中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (28): 7465-7474.doi: 10.12307/2026.815
• 组织工程相关大数据分析 Big data analysis in tissue engineering • 上一篇 下一篇
特发性肺纤维化发病机制及潜在治疗靶点:大规模全基因组关联研究数据分析
范志梁1,柴艺汇1,陈光磊1,李 倩1,顾春松2,陈云志1,李 文1,吴大梅1,蒲 翔1
- 1贵州中医药大学基础中医学院,贵州省贵阳市 550025;2贵州中医药大学第二附属医院,贵州省贵阳市 550003
-
收稿日期:2025-08-01修回日期:2025-12-11出版日期:2026-10-08发布日期:2026-02-26 -
通讯作者:蒲翔,博士,教授,贵州中医药大学基础中医学院,贵州省贵阳市 550025 -
作者简介:第一作者:范志梁,男,1997年生,河南省郑州市人,汉族,贵州中医药大学在读博士,主要从事中药民族药药理物质基础研究。 共同第一作者:柴艺汇,女,1985年生,山西省临汾市人,汉族,2024年菲律宾永恒大学毕业,博士,高级实验师,主要从事中药民族药药理研究。 -
基金资助:贵州省科技计划项目(黔科合基础-ZK[2022]一般456),项目负责人:柴艺汇;国家自然科学基金项目(82060913),
项目负责人:蒲翔;贵州省苗医药全省重点实验室(黔科合平台[2025]018),项目负责人:蒲翔;贵州中医药大学校内科研项目
(贵中医科余[2025]48号),项目负责人:蒲翔
Pathogenesis and potential therapeutic targets of idiopathic pulmonary fibrosis: analysis of data from a large-scale genome-wide association study
Fan Zhiliang1, Chai Yihui1, Chen Guanglei1, Li Qian1, Gu Chunsong2, Chen Yunzhi1, Li Wen1, Wu Damei1, Pu Xiang1
- 1School of Basic Chinese Medicine, Guizhou University of Traditional Chinese Medicine, Guiyang 550025, Guizhou Province, China; 2Second Affiliated Hospital, Guizhou University of Traditional Chinese Medicine, Guiyang 550003, Guizhou Province, China
-
Received:2025-08-01Revised:2025-12-11Online:2026-10-08Published:2026-02-26 -
Contact:Pu Xiang, MD, Professor, School of Basic Chinese Medicine, Guizhou University of Traditional Chinese Medicine, Guiyang 550025, Guizhou Province, China -
About author:Fan Zhiliang, MD candidate, School of Basic Chinese Medicine, Guizhou University of Traditional Chinese Medicine, Guiyang 550025, Guizhou Province, China Chai Yihui, MD, Senior experimentalist, School of Basic Chinese Medicine, Guizhou University of Traditional Chinese Medicine, Guiyang 550025, Guizhou Province, China Fan Zhiliang and Chai Yihui contributed equally to this article. -
Supported by:Guizhou Provincial Science and Technology Program, No. ZK[2022]456 (to CYH); National Natural Science Foundation of China, No. 82060913 (to PX); Guizhou Provincial Key Laboratory of Miao Medicine, No. [2025]018 (to PX); Scientific Research Project of Guizhou University of Traditional Chinese Medicine, No. [2025]48 (to PX)
摘要:
文题释义:
特发性肺纤维化:是一种病因不明的间质性肺疾病,主要特征为细胞外间质胶原沉积和肺功能下降,最终导致呼吸衰竭和死亡。
肠-肺轴:是指肠道菌群通过免疫调节、炎症因子释放及代谢物运输等机制影响肺部健康的一种双向生理联系,在多种肺部疾病中发挥重要作用。
多组学研究:是整合基因组、转录组、代谢组、微生物组等多种生物学数据类型,以系统方式揭示复杂疾病发生机制的一种研究策略。
背景:“肠-肺”轴在多种肺部疾病的发展中扮演重要角色,但在特发性肺纤维化方面缺乏深入探讨,因果关系尚不明确。为深入探讨肠道菌群对特发性肺纤维化的调控机制,该研究引入全基因组关联研究、表达数量性状位点分析以及共定位分子对接技术,旨在从遗传表型、转录调控与药靶分析等多个层面,系统评估肠道菌群通过免疫细胞、炎症因子及代谢物影响特发性肺纤维化的潜在机制,有望进一步阐明特发性肺纤维化的发病机制。
目的:探讨肠道菌群与特发性肺纤维化之间的因果关系,并阐述免疫细胞、炎症蛋白和血液代谢物的中介作用,筛选关键肠道菌群及潜在靶基因,同时预测潜在靶向药物,为特发性肺纤维化的早期诊断与新药开发提供思路。
方法:应用公开发表的肠道菌群的全基因组关联分析汇总统计数据以及芬兰数据库、eQTLGen等全基因组关联研究的公开数据库,分析了473种肠道菌群物种、731种免疫细胞类型、91种炎症蛋白、233种代谢物及特发性肺纤维化数据。通过单变量孟德尔随机化分析,应用逆方差加权法及敏感性分析,探索肠道菌群与特发性肺纤维化的因果关系。采用两步中介孟德尔随机化分析评估免疫细胞、炎症蛋白和代谢物是否在肠道菌群-特发性肺纤维化关系中起中介作用。在此基础上,进一步利用孟德尔随机化与汇总数据的孟德尔随机化方法探讨肠道菌群与基因表达之间的因果关系,并通过共定位分析与可成药性预测筛选潜在药物候选分子,结合分子对接验证。
结果与结论:Bacteroides faecis、Megasphaera和Pandoraea的丰度与特发性肺纤维化存在因果关系。其中Bacteroides faecis通过18∶2亚油酸比率及多个CD4+ T细胞亚群介导特发性肺纤维化风险;Pandoraea的风险同样受到不同的CD4+ T细胞亚群影响;Megasphaera的保护作用则主要通过白细胞介素33、低密度脂蛋白相关代谢物和CD4-CD8- T细胞亚群发挥作用。进一步研究确定了GNF-Pf-2272、5155877和PpIX是KDM4C、CBR3和YWHAG的潜在靶向药物。尽管此研究所用的肺纤维化全基因组关联研究数据主要来源于欧洲人群,但考虑到人类遗传背景在多个核心通路上存在共性,研究结果对于探索通过调控肠道菌群改善中国人群肺纤维化风险仍具有重要的参考价值。
https://orcid.org/0000-0003-3566-5363(蒲翔)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
范志梁, 柴艺汇, 陈光磊, 李 倩, 顾春松, 陈云志, 李 文, 吴大梅, 蒲 翔. 特发性肺纤维化发病机制及潜在治疗靶点:大规模全基因组关联研究数据分析[J]. 中国组织工程研究, 2026, 30(28): 7465-7474.
Fan Zhiliang, Chai Yihui, Chen Guanglei, Li Qian, Gu Chunsong, Chen Yunzhi, Li Wen, Wu Damei, Pu Xiang. Pathogenesis and potential therapeutic targets of idiopathic pulmonary fibrosis: analysis of data from a large-scale genome-wide association study[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7465-7474.
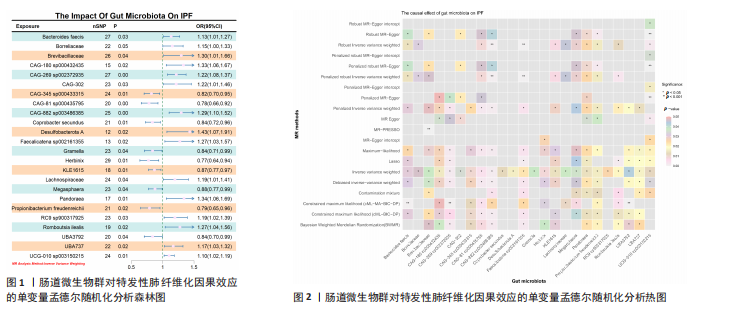
2.2 特发性肺纤维化对肠道微生物群的因果效应 通过逆方差加权法初步筛选出11种与特发性肺纤维化具有反向因果关系的肠道微生物群(图3,4),随后采用了20种不同的方法来排除异常值。经严格筛选有2种显著肠道微生物群被认为对特发性肺纤维化具有显著的反向因果效应,但与2.1结果无重叠,提示先前鉴定的3种肠道微生物群不存在与特发性肺纤维化的反向因果关系。
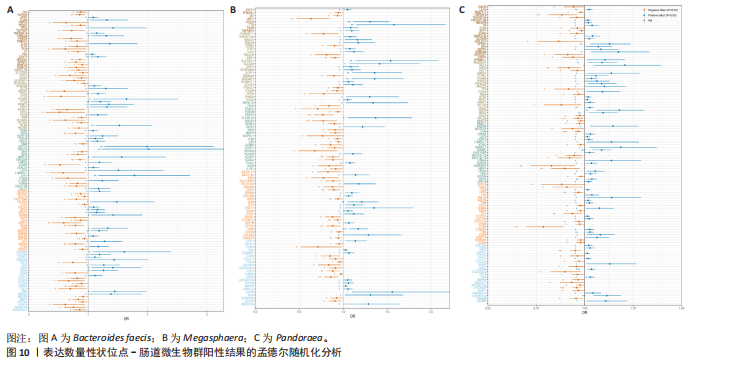
2.3 肠道微生物群对免疫细胞、炎症蛋白及血清代谢物的因果效应 为进一步探究肠道微生物群与特发性肺纤维化因果关系中可能存在的机制,该研究引入了233种代谢物、731种免疫细胞和91种炎症蛋白进行单变量孟德尔随机化分析。结果显示,Bacteroides faecis、Megasphaera、Pandoraea与以下生物标志物存在因果关系:代谢物方面包括18∶2亚油酸/总脂肪酸比值、中等粒径低密度脂蛋白中的游离胆固醇与磷脂;免疫细胞方面涉及包括CD4在多种调节性T细胞亚群中的表达水平、中央记忆CD4?CD8? T细胞的比例;炎症蛋白方面则以白细胞介素33为主。相关性分析显示,Bacteroides faecis与亚油酸比值及调节性T细胞中CD4表达均呈负相关;而Pandoraea在免疫细胞表达方面亦显示出负相关趋势;Megasphaera与白细胞介素33、低密度脂蛋白相关代谢物呈负相关,与中央记忆T细胞呈正相关。这些结果提示肠道微生物群可能通过调控代谢途径和免疫炎症通路影响特发性肺纤维化的发展(图5-8)。
2.4 免疫细胞、炎症蛋白及血清代谢物的中介效应 在该研究中,中央记忆CD4?CD8? T细胞占CD4?CD8? T细胞的比率(10.49%)以及中等粒径低密度脂蛋白中的游离胆固醇(1.26%)被鉴定为Megasphaera影响特发性肺纤维化风险的介导因子。
而Bacteroides faecis的中介结果显示CD4在CD28+ CD4+ T细胞(13.49%)的表达水平、CD4在CD4 Tregs(13.10%)的表达水平、CD4在活化的CD4 Tregs (11.53%)的表达水平、CD4在活化并分泌的CD4 Tregs (8.49%)的表达水平以及18∶2亚油酸/总脂肪酸比率(10.99%)均
介导了其对特发性肺纤维化风险的预测。介导效应比率均超过8%,提示T细胞和脂肪酸可能是Bacteroides faecis影响特发性肺纤维化的关键介导因素。值得注意的是,这4种免疫细胞均与CD4 T细胞相关。此外,CD4在CD4 Tregs (10.09%)的表达水平、CD4在活化的CD4 Tregs (7.00%)的表达水平、CD4在活化并分泌的CD4 Tregs (6.82%)的表达水平介导了Pandoraea对特发性肺纤维化风险的影响。综合考虑结果认为CD4 T细胞在Bacteroides faecis、Megasphaera、Pandoraea与特发性肺纤维化的因果关系中起到了介导作用(表1)。
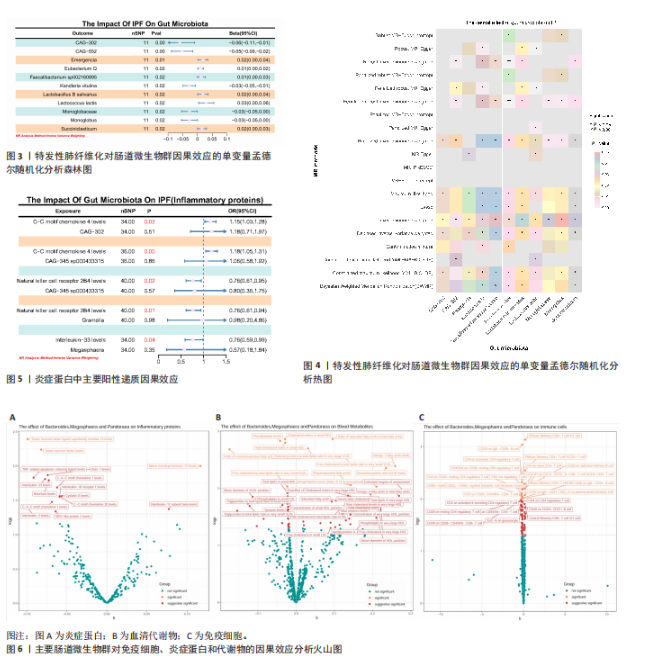
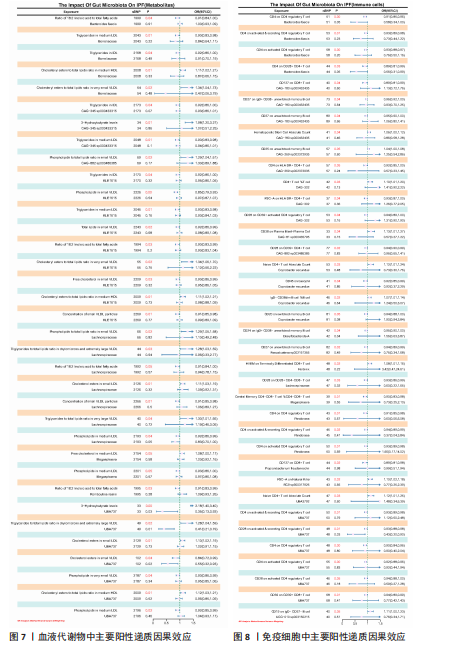
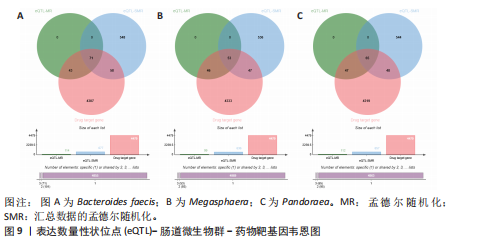
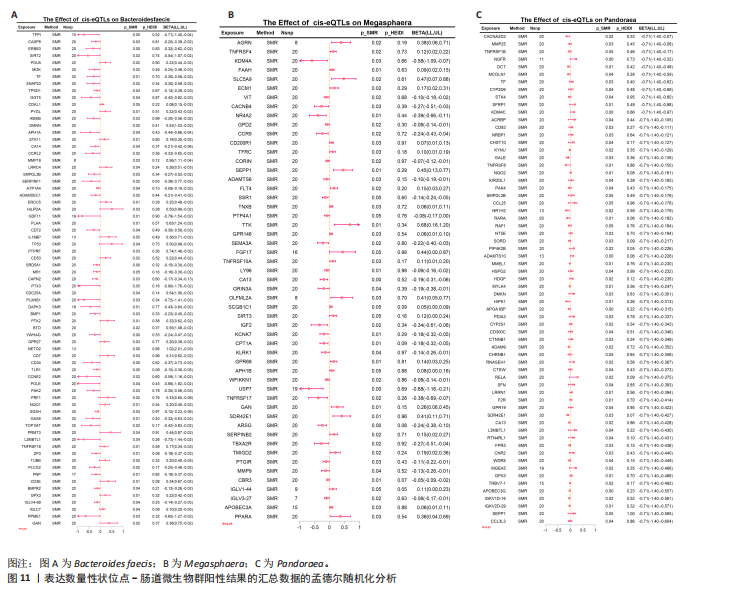
2.5 孟德尔随机化-汇总数据的孟德尔随机化-药物靶点交集分析及贝叶斯共定位分析 基于孟德尔随机化-汇总数据的孟德尔随机化的药靶分析研究结果发现,与Bacteroides faecis相关的基因表达产生了114个显著的孟德尔随机化结果,汇总数据的孟德尔随机化分析则识别出677个显著结果。将这些结果与4 479个药物靶点基因交集后,筛选出71个关键基因。对于Megasphaera,孟德尔随机化分析识别出99个显著结果,汇总数据的孟德尔随机化分析获得636个显著结果,交集后筛选出53个关键基因。对于Pandoraea,孟德尔随机化分析识别出112个显著结果,汇总数据的孟德尔随机化分析识别出657个显著结果,交集后筛选出65个关键基因(图9-11)。
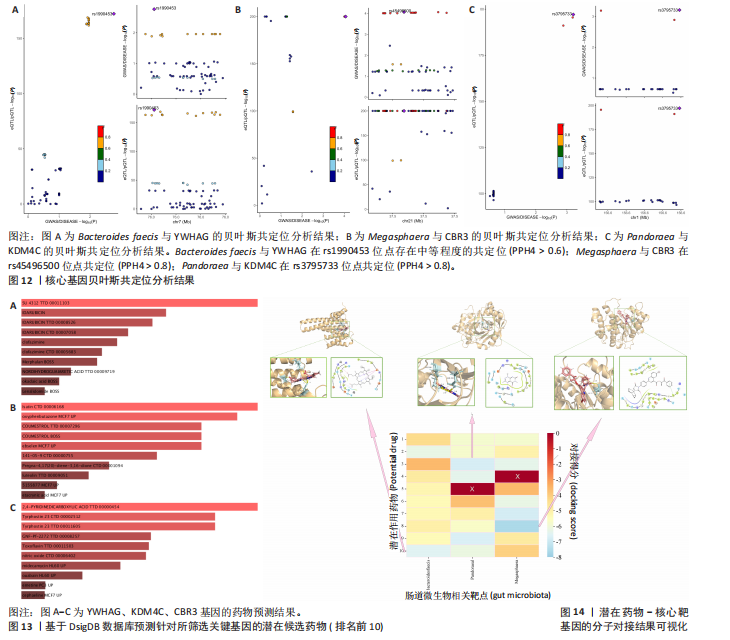
贝叶斯共定位分析结果表明,Bacteroides faecis与YWHAG在rs1990453位点存在中等程度的共定位(PPH4 > 0.6);Megasphaera与CBR3在rs45496500位点共定位(PPH4 > 0.8);Pandoraea与KDM4C在rs3795733位点共定位(PPH4 > 0.8)(图12)。
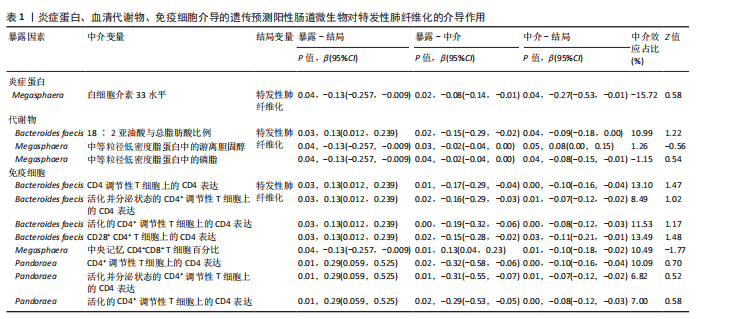
2.6 治疗靶点及药物预测 评估药物-蛋白互作是判断基因能否作为潜在治疗靶点的重要途径,该研究借助DSigDB数据库预测了针对所筛选关键基因的潜在候选药物(预测结果见图13),对排名前10的组合进行了分子对接和可视化分析,结果提示大多数对接组合显示出良好的对接得分。基于分子对接结果推测YWHAG的潜在靶向药物为protoporphyrin IX,KDM4C的潜在靶向药物为GNF-Pf-2272,CBR3的潜在靶向药物为6-溴-2-[4-(3,5-二氧亚基-4-氮杂四环[5.3.2.08,10.02,6]十二-11-烯-4-基)苯基]喹啉-4-甲酸-1-氧亚基-1-苯基丙-2-基酯(5155877)(图14)。
| [1] GLASPOLE I, BONELLA F, BARGAGLI E, et al. Efficacy and safety of nintedanib in patients with idiopathic pulmonary fibrosis who are elderly or have comorbidities. Respir Res. 2021;22(1):125. [2] 程雪,荆焕熙,张运克,等.肺痹方干预肺纤维化小鼠肺泡上皮细胞线粒体途径凋亡的机制[J].中国组织工程研究,2025,29(11):2334-2339. [3] CANI PD. Human gut microbiome: hopes, threats and promises. Gut. 2018;67(9):1716-1725. [4] ROOKS MG, GARRETT WS. Gut microbiota, metabolites and host immunity. Nat Rev Immunol. 2016;16(6):341-352. [5] CLARKE TB. Early innate immunity to bacterial infection in the lung is regulated systemically by the commensal microbiota via nod-like receptor ligands. Infect Immun. 2014;82(11):4596-4606. [6] YANG D, CHEN X, WANG J, et al. Dysregulated Lung Commensal Bacteria Drive Interleukin-17B Production to Promote Pulmonary Fibrosis through Their Outer Membrane Vesicles. Immunity. 2019;50(3):692-706.e7. [7] KLOEPFER KM, LEE WM, PAPPAS TE, et al. Detection of pathogenic bacteria during rhinovirus infection is associated with increased respiratory symptoms and asthma exacerbations. J Allergy Clin Immunol. 2014;133(5):1301-1307. [8] VASAKOVA M, STRIZ I, SLAVCEV A, et al. Th1/Th2 cytokine gene polymorphisms in patients with idiopathic pulmonary fibrosis. Tissue Antigens. 2006;67(3):229-232. [9] MUTSAERS SE, MILES T, PRÊLE CM, et al. Emerging role of immune cells as drivers of pulmonary fibrosis. Pharmacol Ther. 2023;252:108562. [10] ROQUE W, ROMERO F. Cellular metabolomics of pulmonary fibrosis, from amino acids to lipids. Am J Physiol Cell Physiol. 2021;320(5):C689-C695. [11] YAMADA T, NAKASHIMA T, MASUDA T, et al. Intestinal overgrowth of Candida albicans exacerbates bleomycin-induced pulmonary fibrosis in mice with dysbiosis. J Pathol. 2023;261(2):227-237. [12] 殷月,冷思逸,靳攀,等.肠道菌群、运动干预与呼吸系统疾病[J].中国组织工程研究,2025, 29(14):3034-3042. [13] VÕSA U, CLARINGBOULD A, WESTRA HJ, et al. Large-scale cis- and trans-eQTL analyses identify thousands of genetic loci and polygenic scores that regulate blood gene expression. Nat Genet. 2021;53(9):1300-1310. [14] FINAN C, GAULTON A, KRUGER FA, et al. The druggable genome and support for target identification and validation in drug development. Sci Transl Med. 2017;9(383):eaag1166. [15] QIN Y, HAVULINNA AS, LIU Y, et al. Combined effects of host genetics and diet on human gut microbiota and incident disease in a single population cohort. Nat Genet. 2022;54(2):134-142. [16] ORRÙ V, STERI M, SIDORE C, et al. Complex genetic signatures in immune cells underlie autoimmunity and inform therapy. Nat Genet. 2020;52(10): 1036-1045. [17] ZHAO JH, STACEY D, ERIKSSON N, et al. Genetics of circulating inflammatory proteins identifies drivers of immune-mediated disease risk and therapeutic targets. Nat Immunol. 2023;24(9):1540-1551. [18] KARJALAINEN MK, KARTHIKEYAN S, OLIVER-WILLIAMS C, et al. Genome-wide characterization of circulating metabolic biomarkers. Nature. 2024;628(8006):130-138. [19] ZHAO G, CAI Y, WANG Y, et al. Genetically predicted blood metabolites mediate the association between circulating immune cells and pancreatic cancer: A Mendelian randomization study. J Gene Med. 2024;26(5):e3691. [20] GONG Y, WANG J, PAN M, et al. Harmine inhibits pulmonary fibrosis through regulating DNA damage repair-related genes and activation of TP53-Gadd45α pathway. Int Immunopharmacol. 2024;138:112542. [21] QI T, WU Y, ZENG J, et al. Identifying gene targets for brain-related traits using transcriptomic and methylomic data from blood. Nat Commun. 2018; 9(1):2282. [22] ZHU Z, ZHANG F, HU H, et al. Integration of summary data from GWAS and eQTL studies predicts complex trait gene targets. Nat Genet. 2016;48(5):481-487. [23] GIAMBARTOLOMEI C, VUKCEVIC D, SCHADT EE, et al. Bayesian test for colocalisation between pairs of genetic association studies using summary statistics. PLoS Genet. 2014;10(5):e1004383. [24] SI S, LIU H, XU L, et al. Identification of novel therapeutic targets for chronic kidney disease and kidney function by integrating multi-omics proteome with transcriptome. Genome Med. 2024;16(1):84. [25] YOO M, SHIN J, KIM J, et al. DSigDB: drug signatures database for gene set analysis. Bioinformatics. 2015;31(18):3069-3071. [26] KULESHOV MV, JONES MR, ROUILLARD AD, et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 2016;44(W1):W90-97. [27] WONGKARNJANA A, YANAGIHARA T, KOLB MR. Treatment of idiopathic pulmonary fibrosis with Nintedanib: an update. Expert Rev Respir Med. 2019;13(12):1139-1146. [28] 陈雯,陈丽萍.肺部微生物群在肺纤维化疾病中的研究进展[J].中国当代医药,2023,30(12):44-49. [29] TANG J, XU L, ZENG Y, et al. Effect of gut microbiota on LPS-induced acute lung injury by regulating the TLR4/NF-kB signaling pathway. Int Immunopharmacol. 2021;91:107272. [30] HILTY M, BURKE C, PEDRO H, et al. Disordered microbial communities in asthmatic airways. PLoS One. 2010;5(1):e8578. [31] SEELIGER B, CARLEO A, WENDEL-GARCIA PD, et al. Changes in serum metabolomics in idiopathic pulmonary fibrosis and effect of approved antifibrotic medication. Front Pharmacol. 2022; 13:837680. [32] MOHEBALI N, WEIGEL M, HAIN T, et al. Faecalibacterium prausnitzii, Bacteroides faecis and Roseburia intestinalis attenuate clinical symptoms of experimental colitis by regulating Treg/Th17 cell balance and intestinal barrier integrity. Biomed Pharmacother. 2023;167:115568. [33] NARAYANA JK, ALIBERTI S, MAC AOGÁIN M, et al. Microbial Dysregulation of the Gut-Lung Axis in Bronchiectasis. Am J Respir Crit Care Med. 2023;207(7):908-920. [34] DICKSON RP, SINGER BH, NEWSTEAD MW, et al. Enrichment of the lung microbiome with gut bacteria in sepsis and the acute respiratory distress syndrome. Nat Microbiol. 2016;1(10):16113. [35] LU X, XIONG L, ZHENG X, et al. Structure of gut microbiota and characteristics of fecal metabolites in patients with lung cancer. Front Cell Infect Microbiol. 2023;13:1170326. [36] WANG Z, LIU J, LI F, et al. Mechanisms of Qingyi Decoction in Severe Acute Pancreatitis-Associated Acute Lung Injury via Gut Microbiota: Targeting the Short-Chain Fatty Acids-Mediated AMPK/NF-κB/NLRP3 Pathway. Microbiol Spectr. 2023;11(4):e0366422. [37] ZHANG M, SHEN L, ZHOU X, et al. The Microbiota of Human Lung of Pulmonary Tuberculosis and the Alteration Caused by Anti-tuberculosis Drugs. Curr Microbiol. 2022;79(11):321. [38] WU X, LI RF, LIN ZS, et al. Coinfection with influenza virus and non-typeable Haemophilus influenzae aggregates inflammatory lung injury and alters gut microbiota in COPD mice. Front Microbiol. 2023;14:1137369. [39] FARSI Y, TAHVILDARI A, ARBABI M, et al. Diagnostic, Prognostic, and Therapeutic Roles of Gut Microbiota in COVID-19: A Comprehensive Systematic Review. Front Cell Infect Microbiol. 2022;12:804644. [40] CHAU J, YADAV M, LIU B, et al. Prospective correlation between the patient microbiome with response to and development of immune-mediated adverse effects to immunotherapy in lung cancer. BMC Cancer. 2021;21(1):808. [41] LU HF, LI A, ZHANG T, et al. Disordered oropharyngeal microbial communities in H7N9 patients with or without secondary bacterial lung infection. Emerg Microbes Infect. 2017;6(12):e112. [42] MA S, ZHANG F, ZHOU F, et al. Metagenomic analysis reveals oropharyngeal microbiota alterations in patients with COVID-19. Signal Transduct Target Ther. 2021;6(1):191. [43] LU KY, ALQADERI H, BIN HASAN S, et al. Sputum production and salivary microbiome in COVID-19 patients reveals oral-lung axis. PLoS One. 2024; 19(7):e0300408. [44] DRUMMOND MH. The base sequence of the nifF gene of Klebsiella pneumoniae and homology of the predicted amino acid sequence of its protein product to other flavodoxins. Biochem J. 1985;232(3):891-896. [45] GREEN H, JONES AM. The microbiome and emerging pathogens in cystic fibrosis and non-cystic fibrosis bronchiectasis. Semin Respir Crit Care Med. 2015;36(2):225-235. [46] DEGAND N, LOTTE R, DECONDÉ LE BUTOR C, et al. Epidemic spread of Pandoraea pulmonicola in a cystic fibrosis center. BMC Infect Dis. 2015;15:583. [47] COSTELLO A, HERBERT G, FABUNMI L, et al. Virulence of an emerging respiratory pathogen, genus Pandoraea, in vivo and its interactions with lung epithelial cells. J Med Microbiol. 2011;60(Pt 3):289-299. [48] MA Z, ZOU X, LIN J, et al. Sepsis Due to Pandoraea sputorum Infection After Multiple Trauma in a Non-Cystic Fibrosis Patient: A Case Report from Southeast China. Infect Drug Resist. 2022;15:7043-7052. [49] KOLAHIAN S, FERNANDEZ IE, EICKELBERG O, et al. Immune Mechanisms in Pulmonary Fibrosis. Am J Respir Cell Mol Biol. 2016;55(3):309-322. [50] HEUKELS P, MOOR CC, VON DER THÜSEN JH, et al. Inflammation and immunity in IPF pathogenesis and treatment. Respir Med. 2019;147:79-91. [51] SHI X, PAN Z, CAI W, et al. Identification and immunological characterization of cuproptosis-related molecular clusters in idiopathic pulmonary fibrosis disease. Front Immunol. 2023;14:1171445. [52] CELADA LJ, KROPSKI JA, HERAZO-MAYA JD, et al. PD-1 up-regulation on CD4+ T cells promotes pulmonary fibrosis through STAT3-mediated IL-17A and TGF-β1 production. Sci Transl Med. 2018; 10(460):eaar8356. [53] GILANI SR, VUGA LJ, LINDELL KO, et al. CD28 down-regulation on circulating CD4 T-cells is associated with poor prognoses of patients with idiopathic pulmonary fibrosis. PLoS One. 2010;5(1):e8959. [54] BARDE F, LORENZON R, VICAUT E, et al. Induction of regulatory T cells and efficacy of low-dose interleukin-2 in systemic sclerosis: interventional open-label phase 1-phase 2a study. RMD Open. 2024;10(2):e003500. [55] KOTSIANIDIS I, NAKOU E, BOUCHLIOU I, et al. Global impairment of CD4+CD25+FOXP3+ regulatory T cells in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2009;179(12):1121-1130. [56] BIRJANDI SZ, PALCHEVSKIY V, XUE YY, et al. CD4(+)CD25(hi)Foxp3(+) Cells Exacerbate Bleomycin-Induced Pulmonary Fibrosis. Am J Pathol. 2016; 186(8):2008-2020. [57] ANDUGULAPATI SB, GOURISHETTI K, TIRUNAVALLI SK, et al. Biochanin-A ameliorates pulmonary fibrosis by suppressing the TGF-β mediated EMT, myofibroblasts differentiation and collagen deposition in in vitro and in vivo systems. Phytomedicine. 2020;78:153298. [58] LI ZY, XIAO L, LIN G, et al. Contribution of tissue transglutaminase to the severity of hepatic fibrosis resulting from Schistosoma japonicum infection through the regulation of IL-33/ST2 expression. Parasit Vectors. 2019;12(1):302. [59] WANG QR, LIU SS, MIN JL, et al. CCL17 drives fibroblast activation in the progression of pulmonary fibrosis by enhancing the TGF-β/Smad signaling. Biochem Pharmacol. 2023;210:115475. [60] 王璐,徐婷贞,周林水,等.M2型巨噬细胞通过CCL22-CCR4信号促进肺泡上皮细胞EMT[J].中国病理生理杂志,2022,38(10):1848-1855. [61] 汪泽婷,蒋钰玉,王晓慧,等.巨噬细胞在肺纤维化中的作用研究进展[J].中国免疫学杂志, 2025,41(3):513-521. [62] CHANDA D, OTOUPALOVA E, SMITH SR, et al. Developmental pathways in the pathogenesis of lung fibrosis. Mol Aspects Med. 2019;65:56-69. [63] SHI X, CHEN Y, LIU Q, et al. LDLR dysfunction induces LDL accumulation and promotes pulmonary fibrosis. Clin Transl Med. 2022;12(1):e711. [64] CHU SG, VILLALBA JA, LIANG X, et al. Palmitic Acid-Rich High-Fat Diet Exacerbates Experimental Pulmonary Fibrosis by Modulating Endoplasmic Reticulum Stress. Am J Respir Cell Mol Biol. 2019; 61(6):737-746. [65] ROMERO F, HONG X, SHAH D, et al. Lipid Synthesis Is Required to Resolve Endoplasmic Reticulum Stress and Limit Fibrotic Responses in the Lung. Am J Respir Cell Mol Biol. 2018;59(2):225-236. [66] NEIGHBORS M, LI Q, ZHU SJ, et al. Bioactive lipid lysophosphatidic acid species are associated with disease progression in idiopathic pulmonary fibrosis. J Lipid Res. 2023;64(6):100375. [67] EL AGHA E, MOISEENKO A, KHEIROLLAHI V, et al. Two-Way Conversion between Lipogenic and Myogenic Fibroblastic Phenotypes Marks the Progression and Resolution of Lung Fibrosis. Cell Stem Cell. 2017;20(2):261-273.e3. [68] ONAL G, KUTLU O, GOZUACIK D, et al. Lipid Droplets in Health and Disease. Lipids Health Dis. 2017;16(1):128. [69] WANG P, DENG Y, FU X. MiR-509-5p suppresses the proliferation, migration, and invasion of non-small cell lung cancer by targeting YWHAG. Biochem Biophys Res Commun. 2017;482(4):935-941. [70] WANG J, PAN X, LI J, et al. TXNDC9 knockdown inhibits lung adenocarcinoma progression by targeting YWHAG. Mol Med Rep. 2022;25(6):203. [71] YOO JO, KWAK SY, AN HJ, et al. miR-181b-3p promotes epithelial-mesenchymal transition in breast cancer cells through Snail stabilization by directly targeting YWHAG. Biochim Biophys Acta. 2016;1863(7 Pt A):1601-1611. [72] FUJITA H, NAGAKAWA K, KOBUCHI H, et al. Phytoestrogen Suppresses Efflux of the Diagnostic Marker Protoporphyrin IX in Lung Carcinoma. Cancer Res. 2016;76(7):1837-1846. [73] HYUN DS, KIM HT, JHEON SH, et al. A preliminary study of protoporphyrin-IX as a potential candidate for identification of lung cancer cells using fluorescence microscopy. Photodiagnosis Photodyn Ther. 2009;6(3-4):221-226. [74] SHU Y, MA M, PAN X, et al. Cobalt protoporphyrin-induced nano-self-assembly for CT imaging, magnetic-guidance, and antioxidative protection of stem cells in pulmonary fibrosis treatment. Bioact Mater. 2022;21:129-141. [75] MINGONE CJ, GUPTE SA, CHOW JL, et al. Protoporphyrin IX generation from delta-aminolevulinic acid elicits pulmonary artery relaxation and soluble guanylate cyclase activation. Am J Physiol Lung Cell Mol Physiol. 2006;291(3):L337-L344. [76] ALZAHRANI AR, MOHAMED DI, ABO NAHAS HH, et al. Trimetazidine Alleviates Bleomycin-Induced Pulmonary Fibrosis by Targeting the Long Noncoding RNA CBR3-AS1-Mediated miRNA-29 and Resistin-Like Molecule alpha 1: Deciphering a Novel Trifecta Role of LncRNA CBR3-AS1/miRNA-29/FIZZ1 Axis in Lung Fibrosis. Drug Des Devel Ther. 2024;18:3959-3986. [77] HOU M, WU N, YAO L. LncRNA CBR3-AS1 potentiates Wnt/β-catenin signaling to regulate lung adenocarcinoma cells proliferation, migration and invasion. Cancer Cell Int. 2021;21(1):36. [78] EBERT B, KISIELA M, MALÁTKOVÁ P, et al. Regulation of human carbonyl reductase 3 (CBR3; SDR21C2) expression by Nrf2 in cultured cancer cells. Biochemistry. 2010;49(39):8499-8511. [79] JIE X, FONG WP, ZHOU R, et al. USP9X-mediated KDM4C deubiquitination promotes lung cancer radioresistance by epigenetically inducing TGF-β2 transcription. Cell Death Differ. 2021;28(7):2095-2111. [80] JIE X, CHEN Y, ZHAO Y, et al. Targeting KDM4C enhances CD8+ T cell mediated antitumor immunity by activating chemokine CXCL10 transcription in lung cancer. J Immunother Cancer. 2022;10(2):e003716. [81] SALMINEN A, KAARNIRANTA K, KAUPPINEN A. Hypoxia-Inducible Histone Lysine Demethylases: Impact on the Aging Process and Age-Related Diseases. Aging Dis. 2016;7(2):180-200. [82] PUHL AC, FERNANDES RS, GODOY AS, et al. The protein disulfide isomerase inhibitor 3-methyltoxoflavin inhibits Chikungunya virus. Bioorg Med Chem. 2023;83:117239. |
| [1] | 刘凤芝, 董玉娜, 田文艺, 王春雷, 梁晓东, 鲍 霖. 基因预测731种免疫细胞表型与类风湿性关节炎的关系[J]. 中国组织工程研究, 2026, 30(5): 1311-1319. |
| [2] | 容向宾, 郑海波, 莫学燊, 侯 坤, 曾 平, . 血浆代谢物、免疫细胞与髋骨关节炎的因果推断:GWAS数据欧洲群体资料分析[J]. 中国组织工程研究, 2026, 30(4): 1028-1035. |
| [3] | 丁 宇, 陈婧雯, 陈秀燕, 施慧敏, 杨雨蝶, 周美启, 崔 帅, . 循环炎症蛋白与心肌肥厚:来自GWAS Catalog与芬兰数据库欧洲群体的大样本分析[J]. 中国组织工程研究, 2026, 30(4): 1047-1057. |
| [4] | 赵非凡, 曹玉净. 强直性脊柱炎与银屑病共有基因人工神经网络模型及基于机器学习的挖掘和验证[J]. 中国组织工程研究, 2026, 30(3): 770-784. |
| [5] | 刘 楚, 丘博元, 童思文, 何林宇威, 陈浩博, 欧志学. 遗传学视角揭示血液代谢物与骨坏死的关系:基于芬兰FinnGen数据库信息分析[J]. 中国组织工程研究, 2026, 30(3): 785-794. |
| [6] | 黄 哲, 尚宝令, 姚耿圳, 潘光明, . 免疫细胞与心血管疾病风险的关联:欧洲人群全基因组关联研究[J]. 中国组织工程研究, 2026, 30(23): 5992-5999. |
| [7] | 张 政, 张艺博, 许 斌, 颜士朝, 郭 徽. 肌肉减少症与非酒精性脂肪肝:肠道微生物群分析[J]. 中国组织工程研究, 2026, 30(23): 6000-6009. |
| [8] | 仇雪笛, 郭 超, 何佳玥, 周 政. 单细胞测序数据识别牙周炎患者的差异表达基因和免疫细胞亚型[J]. 中国组织工程研究, 2026, 30(19): 4949-4964. |
| [9] | 梁 周, 潘成镇, 陈 锋, 张 驰, 杨 博, 韦宗波, 蒙建华, 周 砫. 骨质疏松相关外泌体诊断标志物的鉴定与药物初筛[J]. 中国组织工程研究, 2026, 30(13): 3458-3473. |
| [10] | 祁 祥, 曹 珊, 陈 健, 张艺嘉, 刘珂珂, 徐子福, 刘 往, 付晓霄, 殷晓磊. 筛选动脉粥样硬化线粒体功能障碍与铁死亡相关基因及调控的中药预测[J]. 中国组织工程研究, 2026, 30(10): 2641-2652. |
| [11] | 李加根, 陈跃平, 黄柯琪, 陈尚桐, 黄川洪. 线粒体自噬视域下的类风湿关节炎:多机器学习算法构建预测模型及验证并免疫调控分析[J]. 中国组织工程研究, 2025, 29(在线): 1-15. |
| [12] | 吴广涛, 秦 刚, 何凯毅, 范以东, 李威材, 朱宝刚, 曹 英. 免疫细胞与膝骨关节炎之间因果作用:一项两样本双向孟德尔随机化分析[J]. 中国组织工程研究, 2025, 29(5): 1081-1090. |
| [13] | 王建旭, 董恣豪, 黄子帅, 李思颖, 杨 光. 免疫微环境与骨衰老的相互作用及治疗策略[J]. 中国组织工程研究, 2025, 29(30): 6509-6519. |
| [14] | 陈天鑫, 张智龙, 张 帅, 高 云, 朱瑜琪, 杨胜平. 免疫细胞与代谢性骨病的因果关系:国际数据库欧洲群体的孟德尔随机化分析[J]. 中国组织工程研究, 2025, 29(29): 6326-6332. |
| [15] | 李加根, 陈跃平, 黄柯琪, 陈尚桐, 黄川洪. 线粒体自噬视域下的类风湿关节炎:基于多机器学习算法的互作分析[J]. 中国组织工程研究, 2025, 29(26): 5595-5607. |
肠-肺轴的动态联系与胃肠道、呼吸道黏膜来源自共同的胚胎发育起源有关。其中肠道微生物群在调控宿主生理、代谢及免疫功能中发挥着关键作用,在特发性肺纤维化的发生与发展过程中扮演重要角色[3-4]。具体而言,肠道微生物群通过代谢产物及抗原与宿主肠道微环境相互作用,改变免疫细胞行为从而调控炎症反应[5]。已有研究表明,肠道内白色念珠菌过度生长可通过动员Th17细胞迁移至肺部并分泌白细胞介素17A,促进内皮-间质转化,加速特发性肺纤维化进程;同样,流感嗜血杆菌和卡他莫拉菌等病原体在上呼吸道疾病患者中丰度较高[6-7]。相反,补充特定肠道微生物可调节Th1/Th2/Th17细胞免疫反应,缓解呼吸道症状并改善特发性肺纤维化预后[8]。此外,特发性肺纤维化患者肺组织中的慢性炎症也将进一步促进转化生长因子β和白细胞介素1等炎症蛋白的释放,导致代谢及脂质通路异常[9-10]。
研究表明,补充α-亚麻酸等代谢物能够恢复肠道微生物群平衡,提升抗炎脂肪酸水平,进而改善特发性肺纤维化[11-12]。但代谢物、免疫细胞及炎症蛋白在肠道微生物群与特发性肺纤维化关系中发挥作用的具体机制仍未被完全阐明。因此,需要采用多组学方法来发现新的生物标志物和治疗靶点。
孟德尔随机化已成为探索肠道微生物群与特发性肺纤维化因果关系的重要分析方法。汇总数据的孟德尔随机化方法对全基因组关联研究数据及表达数量性状位点数据进行深入挖掘,可筛选出表达水平发生变化的关键因子[13]。该研究利用大规模基因组数据及可成药基因数据库,系统分析了肠道微生物群与特发性肺纤维化之间的因果关系,同时评估了免疫细胞、炎症蛋白及代谢物在其中的作用。鉴于单独应用孟德尔随机化存在识别可靠因果基因的局限性,该研究结合汇总数据的孟德尔随机化、HEIDI检验、共定位分析,进一步验证所筛选基因作为特发性肺纤维化治疗靶点的潜力[14]。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 设计 采用单变量孟德尔随机化方法评估肠道微生物群与特发性肺纤维化之间的正向因果关系。随后,以特发性肺纤维化为暴露进行反向因果研究,评估特发性肺纤维化对肠道微生物群的影响。接着,探讨免疫细胞、炎症蛋白和代谢物在肠道微生物群-特发性肺纤维化因果关系中的中介作用。最后,应用汇总数据的孟德尔随机化/孟德尔随机化方法分析表达数量性状位点对肠道微生物群的影响,并结合贝叶斯共定位分析、药靶分析及分子对接探讨潜在治疗靶点。
1.2 时间及地点 研究于2024年9-11月在贵州中医药大学完成。
1.3 数据来源
肠道微生物组数据来源:该研究使用了473个肠道菌群的公开数据集[15],该数据集纳入5 959名个体的肠道微生物特征,鉴定出567个独立的单核苷酸多态性遗传变异单位,提供了肠道微生物组遗传基础的重要见解。
特发性肺纤维化数据来源:特发性肺纤维化全基因组关联研究数据来源于FinnGen数据库。该研究采用的特发性肺纤维化数据集包括375 082名个体,其中特发性肺纤维化患者2 018例,健康对照373 064例,是研究特发性肺纤维化遗传变异与疾病演变的重要数据基础。特发性肺纤维化相关数据可通过Finngen (https://r9.risteys.finngen.fi/endpoints/IPF)获取。
免疫细胞数据来源:免疫细胞的全基因组关联研究数据来源于一项大规模免疫细胞遗传特征研究的公开数据集。该数据集采用流式细胞术方法检测了3 757名欧洲人样本,鉴定出731种免疫细胞表型[16]。这项研究揭示了免疫细胞亚群之间的复杂遗传调控机制,为相关疾病提供了重要参考。原始数据可通过GWAS Catalog(https://www.ebi.ac.uk/gwas/publications/32929287,GCST0001391-GCST0002121)获取。
炎症蛋白数据来源:炎症蛋白的全基因组关联研究数据来源于公开数据集[17],该研究使用Olink Target平台在14 824名欧洲个体中分析了91种血浆蛋白并鉴定出180个蛋白质数量性状位点。该研究将蛋白质及基因的数量性状位点信息与全基因组关联研究数据整合,揭示了炎症蛋白与疾病病理之间的关联。相关数据可通过GWAS Catalog (GCST90274758-GCST90274848)在https://www.ebi.ac.uk/gwas/publications/37563310获取。
代谢物数据来源:代谢物全基因组关联研究数据来源于KARJALAINEN等[18]研究的公开数据,该研究分析了136 016名参与者中233种循环代谢物质特征,共鉴定出400多个独立的遗传位点,强调了代谢性状的遗传基础及其在复杂疾病中的意义。相关数据可
通过https://www.ebi.ac.uk/ gwas/publications/38448586 (GCST90302105-GCST90302051)获取。
基因表达数据来源:用于验证的表达数量性状位点数据来源于eQTLGen联盟,该项目分析了31 684名个体外周血中的表达数量性状位点特征,提供了在基因层面表达调控的全面见解。数据可通过eQTLGen平台(https://eqtlgen.org/)获取。
以上数据库均符合伦理和法律要求,且均对中国作者开放。
1.4 工具变量筛选 为了确保以肠道微生物群作为暴露、特发性肺纤维化作为结局研究的数据稳健,该研究对工具变量的筛选制定了以下多项严格标准。首先,与肠道微生物群相关的工具变量必须达到全基因组显著性水平(P < 1× 10⁻⁵)。其次,进行连锁不平衡分析,要求R² < 0.001,LD=10 000 kb。最后,为减少混杂效应,该研究将F ≤ 10的工具变量视为弱工具变量,由于弱工具变量可能导致结果偏倚,因此排除了F < 10的工具变量。
在反向孟德尔随机化分析中,特发性肺纤维化的工具变量筛选标准为:P < 5×10⁻⁸,R² < 0.001,LD=10 000 kb,且F统计量亦需> 10。对于两步中介孟德尔随机化分析和多变量孟德尔随机化(MVMR)分析,代谢物的工具变量需满足以下标准:P < 1×10⁻⁵,R² < 0.001,LD=500 kb;免疫细胞的工具变量需满足:P < 1×10⁻⁵,R² < 0.1,LD=500 kb;炎症蛋白的工具变量需满足:P < 5×10⁻⁶,R² < 0.001,LD=10 000 kb。同时排除F < 10的工具变量。对于顺式表达数量性状位点工具变量,筛选标准为:R² < 0.001,LD=10 000 kb。鉴于每次只能使用一个独立的工具变量,因此采用沃尔德比值作为主要孟德尔随机化效应估计方法,显著性阈值设定为P < 0.05。
1.5 统计学分析
1.5.1 单变量孟德尔随机化分析 在单变量孟德尔随机化分析中,将逆方差加权法作为“金标准”验证所有工具变量的有效性并计算加权总效应[19]。为确保结果的稳健性,该研究还采取了多项敏感性和多效性分析作为补充措施。
1.5.2 敏感性检验及多效性检验 该研究采用了多种方法来提高结果估计的稳健性,减少因异常值及强多效性工具变量产生的敏感性影响,包括稳健逆方差加权、惩罚逆方差加权、惩罚稳健逆方差加权、MR-Egger及其稳健和惩罚版本、MR-PRESSO以及MR-Lasso检验等,进一步提高了结果估计的准确性与可靠性。
1.5.3 两步中介孟德尔随机化分析 在两步中介孟德尔随机化分析中,第一步采用单变量孟德尔随机化分析肠道微生物群对中介变量(免疫细胞、炎症蛋白以及血液代谢物)的因果效应,获得效应估计值BETA1;第二步针对第一步筛选出的显著中介变量,采用多变量孟德尔随机化分析,分别评估中介变量对特发性肺纤维化的独立因果效应BETA2。肠道微生物群对特发性肺纤维化的总效应BETA由单变量孟德尔随机化分析确定,中介效应比例为(BETA1×BETA2)/BETA[20]。
1.5.4 汇总数据的孟德尔随机化多效性关联及共定位分析 在汇总数据的孟德尔随机化分析中,基于总结水平数据研究方法探讨关键微生物及表达数量性状位点的基因表达与性状之间的多效性关联,进一步了解基因与性状之间的关系[21-22]。筛选标准为汇总数据的孟德尔随机化检验P值< 0.05,同时应用HEDI检验确保P值> 0.05。随后,使用“coloc” R包(版本5.2.3)进行贝叶斯共定位分析,以确定全基因组关联研究数据与表达数量性状位点信号座是否在致病位点重叠[23-24]。分析涉及5种后验概率假设(PPH):①与两种性状均无关联(H0);②仅与性状1相关(H1);③仅与性状2相关(H2);④两种性状分别由不同的致病变异位点导致(H3);⑤两种性状由相同的致病变异位点导致(H4)。共定位标准为:当PPH4 > 0.8时,定义为共定位,当PPH4 > 0.6时,定义为中度共定位。
1.5.5 药物靶点预测与分子对接分析 将筛选得到的目标基因上传至DSigDB数据库[25],并通过Enrichr基因集富集分析平台预测潜在靶向药物[26]。随后选取关键蛋白与潜在药物开展分子对接分析,采用Schrödinger 12.8与PyMOL软件,基于对接得分评价药物与靶点的结合活性,同时对结果进行可视化。
特发性肺纤维化是一种严重的慢性间质性肺病,缺乏特效的治疗方法且难以早期诊断。患者通常出现呼吸困难、通气功能障碍、胸闷和胸痛等症状,且随着病程进展会愈发加重[27]。特发性肺纤维化的病因复杂,但归根到底是肺外基质的异常增殖和过度胶原沉积,在这一过程中,细胞外基质沉积主要由胶原蛋白、弹性蛋白以及浸润的成纤维细胞、上皮细胞、巨噬细胞和淋巴细胞组成[2],导致局部肺组织瘢痕纤维化并促进免疫机制过度应答,引发严重的炎症反应。炎症改变局部代谢导致代谢物的异常积累,不仅加剧了肺组织损伤,还促进了病理扩散至周围组织,推动了特发性肺纤维化的进展[28]。
肠-肺轴是指由免疫、神经和代谢物等连接的肠道和肺部相互作用的调节网络。肠道作为人体最大免疫器官在维持局部免疫稳态、调节炎症反应、抵抗病原体等方面发挥着重要作用。研究表明,肠道微生物群通过代谢产物可与宿主免疫系统产生联系以调节免疫细胞的作用,改变免疫反应和炎症过程[29]。在这个过程中肠道微生物群通过影响免疫细胞激活与分化促进内皮-间充质转化,加剧特发性肺纤维化的发展[30]。此外,肠道微生物群还可通过调节转化生长因子β和白细胞介素1等炎症因子释放,影响肺局部脂质代谢途径,进而加速慢性炎症和纤维化的形成[31]。因此,全面开展多组学研究以探讨肠道微生物群在特发性肺纤维化发病机制中的作用,对于发现新型生物标志物和治疗靶点有重要意义。
根据单变量孟德尔随机化/多变量孟德尔随机化的分析结果,Bacteroides faecis、Megasphaera和Pandoraea是通过免疫细胞、炎症蛋白和血清代谢物影响特发性肺纤维化发生发展的关键物种。文章重点讨论这3种菌及其相应中介因子的关系。
Bacteroides faecis是Bacteroides属的重要成员,已有研究表明肠道中Bacteroides faecis的低丰度会增加肠道炎症的风险,作用机制可能与改变调节性T细胞的比率,特别是抑制CD4+ CD25+调节性T细胞的损失有关[32]。随着“肠-肺”轴理论的拓展,肠道中的Bacteroides与肺部常见致病菌——假单胞菌属之间的强关联得到了越来越多的验证[33]。值得注意的是,Bacteroides已在肺病患者的肺泡灌洗液中检测到[34],且肠道和肺部Bacteroides的微生物群失调促进白细胞介素17B的产生,招募中性粒细胞并激活Th17细胞,这些都加速了特发性肺纤维化的发展[6]。此外,Bacteroides在肺癌[35]、急性肺损伤[36]、结核和慢性阻塞性肺疾病等疾病患者中也更为丰富[37-38],提示Bacteroides与肺部炎症程度存在关联。
Megasphaera是Veillonellaceae科中的严格厌氧革兰阴性菌,被认为是与特发性肺纤维化严重程度和不良预后最相关的球菌之一,它在肠道中丰度的变化与COVID-19引起的病毒性肺炎以及特发性肺纤维化的严重性相关,被定位为评价特发性肺纤维化和肺癌预后的潜在生物标志物[39]。虽然不同免疫疗法对Megasphaera相关疾病的影响各异[40],但Megasphaera已被证明能降低H7N9病毒感染个体的肺炎风险[41],且Megasphaera丰度在COVID-19感染肺炎患者中更高[42]。有趣的是,Megasphaera丰度的增加与患者咳痰量增加正有关[43],这可能与氨基酸代谢和外源性生物降解相关。考虑到特发性肺纤维化常常是肺炎、肺癌和慢性阻塞性肺疾病等疾病的终末状态,且Megasphaera与肺炎克雷伯菌的短链黄酮毒素存在同源性[44],提示Megasphaera可能作为特发性肺纤维化的早期诊断标志物。
Pandoraea是Burkholderiaceae科中代表性的革兰阴性菌,因与特发性肺纤维化存在关联而受到广泛关注。业界普遍认为Pandoraea与囊性纤维化相关,后者是一种因反复肺部感染而引发的肺局部纤维化病变[45]。多项研究指出Pandoraea大量存在囊性纤维化患者的痰液和肺部中,并可与假单胞菌共存导致败血症和包括特发性肺纤维化在内的慢性呼吸道疾病[46]。
此外,Pandoraea菌株能够在不形成生物膜的情况下透过单层上皮细胞并表现出细胞毒性,显著诱导白细胞介素6和白细胞介素8等促炎因子分泌,且效果远强于大肠埃希氏菌[47]。由于特发性肺纤维化的特征是间质基质过度沉积,沉积物主要来源于上皮细胞或纤维细胞,而Pandoraea可激活这些因素,提示Pandoraea是特发性肺纤维化的潜在致病物种[48]。
免疫细胞异常可通过多种机制参与特发性肺纤维化的发病过程。首先,巨噬细胞在受到刺激后可分泌白细胞介素1、白细胞介素6和肿瘤坏死因子α等促炎因子,激活成纤维细胞导致局部炎症并招募T细胞和B细胞等免疫细胞至损伤部位[49]。被活化的成纤维细胞通过转化生长因子β促进胶原蛋白的合成,导致胶原蛋白和细胞外基质过度沉积,最终引发肺组织纤维化。此外,T细胞的过度激活可直接作用于局部组织加剧炎症反应,进一步加速纤维化进程。因此,免疫细胞的异常行为被认为是特发性肺纤维化发生发展的关键因素之一[50]。
CD4⁺ T细胞是重要的辅助性T细胞,参与辅助B细胞和CD8⁺ T细胞的激活与增殖,并通过细胞因子分泌调节局部免疫反应。CD28作为共刺激分子,通过与其配体结合,在T细胞活化与增殖中发挥关键作用,从而放大T细胞激活信号。有研究发现,特发性肺纤维化患者外周血中CD4⁺ T细胞及单核细胞浸润水平升高[51];在博来霉素诱导的特发性肺纤维化动物模型中,CD4⁺ T细胞中转化生长因子β和白细胞介素17表达升高,进一步证实了此类T细胞与特发性肺纤维化的相关性[52]。此外,特发性肺纤维化患者中CCR2⁺ CD4⁺ T细胞亚群增多,该亚群具有免疫抑制功能,可缓解肺部炎症与纤维化,研究认为外周血中CCR2⁺ CD4⁺ T细胞亚群下调与特发性肺纤维化的不良预后相关[53]。在特发性肺纤维化患者中,Th1和Th2相关细胞因子的分泌受到抑制,CD4⁺ T淋巴细胞中调节性T细胞比例呈负向调节[54];动物实验也发现类似的情况,博来霉素诱导的特发性肺纤维化小鼠中CD4⁺ CD25hi Foxp3⁺调节性T细胞功能受损可加重肺纤维化,进一步验证了调节性T细胞损伤与特发性肺纤维化的发生及其严重程度密切相关[55]。以上发现强调了调节性T细胞在特发性肺纤维化进程中的关键作用。
在肺部炎症期间由免疫细胞释放的细胞因子和生长因子可促进成纤维细胞的增殖与活化,造成胶原蛋白过度沉积,这是特发性肺纤维化的一个标志性特征[56-57]。慢性炎症引发持续的肺组织损伤,进一步加重炎症反应,形成促纤维化的恶性循环,其中影响特发性肺纤维化的关键炎症蛋白包括白细胞介素家族成员、C-C趋化因子配体以及转化生长因子β等。
白细胞介素33作为白细胞介素家族细胞因子,可通过巨噬细胞增强促纤维化的Th2细胞因子表达,激活白细胞介素33/ST2信号轴诱导骨髓源细胞极化为M2型巨噬细胞,进而分泌白细胞介素13和转化生长因子β,并与白细胞介素25、胸腺基质淋巴细胞生成素等因子协同促进嗜酸性粒细胞浸润及胶原蛋白生成[58]。此外,C-C趋化因子受体4也在特发性肺纤维化患者中存在高表达,敲除或抑制C-C趋化因子受体4活性可改善甚至逆转模型小鼠的肺组织病理进程[59]。临床研究发现,特发性肺纤维化患者中CD68⁺肺泡巨噬细胞与纤维化高度相关,提示C-C趋化因子受体4可能通过调控巨噬细胞极化从而影响特发性肺纤维化进程[60]。转化生长因子β是特发性肺纤维化中最具代表性的蛋白之一,主要由肺泡上皮细胞、巨噬细胞、血小板及调节性T细胞分泌,参与细胞外基质生成、炎症细胞招募以及成纤维细胞分化等关键过程[61]。在正常生理条件下,转化生长因子β有助于维持肺部稳态,但信号异常则可诱导上皮-间充质转化及细胞内转录因子活化,导致胶原蛋白过度沉积,从而触发特发性肺纤维化[62],目前的研究也证实白细胞介素33和C-C趋化因子受体4均与转化生长因子β密切相关。
血清代谢物与特发性肺纤维化密切相关,特定代谢改变可反映纤维化的发生与进展。在炎症过程中,脂肪酸及氨基酸代谢紊乱可加剧炎症反应,影响细胞增殖与修复过程,最终促进成纤维细胞活化和胶原蛋白沉积,加速肺纤维化进展[31]。此外,低密度脂蛋白、总脂肪酸和三酰甘油是脂肪酸/氨基酸代谢中的关键中间产物,研究显示,特发性肺纤维化患者血浆及内皮细胞中低密度脂蛋白及其受体表达水平升高,Ldlr-/-特发性肺纤维化小鼠也表现出相同的趋势变化,而低密度脂蛋白的异常积累可损伤并激活内皮细胞与上皮细胞,促使其转分化为成纤维细胞,进一步加重纤维化。这提示低密度脂蛋白及低密度脂蛋白受体代谢异常可能参与特发性肺纤维化的发病机制[63]。
关于特发性肺纤维化患者肺组织中脂肪酸代谢障碍及促纤维化脂质过量的机制已有充分阐明:特发性肺纤维化患者血清总脂肪酸水平较对照组升高63%,肺组织与支气管肺泡灌洗液中棕榈酸与硬脂酸含量亦显著升高[64],提示总脂肪酸可能在疾病病理过程中发挥重要作用。目前业界普遍认为代谢通路异常可促使上皮细胞、巨噬细胞及成纤维细胞/肌成纤维细胞呈现促纤维化表型[65],其中三酰甘油是人体内最主要的能量储存形式,特发性肺纤维化患者血浆三酰甘油水平显著降低,且在特发性肺纤维化的不同阶段三酰甘油水平存在显著差异,并与死亡风险密切相关[66]。已知特发性肺纤维化的关键致病机制是成纤维细胞向肌成纤维细胞的转分化,而三酰甘油可调控脂肪成纤维细胞的分化过程[67]。此外,三酰甘油参与多种生理和病理过程,包括内质网应激、胰岛素抵抗、自噬、线粒体与核功能调节、炎症以及病毒感染,提示三酰甘油可能作为调控肺部成纤维细胞稳态的关键因子[68]。
基于孟德尔随机化-汇总数据的孟德尔随机化及共定位分析的遗传与药物预测结果显示,YWHAG、KDM4C与CBR3为关键靶向基因,而PpIX、GNF-Pf-2272及5155877为最具潜力的治疗候选药物。
YWHAG是14-3-3蛋白家族成员之一,在细胞增殖、迁移、氧化应激及炎症中发挥重要作用,YWHAG在非小细胞肺癌和肺腺癌细胞中高表达,可促进癌细胞增殖、迁移并增强抗凋亡能力[69]。抑制YWHAG上游蛋白TXNDC9可阻碍肺腺癌细胞的恶性行为,而YWHAG过表达可逆转该效应[70]。此外,YWHAG也是miR-509-5p的直接靶点,后者可有效抑制非小细胞肺癌的细胞增殖与侵袭[71]。虽然目前尚无直接证据将YWHAG与特发性肺纤维化联系起来,但YWHAG在肺癌中的治疗潜力提示它亦可能参与特发性肺纤维化病理过程,尤其考虑到特发性肺纤维化中持续的炎症与组织损伤可增加肺癌发生风险。
PpIX是通过5-氨基乙酰丙酸合成的潜在肺癌生物标志物,可通过ABCG2跨膜蛋白的胞外转运系统调控在癌细胞中富集[72]。目前认为PpIX的积累可显著提高肺癌荧光成像诊断灵敏性,尤其在肺癌细胞特异性染色中表现出良好效果[73],在临床上可增强间充质干细胞的治疗效果,促进PpIX在肺中的滞留与存活,改善肺功能并缓解纤维化进程[74]。此外,PpIX可调控铁的可利用性,进而影响肺动脉扩张功能,这一作用在特发性肺纤维化及其他肺部疾病治疗中具有潜在意义[75]。
CBR3是脱氢酶/还原酶酶家族成员,可通过调控miR-29表达影响氧化应激与炎症信号通路,从而调节特发性肺纤维化的进展[76]。目前研究认为CBR3-AS1通过激活Wnt/β-catenin信号通路参与肺腺癌细胞的增殖与迁移并受核因子E2相关因子2调控[77],后者是氧化应激反应的关键因子,提示CBR3可能通过维持肺细胞的氧化还原平衡参与特发性肺纤维化的发生发展[78]。因此,CBR3及其调控机制可能成为特发性肺纤维化潜在的治疗靶点。然而,对于化合物5155877的研究仍然匮乏,药理作用仍需进一步探索。
KDM4C是含JmjC结构域蛋白家族的关键成员,参与表观遗传修饰及转化生长因子β信号调控。在肺癌中,KDM4C去甲基化功能可通过降低H3K9me3水平,上调转化生长因子β与CXC趋化因子配体10表达,从而调节纤维化机制及免疫微环境直接影响基因转录[79-80]。此外,在慢性缺氧条件下,KDM4C表达受低氧诱导因子1α诱导促进染色质去甲基化与基因结构改变,从而影响细胞增殖与纤维化进程[81]。
3-methyltoxoflavin是一种具有抗病毒活性的蛋白二硫异构酶抑制剂,表现出良好的代谢稳定性和细胞通透性[82]。但目前尚无研究揭示KDM4C与特发性肺纤维化的关联,其在肺部疾病中的应用仍需进一步研究。
此研究虽在数据量、分析手段和药靶构建方面有一定的优势,但仍存在一定局限性。首先,特发性肺纤维化是一种病因复杂、进展缓慢的间质性肺病,在患者不同时期、病机病因及个体免疫状态等方面存在显著异质性。这种异质性可能影响肠道微生物群的表达模式,从而对因果关系结果稳定性产生影响。其次,此研究选择的数据集未能对特发性肺纤维化患者的分期和病因进行划分,可能导致结果在群体层面具有一定泛化倾向,缺乏精细诊断依据。因此,后续研究将结合更细化的临床表型与队列研究数据,以提高结果的临床适用性与转化价值。
该研究系统揭示了肠道微生物群(如Bacteroides faecis、Megasphaera与Pandoraea)通过免疫细胞、炎症因子及代谢物影响特发性肺纤维化发病风险的因果关系和潜在机制,对中国医学研究具有重要借鉴意义:①在基础医学领域,提供了“肠-肺轴”视角下的遗传变异和免疫调控的完整机制,为阐述特发性肺纤维化免疫代谢机制指出了新方向;②该研究结果提示CD4+ T细胞亚群、白细胞介素33水平及脂质代谢紊乱可作为疾病诊疗的潜在生物标志物,有助于临床对高风险人群的精准早期诊断;③在药物研发方面,识别出的关键靶基因和潜在药物,为开发特发性肺纤维化新药提供参考。尽管所用全基因组关联研究数据源于欧洲人群,但考虑人类的同源性,该研究结果在中国人群中同样具有较强适应性与推广价值。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
“肠-肺轴”作为微生物与肺部疾病交叉领域的重要研究方向。越来越多的研究证明,肠道菌群不仅参与局部肠道免疫调控,还通过影响相关代谢产物改变远端肺部微环境,可能在特发性肺纤维化等肺病的发生发展中发挥关键调控作用。目前关于“肠-肺轴”在特发性肺纤维化中的作用机制缺乏系统性多组学整合研究,尤其在机制的因果判断、药物靶点识别方面相对空白。#br# 本研究通过整合全基因组关联研究、表达数量性状位点、中介分析和分子对接等多种方法,系统评估肠道菌群通过免疫细胞、炎症因子及代谢物影响特发性肺纤维化的因果关系和机制链条,探讨了关键菌群与潜在治疗靶点,在机制探索和靶向药物预测方面具有理论意义。随着多组学技术和人工智能大数据算法的不断进步,未来有望进一步揭示肠道菌群在特发性肺纤维化及其他慢性肺病中的作用机制,为精准靶向防治提供理论基础。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||