宫组织萎缩,形态不规则,呈棕黄色,表面凹凸不平,组织僵硬,切面观内膜变薄,血管分布稀疏,大部分血管扩张显露出淤血,部分可见输卵管积水,由此说明通过体积分数95%乙醇维持灌注子宫,可造成实质性损伤。与假手术组相比,模型组子宫和脾脏脏器系数具有显著差异(P < 0.001)。hUCB-PRP组子宫较模型组有所改善,形态较不规则,粗细不均,部分区域呈现出水肿,颜色略显苍白,切面观子宫内膜厚度和血管分布中等,无明显淤血坏死区域。hUCB-MNCs组子宫组织大小欠佳,形态欠规则,颜色趋于正常,表面欠光滑,切面观内膜厚度和血管分布有所改善,可见少量淤血坏死区域。
hUCB-MSCs组子宫组织大小接近正常,形态较规则,呈淡粉色,组织表面光滑,有一定弹性,切面观内膜厚度趋向正常,血管分布丰富,未见明显淤血坏死区域,见图1B。此外,与模型组相比,脐血衍生物各组在子宫和脾脏脏器系数上呈现出差异性的改善(P < 0.01),但脐血衍生物组间差异不显著。与模型组相比,各组卵巢、脾脏脏器系数尚未发现有统计学意义,见表2,图1C。
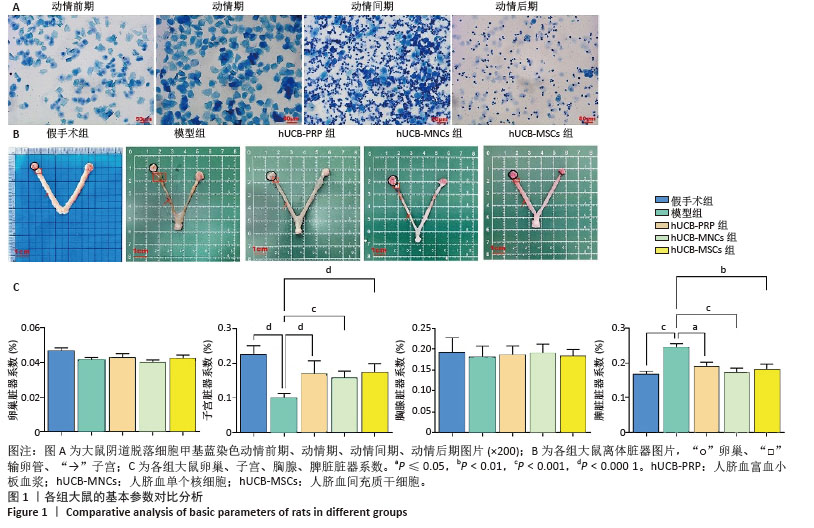
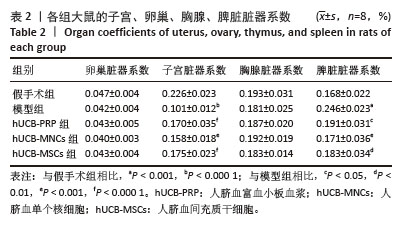
2.3 各组大鼠子宫内膜形态 人脐血衍生物对受损后的大鼠子宫内膜具有不同程度的修复作用,主要表现在增加子宫内膜腺体与血管的生成,促进生长因子、激素调节因子、营养物质和黏液的分泌,支持胚胎着床和早期妊娠。实验研究结果显示,假手术组子宫内膜显示正常的多层上皮细胞,结构完整,表面由柱状上皮覆盖,细胞核形态规则,细胞质均匀;基质细胞分布均匀,无明显炎症反应;腺体呈现良好的圆形或卵形结构,开口清晰,腺上皮细胞呈柱状;血管结构清晰,排列有序,无明显增生或退化。与假手术组相比,模型组子宫内膜厚度减少,上皮细胞层变薄,细胞核形态不规则,部分细胞凋亡;基质细胞减少,可见炎性细胞浸润;腺体数量明显减少,部分腺体形态不规则,腺腔萎缩,腺上皮细胞减少;血管结构紊乱,部分血管壁增厚,内皮细胞损伤。经过hUCB-PRP、hUCB-MSCs、hUCB-MNCs宫腔灌注治疗后,3个治疗组的子宫内膜厚度均有所增加(P < 0.01),上皮细胞层修复良好,细胞核形态逐渐规则;基质细胞增加,纤维化减轻,炎性细胞浸润减少,见图2A。组间比较显示,与hUCB-PRP组相比,hUCB-MSCs组和hUCB-MNCs组腺体数量均降低(P < 0.05)。除此之外,子宫内膜厚度、血管数目尚未发现显著差异,见图2B。
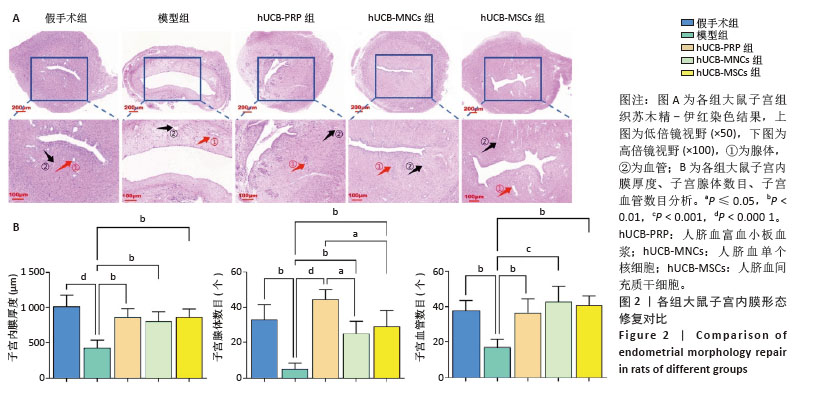
2.4 各组大鼠子宫内膜抗纤维化能力 Masson染色后大鼠子宫组织胶原纤维呈蓝色,肌纤维呈现红色。在正常的大鼠子宫内膜中,肌纤维主要位于子宫肌层中,胶原纤维主要位于大鼠子宫内膜的间质以及最外层的包膜中。当子宫的抗纤维化能力降低后,子宫组织中细胞外基质的过度积累,导致组织结构和功能改变,进而通过基质微环境、子宫内膜容受性、血管生成、免疫调节等方面对胚胎着床产生影响[54-57]。
假手术组胶原纤维少量均匀分布在子宫内膜间质,无明显过度增生或缺失;细胞核呈红色,基质细胞分布正常,无明显纤维化迹象。与假手术组相比,模型组胶原纤维过度增生,分布不均匀;细胞核红色变淡,基质细胞明显减少。
hUCB-PRP组与hUCB-MSCs组胶原纤维增生减少,分布欠均匀,主要分布于子宫内膜基质层;细胞核红色较浓,基质细胞增多。hUCB-MNCs组胶原纤维分布均匀,纤维化显著减轻;细胞核红色浓密,基质细胞数量显著增加,见图3A。以胶原容积分数来量化各组子宫组织纤维化程度,结果显示:与假手术组相比,模型组出现显著纤维化,差异有显著性意义(P < 0.000 1)。与模型组相比,hUCB-PRP、hUCB-MSCs与hUCB-MNCs宫腔灌注治疗后,纤维化水平明显改善,以hUCB-MNCs组最为明显,差异有显著性意义(P < 0.05),见图3B。
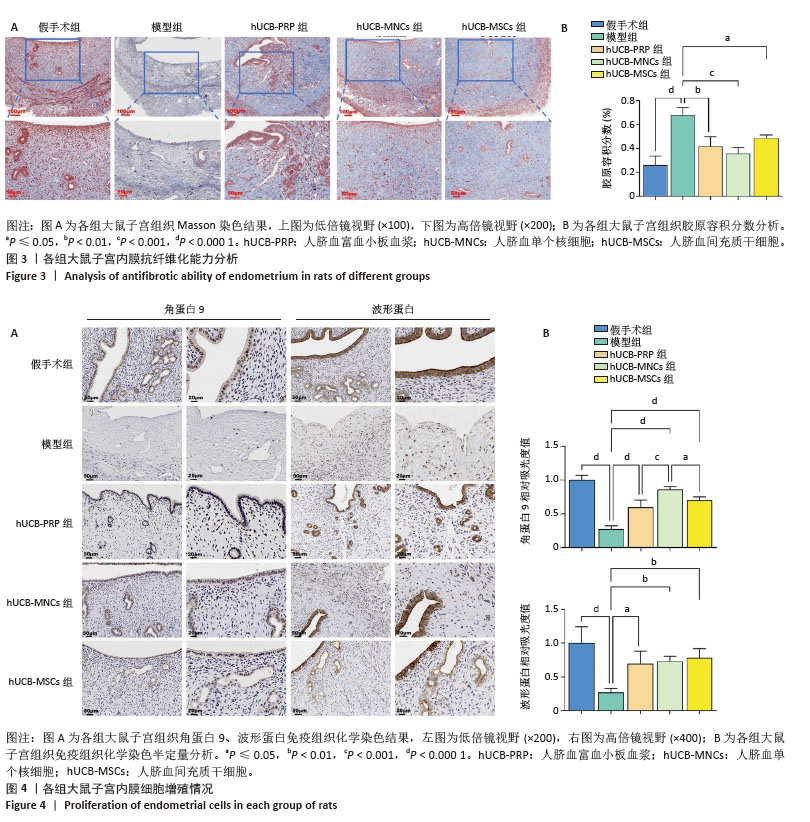
2.5 各组大鼠子宫内膜细胞增殖情况 角蛋白在子宫内膜中的表达主要局限于上皮细胞和腺上皮细胞中,是细胞骨架的重要组成成分,主要功能是维持子宫上皮细胞的组织连续性和完整性。而波形蛋白的表达局限于子宫内膜的间质,是间质细胞骨架的重要组成成分,不仅可以维持细胞结构稳定,还参与细胞有丝分裂、增殖分化及胞内信号转导[53]。免疫组化结果显示,与假手术组相比,模型组角蛋白9、波形蛋白表达水平显著降低(P < 0.000 1)。与模型组相比,hUCB-PRP、hUCB-MNCs与hUCB-MSCs宫腔灌注治疗后,角蛋白9、波形蛋白表达水平均有一定程度的上升(P < 0.01),见图4A。进一步组间比较,与hUCB-MNCs组相比,hUCB-PRP组与hUCB-MSCs组角蛋白9表达水平均降低(P < 0.05)。波形蛋白表达水平组间比较尚未发现统计学差异,见图4B。
2.6 各组大鼠子宫内膜容受性 整合素αγβ3是一种细胞黏附分子,广泛表达于子宫内膜上皮细胞和基质细胞中,特别是在子宫内膜的种植窗口期表达最高。白血病抑制因子作为一种多效性细胞因子,与整合素αγβ3具有协同作用,故整合素αγβ3蛋白和白血病抑制因子蛋白表达升高有利于胚胎的着床与植入。与假手术组相比,模型组整合素αγβ3蛋白和白血病抑制因子蛋白表达水平显著下降(P < 0.05)。经人脐血衍生物治疗后,与模型组相比,除hUCB-MNCs组整合素αγβ3蛋白表达尚未发现显著升高外,其余组别的整合素αγβ3蛋白和白血病抑制因子蛋白表达水平均有所升高,差异有显著性意义(P < 0.05),见图5A-D。
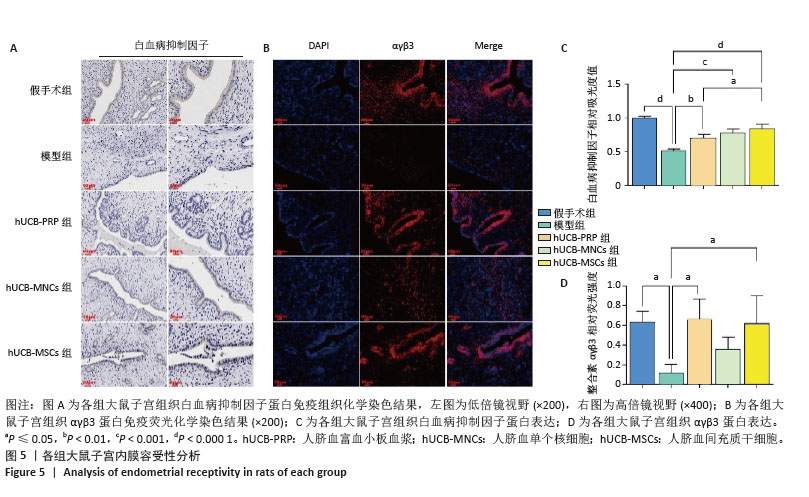
2.7 各组大鼠子宫内膜血管生成情况 血管内皮生长因子和碱性成纤维细胞生长因子是重要的血管生成因子,能够促进血管内皮细胞的增殖和迁移。此外,作为存在于内皮细胞表面的一种黏附分子,血小板内皮细胞黏附分子(CD31)也参与内皮细胞之间的黏附及新生血管的形成。在受损的子宫内膜中,血管生成是修复的关键机制,为再生组织提供必要的氧气和营养物质。与假手术组相比,模型组血管内皮生长因子、碱性成纤维细胞生长因子和CD31蛋白表达水平均有所下降,而经过人脐血衍生物治疗后,血管内皮生长因子、碱性成纤维细胞生长因子和CD31蛋白表达水平均有回升,差异有显著性意义(P < 0.05),见图6A,B。其中血管内皮生长因子蛋白表达水平以hUCB-MNCs和hUCB-MSCs组回升较为明显(P < 0.001),而CD31蛋白表达水平从hUCB-PRP组到hUCB-MNCs组再到hUCB-MSCs组逐渐降低,但与模型组相比,均有所升高,差异有显著性意义(P < 0.05)。碱性成纤维细胞生长因子则以hUCB-MNCs组增加最显著(P < 0.001),见图6C,D。
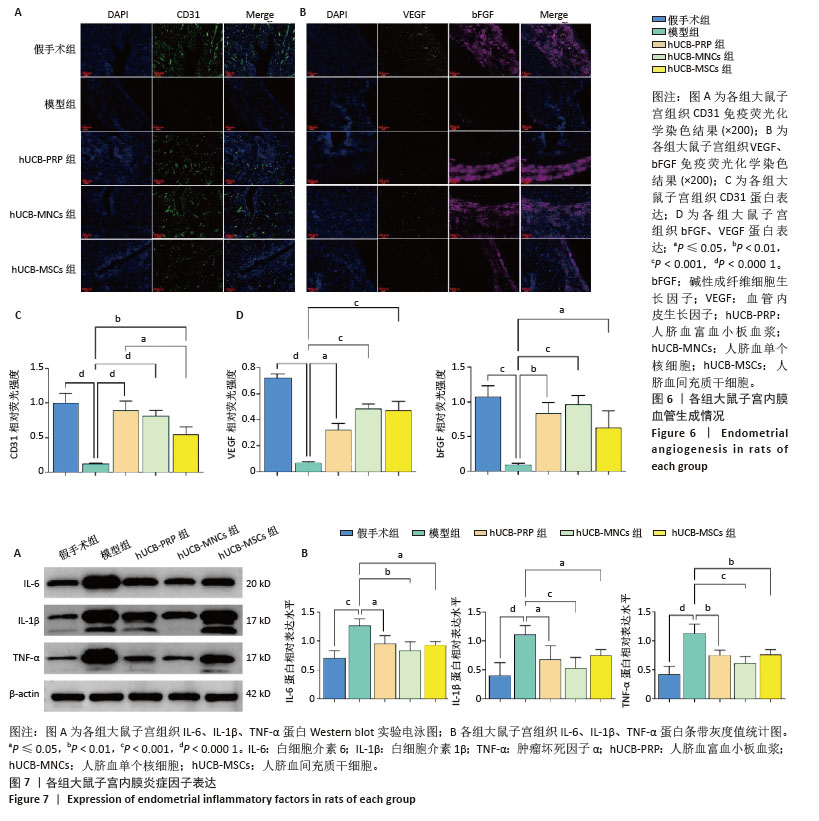
2.8 各组大鼠子宫内膜炎症反应 在子宫内膜的修复过程中,炎症反应扮演着重要角色。白细胞介素6、白细胞介素1β和肿瘤坏死因子α等炎症因子的适度表达有助于清除损伤并促进组织修复,但过度的炎症反应可能干扰修复过程,导致不良后果。因此,通过人脐血衍生物来平衡炎症反应,能够有效修复子宫内膜。与假手术组相比,模型组白细胞介素6、白细胞介素1β和肿瘤坏死因子α蛋白表达水平均显著升高(P < 0.001)。经人脐血衍生物灌注治疗后,白细胞介素6、白细胞介素1β和肿瘤坏死因子α蛋白表达水平均降低(P < 0.05),其中以hUCB-MNCs组抑制炎症与免疫调控作用最为显著(P < 0.01)。白细胞介素6、白细胞介素1β和肿瘤坏死因子α蛋白表达水平在3个治疗组间未见差异(P > 0.05),见图7A,B。
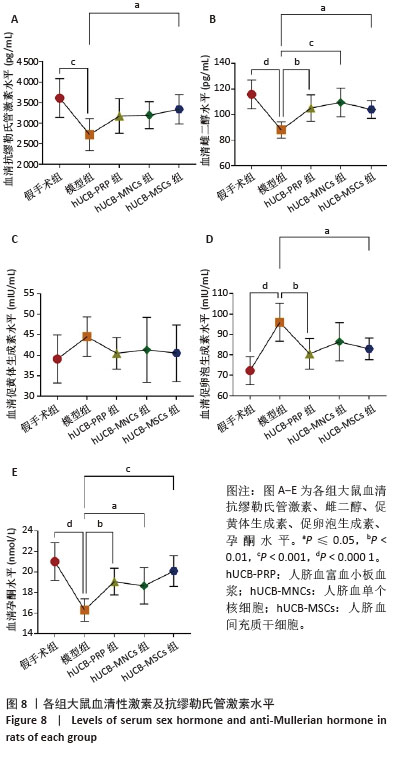
2.9 各组大鼠血清性激素及抗缪勒氏管激素水平 大鼠血清中性激素及抗缪勒氏管激素水平的变化与子宫内膜的修复过程密切相关,这些激素的平衡状态有助于促进子宫内膜的再生和修复,而异常的激素水平可能影响修复效果,甚至导致不良后果。因此,监测这些激素水平可帮助评估和调节子宫内膜的修复过程。与假手术组相比,模型组血清雌二醇、促黄体生成素、促卵泡生成素、孕酮及抗缪勒氏管激素水平均向不良方向增加,除促黄体生成素尚未发现显著变化外,其余指标差异均有显著性意义(P < 0.001)。经过人脐血衍生物治疗后,与模型组相比,hUCB-MSCs组抗缪勒氏管激素水平增加,hUCB-PRP组和hUCB-MSCs组促卵泡生成素水平稍有降低,hUCB-PRP组、hUCB-MNCs组和hUCB-MSCs组雌二醇与孕酮水平均升高,差异有显著性意义(P < 0.05),见图8。
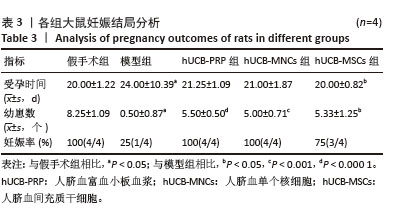
2.10 各组大鼠的生育力 与假手术组相比,模型组受孕时间有所增加,产仔数明显降低,差异有显著性意义(P < 0.05)。经人脐血衍生物治疗后,与模型组相比,3组产仔数均有所增加,差异有显著性意义(P < 0.05)。人脐血衍生物3组间比较,尚未发现差异有显著性意义,这可能与样本量不够大有一定联系。此外,与模型组相比,hUCB-MSCs组受孕时间降低,差异有显著性意义(P < 0.05),见表3。
| [1] SAAD-NAGUIB MH, KENFACK Y, SHERMAN LS, et al. Impaired receptivity of thin endometrium: therapeutic potential of mesenchymal stem cells. Front Endocrinol (Lausanne). 2024;14:1268990. [2] LIU X, QIAN C, JIANG X, et al. Efficacy of platelet-rich plasma in the treatment of thin endometrium: a meta-analysis of randomized controlled trials. BMC Pregnancy Childbirth. 2024;24(1):567. [3] WEISS NS, VAN VLIET MN, LIMPENS J, et al. Endometrial thickness in women undergoing IUI with ovarian stimulation. How thick is too thin? A systematic review and meta-analysis. Hum Reprod. 2017;32(5): 1009-1018. [4] LIU KE, HARTMAN M, HARTMAN A, et al. The impact of a thin endometrial lining on fresh and frozen-thaw IVF outcomes: an analysis of over 40 000 embryo transfers. Hum Reprod. 2018;33(10):1883-1888. [5] KASIUS A, SMIT JG, TORRANCE HL, et al. Endometrial thickness and pregnancy rates after IVF: a systematic review and meta-analysis. Hum Reprod Update. 2014;20(4):530-541. [6] MOUHAYAR Y, SHARARA FI. G-CSF and stem cell therapy for the treatment of refractory thin lining in assisted reproductive technology. J Assist Reprod Genet. 2017;34(7):831-837. [7] MAO X, ZHANG J, CAI R, et al. Therapeutic role of granulocyte macrophage colony-stimulating factor (GM-CSF) in patients with persistent thin endometrium: A prospective and randomized study. Int J Gynaecol Obstet. 2020;150(2):194-199. [8] MAKRIGIANNAKIS A, MAKRYGIANNAKIS F, VREKOUSSIS T. Approaches to Improve Endometrial Receptivity in Case of Repeated Implantation Failures. Front Cell Dev Biol. 2021;9:613277. [9] LV H, ZHAO G, JIANG P, et al. Deciphering the endometrial niche of human thin endometrium at single-cell resolution. Proc Natl Acad Sci U S A. 2022;119(8):e2115912119. [10] SHU J, LIU X, LI R. Editorial: Impaired receptivity of thin endometrium: the mechanism, hormone intervention and strategies. Front Endocrinol (Lausanne). 2024;15:1432284. [11] WANG Y, TANG Z, TENG X. New advances in the treatment of thin endometrium. Front Endocrinol (Lausanne). 2024;15:1269382. [12] ZHAO J, ZHANG Q, WANG Y, et al. Uterine infusion with bone marrow mesenchymal stem cells improves endometrium thickness in a rat model of thin endometrium. Reprod Sci. 2015;22(2):181-188. [13] HAO SN, XIA LJ, XI J, et al. Electroacupuncture combined with bone marrow mesenchymal stem cell transplantation promotes repair of thin endometrium by regulating SDF-1/CXCR4 signaling. Zhen Ci Yan Jiu. 2023;48(9):870-880. [14] ARIKAN G, TURAN V, KUREKEKEN M, et al. Autologous bone marrow-derived nucleated cell (aBMNC) transplantation improves endometrial function in patients with refractory Asherman’s syndrome or with thin and dysfunctional endometrium. J Assist Reprod Genet. 2023;40(5):1163-1171. [15] YE MX, YU L, WANG SF, et al. Efficacy of gamma-irradiated adipose-derived stem cells for treatment of thin endometrium in rats. Nan Fang Yi Ke Da Xue Xue Bao. 2017;37(5):575-580. [16] SUDOMA I, PYLYP L, KREMENSKA Y, et al. Application of autologous adipose-derived stem cells for thin endometrium treatment in patients with failed ART programs. J Stem Cell Ther Transplant. 2019;3(1): 001-008. [17] YOTSUMOTO F, YOSHIKAWA K, HIRAKAWA T, et al. Safety and Potential Effect of Intrauterine Infusion of Autologous Adipose Tissue-Derived Regenerative Cells in Patients With Implantation Failure: A Pilot Study. Cureus. 2024;16(3):e57220. [18] ZHAO M, CHI F, ZHANG T, et al. Human menstrual blood-derived mesenchymal stem cells regulation of the EGF/Ras p21 pathway as a potential therapeutic target for thin endometrium. Ann Transl Med. 2021;9(18):1476. [19] WANG H, CHEN K, ZONG L, et al. MALAT1/miR-7-5p/TCF4 Axis Regulating Menstrual Blood Mesenchymal Stem Cells Improve Thin Endometrium Fertility by the Wnt Signaling Pathway. Cell Transplant. 2024;33:9636897241259552. [20] ZHANG Y, SHI L, LIN X, et al. Unresponsive thin endometrium caused by Asherman syndrome treated with umbilical cord mesenchymal stem cells on collagen scaffolds: a pilot study. Stem Cell Res Ther. 2021;12(1):420. [21] ZHOU S, LEI Y, WANG P, et al. Human Umbilical Cord Mesenchymal Stem Cells Encapsulated with Pluronic F-127 Enhance the Regeneration and Angiogenesis of Thin Endometrium in Rat via Local IL-1β Stimulation. Stem Cells Int. 2022;2022:7819234. [22] SAPOZHAK IM, GUBAR ОS, RODNICHENKO AE, et al. Application of autologous endometrial mesenchymal stromal/stem cells increases thin endometrium receptivity: a case report. J Med Case Rep. 2020; 14(1):190. [23] RODRÍGUEZ-EGUREN A, BUENO-FERNANDEZ C, GÓMEZ-ÁLVAREZ M, et al. Evolution of biotechnological advances and regenerative therapies for endometrial disorders: a systematic review. Hum Reprod Update. 2024;30(5):584-613. [24] AGARWAL M, METTLER L, JAIN S, et al. Management of a Thin Endometrium by Hysteroscopic Instillation of Platelet-Rich Plasma Into The Endomyometrial Junction: A Pilot Study. J Clin Med. 2020; 9(9):2795. [25] YUAN G, YU C, DU X, et al. Injectable GelMA Hydrogel Microspheres with Sustained Release of Platelet-Rich Plasma for the Treatment of Thin Endometrium. Small. 2024;20(47):e2403890. [26] WANG X, LIU L, MOU S, et al. Investigation of platelet-rich plasma in increasing proliferation and migration of endometrial mesenchymal stem cells and improving pregnancy outcome of patients with thin endometrium. J Cell Biochem. 2019;120(5):7403-7411. [27] TABEEVA G, SILACHEV D, VISHNYAKOVA P, et al. The Therapeutic Potential of Multipotent Mesenchymal Stromal Cell-Derived Extracellular Vesicles in Endometrial Regeneration. Int J Mol Sci. 2023;24(11):9431. [28] ZHOU Y, LI Q, YOU S, et al. Efficacy of Mesenchymal Stem Cell-Derived Extracellular Vesicles in the Animal Model of Female Reproductive Diseases: A Meta-Analysis. Stem Cell Rev Rep. 2023;19(7):2299-2310. [29] GIBSON OO, MAKARCHUK O, RIMARCHUK M, et al. Role of vascular growth factors as a regulator of angiogenesis processes at the stage of implantation potential formation in women with uterine factor infertility. Health of Woman. 2019;6(142):27-33. [30] WANG S, LIU T, NAN N, et al. Exosomes from Human Umbilical Cord Mesenchymal Stem Cells Facilitates Injured Endometrial Restoring in Early Repair Period through miR-202-3p Mediating Formation of ECM. Stem Cell Rev Rep. 2023;19(6):1954-1964. [31] WANG S, SHI C, CAI X, et al. Human Acellular Amniotic Matrix with Previously Seeded Umbilical Cord Mesenchymal Stem Cells Restores Endometrial Function in a Rat Model of Injury. Mediators Inflamm. 2021;2021:5573594. [32] SONG L, ZHANG Q, ZHU S, et al. Granulocyte Colony-Stimulating Factor Combined With Transcutaneous Electrical Acupoint Stimulation in Treatment of Unresponsive Thin Endometrium in Frozen Embryo Transfer Cycles. Front Reprod Health. 2021;3:647336. [33] PARAJULI R, JAIN V, GAINDER S, et al. An interventional study on the effect of intrauterine granulocyte-colony stimulating factor instillation on thin or damaged endometrium in women undergoing assisted reproduction. Fertil Steril. 2024;122(4):e425. [34] CHEN J, HUANG F, FU J, et al. Hyperbaric oxygen therapy: a possible choice for patients with resistant thin endometrium during frozen embryo transfer treatments. Reprod Biol Endocrinol. 2023;21(1):80. [35] RODRÍGUEZ-EGUREN A, GÓMEZ-ÁLVAREZ M, FRANCÉS-HERRERO E, et al. Human Umbilical Cord-Based Therapeutics: Stem Cells and Blood Derivatives for Female Reproductive Medicine. Int J Mol Sci. 2022;23(24):15942. [36] RODRÍGUEZ-EGUREN A, DE MIGUEL-GÓMEZ L, FRANCÉS-HERRERO E, et al. Human umbilical cord platelet-rich plasma to treat endometrial pathologies: methodology, composition and pre-clinical models. Hum Reprod Open. 2022;2023(1):hoac053. [37] CECERSKA-HERYĆ E, GOSZKA M, SERWIN N, et al. Applications of the regenerative capacity of platelets in modern medicine. Cytokine Growth Factor Rev. 2022;64:84-94. [38] LEE OK, KUO TK, CHEN WM, et al. Isolation of multipotent mesenchymal stem cells from umbilical cord blood. Blood. 2004;103(5):1669-1675. [39] XI Y, YUE G, GAO S, et al. Human umbilical cord blood mononuclear cells transplantation for perinatal brain injury. Stem Cell Res Ther. 2022;13(1):458. [40] BAZANOVICH SA, RYABOV SI, ZVYAGINTSEVA MA, et al. Evaluation of the Effectiveness of Systemic Therapy of Spinal Cord Injury of Moderate Severity with Human Umbilical Cord Placental Blood Mononuclear Cells Using Indicators of Dispersion of Articular Angles in the Swimming Test. Bull Exp Biol Med. 2023;174(6):784-789. [41] SHULMAN I, OGURCOV S, KOSTENNIKOV A, et al. Application of Autologous Peripheral Blood Mononuclear Cells into the Area of Spinal Cord Injury in a Subacute Period: A Feasibility Study in Pigs. Biology (Basel). 2021;10(2):87. [42] LI J, JIANG Y, XUE W, et al. Effects of transplantation of umbilical cord blood mononuclear cells into the scrotum on sexual function in elderly mice. Regen Med. 2023;18(9):695-706. [43] CAI X, LI Y, GAO F, et al. Therapeutic effect and study of human umbilical cord blood mononuclear cells in patients with ischaemic bowel disease. Sci Rep. 2024;14(1):6121. [44] CHEN G, YUE A, YU H, et al. Mesenchymal Stem Cells and Mononuclear Cells From Cord Blood: Cotransplantation Provides a Better Effect in Treating Myocardial Infarction. Stem Cells Transl Med. 2016;5(3):350-357. [45] ZHANG L, LI Y, DONG YC, et al. Transplantation of umbilical cord-derived mesenchymal stem cells promotes the recovery of thin endometrium in rats. Sci Rep. 2022;12(1):412. [46] 冯海艳,邓春雷,张怡君,等.薄型子宫内膜大鼠95%乙醇造模法的实验研究[J].湖北医药学院学报,2024,43(5):490-493+458. [47] WANG J, QIN W, ZHONG Y, et al. Injectable collagen hydrogel combines human umbilical cord mesenchymal stem cells to promote endometrial regeneration in rats with thin endometrium. Int J Biol Macromol. 2024;254(Pt 1):127591. [48] XIA L, MENG Q, XI J, et al. The synergistic effect of electroacupuncture and bone mesenchymal stem cell transplantation on repairing thin endometrial injury in rats. Stem Cell Res Ther. 2019;10(1):244. [49] GUO Q, CHANG Y, LI J, et al. Regenerative Effects of Locally or Intra-Arterially Administered BMSCs on the Thin Endometrium. Front Bioeng Biotechnol. 2022;10:735465. [50] LIN Y, DONG S, YE X, et al. Synergistic regenerative therapy of thin endometrium by human placenta-derived mesenchymal stem cells encapsulated within hyaluronic acid hydrogels. Stem Cell Res Ther. 2022;13(1):66. [51] LI J, HUANG B, DONG L, et al. WJ-MSCs intervention may relieve intrauterine adhesions in female rats via TGF-β1-mediated Rho/ROCK signaling inhibition. Mol Med Rep. 2021;23(1):15. [52] XIE Y, TIAN Z, QI Q, et al. The therapeutic effects and underlying mechanisms of the intrauterine perfusion of granulocyte colony-stimulating factor on a thin-endometrium rat model. Life Sci. 2020; 260:118439. [53] 刘若熹.人脐带间充质干细胞修复大鼠薄型子宫内膜的研究[D].济南:山东大学,2020. [54] GARGETT CE, YE L. Endometrial reconstruction from stem cells. Fertil Steril. 2012;98(1):11-20. [55] LIU F, HU S, WANG S, et al. Cell and biomaterial-based approaches to uterus regeneration. Regen Biomater. 2019;6(3):141-148. [56] ZHANG Z, LI S, DENG J, et al. Aspirin inhibits endometrial fibrosis by suppressing the TGF β1 Smad2/Smad3 pathway in intrauterine adhesions. Int J Mol Med. 2020;45(5):1351-1360. [57] AI Y, CHEN M, LIU J, et al. lncRNA TUG1 promotes endometrial fibrosis and inflammation by sponging miR-590-5p to regulate Fasl in intrauterine adhesions. Int Immunopharmacol. 2020;86:106703. [58] WORLD HEALTH ORGANIZATION. WHO Team Sexual and Reproductive Health and Research Infertility Prevalence Estimates 1990-2021. Geneva: World Health Organization, 2024. [59] CAIAFFA V, IPPOLITO F, ABATE A, et al. Allogenic platelet concentrates from umbilical cord blood for knee osteoarthritis: preliminary results. Med Glas (Zenica). 2021;18(1):260-266. [60] YANG Q, HUANG J, LIU Y, et al. Human Umbilical Cord Mesenchymal Stem Cells Promote Anti-Inflammation and Angiogenesis by Targeting Macrophages in a Rat Uterine Scar Model. Stem Cell Rev Rep. 2024;20(6):1555-1568. [61] SHIN SY, CHUNG N, SHIN JE, et al. Angiogenic factor-driven improvement of refractory thin endometrium with autologous platelet-rich plasma intrauterine infusion in frozen embryo transfer cycles. Front Endocrinol (Lausanne). 2024;15:1431453. [62] JING Z, YI Y, XI H, et al. Therapeutic Effects of VEGF Gene-Transfected BMSCs Transplantation on Thin Endometrium in the Rat Model. Stem Cells Int. 2018;2018:3069741. [63] LV H, XU R, XIE X, et al. Injectable, degradable, and mechanically adaptive hydrogel induced by L-serine and allyl-functionalized chitosan with platelet-rich plasma for treating intrauterine adhesions. Acta Biomater. 2024;184:144-155. [64] KIM MK, YOON JA, YOON SY, et al. Human Platelet-Rich Plasma Facilitates Angiogenesis to Restore Impaired Uterine Environments with Asherman’s Syndrome for Embryo Implantation and Following Pregnancy in Mice. Cells. 2022;11(9):1549. [65] ZADEHMODARRES S, SALEHPOUR S, SAHARKHIZ N, et al. Treatment of thin endometrium with autologous platelet-rich plasma: a pilot study. JBRA Assist Reprod. 2017;21(1):54-56. [66] SFAKIANOUDIS K, SIMOPOULOU M, NITSOS N, et al. Successful Implantation and Live Birth Following Autologous Platelet-rich Plasma Treatment for a Patient with Recurrent Implantation Failure and Chronic Endometritis. In Vivo. 2019;33(2):515-521. [67] MOLINA A, SÁNCHEZ J, SÁNCHEZ W, et al. Platelet-rich plasma as an adjuvant in the endometrial preparation of patients with refractory endometrium. JBRA Assist Reprod. 2018;22(1):42-48. [68] ZHANG S, LI P, YUAN Z, et al. Platelet-rich plasma improves therapeutic effects of menstrual blood-derived stromal cells in rat model of intrauterine adhesion. Stem Cell Res Ther. 2019;10(1):61. [69] TANDULWADKAR S, MISHRA S, GUPTA S. Successful Application of Combined Autologous Bone Marrow-Derived Stem Cells and Platelet-Rich Plasma in a Case of Severe Asherman Syndrome and Subsequent in vitro Fertilization Conception. J Hum Reprod Sci. 2021;14(4):446-449. [70] LI J, LI X, DING J, et al. Analysis of pregnancy outcomes in patients with recurrent implantation failure complicated with chronic endometritis. Front Cell Dev Biol. 2023;11:1088586. [71] CASTELLANO JM, MOSHER KI, ABBEY RJ, et al. Human umbilical cord plasma proteins revitalize hippocampal function in aged mice. Nature. 2017;544(7651):488-492. [72] EHRHART J, SANBERG PR, GARBUZOVA-DAVIS S. Plasma derived from human umbilical cord blood: Potential cell-additive or cell-substitute therapeutic for neurodegenerative diseases. J Cell Mol Med. 2018;22(12):6157-6166. [73] MURPHY MB, BLASHKI D, BUCHANAN RM, et al. Adult and umbilical cord blood-derived platelet-rich plasma for mesenchymal stem cell proliferation, chemotaxis, and cryo-preservation. Biomaterials. 2012;33(21):5308-5316. [74] LEHALLIER B, GATE D, SCHAUM N, et al. Undulating changes in human plasma proteome profiles across the lifespan. Nat Med. 2019;25(12): 1843-1850. [75] NAGAMURA-INOUE T, HE H. Umbilical cord-derived mesenchymal stem cells: Their advantages and potential clinical utility. World J Stem Cells. 2014;6(2):195-202. [76] ALANAZI A, ALASSIRI M, JAWDAT D, et al. Mesenchymal stem cell therapy: A review of clinical trials for multiple sclerosis. Regen Ther. 2022;21:201-209. [77] CHENG H, LIU X, HUA R, et al. Clinical observation of umbilical cord mesenchymal stem cell transplantation in treatment for sequelae of thoracolumbar spinal cord injury. J Transl Med. 2014;12:253. [78] DING DC, CHANG YH, SHYU WC, et al. Human umbilical cord mesenchymal stem cells: a new era for stem cell therapy. Cell Transplant. 2015;24(3):339-347. [79] HE X, WANG Q, ZHAO Y, et al. Effect of Intramyocardial Grafting Collagen Scaffold With Mesenchymal Stromal Cells in Patients With Chronic Ischemic Heart Disease: A Randomized Clinical Trial. JAMA Netw Open. 2020;3(9):e2016236. [80] BARTOLUCCI J, VERDUGO FJ, GONZÁLEZ PL, et al. Safety and Efficacy of the Intravenous Infusion of Umbilical Cord Mesenchymal Stem Cells in Patients With Heart Failure: A Phase 1/2 Randomized Controlled Trial (RIMECARD Trial [Randomized Clinical Trial of Intravenous Infusion Umbilical Cord Mesenchymal Stem Cells on Cardiopathy]). Circ Res. 2017;121(10):1192-1204. [81] ÖZMERT E, ARSLAN U. Management of retinitis pigmentosa by Wharton’s jelly-derived mesenchymal stem cells: prospective analysis of 1-year results. Stem Cell Res Ther. 2020;11(1):353. [82] ZANG L, LI Y, HAO H, et al. Efficacy and safety of umbilical cord-derived mesenchymal stem cells in Chinese adults with type 2 diabetes: a single-center, double-blinded, randomized, placebo-controlled phase II trial. Stem Cell Res Ther. 2022;13(1):180. [83] HUANG J, LI Q, YUAN X, et al. Intrauterine infusion of clinically graded human umbilical cord-derived mesenchymal stem cells for the treatment of poor healing after uterine injury: a phase I clinical trial. Stem Cell Res Ther. 2022;13(1):85. [84] XU Y, HU J, LV Q, et al. Endometrium-derived mesenchymal stem cells suppress progression of endometrial cancer via the DKK1-Wnt/β-catenin signaling pathway. Stem Cell Res Ther. 2023;14(1):159. [85] BENVENUTO F, VOCI A, CARMINATI E, et al. Human mesenchymal stem cells target adhesion molecules and receptors involved in T cell extravasation. Stem Cell Res Ther. 2015;6:245. [86] JIAO Y, LI XY, LIU J. A New Approach to Cerebral Palsy Treatment: Discussion of the Effective Components of Umbilical Cord Blood and its Mechanisms of Action. Cell Transplant. 2019;28(5):497-509. [87] AHN JY, HONG YH, KIM KC, et al. Effect of Human Peripheral Blood Mononuclear Cells on Mouse Endometrial Cell Proliferation: A Potential Therapeutics for Endometrial Regeneration. Gynecol Obstet Invest. 2022;87(2):105-115. [88] ZHAO G, CAO Y, ZHU X, et al. Transplantation of collagen scaffold with autologous bone marrow mononuclear cells promotes functional endometrium reconstruction via downregulating ΔNp63 expression in Asherman’s syndrome. Sci China Life Sci. 2017;60(4):404-416. [89] RALLAPALLI S, GUHATHAKURTA S, NARAYAN S, et al. Generation of clinical-grade red blood cells from human umbilical cord blood mononuclear cells. Cell Tissue Res. 2019;375(2):437-449. [90] ZHANG J, ZHAI H, YU P, et al. Human Umbilical Cord Blood Mononuclear Cells Ameliorate CCl4-Induced Acute Liver Injury in Mice via Inhibiting Inflammatory Responses and Upregulating Peripheral Interleukin-22. Front Pharmacol. 2022; 13:924464. [91] RAMLI Y, ALWAHDY AS, KURNIAWAN M, et al. Intra-arterial Transplantation of Human Umbilical Cord Blood Mononuclear Cells in Sub-acute Ischemic Stroke Increases VEGF Expression in Rats. J Stem Cells Regen Med. 2018;14(2):69-79. [92] GATINA DZ, GAZIZOV IM, ZHURAVLEVA MN, et al. Induction of Angiogenesis by Genetically Modified Human Umbilical Cord Blood Mononuclear Cells. Int J Mol Sci. 2023;24(5):4396. [93] CHO KH, CHOI JI, KIM JO, et al. Therapeutic mechanism of cord blood mononuclear cells via the IL-8-mediated angiogenic pathway in neonatal hypoxic-ischaemic brain injury. Sci Rep. 2020;10(1):4446. [94] EFENDIEVA Z, VISHNYAKOVA P, APOLIKHINA I, et al. Hysteroscopic injections of autologous endometrial cells and platelet-rich plasma in patients with thin endometrium: a pilot randomized study. Sci Rep. 2023;13(1):945. [95] YUAN G, LI D, DU X, et al. Effects of platelet-rich fibrin on human endometrial stromal cells behavior in comparison to platelet-rich plasma. Front Cell Dev Biol. 2024;12:1445928. [96] XIE Q, LIU R, JIANG J, et al. What is the impact of human umbilical cord mesenchymal stem cell transplantation on clinical treatment. Stem Cell Res Ther. 2020;11(1):519. [97] BABA K, YAMAZAKI Y, SONE Y, et al. An in vitro long-term study of cryopreserved umbilical cord blood-derived platelet-rich plasma containing growth factors-PDGF-BB, TGF-β, and VEGF. J Craniomaxillofac Surg. 2019;47(4):668-675. [98] ZHU D, CHENG K. Cardiac Cell Therapy for Heart Repair: Should the Cells Be Left Out. Cells. 2021;10(3):641. [99] CHEN K, WANG H, ZHAO X, et al. A Novel Method to Repair Thin Endometrium and Restore Fertility Based on Menstruation-Derived Stem Cell. Reprod Sci. 2024;31(6):1662-1673. [100] LIN X, FANG Y, MI X, et al. Intrauterine injection of bioengineered hydrogel loaded exosomes derived from HUCM stem cells and spermidine prominently augments the pregnancy rate in thin endometrium rats. Regen Ther. 2024;27:63-72. [101] XIN L, LIN X, PAN Y, et al. A collagen scaffold loaded with human umbilical cord-derived mesenchymal stem cells facilitates endometrial regeneration and restores fertility. Acta Biomater. 2019;92:160-171. [102] XU L, DING L, WANG L, et al. Umbilical cord-derived mesenchymal stem cells on scaffolds facilitate collagen degradation via upregulation of MMP-9 in rat uterine scars. Stem Cell Res Ther. 2017;8(1):84. [103] SHUAI Q, LIANG Y, XU X, et al. Sodium alginate hydrogel integrated with type III collagen and mesenchymal stem cell to promote endometrium regeneration and fertility restoration. Int J Biol Macromol. 2023; 253(Pt 6):127314. [104] KHARBIKAR BN, MOHINDRA P, DESAI TA. Biomaterials to enhance stem cell transplantation. Cell Stem Cell. 2022;29(5):692-721. [105] HE X, HONG W, YANG J, et al. Spontaneous apoptosis of cells in therapeutic stem cell preparation exert immunomodulatory effects through release of phosphatidylserine. Signal Transduct Target Ther. 2021;6(1):270. [106] NÉMETH K, LEELAHAVANICHKUL A, YUEN PS, et al. Bone marrow stromal cells attenuate sepsis via prostaglandin E(2)-dependent reprogramming of host macrophages to increase their interleukin-10 production. Nat Med. 2009;15(1):42-49. |
| [1] | 鄢成波, 罗秋池, 樊佳兵, 顾叶婷, 邓 倩, 张军梅. 2型糖尿病对大鼠正畸牙移动与张力侧骨微结构参数的影响[J]. 中国组织工程研究, 2026, 30(4): 824-831. |
| [2] | 苏晓杨, 陈文婷, 付怡丹, 赵 燕, 兰丹凤, 杨秋萍. Mer受体酪氨酸激酶与SD大鼠糖尿病周围神经病变的相关性[J]. 中国组织工程研究, 2025, 29(8): 1593-1599. |
| [3] | 何龙才, 宋文学, 明 江, 陈光唐, 王军浩, 廖益东, 崔君拴, 徐卡娅. SD大鼠乳鼠原代皮质神经元和小胶质细胞同时提取并培养的实验方法[J]. 中国组织工程研究, 2025, 29(7): 1395-1400. |
| [4] | 卓秋燕, 蒋 群, 夏 思, 卢诗颖, 刘燕娣, 戴 媺. 骨髓增生异常综合征模型大鼠骨髓造血:活髓方干预免疫检查点的作用机制[J]. 中国组织工程研究, 2025, 29(36): 7735-7742. |
| [5] | 钟 敏, 王 成, 范振海, 李林艳, 余丽梅. 围产期间充质干细胞及联合水凝胶治疗宫腔粘连的作用和机制[J]. 中国组织工程研究, 2025, 29(31): 6792-6799. |
| [6] | 梁柔筠, 展立芬, 曾学究, 丁强盛, 罗小精, 卓 越, 艾 坤, 邓石峰, 许 明, 张 泓. 完全性脊髓横断后排尿次数对神经源性膀胱模型大鼠成模率的影响[J]. 中国组织工程研究, 2025, 29(18): 3840-3847. |
| [7] | 闻哲嘉, 吕 芳. 基于微流控芯片评估富血小板血浆促进子宫内膜细胞的增殖[J]. 中国组织工程研究, 2025, 29(10): 2091-2096. |
| [8] | 马艳霞 , 杨严伟 , 马宇航, 李 迪 , 王晓燕 , 邹明明 , 韦善文. 创伤性脑损伤大鼠皮质神经元中 Lnx1 表达及参与继发性脑损伤的机制[J]. 中国组织工程研究, 2025, 29(1): 24-30. |
| [9] | 杨 程, 李玉生, 焦泓焯, 尚 曼, 刘 琪, 李林臻, 樊方阳, 张成龙, 张晓宇, 张君涛. 肾虚血瘀型SD大鼠膝骨关节炎模型的建立与验证[J]. 中国组织工程研究, 2024, 28(27): 4273-4280. |
| [10] | 武晓丹, 王治国, 战 莹, 张国旭. 32P-β射线致急性放射性皮肤损伤模型的建立及损伤机制[J]. 中国组织工程研究, 2024, 28(14): 2173-2179. |
| [11] | 廖益东, 明 江, 宋文学, 王梓力, 张 宇, 廖一飞, 徐卡娅, 杨 华. 一种同时培养原代皮质及海马神经元的实验方法[J]. 中国组织工程研究, 2023, 27(6): 897-902. |
| [12] | 丁 雪, 贾 莹, 刘 纯, 杨世榕, 赖灵妍, 杨 桦, 丁 琪. 慢性氟中毒SD大鼠正畸牙移动的位移及速率变化[J]. 中国组织工程研究, 2022, 26(29): 4687-4692. |
| [13] | 刘星妤, 胡小芳, 徐广立, 王 瑞, 李佩瑶, 王梦园, 彭 丽, 朱香颖. 人脐带间充质干细胞修复子宫内膜损伤[J]. 中国组织工程研究, 2022, 26(24): 3921-3927. |
| [14] | 王文华, 刘 洋, 王巧敏, 吕 洋, 赵亚如, 王海萍. 5-氮胞苷联合骨形态发生蛋白2诱导骨髓间充质干细胞向心肌样细胞分化[J]. 中国组织工程研究, 2022, 26(19): 2958-2963. |
| [15] | 贾麒钰, 郭 建, 魏 琴, 郭晓斌, 陈东生, 冯东伟, 刘彦士, 马 创. 大鼠股骨外固定架螺钉效能验证模型的建立及评价[J]. 中国组织工程研究, 2022, 26(18): 2854-2861. |
尽管目前对于薄型子宫内膜的诊断阈值尚无统一标准,且具体数据在不同研究中存在一定的差异[5-8],但临床和实验证据表明,随着子宫内膜厚度的减少,一系列不良的生殖效应逐渐显现,已被广泛认为是不孕、复发性流产和胎盘发育异常的关键原因[9]。
近年来,越来越多的研究显示脐血细胞组分在组织修复和再生中具有显著效果,应用潜力逐渐显露[10-11]。目前薄型子宫内膜的治疗方法包括传统药物治疗、细胞治疗(骨髓[12-14]、脂肪[15-17]、经血[18-19]、脐血[20-21]、子宫内膜来源间充质干细胞[22],人胚胎干细胞[23])、非细胞治疗(富血小板血浆[24-26]、细胞外囊泡[27-28]、生长因子[29]、miRNAs[30]、替代脱细胞疗法[31]、粒细胞集落刺激因子[32])、生物工程材料治疗(细胞外基质衍生水凝胶、其他生物材料)以及高压氧治疗[33-34]。由于传统药物治疗在子宫内膜修复/再生和生育能力恢复方面的疗效参差不齐,因此国际上正在努力开发基于干细胞方法、脱细胞成分和生物材料(天然或合成)的有效治疗方法[35]。人脐血富血小板血浆(human umbilical cord blood platelet-rich plasma,hUCB-PRP)作为一种来自年轻供者的异基因脐血脱细胞成分[36],被公认为是一种安全有效的促进组织修复与再生的治疗方法[37],激活后会迅速释放更多具有再生能力的蛋白质、生长因子及生物分子。LEE等[38]指出,人脐血间充质干细胞(human umbilical cord blood derived mesenchymal stem cells,hUCB-MSCs)作为一种多功能干细胞,具有分化为多种细胞类型的能力,是治疗子宫内膜疾病最常见的间充质干细胞类型。然而,关于人脐血单个核细胞(human umbilical cord blood mononuclear cells,hUCB-MNCs)治疗薄型子宫内膜的研究报道尚少,但已有文献证明,hUCB-MNCs可以用于治疗颅脑损伤[39]、脊髓损伤[40-41]、性功能障碍[42]、消化系统疾病[43]、心血管疾病等[44],并显示出良好的效果。
新兴的治疗方法结合了细胞、非细胞和生物工程等技术,通过同时恢复体内平衡、调节炎症、增强细胞增殖和促进组织重塑来加速组织再生。但目前关于薄型子宫内膜的细胞疗法与非细胞疗法的对比研究相对匮乏,同时也缺乏关于hUCB-MNCs对薄型子宫内膜影响的相关数据。基于此,该研究系统比较3种人脐血衍生物组分(hUCB-PRP、hUCB-MSCs、hUCB-MNCs)在大鼠薄型子宫内膜修复中的作用,通过分析细胞学和分子生物学机制,揭示在内膜容受性、内膜再生功能及改善妊娠结局方面的差异与优势。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.2 时间及地点 实验于2023年7月至2024年7月在贵州医科大学附属医院临床研究中心SPF级动物房内进行。
1.3 材料
1.3.1 实验动物 7-9周龄SPF级健康且性成熟(未交配)的雌性Sprague-Dawley(SD)大鼠67只,体质量为(220±20) g,购自广东维通利华实验动物技术有限公司,许可证号:SCXK(粤)2022-0063。所有SD大鼠均符合国家实验动物标准,饲养于贵州医科大学附属医院临床研究中心SPF级动物中心。光照时间:08:00 am-20:00 pm,温度:(22±1) ℃,湿度:65%-85%,自由进食和饮水。动物实验通过贵州医科大学实验动物伦理委员会批准(伦理批号:NO.2305161,审查日期:2023-05-22)。实验过程遵循了国际兽医学编辑协会《关于动物伦理与福利的作者指南共识》和本地及国家法规。实验动物在麻醉下进行所有手术,并最大限度地减少其疼痛、痛苦和死亡。
1.3.2 人脐血衍生物 hUCB-PRP、hUCB-MNCs及hUCB-MSCs的制备、纯化、运输均由山东省齐鲁干细胞工程有限公司根据《脐血富血小板血浆的制备工艺、质量控制标准和制定依据及工艺稳定性数据》《脐血单个核细胞制剂的制备工艺、质量控制标准和制定依据及工艺稳定性数据》《干细胞制剂的制备工艺、质量控制标准和制定依据及工艺稳定性数据》文件标准提供。
(1)hUCB-PRP:hUCB-PRP为呈黄色澄清液体,无色泽异常、蛋白析出、气泡及重度乳糜等情况,封装完好,采取-80 ℃保存,避免辐照。血小板数量9.00×109,红细胞数量22.00×107,血型AB+,pH值6.70,体积12 mL,母血检测:HBsAg阴性、Anti-HCV阴性、Anti-CMV-IgM阴性、Anti-TP阴性、Anti-HIVⅠ/Ⅱ阴性、HBV-DNA阴性、HIV-RNA阴性、HCV-RNA阴性;微生物检测:需氧菌阴性,厌氧菌阴性。
(2)hUCB-MNCs:hUCB-MNCs制剂为淡黄色或淡粉色液体,封装完好,放置于液氮保存。细胞悬液体积10 mL,细胞数量3.0×108,细胞活率97.87%;母血检测:HBsAg阴性、Anti-HCV阴性、Anti-CMV-IgM阴性、Anti-TP阴性、Anti-HIVⅠ/Ⅲ阴性、HBV-DNA阴性、HIV-RNA阴性、HCV-RNA阴性;微生物检测:需氧菌阴性,厌氧菌阴性,细菌内毒素< 0.25 EU/mL;细胞流式表型检测:CD34阳性率0.41%;CFU-GEMM克隆形成率16/104。
(3)hUCB-MSCs:hUCB-MSCs制剂为无色或淡黄色液体,封装完好,放置于液氮保存。细胞悬液体积7 mL,基本指标:细胞形态正常、第3代、细胞数量7.00×107、细胞活性98.08%;微生物检测:真菌阴性、需氧菌阴性、支原体阴性、厌氧菌阴性,内毒素检测合格;细胞表型:CD34阳性率0.02%、CD45阳性率0.26%、CD90阳性率99.90%、CD44阳性率99.53%、CD73阳性率99.61%、CD105阳性率99.81%、HLA-DR阳性率0.21%;母血病毒检测:HBsAg阴性、Anti-HIVⅠ/Ⅱ阴性、Anti-HCV阴性、Anti-TP阴性、Anti-CMV-IgM阴性。
1.3.3 主要试剂与仪器 整合素αγβ3抗体(英国,abcam,ab7166);白血病抑制因子抗体(英国,abcam,ab138002);血小板-内皮细胞黏附分子1抗体(英国,abcam,ab9498);荧光二抗(英国,abcam,ab150079/ab150113);HRP标记二抗(中国,武汉三鹰,SA00001-2/SA00001-1);血管内皮生长因子抗体(美国,Thermo Fisher,MA5-13182);碱性成纤维细胞生长因子抗体(中国,华安生物,ER1904-22);角蛋白9抗体(中国,华安生物,ET1612-77);波形蛋白抗体(中国,武汉三鹰,60330-1-Ig);白细胞介素6(中国,正能生物,500286);白细胞介素1β(中国,正能生物,516288);肿瘤坏死因子α抗体(中国,正能生物,346654);ELISA试剂盒(中国,晶美生物,JM-01972R1/JM-01738R1/JM-01981R1/JM-01957R1/ JM-01626R1);DAPI溶液(中国,索莱宝,C0065);抗荧光衰减封片剂(中国,索莱宝,S2100);苏木精-伊红染色试剂盒(中国,索莱宝,G1120);Masson染色试剂盒(中国,索莱宝,G1340);激光共聚焦显微镜(德国,蔡司,LSM710型);生物安全柜(新加坡,ESCO,AC2-4S1);动物麻醉机(中国,瑞沃德生命,R650);低温超速离心机(美国,贝尔曼库尔特,Allegra X-64R);细胞计数仪(美国,DeNovix,CellDrop FL型);倒置显微镜(德国,徕卡,DMILLED型);电泳槽(美国,Bio Rad,Mini-PROTEAN Tetra);化学发光成像系统(美国,Bio Rad,ChemiDoc);酶标仪(美国,Thermo Fisher,Varioskan LUX)。
1.4 实验方法
1.4.1 hUCB-PRP的准备 将冻结的hUCB-PRP从-80 ℃取出,体积分数75%乙醇表面消毒后,放置在无菌冻容器中,置于4 ℃冰箱中缓慢解冻。缓慢解冻有助于减少冰晶对细胞和蛋白质的损伤。在解冻过程中,轻轻摇动容器,使内容物均匀受热,避免局部过热或过冷。确保hUCB-PRP完全解冻,没有残留冰块。解冻过程需要6-8 h。解冻后,将hUCB-PRP分装为0.5 mL(盛于1.5 mL EP管)置于4 ℃冷藏备用。
1.4.2 hUCB-MNCs的准备 将存放于液氮中的hUCB-MNCs取出,迅速转移至37 ℃恒温水浴箱内,边融化边晃动,注意不要让水面没过冻存管口,待细胞融化至冰水混合物状态时,迅速从水中取出。然后,将复苏后的细胞外包装用体积分数75%乙醇表面消毒后,移入生物安全柜。在15 mL离心管中提前加入复苏Buffer,用移液管吸取冻存管中的细胞悬液,转移至加有复苏Buffer的离心管中,补加Buffer,混匀后离心。预实验设置5×106 hUCB-MNCs/0.5 mL PBS、1×107 hUCB-MNCs/ 0.5 mL PBS、5×107 hUCB-MNCs/0.5 mL PBS 3个剂量梯度进行了再灌注细胞治疗的效果摸索,根据结果的分析统计,得出在 hUCB-MNCs剂量达到1×107/0.5 mL PBS前后治疗薄型子宫内膜效果最佳。故将细胞浓度调整为(1×107/0.5 mL PBS)用于宫角再灌注治疗备用。
1.4.3 hUCB-MSCs的准备 将存放于液氮中的hUCB-MSCs取出,迅速转移至37 ℃恒温水浴锅中,不断摇动使之融化,达到合适的融化体积即可取出,复苏过程中注意保持冻存管垂直放置,并避免水面没过管口。然后,用体积分数75%乙醇消毒冻存管表面后,移入生物安全柜,将细胞样本转至无菌离心管中,补加完全培养基,离心后弃上清,用经过滤器过滤的完全培养基调整细胞浓度至 1×107 L-1后接种于25 cm2培养瓶中,做好标记,置于培养箱中培养;培养24 h后更换培养基,以后每隔3 d换液1次,达到合适汇合度后进行传代操作。传到第5代,将细胞浓度调整为(1×107/0.5 mL PBS)用于宫角再灌注治疗备用[45]。
1.4.4 动物实验分组 选择60只动情周期规律的SD大鼠入组(有7只不正常的被舍弃),按照随机数字表随机分为5组:假手术组、模型组、hUCB-PRP组、hUCB-MSCs组和hUCB-MNCs组,每组12只,24个子宫。
1.4.5 模型制备、给药方法与剂量 采用阴道涂片染色连续观察大鼠的动情周期,对3个连续动情周期规律的大鼠进行造模。造模前大鼠禁食、禁饮6-8 h。大鼠经异氟醚诱导麻醉昏迷后,放于手术板上异氟醚维持麻醉,将大鼠固定、剃毛备皮、碘伏消毒,于耻骨联合上方2.0-2.5 cm处(尿道口上方约3 cm)纵行切开腹壁2 cm,沿腹白线逐步分离,找到并暴露子宫,分离出“V”型子宫后,在子宫周围垫无菌纱布,保护进针部位的液体渗漏,两侧子宫末端交界处用微血管夹夹闭,防止液体流出。用无齿镊将一侧子宫轻轻拉直,1 mL注射器于宫角处沿子宫长轴缓慢注射生理盐水或体积分数95%乙醇[46-53]。为保证宫腔持续充盈状态,先缓慢注入
0.2 mL,维持2 min后,继续宫腔注射0.3 mL,维持3 min,共维持5 min。维持后需抽出剩余未吸收液体,对于注射体积分数95%乙醇的大鼠,需换生理盐水冲洗宫腔2次。然后,根据分组缓慢维持再灌注PBS或脐血衍生物0.5 mL,见表1。另一侧子宫操作相同。注射完成后退出注射器针头,用医用胶迅速封闭针头穿刺孔,防止外渗。操作过程尽量不移动注射器针头,避免子宫穿孔。最后恢复子宫解剖结构,依次缝合关腹。术后注意保温,术后当日在大鼠饮用水中放入阿莫西林胶囊0.25 g,连续3 d,预防术后感染。严密观察大鼠行为有无异常。
1.4.6 体质量与动情周期的测定 自7-9周龄SD大鼠被引入动物房缓冲间后,从第2日起,直到大鼠连续3个动情周期保持规律为止,每日上午8:00-9:00称体质量。小棉签蘸取阴道分泌物涂抹在载玻片上,室温下自然干燥后以亚甲蓝溶液染色10-12 min,蒸馏水缓慢冲洗染液。光学显微镜下观察阴道脱落细胞,依据脱落细胞形态确定具体的动情周期时期。
1.4.7 脏器系数的测定 在宫角再灌注后第3个正常动情周期的动情间期,每组8只大鼠均用异氟烷麻醉后实施安乐死处置。随后,取出“V”型子宫组织、卵巢和胸腺、脾脏。使用万分之一天平称取各自脏器质量,普通天平称取大鼠质量并记录。计算脏器系数=(脏器的质量)/(大鼠总体质量)×100%。
1.4.8 石蜡切片与苏木精-伊红染色 取上述大鼠子宫组织,用小剪刀从阴道汇合处精准分割为两部分,每部分均分为两段,其中一段子宫组织(16个子宫半段,上下半段随机分配)固定于40 g/L多聚甲醛中24 h,经梯度乙醇脱水后,分别浸泡二甲苯、液态石蜡进行透化和包埋,用石蜡切片机将石蜡块切成5 μm的薄片,经脱蜡、水化、染色、分化、反蓝、透明和封固后,通过光学显微镜观察大鼠子宫组织病理形态改变。将染色完成的玻片进行全景扫描,使用CaseViewer软件计算出子宫内膜的厚度和子宫腺体、血管数量。
1.4.9 Masson染色 石蜡切片均脱蜡至水,滴加 Weigert 铁苏木精染色液染色8 min,洗净后酸性分化液分化10 s,蓝化液返蓝3 min,滴加丽春红品红染色液染色10 min,再滴加弱酸工作液洗30 s,磷钼酸溶液处理2 min,洗净,苯胺蓝染色液染色1 min,再次洗净,之后用体积分数95%乙醇快速脱水1次,无水乙醇脱水2次,二甲苯透明2次,最后中性树胶封固后显微镜下观察。用ZEN软件进行图像捕捉,Image J软件分析计算胶原容积分数,胶原容积分数=(胶原区域面积/总观察区域面积)×100%。
1.4.10 免疫组织化学染色 首先将石蜡切片脱蜡水化,然后按照枸橼酸钠修复→过氧化氢阻断→山羊血清封闭→波形蛋白抗体(1∶200)、角蛋白9抗体(1∶100)和白血病抑制因子抗体(1∶100)于4 ℃孵育过夜→二抗HRP山羊抗兔IgG(1∶5 000)和HRP山羊抗小鼠IgG(1∶5 000)室温孵育30 min→DAB显色→苏木精复染→脱水、透明、封固。通过光学显微镜观察染色情况,使用Image J软件计算相对吸光度值,相对吸光度值=目标区域的平均吸光度值/对照样本目标区域的平均吸光度值。
1.4.11 免疫荧光染色 大致步骤与免疫组织染色相同。整合素αγβ3抗体(1∶100)、血小板-内皮细胞黏附分子1抗体(1∶200)、血管内皮生长因子抗体(1∶100)和碱性成纤维细胞生长因子抗体(1∶200)于4 ℃孵育过夜→荧光二抗HRP山羊抗兔IgG[Alexa Fluor® 647](1∶500)和HRP山羊抗小鼠IgG[Alexa Fluor® 488](1∶500)避光孵育2 h→PBS清洗3次→DAPI染细胞核→抗荧光淬灭剂和中性树胶封固。在荧光显微镜下观察,使用Image J软件计算出各组相对荧光强度,相对荧光强度=目标区域的平均荧光强度/对照样本目标区域的平均荧光强度。
1.4.12 Western blot检测 精确称量子宫组织(剩余16个子宫半段),迅速放入预冷的玻璃匀浆器中,加入适量的组织蛋白裂解液,在冰上裂解30 min,4 ℃、12 000 r/min离心10 min,取上清液。根据BCA法测定蛋白浓度,计算出上样量,随后进行蛋白质电泳分离,将蛋白质转印至PVDF膜上,用封闭液封闭非特异性结合位点,4 ℃孵育一抗白细胞介素1β(1∶1 000)、肿瘤坏死因子α(1∶1 000)、白细胞介素6(1∶1 000)、β-actin(1∶5 000)过夜,次日TBST清洗3次后室温孵育二抗HRP山羊抗兔IgG(1∶5 000)和HRP山羊抗小鼠IgG(1∶5 000) 2 h,最后使用ECL化学发光试剂进行曝光。通过Image J软件对条带的灰度值进行分析,结果用蛋白相对表达量表示,蛋白相对表达量=蛋白灰度值/内参灰度值。
1.4.13 血清ELISA测定 大鼠在异氟烷维持麻醉过程中进行心脏取血,室温下静置30-60 min,2 500 r/min离心15 min后收集血清。按照ELISA试剂盒说明书操作检测抗缪勒氏管激素、促卵泡生成素、促黄体生成素、雌二醇、孕酮水平。
1.4.14 生育力测定 宫角灌注后第3个正常动情周期的动情间期,将每组4只雌性大鼠与性成熟的雄性大鼠按照1∶2的比例合笼。从次日开始,每日早晨检查雌性大鼠是否有阴栓,若有,则记为受孕第1天,并将雌雄大鼠分笼。待大鼠成功分娩幼崽后,统计幼崽数量及妊娠率。
1.5 主要观察指标 ①各组大鼠子宫、卵巢、脾脏和胸腺组织的脏器系数;②各组大鼠子宫内膜厚度与子宫腺体、血管的数量;③各组大鼠子宫内膜纤维化程度;④各组大鼠子宫内膜角蛋白9、波形蛋白表达;⑤各组大鼠子宫内膜整合素αγβ3、白血病抑制因子蛋白表达;⑥各组大鼠子宫内膜CD31、血管内皮生长因子、碱性成纤维细胞生长因子蛋白表达;⑦各组大鼠子宫内膜白细胞介素6、白细胞介素1β和肿瘤坏死因子α蛋白表达;⑧各组大鼠血清性激素与抗缪勒氏管激素水平。
1.6 统计学分析 采用IBM SPSS Statistics 26.0和GraphPad Prism 10.2.3软件进行统计学分析。在对计量资料进行集中趋势和离散程度描述时,首先需要进行正态性检验(Shapiro-Wilk检验)和方差齐性检验(Levene’s检验)以评估数据的分布特征。如果数据符合正态分布,则使用x±s来描述;如数据不符合正态分布,则使用中位数(四分位数间距)来描述。若数据符合正态分布并且各组方差齐性,则采用单因素ANOVA用于多个组间比较,当结果显示总体有显著差异时,则进一步进行“Tukey’s HSD”事后多重比较;若数据不符合正态分布或方差不齐,则采用非参数检验方法Kruskal-Wallis H检验。在进行多组计数资料间的比较时,当预期频数≥5时,采用卡方检验;当最小频数< 5时,则采用费舍尔精确检验(Fisher’s Exact Test)。P < 0.05为差异有显著性意义。该研究的统计方法已获得贵州医科大学统计学专家的审核。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
hUCB-PRP作为一种容易获得的脐血衍生物,一旦被激活,就会释放出多种具有再生特性的蛋白质、生长因子和生物分子[37],可从多方面有效修复受损子宫内膜。该研究观察到hUCB-PRP可以使子宫内膜增厚、促进血管生成、减少内膜纤维化、调节局部炎症、提高妊娠率等。这些观点与ZADEHMODARRES等[65-67]的观点保持一致,他们指出hUCB-PRP较其他PRP有着更强的促进血管生成作用[36,68-69],可以有效减弱免疫反应[70]。此外,对hUCB-PRP的研究报告也进一步证实了此观点,报告显示hUCB-PRP比异体外周PRP产生更高浓度的生长因子、趋化因子和细胞因子,并能通过增强血管内皮生长因子促进组织再生[71-74]。基于hUCB-MSCs的细胞治疗已应用于众多医学学科,大量用于再生和免疫调节目的[75-78],展现出显著的免疫抑制和抗炎能力[79-82],对此,妇科也不例外[83]。在该研究中发现hUCB-MSCs不仅能够促进生长因子、生物分子(增殖相关指标角蛋白9、波形蛋白,内膜容受性相关指标白血病抑制因子、整合素αγβ3,血管生成相关指标CD31、血管内皮生长因子、碱性成纤维细胞生长因子)、激素的合成和分泌,还可以通过旁分泌信号影响邻近细胞[84],共同在细胞增殖、内膜容受性、免疫调节和抗纤维化等方面发挥重要作用,从而促进损伤组织的再生。有证据表明,hUCB-MSCs在细胞治疗过程中主要通过激活整合素定植于损伤部位(归巢),并通过释放旁分泌因子(如细胞因子、生长因子和外泌体)参与细胞增殖和血管生成,调节免疫系统以控制炎症和促进组织再生[85]。这与此研究结果高度吻合,目前大量的临床试验也支持这种说法[86-87],且预后效果较好,暂未发现不良事件的报道。人外周血单核细胞促进小鼠子宫内膜细胞增殖,显示出了人外周血单核细胞诱导子宫内膜再生的潜力[88]。此外,也有研究指出自体骨髓单个核细胞移植到阿舍曼综合征(表现为子宫内膜再生障碍)患者子宫内膜,可以恢复子宫内膜再生[88]。而考虑到脐血是比成人骨髓或外周血更好的造血干细胞来源[89],该研究有理由认为hUCB-MNCs可以作为一种治疗薄型子宫内膜的潜能细胞。hUCB-MNCs作为一种由多种免疫细胞类型组成的脐血衍生物,不仅具有细胞成分的多样性,还具有较低的免疫排斥反应特性[90],该研究结果显示,hUCB-MNCs可以通过恢复子宫内膜形态、抗纤维化、促细胞增殖、诱导血管生成、抗炎、内膜容受性与免疫调节等对受损的子宫内膜进行修复。这与hUCB-MNCs治疗心脑血管疾病展现出的作用机制相一致[91-93]。
hUCB-PRP、hUCB-MSCs、hUCB-MNCs的制备过程具有不同的简便性、成本效益和效能。整体来看,hUCB-PRP的制备过程最为简便和经济,优点在于生物活性较高,能促进组织修复,广泛应用于伤口愈合和骨关节炎治疗[94-95]。同时,hUCB-PRP属于脐血中的脱细胞成分,表现出较低的免疫原性和致瘤性。除了长期使用更经济外[96],这些成分在微生物学上是安全的,并且避免了自体治疗带来的额外负担[59]。
此外,hUCB-PRP中的可溶性生长因子在长时间冷冻保存后保持稳定[97]。然而,hUCB-PRP的成分可能存在不均一性,影响疗效,未来的研究应该制定策略,减少潜在的问题 [98]。hUCB-MNCs包含多种类型的免疫细胞,具有较强的免疫调节能力,适合用于免疫治疗。尽管hUCB-MNCs制备简单且成本较低,无需激素预处理或其他步骤,可在1 d内完成提取[87],但目前没有合适的动物模型来评估细胞效力,加之成分复杂可能影响治疗效果,且数量有限;在安全性上,需确保制备和存储符合标准,需更多实验支撑,以降低潜在风险。相比之下,hUCB-MSCs的制备过程较为复杂和耗时,需要细胞培养和扩增,虽然制备技术要求高,但hUCB-MSCs具备多向分化潜力和免疫调节功能,已应用于众多医学学科。尽管如此,目前对于hUC-MSCs的安全性研究结果却呈现出多样性。关于它们的使用和长期安全性、高成本和有效性存在许多担忧[99],仍需要进一步的研究来评估hUCB-MSCs在临床环境中的疗效和长期安全性。
值得注意的是,多项研究将生物工程材料(例如水凝胶、支架或纳米材料)与人脐血衍生物结合[100],这些材料可以促进细胞黏附到受伤区域[101-102],提高保留时间和治疗效果[103-104],达到了增强脐血源性衍生物的再生效果[36]。因此,生物工程材料与人脐血衍生物或细胞的结合将在临床应用中展现出巨大的潜力。此外,免疫细胞可能是间充质干细胞促进组织修复的重要递质[105]。有研究证实巨噬细胞的耗竭使移植间充质干细胞的有益作用消失,揭示巨噬细胞可能是间充质干细胞诱导免疫调节的主要靶点[106]。因此,hUCB-PRP、hUCB-MSCs、hUCB-MNCs之间可能存在相互促进的协同效应,如果它们的协同作用得到证实,这将对子宫内膜策略的临床管理有着重要意义。综上所述,该研究初步探索了3种脐血衍生物在内膜增殖与容受性、血管生成、抗炎症和抗纤维化等方面的作用效果,并简要比较了它们治疗薄型子宫内膜的疗效。然而,该研究仍存在一定局限性,例如缺乏3种脐血衍生物促进子宫内膜修复的具体作用机制以及三者之间的交互作用。未来研究应致力于揭示具体的机制,探索组合疗法的潜力,并通过临床前和临床试验验证安全性和有效性,以期为薄型子宫内膜治疗提供更优方案。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
文题释义:#br#
人脐血衍生物:指从人脐血中提取的多种生物活性物质和细胞成分,主要包括单个核细胞、间充质干细胞和造血细胞,以及各种生长因子和抗炎/免疫调节因子,还有血液衍生因子富血小板血浆。这些衍生物展现出多样化且重要的生物学功能,如促进细胞增殖、分化、迁移和免疫调节等,在血液疾病治疗、再生医学、组织修复和免疫调节等领域具有广泛的应用前景。
薄型子宫内膜:是指在特定生理或病理情况下,子宫内膜厚度相对较薄,无法满足正常妊娠的必要条件,主要表现为细胞增殖不足和功能失衡,是导致女性月经异常、不孕症和反复流产的重要因素,能够影响子宫内膜的耐受性,进而降低胚胎的着床率和临床妊娠率,同时与辅助生殖治疗的预后质量下降相关。
#br#
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
根据世界卫生组织的统计,全球约有1.86亿人无法生育,占育龄人口的15%-20%。然而,妊娠的建立和维持在很大程度上取决于子宫内膜的功能。薄型子宫内膜通常对生长刺激无反应,是取消胚胎移植的重要指标。鉴于子宫内膜在育龄期女性和孕产妇健康及生殖中的重要作用,维持其生理结构、消除缺陷并在受伤后恢复其正常功能至关重要。本研究旨在探索和对比3种脐血衍生物(hUCB-PRP、hUCB-MSCs 和 hUCB-MNCs)治疗薄型子宫内膜的效果及对妊娠结局的影响,以期为薄型子宫内膜的临床应用提供更科学的依据。
#br#
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||