[1] BEJARANO L, JORDĀO M, JOYCE J. Therapeutic targeting of the tumor microenvironment. Cancer Discov. 2021;11(4):933-959.
[2] HUANG Y, HUANG J, WANG Y, et al. Progressive aggregation-induced emission strategy for imaging of aluminum ions in cellular microenvironment. Talanta. 2020;211:120699.
[3] STUDENT S, MILEWSKA M, OSTROWSKI Z, et al. Microchamber microfluidics combined with thermogellable glycomicrogels - Platform for single cells study in an artificial cellular microenvironment. Mater Sci Eng C Mater Biol Appl. 2021; 119:111647.
[4] HIRANO N, KUSUHARA H, SUEYOSHI Y, et al. Ethanol treatment of nanoPGA/PCL composite scaffolds enhances human chondrocyte development in the cellular microenvironment of tissue-engineered auricle constructs. PLoS One. 2021;16(7):e0253149.
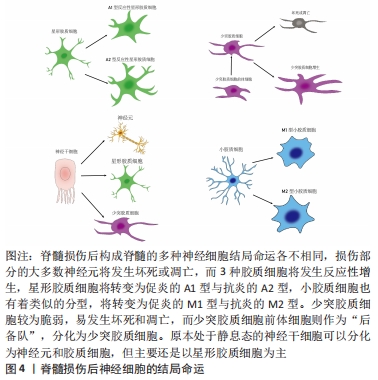
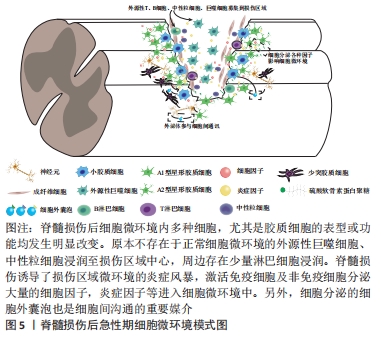
[5] ZHAO Y, XIAO Z, CHEN B, et al. The neuronal differentiation microenvironment is essential for spinal cord injury repair. Organogenesis. 2017;13(3):63-70.
[6] SHU J, CHENG F, GONG Z, et al. Transplantation strategies for spinal cord injury based on microenvironment modulation. Curr Stem Cell Res Ther. 2020;15(6):522-530.
[7] TRAN A, WARREN P, SILVER J. The biology of regeneration failure and success after spinal cord injury. Physiol Rev. 2018;98(2):881-917.
[8] LIDDELOW S, BARRES B. SnapShot: astrocytes in health and disease. Cell. 2015; 162(5):1170-1170.e1.
[9] KETTENMANN H, HANISCH UK, NODA M, et al. Physiology of microglia. Physiol Rev. 2011;91(2):461-553.
[10] MELETIS K, BARNABE-HEIDER F, CARLEN M, et al. Spinal cord injury reveals multilineage differentiation of ependymal cells. PLoS Biol. 2008;6(7):e182.
[11] BONNANS C, CHOU J, WERB Z. Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol. 2014;15(12):786-801.
[12] JAUDON F, CHIACCHIARETTA M, ALBINI M, et al. Kidins220/ARMS controls astrocyte calcium signaling and neuron-astrocyte communication. Cell Death Differ. 2020;27(5):1505-1519.
[13] KITCHEN P, SALMAN MM, HALSEY AM, et al. Targeting aquaporin-4 subcellular localization to treat central nervous system edema. Cell. 2020;181(4):784-799.e19.
[14] ZAMANIAN JL, XU L, FOO LC, et al. Genomic analysis of reactive astrogliosis. J Neurosci. 2012;32(18):6391-6410.
[15] ANDERSON MA, BURDA JE, REN Y, et al. Astrocyte scar formation aids central nervous system axon regeneration. Nature. 2016;532(7598):195-200.
[16] LI Y, HE X, KAWAGUCHI R, et al. Microglia-organized scar-free spinal cord repair in neonatal mice. Nature. 2020;587(7835):613-618.
[17] ZRZAVY T, SCHWAIGER C, WIMMER I, et al. Acute and non-resolving inflammation associate with oxidative injury after human spinal cord injury. Brain. 2021;144(1): 144-161.
[18] WANG C, XU T, LACHANCE BB, et al. Critical roles of sphingosine kinase 1 in the regulation of neuroinflammation and neuronal injury after spinal cord injury. J Neuroinflammation. 2021;18(1):50.
[19] BELLVER-LANDETE V, BRETHEAU F, MAILHOT B, et al. Microglia are an essential component of the neuroprotective scar that forms after spinal cord injury. Nat Commun. 2019;10(1):518.
[20] MCTIGUE DM, WEI P, STOKES BT. Proliferation of NG2-positive cells and altered oligodendrocyte numbers in the contused rat spinal cord. J Neurosci. 2001;21(10):3392-3400.
[21] GIACCI MK, BARTLETT CA, SMITH NM, et al. Oligodendroglia are particularly vulnerable to oxidative damage after neurotrauma in vivo. J Neurosci. 2018; 38(29):6491-6504.
[22] ASSINCK P, DUNCAN GJ, PLEMEL JR, et al. Myelinogenic Plasticity of Oligodendrocyte Precursor Cells following Spinal Cord Contusion Injury. J Neurosci. 2017;37(36): 8635-8654.
[23] LIDDELOW SA, GUTTENPLAN KA, CLARKE LE, et al. Neurotoxic reactive astrocytes are induced by activated microglia. Nature. 2017;541(7638):481-487.
[24] SONN I, NAKAMURA M, RENAULT-MIHARA F, et al. Polarization of reactive astrocytes in response to spinal cord injury is enhanced by M2 macrophage-mediated activation of wnt/beta-catenin pathway. Mol Neurobiol. 2020;57(4): 1847-1862.
[25] WENZEL TJ, BAJWA E, KLEGERIS A. Cytochrome c can be released into extracellular space and modulate functions of human astrocytes in a toll-like receptor 4-dependent manner. Biochim Biophys Acta Gen Subj. 2019;1863(11):129400.
[26] KIM S, SON Y. Astrocytes stimulate microglial proliferation and m2 polarization in vitro through crosstalk between astrocytes and microglia. Int J Mol Sci. 2021; 22(16):8800.
[27] TANG Y, LIU L, XU D, et al. Interaction between astrocytic colony stimulating factor and its receptor on microglia mediates central sensitization and behavioral hypersensitivity in chronic post ischemic pain model. Brain Behav Immun. 2018; 68:248-260.
[28] TRAN AP, SUNDAR S, YU M, et al. Modulation of receptor protein tyrosine phosphatase sigma increases chondroitin sulfate proteoglycan degradation through cathepsin b secretion to enhance axon outgrowth. J Neurosci. 2018;38(23):5399-5414.
[29] MOORE CS, MILNER R, NISHIYAMA A, et al. Astrocytic tissue inhibitor of metalloproteinase-1 (TIMP-1) promotes oligodendrocyte differentiation and enhances CNS myelination. J Neurosci. 2011;31(16):6247-6254.
[30] DING Z, DAI C, ZHONG L, et al. Neuregulin-1 converts reactive astrocytes toward oligodendrocyte lineage cells via upregulating the PI3K-AKT-mTOR pathway to repair spinal cord injury. Biomed Pharmacother. 2021;134:111168.
[31] ZUO H, WOOD WM, SHERAFAT A, et al. Age-dependent decline in fate switch from ng2 cells to astrocytes after olig2 deletion. J Neurosci. 2018;38(9):2359-2371.
[32] MIRON VE, BOYD A, ZHAO JW, et al. M2 microglia and macrophages drive oligodendrocyte differentiation during CNS remyelination. Nat Neurosci. 2013; 16(9):1211-1218.
[33] SAKAKIBARA S, NAKADATE K, OOKAWARA S, et al. Non-cell autonomous impairment of oligodendrocyte differentiation precedes CNS degeneration in the Zitter rat: implications of macrophage/microglial activation in the pathogenesis. BMC Neurosci. 2008;9:35.
[34] SAS AR, CARBAJAL KS, JEROME AD, et al. A new neutrophil subset promotes CNS neuron survival and axon regeneration. Nat Immunol. 2020;21(12):1496-505.
[35] KUCHAROVA K, STALLCUP WB. NG2-proteoglycan-dependent contributions of oligodendrocyte progenitors and myeloid cells to myelin damage and repair. J Neuroinflammation. 2015;12:161.
[36] MARTIñóN S, GARCíA-VENCES E, TOSCANO-TEJEIDA D, et al. Long-term production of BDNF and NT-3 induced by A91-immunization after spinal cord injury. BMC Neurosci. 2016;17(1):42.
[37] TIAN L, RAUVALA H, GAHMBERG CG. Neuronal regulation of immune responses in the central nervous system. Trends Immunol. 2009;30(2):91-99.
[38] LEHRMAN EK, WILTON DK, LITVINA EY, et al. CD47 protects synapses from excess microglia-mediated pruning during development. Neuron. 2018;100(1):120-134.e6.
[39] SAINI V, LOERS G, KAUR G, et al. Impact of neural cell adhesion molecule deletion on regeneration after mouse spinal cord injury. Eur J Neurosci. 2016;44(1):1734-1746.
[40] GAUTIER HO, EVANS KA, VOLBRACHT K, et al. Neuronal activity regulates remyelination via glutamate signalling to oligodendrocyte progenitors. Nat Commun. 2015;6:8518.
[41] CIZKOVA D, NAGYOVA M, SLOVINSKA L, et al. Response of ependymal progenitors to spinal cord injury or enhanced physical activity in adult rat. Cell Mol Neurobiol. 2009;29(6-7):999-1013.
[42] LACROIX S, HAMILTON LK, VAUGEOIS A, et al. Central canal ependymal cells proliferate extensively in response to traumatic spinal cord injury but not demyelinating lesions. PLoS One. 2014;9(1):e85916.
[43] SALEWSKI RP, MITCHELL RA, LI L, et al. Transplantation of induced pluripotent stem cell-derived neural stem cells mediate functional recovery following thoracic spinal cord injury through remyelination of axons. Stem Cells Transl Med. 2015;4(7):743-754.
[44] WU G, SHI H, CHE M, et al. Recovery of paralyzed limb motor function in canine with complete spinal cord injury following implantation of MSC-derived neural network tissue. Biomaterials. 2018;181:15-34.
[45] JONES I, NOVIKOVA L, WIBERG M, et al. Human embryonic stem cell-derived neural crest cells promote sprouting and motor recovery following spinal cord injury in adult rats. Cell transplantation. 2021;30:963689720988245.
[46] LIU X, HAO M, CHEN Z, et al. 3D bioprinted neural tissue constructs for spinal cord injury repair. Biomaterials. 2021;272:120771.
[47] COFANO F, BOIDO M, MONTICELLI M, et al. Mesenchymal stem cells for spinal cord injury: current options, limitations, and future of cell therapy. Int J Mol Sci. 2019;20(11):2689.
[48] BARTANUSZ V, JEZOVA D, ALAJAJIAN B, et al. The blood-spinal cord barrier: morphology and clinical implications. Ann Neurol. 2011;70(2):194-206.
[49] DAVID S, LACROIX S. Molecular approaches to spinal cord repair. Annu Rev Neurosci. 2003;26:411-440.
[50] TSATA V, MOLLMERT S, SCHWEITZER C, et al. A switch in pdgfrb(+) cell-derived ECM composition prevents inhibitory scarring and promotes axon regeneration in the zebrafish spinal cord. Dev Cell. 2021;56(4):509-524.e9.
[51] TOM VJ, DOLLER CM, MALOUF AT, et al. Astrocyte-associated fibronectin is critical for axonal regeneration in adult white matter. J Neurosci. 2004;24(42):9282-9290.
[52] LI X, LIU X, JOSEY B, et al. Short laminin peptide for improved neural stem cell growth. Stem Cells Transl Med. 2014;3(5):662-670.
[53] WISEMAN TM, BARON-HEERIS D, HOUWERS IGJ, et al. Peptide hydrogel scaffold for mesenchymal precursor cells implanted to injured adult rat spinal cord. Tissue Eng Part A. 2021;27(15-16):993-1007.
[54] YANG Y, FAN Y, ZHANG H, et al. Small molecules combined with collagen hydrogel direct neurogenesis and migration of neural stem cells after spinal cord injury. Biomaterials. 2021;269:120479.
[55] THOMPSON C, ZURKO J, HANNA B, et al. The therapeutic role of interleukin-10 after spinal cord injury. J Neurotrauma. 2013;30(15):1311-1324.
[56] YATES A, JOGIA T, GILLESPIE E, et al. Acute IL-1RA treatment suppresses the peripheral and central inflammatory response to spinal cord injury. J Neuroinflamm. 2021;18(1):15.
[57] SUN F, ZHANG H, HUANG T, et al. miRNA-221 Regulates spinal cord injury-induced inflammatory response through targeting tnf-α expression. Biomed Res Int. 2021; 2021:6687963.
[58] LEIBINGER M, ZEITLER C, GOBRECHT P, et al. Transneuronal delivery of hyper-interleukin-6 enables functional recovery after severe spinal cord injury in mice. Nat Commun. 2021;12(1):391.
[59] EL ANDALOUSSI S, MAGER I, BREAKEFIELD XO, et al. Extracellular vesicles: biology and emerging therapeutic opportunities. Nat Rev Drug Discov. 2013;12(5):347-357.
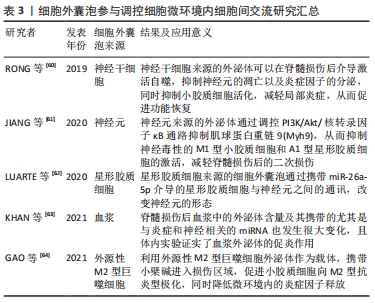
[60] RONG Y, LIU W, WANG J, et al. Neural stem cell-derived small extracellular vesicles attenuate apoptosis and neuroinflammation after traumatic spinal cord injury by activating autophagy. Cell Death Dis. 2019;10(5):340.
[61] JIANG D, GONG F, GE X, et al. Neuron-derived exosomes-transmitted miR-124-3p protect traumatically injured spinal cord by suppressing the activation of neurotoxic microglia and astrocytes. J Nanobiotechnol. 2020;18(1):105.
[62] LUARTE A, HENZI R, FERNANDEZ A, et al. Astrocyte-derived small extracellular vesicles regulate dendritic complexity through mir-26a-5p activity. Cells. 2020; 9(4):930.
[63] KHAN N, CAO T, HE J, et al. Spinal cord injury alters microRNA and CD81+ exosome levels in plasma extracellular nanoparticles with neuroinflammatory potential. Brain Behav Immun. 2021;92:165-183.
[64] GAO Z, ZHANG C, XIA N, et al. Berberine-loaded M2 macrophage-derived exosomes for spinal cord injury therapy. Acta Biomateri. 2021;126:211-223.
|