中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (28): 7347-7354.doi: 10.12307/2026.373
• 组织构建综述 tissue construction review • 上一篇 下一篇
运动调控细胞焦亡防治骨代谢异常疾病
昝俊浩1,胡淑娟1,袁新国1,蒲 锐1,2
- 长江大学,1教育与体育学院,2运动人体科学实验室,湖北省荆州市 434023
-
收稿日期:2025-07-09修回日期:2025-09-10出版日期:2026-10-08发布日期:2026-02-12 -
通讯作者:袁新国,教授,博士生导师,长江大学教育与体育学院,湖北省荆州市 434023 共同通讯作者:蒲锐,讲师,硕士生导师,长江大学,教育与体育学院,运动人体科学实验室,湖北省荆州市 434023 -
作者简介:昝俊浩,男,2002年生,湖北省襄阳市人,汉族,长江大学在读硕士,主要从事运动健康促进方面的研究。 -
基金资助:湖北省自然科学基金项目(2025AFB925),项目负责人:胡淑娟
Exercise regulation of pyroptosis for the prevention and treatment of bone metabolic disorders
Zan Junhao1, Hu Shujuan1, Yuan Xinguo1, Pu Rui1, 2
- 1College of Education and Physical Education, 2Human Science Laboratory, Yangtze University, Jingzhou 434023, Hubei Province, China
-
Received:2025-07-09Revised:2025-09-10Online:2026-10-08Published:2026-02-12 -
Contact:Yuan Xinguo, Professor, Doctoral supervisor, College of Education and Physical Education, Yangtze University, Jingzhou 434023, Hubei Province, China Co-corresponding author: Pu Rui, Lecturer, Master’s supervisor, College of Education and Physical Education, Yangtze University, Jingzhou 434023, Hubei Province, China; Human Science Laboratory, Yangtze University, Jingzhou 434023, Hubei Province, China -
About author:Zan Junhao, MS candidate, College of Education and Physical Education, Yangtze University, Jingzhou 434023, Hubei Province, China -
Supported by:Hubei Provincial Natural Science Foundation, No. 2025AFB925 (to HSJ)
摘要:
文题释义:
细胞焦亡:一种程序性细胞死亡方式,由炎症刺激引发细胞膜裂解死亡,细胞内容物流出并引起强烈炎症反应。
运动介导细胞焦亡在骨代谢异常疾病中的作用机制:运动可抑制细胞焦亡蛋白表达、炎症因子分泌,改善氧化应激水平和调节肌因子的表达,抑制破骨细胞活性,增加成骨细胞活性,在调控骨代谢异常疾病中发挥有益作用。
背景:细胞焦亡广泛参与成骨细胞介导的骨形成与破骨细胞介导的骨吸收过程,细胞焦亡释放的炎症因子导致骨代谢失衡。运动可抑制细胞焦亡的发生、调控骨代谢异常疾病进展,已成为防治骨代谢异常疾病的研究热点。
目的:总结细胞焦亡在骨代谢异常疾病中的调控作用以及运动介导细胞焦亡在改善骨代谢异常疾病中的分子机制。
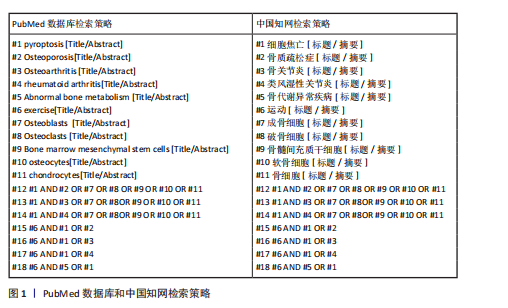
方法:以“exercise,pyroptosis,Osteoporosis,Osteoarthritis,rheumatoid arthritis,Abnormal bone metabolism,Osteoblasts,Osteoclasts,Bone marrow mesenchymal stem cells,osteocytes,chondrocytes”为英文检索词,以“运动,细胞焦亡,骨关节炎,骨质疏松症,类风湿性关节炎,骨代谢异常,成骨细胞,破骨细胞,骨髓间充质干细胞,骨细胞及软骨细胞”为中文检索词,检索中国知网与PubMed数据库1992-2025年间的相关文献,根据入选标准选择80篇文献进行综述。
结果与结论:细胞焦亡及其炎症因子在骨代谢异常疾病的发生发展中发挥着重要作用。近年来研究表明,运动作为一种安全有效的非药物干预手段,可通过抑制炎症小体活化、抑制焦亡蛋白表达、调节肌因子分泌以及降低氧化应激水平来缓解细胞焦亡,致使骨吸收减少、骨形成增加。但当前细胞焦亡与骨代谢异常疾病的研究存在局限,运动介导细胞焦亡参与骨代谢异常疾病的具体通路和调控机制有待进一步研究。
https://orcid.org/0009-0001-7133-5380 (昝俊浩)
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
中图分类号:
引用本文
昝俊浩, 胡淑娟, 袁新国, 蒲 锐. 运动调控细胞焦亡防治骨代谢异常疾病[J]. 中国组织工程研究, 2026, 30(28): 7347-7354.
Zan Junhao, Hu Shujuan, Yuan Xinguo, Pu Rui. Exercise regulation of pyroptosis for the prevention and treatment of bone metabolic disorders[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(28): 7347-7354.
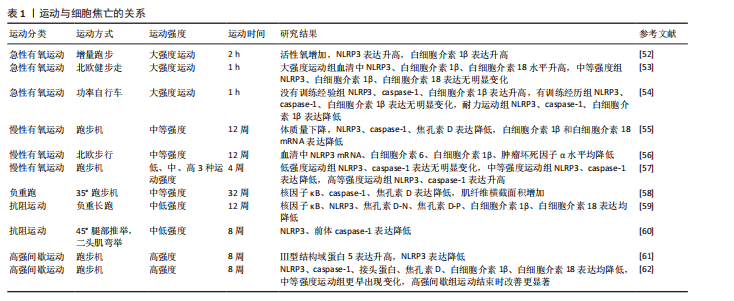
细胞焦亡通常由炎症小体触发,包括经典途径和非经典途径。炎症小体是caspase-1或其他炎性caspase的胞质超分子复合体,NLRP3是目前研究最多的炎性小体之一[10]。
在经典途径中,NLRP3表达可在识别病原分子模式时由炎症反应的细胞因子引起,激活核因子κB进行核转录,NLRP3和其他细胞因子表达转录上调[11];NLRP3激活后组装并募集下游炎症小体形成复合体,随后活化并裂解前体caspase-1形成具有活性的caspase-1[12-13],caspase-1通过蛋白水解作用把前体白细胞介素1β和前体白细胞介素18加工成熟白细胞介素1β和白细胞介素18;同时caspase-1切割焦孔素D产生焦孔素D-N末端结构,转移至细胞膜形成膜孔[14],细胞外的水分子和游离小分子进入细胞内,细胞吸水涨破死亡,炎症因子白细胞介素β和白细胞介素18以及胞内物质释放,从而引发细胞焦亡。
在非经典途径中,依赖caspase-4/ 5和caspase-11,对革兰阴性菌脂多糖具有高度亲和性,可刺激巨噬细胞产生炎症反应[15]。
炎症因子可直接识别细胞内脂多糖有助于前体caspase-4/5/11 的快速寡聚化。形成caspase-4/5/11非经典炎性小体,活化后的caspase-4/5/11激活焦孔素,导致细胞膜上形成穿孔,并产生气泡,此过程不依赖于caspase-1。体液中的水分子和小分子物质不断进入细胞内,细胞持续增大直至破裂,细胞内容物和炎性因子不断流出,引发细胞焦亡非经典途径。细胞焦亡机制见图4。
2.2 细胞焦亡在骨代谢异常疾病中的作用 近年来,以细胞焦亡为靶点诊疗骨代谢异常疾病已成为研究热点。
2.2.1 细胞焦亡与骨质疏松症 骨质疏松症是一种由于成骨细胞与破骨细胞代谢失衡引起的全身性进行性代谢性骨病,表现为骨密度降低、骨量减少及骨微结构破坏,易导致骨脆性增加与骨折风险升高,常见于中老年人及绝经后女性[16]。TAO等[17]首次提出细胞焦亡作为骨质疏松症发病机制的新假说,指出在病理条件下成骨细胞发生焦亡可抑制其增殖与分化,进而影响骨形成;同时,焦亡诱导的炎症反应可激活破骨细胞,加速骨质流失。炎症是在无感染情况下由内源性信号驱动的慢性、无菌、低度炎症状态[18],炎症因子可诱导成骨细胞焦亡。高浓度白细胞介素17可上调NLRP3、caspase-1、焦孔素D表达,促进白细胞介素1β释放并引发成骨细胞破裂[19]。在粪肠球菌或金黄色葡萄球菌感染模型中,炎性小体和焦亡蛋白被激活,而焦亡抑制剂可减轻焦亡程度并恢复骨结构[20-21]。此外,高糖环境可通过caspase-1/焦孔素D/白细胞介素1β通路抑制成骨细胞功能[22]。焦亡抑制剂如坏死磺酰胺可降低相关基因表达、减少炎症因子释放,改善成骨细胞活性[23]。由此可见,靶向焦亡通路有望成为改善骨质疏松症的一种潜在治疗策略。因此,在炎症或病原体感染等病理条件下,NLRP3炎性小体、caspase-1和焦孔素D通路被激活,导致成骨细胞破裂死亡并释放炎症因子如白细胞介素1β,抑制骨形成。另外,不同致病因素如高糖环境、金黄色葡萄球菌感染、肠道菌群失调等均能诱导焦亡,损害骨结构。
细胞焦亡释放的炎症因子可过度激活破骨细胞。肿瘤坏死因子α是成骨细胞分化的抑制因子和破骨细胞生成的促进因子,在炎症性
关节疾病中对骨稳态具有重要调控作用[24]。肿瘤坏死因子α通过激活骨细胞内的核因子κB通路直接增强核因子κB受体活化因子配体(RANKL)表达,促进破骨细胞生成。MARAHLEH等[25]研究发现,经肿瘤坏死因子α处理的骨细胞核因子κB受体活化因子配体(RANKL) mRNA表达显著增加,添加抑制剂后该表达水平明显降低。一方面,核因子κB上调T细胞核因子的表达,促进破骨细胞基因转录和分化;另一方面,核因子κB进入细胞核后促进NLRP3炎性小体转录,诱导巨噬细胞与成骨细胞发生焦亡并释放炎症因子。在牙周炎等感染性炎症引起的骨吸收过程中,NLRP3可促进破骨细胞分化,敲除NLRP3基因可阻断白细胞介素1β的释放并抑制破骨细胞分化,而NLRP3炎性小体特异性抑制剂MCC950可有效抑制破骨前体细胞的分化,显著减缓牙槽骨丢失,降低白细胞介素1β活化水平及破骨细胞分化[26]。另外,NLRP3不仅增强破骨细胞的骨吸收功能,还可促进破骨细胞生成、抑制因子聚合酶1的降解,因此,NLRP3炎性小体的激活不仅引发炎症递质的释放,还可诱导细胞因子的自主调控反应[27]。由此可见,成骨细胞焦亡减弱骨组织形成,同时产生的炎症因子刺激破骨细胞活性,加重病理进程,但多数研究以体外细胞实验为主,缺乏系统性体内实验或长期动物模型研究。此外,焦亡抑制剂的作用机制多为初步观察,具体剂量、安全性、长期效应仍需系统验证。
2.2.2 细胞焦亡与骨关节炎 骨关节炎是关节组织修复与破坏不平衡所引起的关节退行性疾病[28],受年龄、肥胖和机械因素影响,病理表现为软骨基质降解、滑膜炎症等,易导致关节功能障碍、疼痛、僵硬以及丧失活动能力[29]。通常软骨处于稳定状态,当促炎因子、机械过载或代谢紊乱时引起软骨细胞外基质降解,由于软骨中血管不足其再生和修复就会受到限制,导致细胞功能障碍炎症反应引发骨关节炎;另一方面,滑膜炎症是骨关节炎常见的病理表现,严重的滑膜炎会加剧软骨侵蚀[30-31]。软骨细胞焦亡加重骨关节炎进展,同时细胞焦亡引起的炎症反应导致更严重的滑膜炎症和疼痛,因此,细胞焦亡已成为骨关节炎防治的研究热点。
软骨细胞焦亡是导致骨关节炎发生的重要因素。基质金属蛋白酶13具有裂解Ⅱ型胶原蛋白的特异能力,是介导软骨细胞外基质降解的关键因子[32]。PAN等[33]研究发现,白细胞介素1β刺激可显著增加软骨细胞和巨噬细胞中基质金属蛋白酶13、白细胞介素6、白细胞介素1β、NLRP3 和 caspase-1表达,同时下调蛋白聚糖和Ⅱ型胶原蛋白表达,提示细胞焦亡加速基质降解并促进炎症因子的释放。有研究表明,厚实酸可抑制白细胞介素1β诱导的软骨细胞炎症反应,包括一氧化氮、前列腺激素、肿瘤坏死因子α和白细胞介素6的产生,同时逆转白细胞介素1β诱导的基质金属蛋白酶13上调和Ⅱ型胶原及聚集蛋白聚糖下调,该作用机制可能与抑制核因子κB信号通路、减缓炎症反应及细胞外基质变性有关[34]。此外,抑制软骨细胞焦亡可有效延缓骨关节炎的进程。研究表明,石斛碱[35]、转录因子Sox9[36]、姜黄素及隐丹参酮等活性物质可通过抑制核因子κB通路和下调白细胞介素1β等炎症因子的表达[37-38],抑制软骨细胞焦亡,具有作为骨关节炎潜在治疗药物的研究价值。
滑膜炎症是骨关节炎发生和进展的重要致病因素之一。在多种炎性细胞因子中,白细胞介素1β是具有代表性的炎症递质,能够显著促进骨组织与软骨的破坏进程;而白细胞介素18通过延长白细胞介素1β的作用时间进一步加剧炎症反应。有研究表明,在大鼠滑膜细胞焦亡模型中,滑膜组织中NLRP3、白细胞介素1β和白细胞介素18的表达水平均显著升高,通过靶向抑制白细胞介素1β和白细胞介素18的表达可有效降低NLRP3、caspase-1及焦孔素D的含量,从而缓解膝骨关节炎相关的滑膜炎症[39]。此外,高迁移率族蛋白B1是由巨噬细胞分泌的重要炎症因子,在滑膜炎症中发挥关键作用。研究发现,骨关节炎大鼠滑膜细胞中高迁移率族蛋白B1、NLRP3和caspase-1表达水平均呈现上调趋势,提示滑膜细胞焦亡可诱导高迁移率族蛋白B1的过度产生,进而加剧炎症反应,为改善滑膜炎症提供了新的干预靶点[40]。上述研究初步揭示软骨细胞和滑膜细胞焦亡在骨关节炎中的作用,但软骨细胞和滑膜细胞之间是否存在炎症信号的交互作用还需进一步探讨。此外,当前以大鼠模型为主,未来可探索多种实验模型,推进临床试验。
2.2.3 细胞焦亡与类风湿关节炎 类风湿关节炎是一种以持续性滑膜炎、软骨破坏和骨侵蚀为特征的自身免疫性疾病,主要临床表现为关节疼痛、肿胀和僵硬。类风湿关节炎的病理过程主要由促炎与抗炎因子失衡所驱动,慢性滑膜炎和关节破坏是该病的核心特征[41]。研究表明,在类风湿关节炎进展过程中,细胞焦亡可引发大量炎症因子的释放,进一步加剧慢性滑膜炎症及骨与关节组织损伤,同时刺激免疫系统产生一定的抗炎反应[42]。滑膜巨噬细胞在类风湿关节炎中的作用尤为关键,主要包括M1型和M2型两类:M1型分泌促炎因子,激活机体免疫反应;M2型释放抗炎因子,有助于抑制炎症反应。M1型巨噬细胞在一定条件下可极化转化为M2型巨噬细胞。ZHAI等[43]的研究发现,类风湿关节炎患者巨噬细胞中M1型滑膜巨噬细胞、caspase-3和焦孔素D的表达显著升高,并且焦孔素D表达水平与细胞焦亡程度呈正相关,提示抑制炎性细胞因子的表达已成为类风湿关节炎潜在的靶向治疗策略之一。
缺氧是类风湿关节炎滑膜炎发展的重要促进因素。由于炎症反应期细胞氧消耗增加,导致线粒体功能障碍和氧化应激增强,进而促使缺氧诱导因子1α的积聚[44]。成纤维样滑膜细胞在缺氧环境中因线粒体损伤产生过量活性氧,促进缺氧诱导因子1α的合成,缺氧诱导因子1α不仅是低氧应答的关键因子,也是诱导细胞焦亡的重要调控分子。在低氧状态下,缺氧诱导因子1α介导NLRP3炎性小体的表达,通过激活caspase-1裂解焦孔素D、白细胞介素1β和白细胞介素18诱导成纤维样滑膜细胞发生焦亡[45]。HONG等[46]研究证实,缺氧诱导因子1α可通过激活NLRP3炎性小体促进caspase-1和焦孔素D的表达,从而加剧炎症反应。类风湿关节炎的另一个显著病理特征是关节腔内炎症代谢物积累导致的酸性环境(pH值降低),从而引起软骨细胞的过度死亡,表明酸敏感离子通道在类风湿关节炎病理进展中起重要作用。酸中毒可通过调控NLRP3炎性小体的表达促进巨噬细胞合成和分泌白细胞介素1β,从而加剧炎症反应[47]。在类风湿关节炎大鼠模型中,NLRP3、caspase-1、白细胞介素1β和白细胞介素18的表达显著上调,而酸敏感通道抑制剂阿米洛利可有效逆转该现象,提示阿米洛利可能通过抑制炎症通路减缓类风湿关节炎的进展[48]。
植物活性成分在类风湿关节炎防治中展现出良好的潜力。例如,安石榴苷是从石榴皮中提取的一种天然物质,能够通过下调NLRP3和caspase-1的表达、减少白细胞介素1β和白细胞介素18的释放,从而抑制滑膜组织的细胞焦亡,该机制可能与安石榴苷对核因子κB信号通路的负向调控有关[49]。除此之外,安石榴苷还可促进巨噬细胞由M1型向M2型极化,从而缓解病理性炎症反应。白虎桂枝汤联合芒果苷与肉桂酸对佐剂诱导的类风湿关节炎大鼠模型具有显著治疗效果,该组合通过抑制核因子κB介导的NLRP3炎性小体激活途径下调caspase-1表达、减少白细胞介素1β与白细胞介素18的释放,从而提高疼痛阈值,缓解关节炎症及骨侵蚀,同时调控焦孔素D介导的焦亡[50]。因此,安石榴苷、白虎桂枝汤、芒果苷及肉桂酸均具有成为类风湿关节炎潜在治疗手段的应用前景。此外,
MicroRNA(miRNA)在类风湿关节炎发病机制中的调控作用也逐渐受到关注。在白细胞介素1β刺激构建的N1511细胞类风湿关节炎模型中,NLRP3、caspase-1、焦孔素D及白细胞介素1β表达显著升高,而敲除miR-144-3p可有效抑制白细胞介素1β诱导的细胞焦亡,为类风湿关节炎的基因治疗提供新思路[51]。已有多项研究探索天然植物活性成分在调控细胞焦亡中的作用,天然药物和miRNA的干预效果虽初见成效,但它们在体内的作用通路仍不明确,缺乏精准靶点阐明,尤其在安全性、靶向效率等方面仍有待深入评估。此外,未来可推进miRNA与非编码RNA调控网络研究,深入挖掘其作为诊断标志物和治疗靶点的潜力。
2.3 运动与细胞焦亡 运动对细胞焦亡的影响备受学者关注。运动对细胞焦亡影响的研究尚处于初始阶段,不同运动方式、强度、持续时间及个体差异对细胞焦亡的影响也具有差异性。下文就不同运动类型对细胞焦亡作用的相关研究进行归纳总结。
2.3.1 有氧运动与细胞焦亡 在不同运动类型中,有氧运动对细胞焦亡的影响研究较为广泛。急性有氧运动指单次力竭性运动,通常持续几分钟到数小时,这类运动的效果主要体现在运动后的立即改变,如功率自行车、力竭性游泳、马拉松等,能使机体产生应激反应。慢性有氧运动是指长期、持续进行的有氧运动,通常保持在一定的频率和强度。急性有氧运动对细胞焦亡的影响可能与机体应激机制有关。在大鼠模型中,急性有氧运动导致心肌组织中NLRP3和白细胞介素1β以及线粒体自噬基因表达增加,线粒体活性氧含量上升,出现明显的炎症细胞浸润现象,提示急性有氧运动促进活性氧的累积,诱导NLRP3寡聚化,增加焦亡蛋白的表达,增加炎症反应[52]。KHAKROO ABKENAR等[53]研究表明,高强度急性有氧运动会激活炎症复合物NLRP3的活性,中等强度急性有氧运动对 NLRP3 基因的表达以及血清白细胞介素1β和白细胞介素18水平没有显著影响,可能是中等强度急性有氧运动不足以达到氧化应激水平。在急性递增有氧运动后,未训练个体caspase-1、NLRP3、白细胞介素1β表达增加,在训练经历丰富的运动员中该指标表现为降低,表明机体炎症表达与训练水平密切相关[54]。急性有氧运动随个体训练水平的增加由促炎反应转变为抗炎反应,急性有氧运动通过激活氧化应激水平促进细胞焦亡蛋白的表达和炎症因子的分泌,进而引发细胞焦亡,然而机体对急性有氧运动会产生适应性变化,训练水平较高的运动员在进行急性有氧运动时炎症反应会受到抑制。但目前尚未见相关研究报道急性有氧运动过程中促炎反应转变为抗炎反应的原因及具体机制,因此需要进一步深入探讨。
中等强度的慢性有氧运动可有效降低炎症小体活化、抑制细胞焦亡。动物研究表明,12周中等强度跑台运动可显著抑制高脂饮食小鼠caspase-1、焦孔素D和NLRP3的表达,血清中白细胞介素1β和白细胞介素18的mRNA水平降低,抑制机体炎症反应;慢性有氧运动通过调节成纤维细胞生长因子的敏感性以及抑制NLRP3炎性小体介导的细胞焦亡相关基因表达,从而减缓炎症反应[55]。人体研究显示,经过12周的北欧步行训练,久坐的老年女性血清中NLRP3 mRNA表达及白细胞介素6、白细胞介素1β和肿瘤坏死因子α水平显著降低,提示中等强度有氧运动能够有效抑制细胞焦亡的发生[56]。另有研究表明,中等强度运动组NLRP3和caspase-1等焦亡相关指标的表达水平下降,而高强度有氧运动组上述指标提高,并且对骨关节造成更严重的负担,低强度运动组上述指标无显著变化,收益较低[57],因此,中等强度的慢性有氧运动可抑制炎症反应、降低焦亡蛋白表达。
由此可见,细胞焦亡的缓解是对有氧运动的适应变化,抑制细胞焦亡作为有效的“媒介”,在缓解炎症反应和疾病进展中发挥关键作用。基于此,中等强度有氧运动调节细胞焦亡的效果最佳,但不同运动时间和运动频率对细胞焦亡的影响鲜有相关研究报道,因此,不同形势下慢性有氧运动与细胞焦亡之间的相互关系需要大量实验进一步探究和验证。
2.3.2 抗阻运动与细胞焦亡 抗阻运动不仅有助于缓解慢性疼痛、改善身体功能,还能显著提升肌肉质量,进而间接促进骨健康。现有研究表明,不同形式、强度和持续时间的无氧运动对细胞焦亡的调控机制存在一定差异性,尚未形成统一结论。研究表明,32周的抗阻训练可显著减轻细胞焦亡程度、抑制核因子κB和caspase-1的表达,在一定程度上缓解胫骨前肌的病理改变,提示长期抗阻训练具有抑制焦亡相关蛋白和炎症因子产生的潜力[58]。另有研究发现,连续12周的抗阻训练可有效降低胰岛素抵抗小鼠海马组织中核因子κB及NLRP3炎性小体的表达水平,进而下调焦亡相关蛋白焦孔素D-N、焦孔素D以及炎症因子白细胞介素1β和白细胞介素18的表达,提示系统性抗阻训练可能通过抑制炎症通路的激活缓解中枢神经系统的细胞焦亡与炎症反应[59]。一项为期8周的抗阻训练研究显示,健康老年人血清中NLRP3和前体caspase-1水平显著下降,推测该运动方式或可通过促进细胞自噬、阻断NLRP3炎性小体的激活,从而发挥抗炎及抗焦亡作用[60]。由此可见,8-32周的抗阻运动降低焦亡相关蛋白和炎症因子的水平,同时抗阻运动通过激活细胞自噬过程,进一步抑制细胞焦亡及炎症反应,表现出潜在的抗炎和组织保护作用。但目前关于抗阻训练对细胞焦亡调控骨代谢的实验研究较少,并且不同运动强度、频率类型及个体基础状态等方面实验研究相对有限,未来需进一步明确不同抗阻运动方案对细胞焦亡的调控效应及其分子机制,建立系统的“运动-细胞焦亡-骨代谢”调控通路,为抗阻运动在骨代谢异常疾病干预中的精准应用提供理论依据。
2.3.3 高强间歇运动与细胞焦亡 高强度间歇运动可抑制炎症因子的表达。ARMANNIA等[61]研究发现,8周高强间歇和中等强度有氧训练均可降低NLRP3的表达,增加Ⅲ型结构域蛋白5的表达,Ⅲ型结构域蛋白5是一种具有调节糖脂代谢、抗氧化及抗炎等功能的肌因子,提示高强度间歇运动可能通过促进Ⅲ型结构域蛋白5分泌来抑制NLRP3炎症小体活化。LIU等[62]研究结果显示,高强度间歇训练与中等强度持续性训练两种方案均能抑制焦亡蛋白(如NLRP3、caspase-1、焦孔素D)和炎症因子(如白细胞介素1β、白细胞介素18)的表达,然而高强间歇训练更早改善神经功能,但最终结果显示中等强度持续性训练的改善效果更显著,可促进M1型促炎细胞向M2型抗炎细胞转化、抑制NLRP3表达。
综上所述,长期中等强度有氧运动在抑制炎症反应、缓解细胞焦亡发生以及维持骨代谢稳态方面具有显著优势,可能是目前较为理想的干预方式之一。然而,目前相关研究多集中于动物模型中有氧运动对细胞焦亡的调控作用,对于抗阻运动、高强度间歇运动及其他运动形式的研究相对较少,尤其缺乏系统性的机制探讨与比较分析,因此,不同类型、强度、周期及个体化的运动方案对细胞焦亡及其在骨代谢异常疾病防治中的具体调控机制,仍有待进一步深入研究。此外,当前运动干预方案多停留于基础实验阶段,它们在临床实践中的适应性和可行性仍需验证,未来可进一步研究,为精准干预骨代谢异常疾病提供科学依据与实践路径。运动与细胞焦亡的关系见表1。
2.4 运动、细胞焦亡与骨代谢异常疾病
2.4.1 运动与细胞焦亡 目前,临床药物治疗骨代谢异常疾病的效果有限,尚未达到全面治愈水平,而运动可有效预防并缓解骨关节类疾病的进展和严重程度。从解剖学角度看,骨与肌肉紧密联系,运动可通过激活肌肉-骨骼-脂肪之间的信号通路刺激细胞生物活性,增强肌肉合成代谢,进而促进骨形成[63]。有研究发现,持续6个月的运动干预可显著减少肌肉流失,对维持骨密度发挥持续的积极作用,提示长期规律运动是延缓肌肉萎缩与骨密度降低的重要策略[64]。屈娅婷等[65]研究表明,中低强度运动联合艾灸干预可显著提高血清钙、磷水平,增强成骨细胞活性,增加骨小梁体积。另有研究指出,连续8周的有氧运动可通过调节肠道微环境抑制核因子κB/NLRP3通路,下调炎症因子白细胞介素1β,减轻细胞焦亡[66]。上述研究表明,中低强度有氧运动联合艾灸可增加成骨细胞活性、提高骨密度。由此可见,长期规律运动不仅能减少肌肉流失、维持骨密度,还能通过调节多种生物标志物增强成骨细胞活性,并且中低强度运动联合重要干预也是延缓骨质流失的有效策略。
2.4.2 运动调控细胞焦亡防治骨代谢异常疾病 细胞焦亡在骨代谢异常疾病的发生发展中发挥重要作用,而科学合理的运动可作为多靶点、低成本和易实施的非药物干预手段,通过运动降低炎症反应、抑制焦亡蛋白表达、调节“肌因子”分泌和减少氧化应激,在骨代谢异常疾病的防治中发挥重要作用。该文在此对运动介导细胞焦亡防治骨代谢异常疾病的相关机制研究进行系统梳理。
(1)运动介导细胞焦亡降低炎症反应:运动干预在改善骨代谢异常方面发挥着重要的抗炎作用,可增强成骨细胞活性、抑制破骨细胞分化与活性。运动通过下调多种促炎因子的表达,如核因子κB、基质金属蛋白酶以及白细胞介素1β和白细胞介素18等,从而减轻骨组织的慢性炎症状态,延缓骨质疏松、滑膜炎症及类风湿关节炎等相关疾病的进展。SABER等[67]实验发现,在接受连续4周的游泳运动干预后,尽管小鼠体质量无明显变化,但炎症因子肿瘤坏死因子α与白细胞介素1β表达水平显著下降,基质金属蛋白酶1和基质金属蛋白酶13表达受到明显抑制,提示有氧运动能够通过降低促炎因子及基质降解酶的水平改善关节活动功能,减轻关节疼痛与肿胀症状。持续12周的跑步机运动可显著抑制白细胞介素1β、白细胞介素6、肿瘤坏死因子α和基质金属蛋白酶13的表达,上调抗炎因子白细胞介素4与白细胞介素10的表达,有效改善滑膜细胞炎症反应,增强局部抗炎能力,从而在缓解关节疼痛方面发挥了积极作用[68]。研究发现,12周的跑台运动可减缓因雌激素水平降低所致的骨小梁体积减少与骨缝宽度增加,下调白细胞介素1和白细胞介素6 mRNA表达水平,进一步说明运动能够通过调控炎症因子水平抑制破骨细胞活性、减少骨吸收、增强成骨细胞功能,从而在绝经后骨质疏松动物模型中有效促进骨量的增加[69]。
此外,破骨细胞的生成与激活受核因子κB受体活化剂和核因子κB受体抑制剂调控,其
中核因子κB受体活化剂是细胞焦亡信号通路的关键启动因子,是诱导破骨细胞分化与成熟的重要调节因子。相关研究指出,绝经后骨质疏松症患者体内肿瘤坏死因子α水平显著升高,该因子不仅参与细胞焦亡导致炎症反应,还可通过激活核因子κ信号通路增强核因子κB活化剂诱导的破骨细胞形成,最终导致骨密度加速流失;运动干预可有效抑制肿瘤坏死因子α的表达,进而阻断细胞焦亡信号的激活,抑制破骨细胞生成与活性,从而在分子层面上发挥骨保护作用[70]。因此,运动通过多重机制干预炎症反应,既可抑制破骨细胞的分化与活性,又可激活成骨相关信号通路,是改善骨代谢异常、预防和干预骨代谢异常疾病的重要策略。
(2)运动介导细胞焦亡抑制焦亡蛋白分泌:运动干预可以通过调控NLRP3/caspase-1/焦孔素D的联级通路抑制细胞焦亡的发生,从而改善骨代谢紊乱状态。过度的细胞焦亡不仅引发组织细胞广泛死亡,其裂解过程中释放的炎症因子等递质可加剧局部炎症反应,进一步激活并放大细胞焦亡的联级效应,抑制NLRP3的表达可作为防止细胞焦亡的靶向干预手段[71]。研究发现,负重有氧跑步可显著改善骨质疏松模型大鼠的身体组成,降低NLRP3、焦孔素D和caspase-1蛋白表达水平,从而抑制细胞焦亡下游通路中炎症因子的释放[72]。运动可从转录和翻译2个层面协同调控焦亡信号,有效缓解组织炎症与细胞损伤,改善细胞焦亡的严重程度。YANG等[73]的研究显示,4周中等强度跑步机运动可抑制核因子κB p65的核转位及NLRP3炎症小体的组装,显著降低下游焦亡蛋白caspase-1的表达,从而减轻软骨细胞的焦亡程度,延缓软骨退变过程,表明有氧运动可通过调节核因子κB/NLRP3信号通路发挥抑制焦亡、保护软骨作用,有助于改善骨关节炎的病理过程。有研究报道,持续6周的高强度间歇运动干预可显著抑制脂多糖诱导的NLRP3炎症小体激活,抑制细胞焦亡相关蛋白的表达,降低炎性细胞因子的释放,从而有效减轻骨组织的炎症损伤[74],进一步拓展了运动干预在调控焦亡通路、干预骨代谢异常疾病中的潜在价值。因此,运动通过调控NLRP3/caspase-1/焦孔素D介导的细胞焦亡信号通路,在降低炎症水平、保护骨组织结构、改善骨代谢异常等方面具有显著作用,具有成为骨代谢相关疾病干预手段的潜力。未来可进一步深入探究不同运动方式、强度及周期对焦亡通路的调控效果,以优化运动处方的个体化干预策略。
(3)运动介导细胞焦亡调节肌因子分泌:运动通过调节肌因子的分泌抑制焦亡蛋白和炎症因子的活化,进而防治骨代谢异常疾病。“肌因子”是机体在运动过程中由肌肉、脂肪等组织器官分泌的生物活性分子,在调节机体代谢和炎症反应方面具有重要作用。目前,在运动介导细胞焦亡防治骨代谢异常疾病的研究中,主要涉及镍纹蛋白样蛋白和鸢尾素。镍纹蛋白样蛋白具有抗炎作用,在骨代谢性疾病中可缓解病程进展。LIU等[75]研究发现,低、中、高3种运动强度均可提高SD大鼠血清和滑液中镍纹蛋白样蛋白含量,其中中等强度运动组镍纹蛋白样蛋白增加幅度最大,同时伴有基质金属蛋白酶13和核因子κB mRNA 表达抑制以及Ⅱ型胶原蛋白表达的恢复。提示运动通过提升镍纹蛋白样蛋白水平发挥抗炎作用,进而抑制NLRP3/caspase-1/焦孔素D联级反应,减轻软骨细胞焦亡。
鸢尾素能够促进成骨细胞的增殖、分化和矿化,调节破骨细胞的分化与成熟[76]。JIA等[77]研究发现,中等强度运动可诱导鸢尾素分泌达到最高水平,对缓解骨代谢异常具有较好的治疗效果;鸢尾素通过抑制核因子κB 信号通路降低 NLRP3 和 caspase-1 的表达,恢复软骨细胞中Ⅱ型胶原蛋白的表达,激活成骨细胞活性。另有研究表明,8 周体育锻炼可改善骨小梁的微观结构、提高鸢尾素表达、促进Ⅱ型肌纤维比例的增加、减弱中枢焦亡反应,从而抑制破骨细胞活性,促进骨形成[78],提示鸢尾素在小鼠中可促进骨形成、增加骨密度,具有较强的合成代谢作用。因此,中等强度的运动有利于镍纹蛋白样蛋白和鸢尾素的分泌,增强成骨细胞活性,提高骨密度,改善骨代谢异常所致的骨质流失。此外,运动过程中肌肉分泌的白细胞介素6也具有抗炎作用。有研究表明,类风湿关节炎患者在运动前1 h白细胞介素6水平迅速升高,随后逐渐下降,并在运动后 24 h恢复至基线水平[79],说明运动可促进抗炎因子的分泌,但肌肉来源的白细胞介素6在运动过程中的具体炎症调节作用仍需进一步研究。目前,关于运动调控肌因子以改善细胞焦亡进而防治类风湿关节炎的直接研究相对较少,尚需更深入的机制探索予以证实。
(4)运动介导细胞焦亡减缓氧化应激水平:运动干预还可通过减轻氧化应激水平抑制细胞焦亡,改善骨代谢异常疾病。嘌呤能受体作为钠钾离子通道,在炎症反应中发挥着“分子开关”的关键调控作用,可导致细胞内钾离子外流,而钾离子外排被认为是激活NLRP3炎性小体的重要信号,进而诱导细胞焦亡联级反应。LI等[80]研究表明,中等强度跑台运动组小鼠中NLRP3、caspase-1及白细胞介素1β的表达显著下降。值得注意的是,嘌呤能受体的表达在不同强度运动之间表现出非线性变化趋势:在中等强度运动组中表达水平最低,而自噬相关指标则明显升高,同时伴随骨赘形成减少和关节间隙变窄程度减轻。研究提示适度激活嘌呤能受体可能通过诱导细胞自噬发挥保护作用,而过度激活则可能促进软骨细胞和滑膜细胞的焦亡,加剧炎症损伤;持续8周的跑步机运动干预可显著抑制小鼠体内NLRP3炎性小体、caspase-1、焦孔素D、白细胞介素1β与白细胞介素18等关键焦亡因子的表达,降低活性氧及脂质过氧化产物丙二醛水平。提示运动能够减缓氧化应激反应,减轻炎症损伤,同时该干预也增强了细胞自噬能力,促进成骨细胞活性,提高骨组织重建与代谢功能,进而对糖尿病相关的代谢性骨质疏松产生积极影响。因此,运动可通过促进细胞自噬、抑制炎症因子及细胞焦亡信号通路的激活从而有效改善骨代谢紊乱,为骨代谢异常疾病的非药物干预提供了重要的理论支持与实践依据。
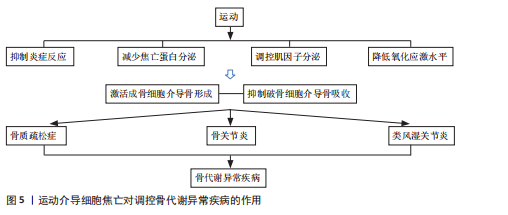
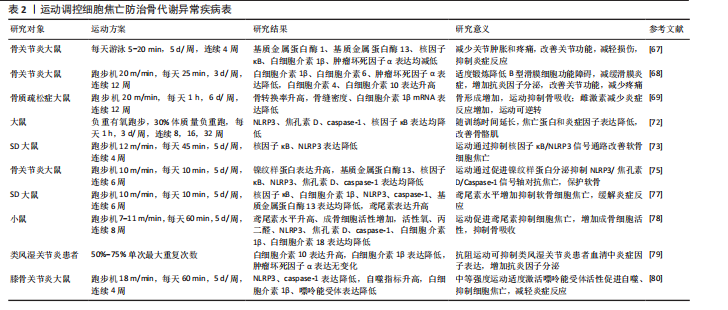
综上所述,运动干预在防治骨代谢异常疾病中展现出多重积极效应,尤其在抑制炎症反应和焦亡相关蛋白表达方面作用显著;同时,运动通过调节肌因子分泌和增强细胞自噬、降低活性氧水平减轻炎症损伤,抑制细胞焦亡。然而,当前多数研究仍集中于动物实验,运动方案在临床中的适应性与推广性仍有待验证。未来亟需构建更贴近人体生理特点的运动干预模式,以期为骨代谢异常疾病的个体化防治提供更科学、精准的理论依据和实践路径。运动调控细胞焦亡防治骨代谢异常疾病的具体机制,详见表2。 运动介导细胞焦亡对调控骨代谢异常疾病的作用,见图5。
| [1] FLORENCIO-SILVA R, SASSO GR, SASSO-CERRI E, et al. Biology of Bone Tissue: Structure, Function, and Factors That Influence Bone Cells. Biomed Res Int. 2015;2015:e421746. [2] 龚卫月,王利敏,朱洁.数据挖掘联合网络药理学与分子对接分析淫羊藿-丹参治疗肾虚血瘀型骨质疏松的作用机制[J].重庆医学, 2025,54(7):1552-1559,1565. [3] 魏福,陈志伟.骨关节炎:一个持续存在的挑战[J].中南医学科学杂志,2023,51(2):157-160. [4] SALEHI S, MAHMOUDINEZHAD DEZFOULIS M, AZADEH H, et al. Immune dysregulation and pathogenic pathways mediated by common infections in rheumatoid arthritis. Folia Microbiol (Praha). 2023;68(3):325-335. [5] 朱跃亚,潘志,王颖航.类风湿关节炎中细胞焦亡的研究现状[J].中国临床药理学杂志, 2025,41(7):1026-1031. [6] LONG F. Building strong bones: molecular regulation of the osteoblast lineage. Nat Rev Mol Cell Biol. 2011;13(1):27-38. [7] ZYCHLINSKY A, PREVOST MC, SANSONETTI PJ. Shigella flexneri induces apoptosis in infected macrophages. Nature. 1992;358(6382):167-169. [8] SHI J, ZHAO Y, WANG K, et al. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature. 2015;526(7575):660-675. [9] KETELUT-CARNEIRO N, FITZGERALD KA. Apoptosis, Pyroptosis, and Necroptosis-Oh My! The Many Ways a Cell Can Die. J Mol Biol. 2022; 434(4):e167378. [10] FU J, WU H. Structural Mechanisms of NLRP3 Inflammasome Assembly and Activation. Annu Rev Immunol. 2023;41:301-316. [11] RAO Z, ZHU Y, YANG P, et al. Pyroptosis in inflammatory diseases and cancer. Theranostics. 2022;12(9):4310-4329. [12] ROZARIO P, PINILLA M, GORSE L, et al. Mechanistic basis for potassium efflux-driven activation of the human NLRP1 inflammasome. Proc Natl Acad Sci U S A. 2024;121(2):e2309579121. [13] MAKONI NJ, NICHOLS MR. The intricate biophysical puzzle of caspase-1 activation. Arch Biochem Biophys. 2021;699:e108753. [14] MAN SM, KARKI R, KANNEGANTI TD. Molecular mechanisms and functions of pyroptosis, inflammatory caspases and inflammasomes in infectious diseases. Immunol Rev. 2017;277(1):61-75. [15] YI YS. Caspase-11 non-canonical inflammasome: a critical sensor of intracellular lipopolysaccharide in macrophage - mediated inflammatory responses. Immunology. 2017;152(2):207-217. [16] QADIR A, LIANG S, WU Z, et al. Senile Osteoporosis: The Involvement of Differen- tiation and Senescence of Bone Marrow Stromal Cells. Int J Mol Sci. 2020;21(1):349-355. [17] TAO Z, WANG J, WEN K, et al. Pyroptosis in Osteoblasts: A Novel Hypothesis Underlying the Pathogenesis of Osteoporosis. Front Endocrinol (Lausanne). 2021;11:e548812. [18] MOTTA F, BARONE E, SICA A, et al. Inflammaging and Osteoarthritis. Clin Rev Allergy Immunol. 2023;64(2):222-238. [19] LEI L, SUN J, HAN J, et al. Interleukin-17 induces pyroptosis in osteoblasts through the NLRP3 inflammasome pathway in vitro. Int Immunopharmacol. 2021;96:e107781. [20] RAN S, CHU M, GU S, et al. Enterococcus faecalis induces apoptosis and pyroptosis of human osteoblastic MG63 cells via the NLRP3 inflammasome. Int Endod J. 2019;52(1):44-53. [21] ZHU X, ZHANG K, LU K, et al. Inhibition of pyroptosis attenuates Staphylococcus aureus-induced bone injury in traumatic osteomyelitis. Ann Transl Med. 2019;7(8):170-175. [22] YANG L, LIU J, SHAN Q, et al. High glucose inhibits proliferation and differentiation of osteoblast in alveolar bone by inducing pyroptosis. Biochem Biophys Res Commun. 2020;522(2):471-478. [23] ZHANG J, WEI K. Necrosulfonamide reverses pyroptosis-induced inhibition of proliferation and differentiation of osteoblasts through the NLRP3/caspase-1/GSDMD pathway. Exp Cell Res. 2021;405(2):e112648. [24] OSTA B, BENEDETTI G, MIOSSEC P. Classical and Paradoxical Effects of TNF-α on Bone Homeostasis. Front Immunol. 2014;5:48-58. [25] MARAHLEH A, KITAURA H, OHORI F, et al. TNF-α Directly Enhances Osteocyte RANKL Expression and Promotes Osteoclast Formation. Front Immunol. 2019;10:23-29. [26] CHEN Y, YANG Q, LV C, et al. NLRP3 regulates alveolar bone loss in ligature-induced periodontitis by promoting osteoclastic differentiation. Cell Prolif. 2021;54(2):e12973. [27] QU C, BONAR SL, HICKMAN-BRECKS CL, et al. NLRP3 mediates osteolysis through inflammation-dependent and -independent mechanisms. FASEB J. 2015;29(4):1269-1279. [28] HUNTER DJ, BIERMA-ZEINSTRA S. Osteoarthritis. Lancet. 2019;393(10182):1745-1759. [29] KATZ JN, ARANT KR, LOESER RF. Diagnosis and Treatment of Hip and Knee Osteoarthritis: A Review. JAMA. 2021;325(6):568-578. [30] SALUCCI S, FALCIERI E, BATTISTELLI M. Chondrocyte death involvement in osteoarthritis. Cell Tissue Res. 2022;389(2):159-170. [31] FRISCIC J, BOTTCHER M, REINWALD C, et al. The complement system drives local inflammatory tissue priming by metabolic reprogramming of synovial fibroblasts. Immunity. 2021;54(5):1002-1021. [32] HU Q, ECKER M. Overview of MMP-13 as a Promising Target for the Treatment of Osteoarthritis. Int J Mol Sci. 2021;22(4):1742-1754. [33] PAN D, YIN P, LI L, et al. Holomycin, a novel NLRP3 inhibitor, attenuates cartilage degeneration and inflammation in osteoarthritis. Biochem Biophys Res Commun. 2023;657:59-68. [34] WU Y, YING J, ZHU X, et al. Pachymic acid suppresses the inflammatory response of chondrocytes and alleviates the progression of osteoarthritis via regulating the Sirtuin 6/NF-κB signal axis. Int Immunopharmacol. 2023;124(Pt A): e110854. [35] CHEN H, TU M, LIU S, et al. Dendrobine Alleviates Cellular Senescence and Osteoarthritis via the ROS/NF-κB Axis. Int J Mol Sci. 2023;24(3):53-65. [36] FUJII Y, LIU L, YAGASAKI L, et al. Cartilage homeostasis and osteoarthritis. Int J Mol Sci. 2022;23(11):63-76. [37] BUHRMANN C, BROCKMUELLER A, MUELLER AL, et al. Curcumin Attenuates Environment-Derived Osteoarthritis by Sox9/NF-kB Signaling Axis. Int J Mol Sci. 2021;22(14):65-76. [38] FENG Z, ZHENG W, LI X, et al. Cryptotanshinone protects against IL-1β-induced inflammation in human osteoarthritis chondrocytes and ameliorates the progression of osteoarthritis in mice. Int Immunopharmacol. 2017;50:161-167. [39] 王毅,姜涛,刘文刚,等.补肾强筋胶囊通过调控NLRP3炎症小体介导的细胞焦亡改善KOA滑膜炎症和纤维化的实验研究[J].世界科学技术-中医药现代化,2024,26(6):1471-1480. [40] XIAO Y, DING L, YIN S, et al. Relationship between the pyroptosis of fibroblast-like synoviocytes and hmgb1 secretion in knee osteoarthritis. Mol Med Rep. 2021;23(2):97-107. [41] JANG S, KWON EJ, LEE JJ. Rheumatoid Arthritis: Pathogenic Roles of Diverse Immune Cells. Int J Mol Sci. 2022;23(2):905-916. [42] RADU AF, BUNGAU SG. Management of Rheumatoid Arthritis: An Overview. Cells. 2021; 10(11):28-37. [43] ZHAI Z, YANG F, XU W, et al. Attenuation of Rheumatoid Arthritis Through the Inhibition of Tumor Necrosis Factor-Induced Caspase 3/Gasdermin E-Mediated Pyroptosis. Arthritis Rheumatol. 2022;74(3):427-440. [44] FEARON U, CANAVAN M, BINIECKA M, et al. Hypoxia, mitochondrial dysfunction and synovial invasiveness in rheumatoid arthritis. Nat Rev Rheumatol. 2016;12(7):385-397. [45] WANG X, CHEN Z, FAN X, et al. Inhibition of DNM1L and mitochondrial fission attenuates inflammatory response in fibroblast-like synoviocytes of rheumatoid arthritis. J Cell Mol Med. 2020;24(2):1516-1528. [46] HONG Z, ZHANG X, ZHANG T, et al. The ROS/GRK2/HIF-1α/NLRP3 Pathway Mediates Pyroptosis of Fibroblast-Like Synoviocytes and the Regulation of Monomer Derivatives of Paeoniflorin. Oxid Med Cell Longev. 2022;2022:e4566851. [47] RAJAMÄKI K, NORDSTRÖM T, NURMI K, et al. Extracellular acidosis is a novel danger signal alerting innate immunity via the NLRP3 inflammasome. J Biol Chem. 2013;288(19):13410-13419. [48] WU X, REN G, ZHOU R, et al. The role of Ca2+ in acid-sensing ion channel 1a-mediated chondrocyte pyroptosis in rat adjuvant arthritis. Lab Invest. 2019;99(4):499-513. [49] GE G, BAI J, WANG Q, et al. Punicalagin ameliorates collagen-induced arthritis by downregulating M1 macrophage and pyroptosis via NF-κB signaling pathway. Sci China Life Sci. 2022;65(3):588-603. [50] LI W, WANG K, LIU Y, et al. A Novel Drug Combination of Mangiferin and Cinnamic Acid Alleviates Rheumatoid Arthritis by Inhibiting TLR4/NFκB/NLRP3 Activation-Induced Pyroptosis. Front Immunol. 2022;13:912-933. [51] JIANG JM, MO ML, LONG XP, et al. MiR-144-3p induced by SP1 promotes IL-1β-induced pyroptosis in chondrocytes via PTEN/PINK1/Parkin axis. Autoimmunity. 2022;55(1):21-31. [52] LI H, MIAO W, MA J, et al. Acute Exercise-Induced Mitochondrial Stress Triggers an Inflammatory Response in the Myocardium via NLRP3 Inflammasome Activation with Mitophagy. Oxid Med Cell Longev. 2016;2016:e1987149. [53] KHAKROO ABKENAR I, RAHMANI-NIA F, LOMBARDI G. The Effects of Acute and Chronic Aerobic Activity on the Signaling Pathway of the Inflammasome NLRP3 Complex in Young Men. Medicina. 2019;55(4):105-114. [54] COMASSI M, SANTINI E, ROSSI C, et al. The level of physical training modulates cytokine levels through P2X7 receptor in healthy subjects. Eur J Clin Invest. 2018;48(2):e29272042. [55] LI XH, LIU LZ, CHEN L, et al. Aerobic exercise regulates FGF21 and NLRP3 inflammasome-mediated pyroptosis and inhibits atherosclerosis in mice. PLoS One. 2022;17(8):e0273527. [56] GOMARASCA M, MICIELSKA K, FARALDI M, et al. Impact of 12-Week Moderate-Intensity Aerobic Training on Inflammasome Complex Activation in Elderly Women. Front Physiol. 2022;13:e792859. [57] LI Z, HUANG Z, ZHANG H, et al. Moderate-intensity exercise alleviates pyroptosis by promoting autophagy in osteoarthritis via the P2X7/AMPK/mTOR axis. Cell Death Discov. 2021;7(1):346-356. [58] 龚丽景,马芳源,杨璐瑶,等.细胞焦亡在长期抗阻训练对增龄大鼠胫骨前肌蛋白质代谢影响中的调节作用[J].中国运动医学杂志, 2022,41(12):956-965. [59] JI RF, BIAN XP, LIU BB, et al. Effects of resistance exercise on pyroptosis-related proteins in hippocampus of insulin resistant mice. Zhongguo Ying Yong Sheng Li Xue Za Zhi. 2020;36(5):456-461. [60] MEJIAS-PENA Y, ESTÉBANEZ B, RODRIGUEZ-MIGUELEZ P, et al. Impact of resistance training on the autophagy-inflammation-apoptosis crosstalk in elderly subjects. Aging (Albany NY). 2017;9(2):408-418. [61] ARMANNIA F, GHAZALIAN F, SHADNOUSH M, et al. Effects of High-Intensity Interval Vs. Moderate-Intensity Continuous Training on Body Composition and Gene Expression of ACE2, NLRP3, and FNDC5 in Obese Adults: A Randomized Controlled Trial. Med J Islam Repub Iran. 2022;36:161-172. [62] LIU MX, LUO L, FU JH, et al. Exercise-induced neuroprotection against cerebral ischemia/reperfusion injury is mediated via alleviating inflammasome-induced pyroptosis. Exp Neurol. 2022;349:e113952. [63] HERRMANN M, ENGELKE K, EBERT R, et al. Interactions between Muscle and Bone-Where Physics Meets Biology. Biomolecules. 2020; 10(3):432-442. [64] 汪聪莹.糖尿病前期人群运动、营养干预后一年随访形态与糖、脂、骨代谢变化研究[D].上海:上海体育学院,2017. [65] 屈娅婷,何军锋,张国民,等.肾虚矮小症雄性大鼠骨代谢异常及艾灸和运动综合方案干预的研究[J].中医药导报,2008,14(11):13-15. [66] 雷森林,谌晓安,宋为正,等.运动通过微生物-肠-脑轴调控NF-κB/NLRP3通路抑制细胞焦亡改善肥胖相关认知障碍[J].微生物学通报, 2025,52(7):3222-3244. [67] SABER MM, MAHMOUD MM, AMIN HM, et al. Therapeutic effects of combining curcumin and swimming in osteoarthritis using a rat model. Biomed Pharmacother. 2023;166:e115309. [68] CASTROGIOVANNI P, DI ROSA M, RAVALLI S, et al. Moderate Physical Activity as a Prevention Method for Knee Osteoarthritis and the Role of Synoviocytes as Biological Key. Int J Mol Sci. 2019;20(3):511-523. [69] LI L, CHEN X, LV S, et al. Influence of exercise on bone remodeling-related hormones and cytokines in ovariectomized rats: a model of postmenopausal osteoporosis. PLoS One. 2014; 9(11):e112845. [70] ZHA L, HE L, LIANG Y, et al. TNF-α contributes to postmenopausal osteoporosis by synergistically promoting RANKL-induced osteoclast formation. Biomed Pharmacother. 2018;102:369-374. [71] 耿珑玉,盛黎,白硕,等.细胞焦亡在运动系统疾病中的作用及相关分子机制[J].中国组织工程研究,2025,29(26):5695-5703. [72] 付鹏宇,杨璐瑶,唐舒宁,等.基于细胞焦亡PCR芯片分析负重跑训练对增龄大鼠骨骼肌丢失的影响[J].中国实验动物学报,2023,31(2):208-216. [73] YANG Y, WANG Y, KONG Y, et al. Moderate Mechanical Stimulation Protects Rats against Osteoarthritis through the Regulation of TRAIL via the NF-κB/NLRP3 Pathway. Oxid Med Cell Longev. 2020;2020:e6196398. [74] 张铭宸,李婷婷,张慧,等.高强度间歇运动通过调控NLRP3炎症小体和M1巨噬细胞极化改善脓毒症心肌损伤[J].免疫学杂志,2024, 40(4):337-345,352. [75] LIU J, JIA S, YANG Y, et al. Exercise induced meteorin-like protects chondrocytes against inflammation and pyroptosis in osteoarthritis by inhibiting PI3K/Akt/NF-κB and NLRP3/caspase-1/GSDMD signaling. Biomed Pharmacother. 2023; 158:e114118. [76] HU X, WANG Z, WANG W, et al. Irisin as an agent for protecting against osteoporosis: A review of the current mechanisms and pathways. J Adv Res. 2024;62:175-186. [77] JIA S, YANG Y, BAI Y, et al. Mechanical Stimulation Protects Against Chondrocyte Pyroptosis Through Irisin-Induced Suppression of PI3K/Akt/NF-κB Signal Pathway in Osteoarthritis. Front Cell Dev Biol. 2022;10:e797855. [78] BEHERA J, ISON J, VOOR MJ, et al. Exercise-Linked Skeletal Irisin Ameliorates Diabetes- Associated Osteoporosis by Inhibiting the Oxidative Damage-Dependent miR -150 -FNDC5 / Pyroptosis Axis. Diabetes. 2022;71(12):2777-2792. [79] PEREIRA NUNES PINTO AC, NATOUR J, DE MOURA CASTRO CH, et al. Acute effect of a resistance exercise session on markers of cartilage breakdown and inflammation in women with rheumatoid arthritis. Int J Rheum Dis. 2017;20(11):1704-1713. [80] LI Z, HUANG Z, ZHANG H, et al. Moderate-intensity exercise alleviates pyroptosis by promoting autophagy in osteoarthritis via the P2X7/AMPK/mTOR axis. Cell Death Discov. 2021;7(1):346-356. |
| [1] | 张庆彤, 陈乐琴, 刘昶, 陈昱廷, 郭睿武. 内源性大麻素系统调控运动动机的神经机制[J]. 中国组织工程研究, 2026, 30(在线): 1-11. |
| [2] | 于晨锜, 刘洋, 余建锋, 康康, 邓垚歌, 夏小伟, 张一健, 朱雪松. 仿生黑磷纳米系统调控滑膜巨噬细胞极化治疗骨关节炎[J]. 中国组织工程研究, 2026, 30(在线): 1-13. |
| [3] | 陈秋函, 杨 龙, 袁代柱, 吴展羽, 邹梓豪, 叶 川. 膝关节周围截骨治疗膝骨关节炎:治疗策略的优化[J]. 中国组织工程研究, 2026, 30(9): 2303-2312. |
| [4] | 张子峥, 罗 旺, 刘长路. 膝内侧间室骨关节炎单髁置换中有限元分析的应用价值[J]. 中国组织工程研究, 2026, 30(9): 2313-2322. |
| [5] | 刘金龙, 阿卜杜吾普尔•海比尔, 白 臻, 苏丹阳, 苗 鑫, 李 菲, 杨晓鹏. 不同非手术方法治疗青少年特发性脊柱侧凸效果的系统综述与网状Meta分析[J]. 中国组织工程研究, 2026, 30(9): 2370-2379. |
| [6] | 张 楠, 孟庆华, 鲍春雨. 踝关节有限元模型的特性及临床应用[J]. 中国组织工程研究, 2026, 30(9): 2343-2349. |
| [7] | 刘文龙, 董 磊, 肖争争, 聂 宇. 骨质疏松患者行固定平台单髁置换后胫骨假体松动的有限元分析[J]. 中国组织工程研究, 2026, 30(9): 2191-2198. |
| [8] | 李智斐, 韩 斌, 柳秋丽, 张展鸣, 韦浩凯, 左匡时, 张翼升. 基于动作捕捉技术分析神经根型颈椎病患者的颈椎运动特征[J]. 中国组织工程研究, 2026, 30(9): 2286-2293. |
| [9] | 黎清斌, 林建辉, 黄文杰, 王明爽, 杜间开, 劳永锵. 膝关节周围骨巨细胞瘤病灶扩大刮除后填充骨水泥:软骨下植骨与不植骨的比较[J]. 中国组织工程研究, 2026, 30(8): 1896-1902. |
| [10] | 胡雄科, 刘少华, 谭 谦, 刘 昆, 朱光辉. 紫草素干预骨髓间充质干细胞改善老年小鼠股骨的微结构[J]. 中国组织工程研究, 2026, 30(7): 1609-1615. |
| [11] | 宋浦蓁, 马贺宾, 陈宏广, 章亚东. 骨髓间充质干细胞外泌体联合转化生长因子β1对巨噬细胞的作用[J]. 中国组织工程研究, 2026, 30(7): 1616-1623. |
| [12] | 潘 冬, 杨加玲, 田 卫, 王东济, 朱 政, 马文超, 刘 娜, 付常喜. 抗阻运动激活衰老大鼠骨骼肌卫星细胞:脂联素受体1途径的作用[J]. 中国组织工程研究, 2026, 30(7): 1736-1746. |
| [13] | 曹 涌, 滕虹良, 邰鹏飞, 李骏达, 朱腾旗, 李兆进. 细胞因子和卫星细胞在肌肉再生中的相互作用[J]. 中国组织工程研究, 2026, 30(7): 1808-1817. |
| [14] | 李林臻, 焦泓焯, 陈伟南, 张铭哲, 王建龙, 张君涛. 淫羊藿苷含药血清对脂多糖诱导人软骨细胞炎症损伤的影响[J]. 中国组织工程研究, 2026, 30(6): 1368-1374. |
| [15] | 钟彩红, 肖晓歌, 李 明, 林剑虹, 洪 靖. 运动相关髌腱炎发病的生物力学机制[J]. 中国组织工程研究, 2026, 30(6): 1417-1423. |
运动干预能够通过多种机制调节细胞焦亡的发生,从而改善骨代谢异常的病理过程。细胞焦亡引起的炎症反应可刺激机体发挥免疫功能,然而过度的炎症反应可增加病理进程,如类风湿关节炎、骨关节炎和滑膜炎症等。运动可通过抑制核因子κB激活减少焦亡蛋白NOD样受体热蛋白结构域相关蛋白3(NOD-like receptor thermal protein domain associated protein 3,NLRP3)活化,减缓caspase-1切割白细胞介素1β和白细胞介素18,抑制焦孔素D激活细胞焦亡,减少炎症因子释放;同时运动诱导抗炎因子白细胞介素6的释放,形成抗炎环境,减少氧化应激状态,有利于抑制细胞焦亡信号通路;此外,运动时产生的多种细胞因子可增加成骨细胞活性,缓解骨密度流失。然而,目前相关综述多聚焦于细胞焦亡在骨代谢异常疾病中的作用机制,关于运动通过调控细胞焦亡改善骨代谢异常疾病的综述性研究仍相对匮乏。因此,该文梳理了细胞焦亡在骨质疏松症、骨关节炎和类风湿关节炎等骨代谢异常疾病中的作用机制,总结不同运动形式、不同运动强度对细胞焦亡的影响以及运动调控细胞焦亡改善骨代谢异常疾病的分子生物学机制,以期为运动干预细胞焦亡防治骨代谢异常疾病提供参考。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
1.1 资料来源
1.1.1 检索人及检索时间 由第一作者在2025年4月进行检索。
1.1.2 文献检索时限 1992年1月至2025年4月。
1.1.3 检索数据库 PubMed数据库和中国知网。
1.1.4 检索词 以“exercise,pyroptosis,Osteoporosis,Osteoarthritis,rheumatoid arthritis,Abnormal bone metabolism,Osteoblasts,Osteoclasts,Bone marrow mesenchymal stem cells,osteocytes,chondrocytes”为英文检索词,以“运动,细胞焦亡,骨关节炎,骨质疏松症,类风湿性关节炎,骨代谢异常,成骨细胞,破骨细胞,骨髓间充质干细胞,骨细胞及软骨细胞”为中文检索词。
1.1.5 检索文献类型 研究原著、综述、荟萃分析、学位论文。
1.1.6 检索策略 采用主题词和自由词结合的方式进行检索,检索策略见图1。
1.2 入组标准
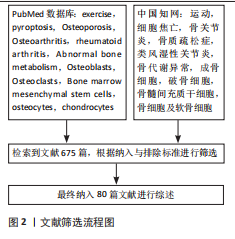
纳入标准:①细胞焦亡与骨代谢异常疾病相关文献;②运动影响细胞焦亡相关文献;③运动介导细胞焦亡调控骨代谢异常疾病相关文献。
排除标准:①讲座、会议摘要;②数据报告不完整或数据无法索取的文章;③重复研究。
1.3 文献质量评估和数据的提取 初步检索得到675篇文献,其中PubMed数据库517篇、中国知网158篇,排除595篇文献,最终纳入80篇文献。文献筛选检索流程见图2。
3.1 既往他人在该领域研究的贡献和存在的问题 研究者们揭示细胞焦亡的分子机制以及在骨质疏松症、骨关节炎、类风湿关节炎等骨代谢异常疾病中的相关通路,认为细胞焦亡与骨代谢异常疾病的发生发展紧密相关,探索通过抑制细胞焦亡来增强骨形成抑制骨吸收的效果。然而,对运动影响细胞焦亡信号通路抑制骨代谢异常疾病发展的作用研究较少。现有证据表明,运动可通过下调肿瘤坏死因子α、白细胞介素1β、白细胞介素18等促炎因子抑制NLRP3/caspase-1/焦孔素D通路的活化,减少骨组织中细胞焦亡的发生,减缓破骨进程并激活成骨细胞功能;同时,运动还能通过调节肌因子的分泌、增强抗氧化能力与细胞自噬,减轻炎症损伤,维持骨代谢稳态。尽管研究聚焦于有氧运动与中强度运动对细胞焦亡的影响,目前尚缺乏关于抗阻运动及高强间歇运动等调控细胞焦亡进而干预骨代谢异常疾病的系统研究,因此,未来研究应在运动方式的基础上进一步聚焦于不同类型、强度和周期的运动,深入挖掘它们在调控细胞焦亡、延缓骨质退化、促进骨健康中的潜力机制。
3.2 该综述区别于他人他篇的特点 既往研究多关注运动干预骨代谢异常疾病的效果,该综述提供更为全面的视角,以细胞焦亡为切入点,深入探讨细胞焦亡在骨质疏松症、骨关节炎和类风湿关节炎中的分子机制,较为全面地总结了运动对细胞焦亡的影响以及运动介导细胞焦亡调控骨代谢异常疾病的研究进展;阐述了运动通过调节细胞焦亡抑制骨代谢异常的可能机制,包括抑制焦亡蛋白NLRP3、caspase-1、核因子κB和炎症因子白细胞介素1β、白细胞介素18、基质金属蛋白酶等表达,调节肌因子分泌以及减少氧化应急水平等及通路间的相互作用,为防治骨代谢异常疾病提供了理论参考。
3.3 该综述的局限性 近年来,关于细胞焦亡在骨质疏松症、骨关节炎和类风湿关节炎中的作用机制相关研究逐渐增多,但运动调控细胞焦亡影响骨质疏松症、骨关节炎和类风湿关节炎的研究相对较少,因此纳入的文献数量有限;现阶段尚缺乏不同运动形式、运动强度及运动时间对细胞焦亡产生影响的相关;此外,实验主要集中在动物实验或细胞实验,有关细胞焦亡对骨代谢异常疾病影响的临床研究较少。因此,纳入的研究中,可能会因实验方法和研究对象不同,结果存在一定偏差。
3.4 该综述的重要意义 文章归纳总结了细胞焦亡在骨代谢异常疾病中的研究进展,为治疗和预防骨代谢异常疾病提供了临床思路,探究新的有效治疗手段具有重要的社会及经济意义。通过分析细胞焦亡在骨质疏松症、骨关节炎和类风湿性关节炎中的分子机制,讨论不同运动方式、强度和时间在骨代谢异常疾病中的用机制及相关治疗策略。此外,运动作为缓解细胞焦亡发生的有效方式,可通过抑制核因子κB转录,抑制NLRP3/caspase-1/焦孔素D信号通路减少炎症因子表达,调控肌因子分泌,降低氧化应激水平带来的影响,进而改善骨代谢异常疾病。进一步明确运动、细胞焦亡和骨代谢异常疾病三者之间的动态相互作用,可为运动通过调节细胞焦亡防治骨代谢异常疾病提供理论依据和新思路。然而,目前介导细胞焦亡治疗骨代谢异常疾病的研究大都基于体外或动物模型实验,而临床试验较少,由于啮齿动物和人类之间焦亡的途径存在差异,相关研究仍具有局限性。
3.5 课题组专家的意见 细胞焦亡广泛参与骨代谢异常相关疾病的发病机制,它们的潜在联系仍有待进一步系统性阐明。目前,运动对细胞焦亡影响的研究主要集中于有氧运动,未来研究应拓展至不同类型的运动形式、运动强度及持续时间对细胞焦亡调控作用的探讨,进一步解析运动潜在的分子机制。现阶段相关研究多数依赖于动物实验,临床研究仍较为匮乏,后续应注重实验成果的临床转化,推动人类研究的发展。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
细胞焦亡抑制成骨细胞活性,激活破骨细胞,引起强烈炎症反应,进而导致骨代谢失衡,加剧骨代谢异常疾病的发展。因此,抑制炎症因子分泌和降低焦亡蛋白转录,对于预防骨代谢异常疾病至关重要。运动作为经济有效的手段有助于成骨细胞激活,加快骨的修复和再生,减缓骨质流失,降低炎症反应,其机制可能与有氧运动抑制NLRP3、caspase-1、GSDMD的表达,减少白细胞介素的释放,调节肌因子分泌和改善氧化应激水平有关,且中等强度的运动效果最佳。抗阻运动可缓解疼痛和增强肌肉力量间接促进骨骼健康。高强间歇运动可调节肌因子分泌,增加抗炎和抗氧化功能。因此,通过合理的运动调节细胞焦亡信号通路,延缓关节退化进程,对骨代谢异常疾病的防治具有重要意义。
中国组织工程研究杂志出版内容重点:干细胞;骨髓干细胞;造血干细胞;脂肪干细胞;肿瘤干细胞;胚胎干细胞;脐带脐血干细胞;干细胞诱导;干细胞分化;组织工程
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||