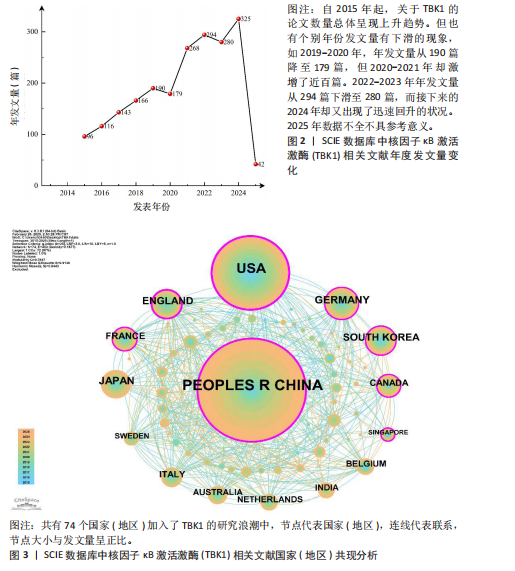
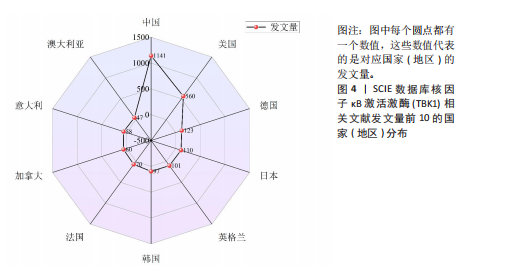
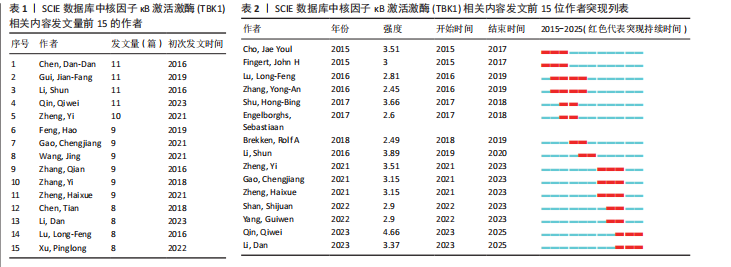
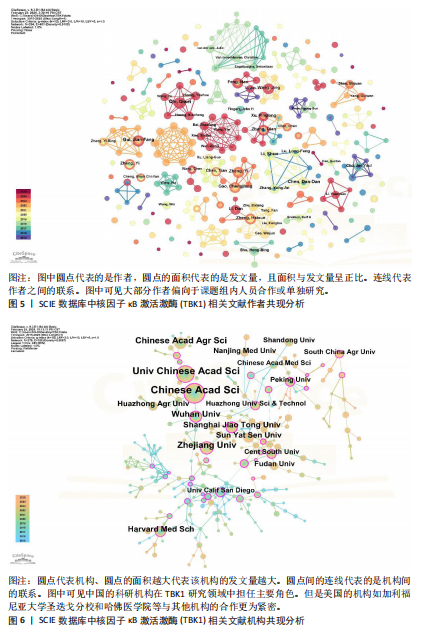
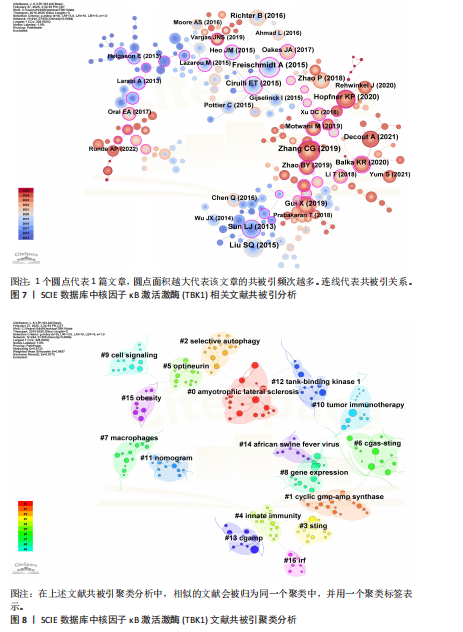
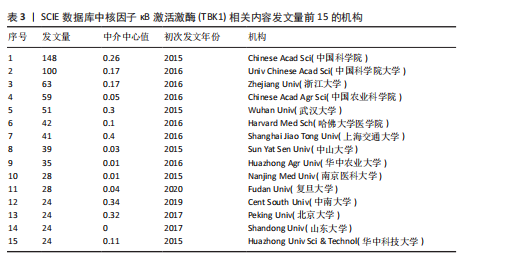
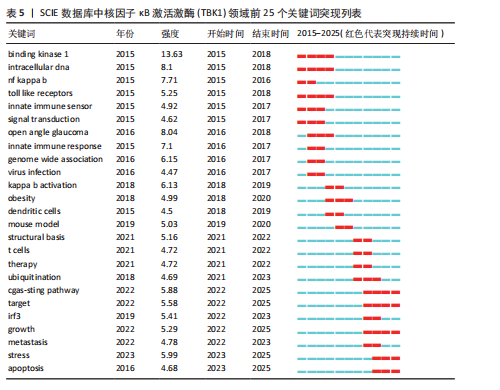
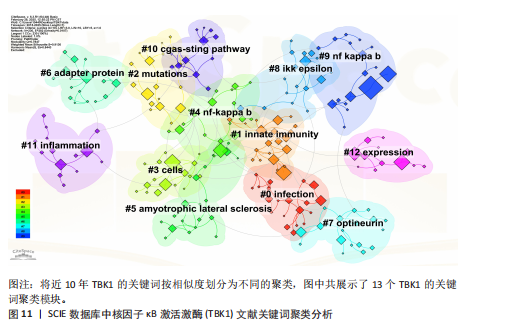
[1] 胡芳茴,李丽蔚,杨霞,等.抑制TBK1通过调节mTORC1信号通路减轻肺泡巨噬细胞中NLRP3介导的细胞焦亡[J/OL].中国免疫学杂志,1-10[2025-02-24].https://link.cnki.net/urlid/22.1126.R.20250124.1616.006.
[2] RUNDE AP, MACK R, S J PB, et al. The role of TBK1 in cancer pathogenesis and anticancer immunity. J Exp Clin Cancer Res. 2022;41(1):135.
[3] MIRANDA A, SHIRLEY CA, JENKINS RW. Emerging roles of TBK1 in cancer immunobiology. Trends Cancer. 2024;10(6):531-540.
[4] ZHANG M, ZOU Y, ZHOU X, et al. Inhibitory targeting cGAS-STING-TBK1 axis: Emerging strategies for autoimmune diseases therapy. Front Immunol. 2022;13:954129.
[5] 王悦宸,侯亚威,王振国.基于文献计量学的丹参研究现状与热点分析[J].中草药,2025,56(4): 1318-1337.
[6] 江宇慧,朱明玉,张景景,等.基于文献计量学分析近10年药用植物组织培养的研究趋势[J].世界科学技术-中医药现代化,2025,27(1):98-109.
[7] 李进鹏,曹妍,赵奕雯,等.基于文献计量学的国内外药食同源及食疗领域研究热点分析[J].护理研究,2024,38(19):3457-3467.
[8] 张建,杨卫华,刘萍,等.海洋固碳研究进展:基于Citespace的可视化分析[J].海洋湖沼通报(中英文),2024,46(4):135-142.
[9] 朱格格,黄安书,覃盈盈.基于Web of Science的国际红树林研究发展态势分析[J].广西师范大学学报(自然科学版), 2024,42(5):1-12.
[10] 徐灿丽,何文星,汪磊,等.肝脏类器官研究的文献计量学分析[J].中国组织工程研究,2024, 28(7):1099-1104.
[11] 谭令,龙霖梓,邓秘,等.抗血小板活化的文献计量学及可视化分析[J].世界科学技术-中医药现代化,2022,24(1):195-208.
[12] 王晗,李春辉,孙海玮.远隔缺血适应相关研究的文献计量学和可视化分析[J].中国脑血管病杂志,2025,22(2):89-98.
[13] 王勇,李宏宇,刘雨航,等.股骨头坏死手术治疗知识图谱:2005-2024数据的文献计量学分析[J].中国组织工程研究,2025,29(33):7250-7260.
[14] 郭萌,吴文,李敬文,等.基于文献计量学的“结核”主题高被引文献特征分析[J].中国防痨杂志,2024,46(5):567-577.
[15] ZHANG C, SHANG G, GUI X, et al. Structural basis of STING binding with and phosphorylation by TBK1. Nature. 2019; 567(7748):394-398.
[16] LIU S, CAI X, WU J, et al. Phosphorylation of innate immune adaptor proteins MAVS, STING, and TRIF induces IRF3 activation. Science. 2015; 347(6227):aaa2630.
[17] FREISCHMIDT A, WIELAND T, RICHTER B, et al. Haploinsufficiency of TBK1 causes familial ALS and fronto-temporal dementia. Nat Neurosci. 2015;18(5):631-636.
[18] HOPFNER KP, HORNUNG V. Molecular mechanisms and cellular functions of cGAS-STING signalling. Nat Rev Mol Cell Biol. 2020;21(9):501-521.
[19] GUI X, YANG H, LI T, et al. Autophagy induction via STING trafficking is a primordial function of the cGAS pathway. Nature. 2019;567(7747):262-266.
[20] REVACH OY, LIU S, JENKINS RW. Targeting TANK-binding kinase 1 (TBK1) in cancer. Expert Opin Ther Targets. 2020;24(11):1065-1078.
[21] HU L, ZHANG Q. Mechanism of TBK1 activation in cancer cells. Cell Insight. 2024;3(5):100197.
[22] HUANG X, HUO L, XIAO B, et al. Activating STING/TBK1 suppresses tumor growth via degrading HPV16/18 E7 oncoproteins in cervical cancer. Cell Death Differ. 2024;31(1):78-89.
[23] ALAM M, HASAN GM, HASSAN MI. A review on the role of TANK-binding kinase 1 signaling in cancer. Int J Biol Macromol. 2021;183:2364-2375.
[24] ZHAO C, ZHAO W. TANK-binding kinase 1 as a novel therapeutic target for viral diseases. Expert Opin Ther Targets. 2019;23(5):437-446.
[25] LAN J, DENG Z, WANG Q, et al. Neuropeptide substance P attenuates colitis by suppressing inflammation and ferroptosis via the cGAS-STING signaling pathway. Int J Biol Sci. 2024;20(7):2507-2531.
[26] DU SS, CHEN GW, YANG P, et al. Radiation Therapy Promotes Hepatocellular Carcinoma Immune Cloaking via PD-L1 Upregulation Induced by cGAS-STING Activation. Int J Radiat Oncol Biol Phys. 2022;112(5):1243-1255.
[27] 龙昊,吴怡林,龚建平.TBK1在炎症性疾病发生机制中的作用[J].重庆医学,2020,49(20):3472-3475+3480.
[28] DUAN QQ, WANG H, SU WM, et al. TBK1, a prioritized drug repurposing target for amyotrophic lateral sclerosis: evidence from druggable genome Mendelian randomization and pharmacological verification in vitro. BMC Med. 2024; 22(1):96.
[29] ZHAO X, CAO Y, LU R, et al. Phosphorylation of AGO2 by TBK1 Promotes the Formation of Oncogenic miRISC in NSCLC. Adv Sci (Weinh). 2024;11(15):e2305541.
[30] GAO CQ, CHU ZZ, ZHANG D, et al. Serine/threonine kinase TBK1 promotes cholangiocarcinoma progression via direct regulation of β-catenin. Oncogene. 2023;42(18):1492-1507.
[31] QIAN Y, YAO W, YANG T, et al. aPKC-ι/P-Sp1/Snail signaling induces epithelial-mesenchymal transition and immunosuppression in cholangiocarcinoma. Hepatology. 2017;66(4): 1165-1182.
[32] YANG S, IMAMURA Y, JENKINS RW, et al. Autophagy Inhibition Dysregulates TBK1 Signaling and Promotes Pancreatic Inflammation. Cancer Immunol Res. 2016;4(6):520-530.
[33] SIDDIQUI AJ, JAMAL A, ZAFAR M, et al. Identification of TBK1 inhibitors against breast cancer using a computational approach supported by machine learning. Front Pharmacol. 2024;15:1342392.
[34] ZHU L, LI Y, XIE X, et al. TBKBP1 and TBK1 form a growth factor signalling axis mediating immunosuppression and tumourigenesis. Nat Cell Biol. 2019;21(12):1604-1614.
[35] CAI H, YAN L, LIU N, et al. IFI16 promotes cervical cancer progression by upregulating PD-L1 in immunomicroenvironment through STING-TBK1-NF-kB pathway. Biomed Pharmacother. 2020;123:109790.
[36] JIANG Y, CHEN S, LI Q, et al. TANK-Binding Kinase 1 (TBK1) Serves as a Potential Target for Hepatocellular Carcinoma by Enhancing Tumor Immune Infiltration. Front Immunol. 2021;12: 612139.
[37] VAN DAELE SH, MOISSE M, VAN VUGT JJFA, et al. Genetic variability in sporadic amyotrophic lateral sclerosis. Brain. 2023;146(9):3760-3769.
[38] GURFINKEL Y, POLAIN N, SONAR K, et al. Functional and structural consequences of TBK1 missense variants in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Neurobiol Dis. 2022;174:105859.
[39] CIRULLI ET, LASSEIGNE BN, PETROVSKI S, et al. Exome sequencing in amyotrophic lateral sclerosis identifies risk genes and pathways. Science. 2015; 347(6229):1436-1441.
[40] WEINREICH M, SHEPHEARD SR, VERBER N, et al. Neuropathological characterization of a novel TANK binding kinase (TBK1) gene loss of function mutation associated with amyotrophic lateral sclerosis. Neuropathol Appl Neurobiol. 2020;46(3):279-291.
[41] AHMAD L, ZHANG SY, CASANOVA JL, et al. Human TBK1: A Gatekeeper of Neuroinflammation. Trends Mol Med. 2016;22(6):511-527.
[42] LI Q, LIU Y, XIA X, et al. Activation of macrophage TBK1-HIF-1α-mediated IL-17/IL-10 signaling by hyperglycemia aggravates the complexity of coronary atherosclerosis: An in vivo and in vitro study. FASEB J. 2021;35(5):e21609.
[43] ZHAO P, SUN X, LIAO Z, et al. The TBK1/IKKε inhibitor amlexanox improves dyslipidemia and prevents atherosclerosis. JCI Insight. 2022; 7(17):e155552.
[44] SUN Y, REVACH OY, ANDERSON S, et al. Targeting TBK1 to overcome resistance to cancer immunotherapy. Nature. 2023;615(7950):158-167.
[45] MAAN M, JAISWAL N, LIU M, et al. TBK1 Reprograms Metabolism in Breast Cancer: An Integrated Omics Approach. J Proteome Res. 2025;24(1):121-133.
[46] MA X, JIA S, WANG G, et al. TRIM28 promotes the escape of gastric cancer cells from immune surveillance by increasing PD-L1 abundance. Signal Transduct Target Ther. 2023;8(1):246.
[47] MAAN M, AGRAWAL NJ, PADMANABHAN J, et al. Tank Binding Kinase 1 modulates spindle assembly checkpoint components to regulate mitosis in breast and lung cancer cells. Biochim Biophys Acta Mol Cell Res. 2021;1868(3):118929.
[48] LEFRANC J, SCHULZE VK, HILLIG RC, et al. Discovery of BAY-985, a Highly Selective TBK1/IKKε Inhibitor. J Med Chem. 2020; 63(2):601-612.
[49] SHIN J, LIM J, HAN D, et al. TBK1 inhibitor amlexanox exerts anti-cancer effects against endometrial cancer by regulating AKT/NF-κB signaling. Int J Biol Sci. 2025;21(1):143-159.
[50] LIN KX, ISTL AC, QUAN D, et al. PD-1 and PD-L1 inhibitors in cold colorectal cancer: challenges and strategies. Cancer Immunol Immunother. 2023;72(12):3875-3893.
[51] VESELY MD, ZHANG T, CHEN L. Resistance Mechanisms to Anti-PD Cancer Immunotherapy. Annu Rev Immunol. 2022;40:45-74.
|