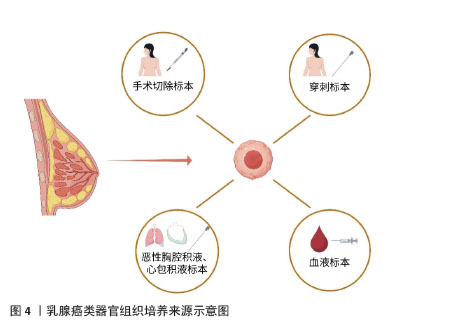
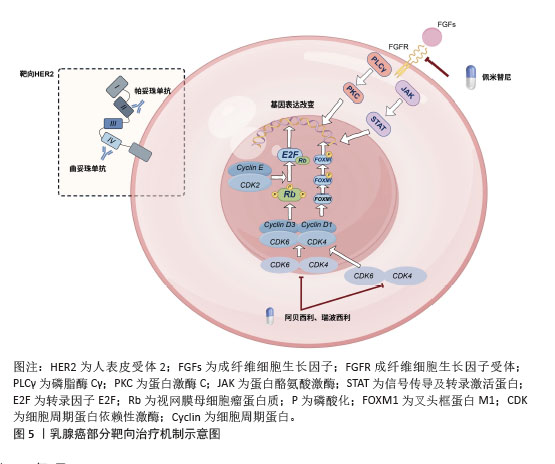
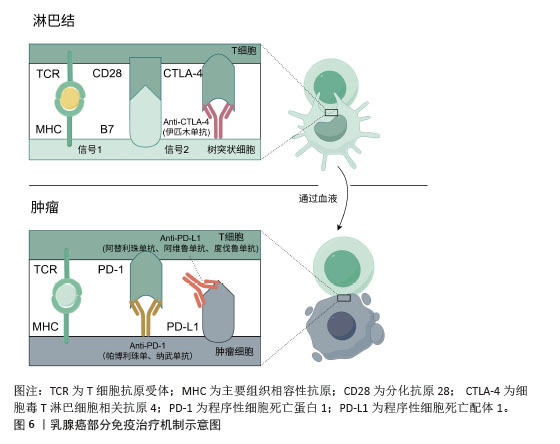
[1] GIAQUINTO AN, SUNG H, MILLER KD, et al. Breast Cancer Statistics,2022. CA Cancer J Clin. 2022;72(6):524-541.
[2] BODAI BI, TUSO P. Breast cancer survivorship: a comprehensive review of long-term medical issues and lifestyle recommendations. Perm J. 2015;19(2):48-79.
[3] BAE SY, KIM S, LEE JH, et al. Poor prognosis of single hormone receptor- positive breast cancer: similar outcome as triple-negative breast cancer.BMC Cancer. 2015;15:138.
[4] 刘继伟,刘伟慈,毛文君.肿瘤类器官在精准肺癌药物筛选中的研发与进展[J].中国组织工程研究,2024,28(31):5078-5084.
[5] DESANTIS CE, MA J, GAUDET MM, et al.Breast cancer statistics,2019. CA Cancer J Clin. 2019; 69(6):438-451.
[6] ROPER J, TAMMELA T, CETINBAS NM, et al.In vivo genome editing and organoid transplantation models of colorectal cancer and metastasis. Nat Biotechnol. 2017;35(6):569-576.
[7] BYRNE AT, ALFÉREZ DG, AMANT F, et al.Interrogating open issues in cancer precision medicine with patient-derived xenografts. Nat Rev Cancer. 2017;17(4):254-268.
[8] SACHS N, DE LIGT J, KOPPER O, et al. A Living Biobank of Breast Cancer Organoids Captures Disease Heterogeneity. Cell. 2018;172(1-2): 373-386.e10.
[9] LEE SH, HU W, MATULAY JT, et al. Tumor Evolution and Drug Response in Patient-Derived Organoid Models of Bladder Cancer. Cell. 2018;173(2): 515-528.e17.
[10] HIDALGO M, AMANT F, BIANKIN AV, et al. Patient-derived xenograft models: an emerging platform for translational cancer research. Cancer Discov. 2014;4(9):998-1013.
[11] YE F, DEWANJEE S, LI Y, et al. Advancements in clinical aspects of targeted therapy and immunotherapy in breast cancer. Mol Cancer. 2023;22(1):105.
[12] GOLDHAMMER N, KIM J, TIMMERMANS-WIELENGA V, et al. Characterization of organoid cultured human breast cancer. Breast Cancer Res. 2019;21(1):141.
[13] CHEN P, ZHANG X, DING R, et al. Patient-Derived Organoids Can Guide Personalized-Therapies for Patients with Advanced Breast Cancer. Adv Sci (Weinh). 2021;8(22):e2101176.
[14] PRANAV P, PALANIYANDI T, BASKAR G, et al. Gene expressions and their significance in organoid cultures obtained from breast cancer patient-derived biopsies. Acta Histochem. 2022; 124(5):151910.
[15] AREF AR, CAMPISI M, IVANOVA E, et al. 3D microfluidic ex vivo culture of organotypic tumor spheroids to model immune checkpoint blockade.Lab Chip. 2018;18(20):3129-3143.
[16] HERNANDEZ-GORDILLO V, KASSIS T, LAMPEJO A, et al. Fully synthetic matrices for in vitro culture of primary human intestinal enteroids and endometrial organoids. Biomaterials. 2020; 254:120125.
[17] PEROU CM, SØRLIE T, EISEN MB, et al. Molecular portraits of human breast tumours. Nature. 2000; 406(6797):747-752.
[18] SLAMON D, LIPATOV O, NOWECKI Z, et al. Ribociclib plus Endocrine Therapy in Early Breast Cancer. N Engl J Med. 2024;390(12):1080-1091.
[19] OLIVEIRA M, POMINCHUK D, NOWECKI Z, et al. Camizestrant, a next-generation oral SERD, versus fulvestrant in post-menopausal women with oestrogen receptor-positive, HER2-negative advanced breast cancer (SERENA-2): a multi-dose, open-label, randomised, phase 2 trial. Lancet Oncol. 2024;25(11):1424-1439.
[20] BIANCHINI G, DE ANGELIS C, LICATA L, et al.Treatment landscape of triple-negative breast cancer - expanded options, evolving needs. Nat Rev Clin Oncol. 2022;19(2):91-113.
[21] JIANG YZ, MA D, SUO C, et al. Genomic and Transcriptomic Landscape of Triple-Negative Breast Cancers: Subtypes and Treatment Strategies. Cancer Cell. 2019;35(3):428-440.e5.
[22] SCHMID P, CORTES J, PUSZTAI L, et al. Pembrolizumab for Early Triple-Negative Breast Cancer. N Engl J Med. 2020;382(9):810-821.
[23] WU H, WANG W, ZHANG Y, et al. Establishment of patient-derived organoids for guiding personalized therapies in breast cancer patients. Int J Cancer. 2024;155(2):324-338.
[24] LIU J, FANG F, YUAN W, et al.Integrating Circulating Tumor DNA Dynamics and Sequential Organoid Drug Screening Enhances Breast Cancer Treatment Efficacy. Cancer Res. 2025;24.doi:10.1158/0008-5472.CAN-25-0608
[25] MAYER EL, BURSTEIN HJ. Chemotherapy for Triple-Negative Breast Cancer: Is More Better?. J Clin Oncol. 2016;34(28):3369-3371.
[26] LIEDTKE C, MAZOUNI C,HESS KR,et al.Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008;26(8):1275-1281.
[27] PSILOPATIS I, MANTZARI A, VRETTOU K, et al. The Role of Patient-Derived Organoids in Triple-Negative Breast Cancer Drug Screening. Biomedicines. 2023;11(3):773.
[28] SAATCI O, KAYMAK A, RAZA U, et al. Targeting lysyl oxidase (LOX) overcomes chemotherapy resistance in triple negative breast cancer. Nat Commun. 2020;11(1):2416.
[29] LIU XY, JIANG W, MA D, et al. SYTL4 downregulates microtubule stability and confers paclitaxel resistance in triple-negative breast cancer.Theranostics. 2020;10(24):10940-10956.
[30] SARI AIP, ROYTRAKUL S, CHITTAVANICH P, et al. Drug repurposing identifies proteasome inhibitors as antiproliferative agents counteracting inflammation-driven chemoresistance in triple-negative breast cancer organoids. Biomed Pharmacother. 2025;190:118359.
[31] WANG X, CHEN T, LI C, et al. CircRNA-CREIT inhibits stress granule assembly and overcomes doxorubicin resistance in TNBC by destabilizing PKR. J Hematol Oncol. 2022;15(1):122.
[32] CRONIN KA, HARLAN LC, DODD KW, et al. Population-based estimate of the prevalence of HER-2 positive breast cancer tumors for early stage patients in the US. Cancer Invest. 2010; 28(9):963-968.
[33] 黄慧琦,伍秋苑,张昆,等.川楝素联合奥拉帕尼在三阴性乳腺癌中的抗肿瘤机制研究[J].天津医药,2025,53(9):897-903.
[34] NAVARRO-YEPES J, KETTNER NM, RAO X, et al. Abemaciclib Is Effective in Palbociclib-Resistant Hormone Receptor-Positive Metastatic Breast Cancers. Cancer Res. 2023;83(19):3264-3283.
[35] TURNER NC, LIU Y, ZHU Z, et al. Cyclin E1 Expression and Palbociclib Efficacy in Previously Treated Hormone Receptor-Positive Metastatic Breast Cancer. J Clin Oncol. 2019;37(14):1169-1178.
[36] KETTNER NM, VIJAYARAGHAVAN S, DURAK MG, et al. Combined Inhibition of STAT3 and DNA Repair in Palbociclib-Resistant ER-Positive Breast Cancer. Clin Cancer Res. 2019;25(13):3996-4013.
[37] BELLI S, ESPOSITO D, ASCIONE CM, et al. EGFR and HER2 hyper-activation mediates resistance to endocrine therapy and CDK4/6 inhibitors in ER+ breast cancer. Cancer Lett. 2024;593:216968.
[38] HILLIS AL, MARTIN TD, MANCHESTER HE, et al. Targeting Cholesterol Biosynthesis with Statins Synergizes with AKT Inhibitors in Triple-Negative Breast Cancer. Cancer Res. 2024;84(19): 3250-3266.
[39] LI YW, DAI LJ, WU XR, et al. Molecular Characterization and Classification of HER2-Positive Breast Cancer Inform Tailored Therapeutic Strategies. Cancer Res. 2024;84(21):3669-3683.
[40] CUI Y, RAN R, DA Y, et al. The combination of breast cancer PDO and mini-PDX platform for drug screening and individualized treatment. J Cell Mol Med. 2024;28(9):e18374.
[41] TAGHIZADEH-TABARSI R, AKBARI-BIRGANI S, AMJADI M, et al. Aptamer-guided graphene oxide quantum dots for targeted suicide gene therapy in an organoid model of luminal breast cancer. Sci Rep. 2024;14(1):24104.
[42] MADORSKY ROWDO FP, XIAO G, KHRAMTSOVA GF, et al. Patient-derived tumor organoids with p53 mutations, and not wild-type p53, are sensitive to synergistic combination PARP inhibitor treatment. Cancer Lett. 2024;584:216608.
[43] LIU Z, SHI Y, LV L, et al. Small Molecular Inhibitors Reverse Cancer Metastasis by Blockading Oncogenic PITPNM3. Adv Sci (Weinh). 2022;9(35): e2204649.
[44] XU Q, JIANG Z, PAN Y, et al.Cucurbitacin B stimulates PD-1 immunotherapy response in malignant breast cancer by covalent targeting MTCH2. Phytomedicine. 2025;145:157017.
[45] YUKI K, CHENG N, NAKANO M, et al. Organoid Models of Tumor Immunology. Trends Immunol. 2020;41(8):652-664.
[46] DIVOUX J, FLORENT R, JACOBS M, et al. The TRIPLEX study: use of patient-derived tumor organoids as an innovative tool for precision medicine in triple-negative breast cancer. BMC Cancer. 2023;23(1):883.
[47] DING JH, XIAO Y, YANG F, et al. Guanosine diphosphate-mannose suppresses homologous recombination repair and potentiates antitumor immunity in triple-negative breast cancer. Sci Transl Med. 2024;16(728):eadg7740.
[48] HUANG L, RONG Y, TANG X, et al. Engineered exosomes as an in situ DC-primed vaccine to boost antitumor immunity in breast cancer. Mol Cancer. 2022;21(1):45.
[49] LIN YZ, LIU CH, WU WR, et al. Memory-promoting function of miR-379-5p attenuates CD8+ T cell exhaustion by targeting immune checkpoints. J Immunother Cancer. 2025;13(4):e010363.
[50] DING JH, XIAO Y, YANG F, et al. Guanosine diphosphate-mannose suppresses homologous recombination repair and potentiates antitumor immunity in triple-negative breast cancer. Sci Transl Med. 2024;16(728):eadg7740.
[51] LI X, PAN B, SONG X, et al. Breast cancer organoids from a patient with giant papillary carcinoma as a high-fidelity model. Cancer Cell Int. 2020;20:86.
[52] SEGATTO I, MATTEVI MC, RAMPIONI VINCIGUERRA GL, et al.A comprehensive luminal breast cancer patient-derived xenografts (PDX) library to capture tumor heterogeneity and explore the mechanisms of resistance to CDK4/6 inhibitors. J Pathol. 2024;264(4):434-447.
[53] 谭景云,兰坚强,白跃宗,等.2024年度肿瘤类器官研究进展[J].肿瘤综合治疗电子杂志, 2025,11(2):77-84.
[54] 中国抗癌协会肿瘤多学科诊疗专业委员会,中国抗癌协会肿瘤内分泌专业委员会,赵冰,等.肿瘤类器官诊治平台的质量控制标准中国专家共识(2022年版)[J].中国癌症杂志,2022, 32(7):657-668.
[55] MAZZUCCHELLI S, PICCOTTI F, ALLEVI R, et al. Establishment and Morphological Characterization of Patient-Derived Organoids from Breast Cancer. Biol Proced Online. 2019;21:12.
[56] MOU J, LI C, ZHENG Q, et al. Research progress in tumor angiogenesis and drug resistance in breast cancer. Cancer Biol Med. 2024;21(7):571-585.
[57] DEKKERS JF, VAN VLIET EJ, SACHS N, et al. Long-term culture, genetic manipulation and xenotransplantation of human normal and breast cancer organoids. Nat Protoc. 2021;16(4):1936-1965.
[58] MAULANA TI, TEUFEL C, CIPRIANO M, et al. Breast cancer-on-chip for patient-specific efficacy and safety testing of CAR-T cells. Cell Stem Cell. 2024;31(7):989-1002.e9.
|