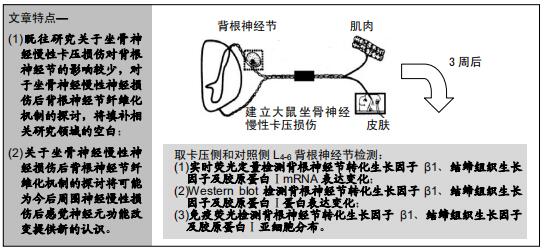
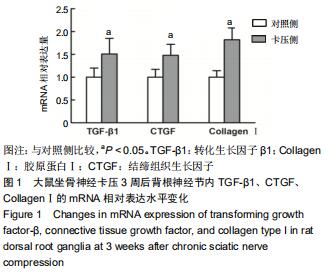
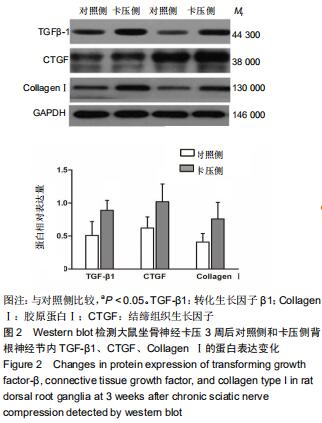
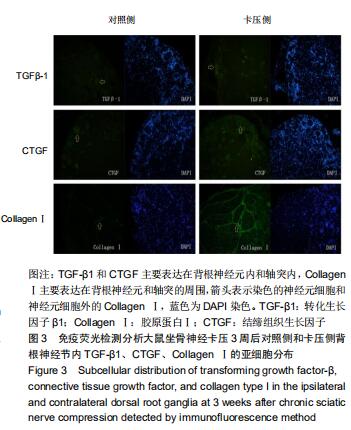
|
[1] WAHAB KW, SANYA EO, ADEBAYO PB, et al. Carpal tunnel syndrome and other entrapment neuropathies. Oman Med J. 2017;32(6):449-454.
[2] PHAM K, GUPTA R. Understanding the mechanisms of entrapment neuropathies. Review article. Neurosurg Focus. 2009;26(2):E7.
[3] MACKINNON SE, Pathophysiology of nerve compression. Hand Clinics. 2002;18(2):231-241.
[4] LIU ZY, CHEN ZB, CHEN JH. A novel chronic nerve compression model in the rat.Neural Regen Res. 2018;13(8): 1477-1485.
[5] FANBIN M, JIANGHAI C, JUAN L, et al. Role of transforming growth factor-beta1 in the process of fibrosis of denervated skeletal muscle. J Huazhong Univ Sci Technolog Med Sci. 2011;31(1):77-82.
[6] CHAO T, PHAM K, STEWARD O, et al. Chronic nerve compression injury induces a phenotypic switch of neurons within the dorsal root ganglia. J Comp Neurol. 2008;506(2): 180-93.
[7] 张烽,顾玉东,徐建光.大鼠坐骨神经损伤后神经元细胞凋亡的初步观察[J].中华手外科杂志,2000,16(1):56-58.
[8] HUANG ZJ, LI HC, COWAN AA, et al. Chronic compression or acute dissociation of dorsal root ganglion induces cAMP-dependent neuronal hyperexcitability through activation of PAR2. Pain. 2012;153(7):1426-1437.
[9] TOMASSONI D, DI CESAVE MANNELLI L, BRAMANTI V, et al. Acetyl-L-carnitine exerts a neuroprotective effect on sciatic nerve axon and myelin following chronic constriction injury. Neural Regen Res .2018;13(4):692-698.
[10] O'BRIEN JP, MACKINNON SE, MACLEAN AR, et al. A model of chronic nerve compression in the rat. Ann Plast Surg. 1987; 19(5):430-435.
[11] SEYER JM, KANG AH, WHITAKER JN. The characterization of type I and type III collagens from human peripheral nerve. Biochim Biophys Acta. 1977;492(2):415-425.
[12] BONNANS C, CHOU J, WERB Z. Remodelling the extracellular matrix in development and disease. Nat Rev Mol Cell Biol. 2014;15(12):786-801.
[13] LEVADA K, OMELYANCHIK A, RODIONOVA V, et al. Magnetic- assisted treatment of liver fibrosis. Cells. 2019;8(10):1279.
[14] DISTLER JHW, GYÖRFI AH, RAMANUJAM M, et al. Shared and distinct mechanisms of fibrosis. Nat Rev Rheumatol. 2019;15(12):705-730.
[15] EL AGHA E, KRAMANN R, SCHNEIDER RK, et al. Mesenchymal stem cells in fibrotic disease. Cell Stem Cell. 2017;21(2):166-177.
[16] MAJO J, KLINKHAMMER BM, BOOR P, et al. Pathology and natural history of organ fibrosis. Curr Opin Pharmacol. 2019; 49:82-89.
[17] GARDET A, ZHENG TS, VINEY JT. Genetic architecture of human fibrotic diseases: disease risk and disease progression. Front Pharmaco. 2013;4:159.
[18] SUN X, NKENNOR B, MASTIKHINA O, et al. Endothelium-mediated contributions to fibrosis. Semin Cell Dev Biol. 2019. doi: 10.1016/j.semcdb.2019.10.015.
[19] HINTERMANN E, CHRISTEN U. The many roles of cell adhesion molecules in hepatic fibrosis. Cells. 2019;8(12): 1503.
[20] BAGALAD BS, MOHAN KUMAR KP, PUNEETH HK. Myofibroblasts: master of disguise. J Oral Maxillofac Pathol. 2017;21(3):462-463.
[21] MANCINI ML, SONIS ST. Mechanisms of cellular fibrosis associated with cancer regimen-related toxicities. Front Pharmacol. 2014;5:51.
[22] CHEN C, WANG YY, WANG YX, et al. Gentiopicroside ameliorates bleomycin-induced pulmonary fibrosis in mice via inhibiting inflammatory and fibrotic process. Biochem Biophys Res Commun. 2018;495(4):2396-2403.
[23] GIMÉNEZ A, DUCH P, PUIG M, et al. Dysregulated Collagen Homeostasis by Matrix Stiffening and TGF-beta1 in fibroblasts from idiopathic pulmonary fibrosis patients: role of FAK/Akt. Int J Mol Sci. 2017;18(11):2431.
[24] MENG XM, NIKOLIC-PATERSON DJ, LAN HY. TGF-β: the master regulator of fibrosis. Nat Rev Nephrol. 2016;12(6): 325-338.
[25] MA TT, MENG XM. TGF-beta/Smad and Renal Fibrosis. Adv Exp Med Biol. 2019;1165:347-364.
[26] HU HH, CHEN DQ, WANG YN, et al. New insights into TGF-β/Smad signaling in tissue fibrosis. Chem Biol Interact. 2018;292:76-83.
[27] FEHRHOLZ M, SPEER CP, KUNZMANN S. Caffeine and rolipram affect Smad signalling and TGF-β1 stimulated CTGF and transgelin expression in lung epithelial cells. PLoS One. 2014;9(5):e97357.
[28] SUN YW, ZHANG YY, KE XJ, et al. Pirfenidone prevents radiation-induced intestinal fibrosis in rats by inhibiting fibroblast proliferation and differentiation and suppressing the TGF-beta1/Smad/CTGF signaling pathway. Eur J Pharmacol. 2018;822:199-206.
[29] GUO J, LIN Q, SHAO Y, et al. miR-29b promotes skin wound healing and reduces excessive scar formation by inhibition of the TGF-β1/Smad/CTGF signaling pathway. Can J Physiol Pharmacol. 2017;95(4):437-442.
[30] GRESSNER OA, GRESSNER AM. Connective tissue growth factor: a fibrogenic master switch in fibrotic liver diseases. Liver Int. 2008;28(8):1065-1079.
[31] 罗涛,李力燕.外周神经系统中背根神经节的研究进展[J].解剖科学进展,2015,21(5):543-546.
[32] GUÉNARD V, GWYNN LA, WOOD PM. Astrocytes inhibit Schwann cell proliferation and myelination of dorsal root ganglion neurons in vitro. J Neurosci. 1994;14(5):2980-2992.
[33] ZHANG YL, CHEN DJ, YANG BL, et al. Microencapsulated Schwann cell transplantation inhibits P2X3 receptor expression in dorsal root ganglia and neuropathic pain.Neural Regen Res. 2018;13(11):1961-1967.
[34] STARK B, CARLSTEDT T, RISLING M. Distribution of TGF-β, the TGF-β type I receptor and the R-II receptor in peripheral nerves and mechanoreceptors; observations on changes after traumatic injury. Brain Res. 2001;913(1):47-56.
[35] DELRÉE P, RIBBENS C, MARTIN D, et al. Plasticity of developing and adult dorsal root ganglion neurons as revealed in vitro. Brain Res Bull. 1993;30(3-4):231.
[36] SUN L, XIU M, WANG S, et al. Lipopolysaccharide enhances TGF-beta1 signalling pathway and rat pancreatic fibrosis. J Cell Mol Med. 2018;22(4):2346-2356.
[37] MEWHORT HE, LIPON BD, SVYSTONYUK DA, et al. Monocytes increase human cardiac myofibroblast-mediated extracellular matrix remodeling through TGF-beta1. Am J Physiol Heart Circ Physiol. 2016;310(6):H716-H724.
[38] IHN H. Pathogenesis of fibrosis: role of TGF-β and CTGF. Curr Opin Rheumatol. 2002;14(6):681-685.
[39] SONNYLAL S, XU S, JONES H, et al. Connective tissue growth factor causes EMT-like cell fate changes in vivo and in vitro. J Cell Sci. 2013;126(Pt 10):2164-2175.
[40] SLAWIK J, ADRIAN L, HOHL M, et al. Irregular pacing of ventricular cardiomyocytes induces pro-fibrotic signalling involving paracrine effects of transforming growth factor beta and connective tissue growth factor. Eur J Heart Fail. 2019; 21(4):482-491.
[41] ABREU JG, KETPURA NI, REVERSADE B, et al. Connective-tissue growth factor (CTGF) modulates cell signalling by BMP and TGF-beta. Nat Cell Biol. 2002;4(8): 599-604.
[42] KAASBØLL OJ, GADICHERLA AK, WANG JH, et al. Connective tissue growth factor (CCN2) is a matricellular preproprotein controlled by proteolytic activation. J Biol Chem. 2018;293(46):17953-17970.
|