中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (32): 8512-8520.doi: 10.12307/2026.286
• 生物材料综述 biomaterial review • 上一篇 下一篇
水凝胶在运动损伤相关骨组织工程修复中的应用与进展
金 媛1,周加兵2
- 1南京体育学院,江苏省南京市 210014;2广东石油化工学院体育学院,广东省茂名市 525000
-
接受日期:2025-12-06出版日期:2026-11-18发布日期:2026-04-29 -
通讯作者:周加兵,讲师,广东石油化工学院体育学院,广东省茂名市 525000 -
作者简介:金媛,女,1993年生,江苏省淮安市人,汉族,硕士,主要从事运动与大众健康方面的研究。 -
基金资助:广东石油化工学院校级科研基金项目(2024rcyj1049,2024rcyj1050),项目负责人:周加兵
Applications and advances of hydrogels in bone tissue engineering repair related to sports injuries
Jin Yuan1, Zhou Jiabing2
- 1Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China; 2College of Physical Education and Sports, Guangdong University of Petrochemical Technology, Maoming 525000, Guangdong Province, China
-
Accepted:2025-12-06Online:2026-11-18Published:2026-04-29 -
Contact:Zhou Jiabing, Lecturer, College of Physical Education and Sports, Guangdong University of Petrochemical Technology, Maoming 525000, Guangdong Province, China -
About author:Jin Yuan, MS, Nanjing Sport Institute, Nanjing 210014, Jiangsu Province, China -
Supported by:Research Fund Project of Guangdong University of Petrochemical Technology, No. 2024rcyj1049, 2024rcyj1050 (to ZJB)
摘要:
文题释义:
水凝胶功能化策略:指通过物理、化学或生物手段对水凝胶结构或组成成分进行有目的地改性和优化,使它具备特定的生物学功能、力学性能或智能响应特性,以满足组织工程、药物递送及再生医学等应用需求的系列方法总称。
骨组织工程:是融合生物学、材料学和工程学等多学科技术,旨在修复、替代或再生受损骨组织的一种前沿生物医学工程技术。骨组织工程的基本原理是在合适的支架材料上接种种子细胞(如间充质干细胞),并结合生长因子或信号分子构建具有良好生物活性和力学性能的三维仿生结构,在体内外诱导新骨形成。
背景:水凝胶是一种高度仿生且可调控的生物材料,在骨组织工程领域展现出广阔的应用前景。
目的:综述水凝胶在骨组织工程中的研究现状与发展趋势。
方法:检索PubMed数据库和中国知网中有关水凝胶在骨组织工程中应用的文献,英文检索词为“Hydrogel,Bone Tissue Engineering,Nanomaterials,Bone Regeneration Mechanism,Bone Defect Repair”,中文检索词为“水凝胶,骨组织工程,纳米材料,骨再生机制,骨缺损修复”。根据纳入与排除标准,最终纳入113篇文献进行综述。
结果与结论:为了增强水凝胶在骨组织工程中的应用特性,研究者实施了多种功能化策略开发,包括物理化学改性、生物功能化及复合增强等,旨在提高水凝胶的生物相容性、机械强度、降解可控特性以及药物递送功能。功能化水凝胶促进骨再生的机制主要包括细胞相互作用机制、生长因子与信号通路调控、机械信号与微环境调控以及药物与纳米递送系统。未来,水凝胶的研究将注重多功能一体化的系统构建,如通过引入温度、pH值、酶或磁场等智能响应模块,实现时空可控的生物因子或药物释放,增强它在骨再生全过程中的动态干预能力;开发具有生物可降解性、力学性能可调和微环境适应性的高分子材料,有效提升它在复杂生理环境中的稳定性与组织整合能力;借助高精度3D打印技术实现结构可控、功能分区、个性定制的支架构建,以匹配患者个体化缺损的结构特征。
https://orcid.org/0009-0009-7060-5359 (金媛)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中图分类号:
引用本文
金 媛, 周加兵. 水凝胶在运动损伤相关骨组织工程修复中的应用与进展[J]. 中国组织工程研究, 2026, 30(32): 8512-8520.
Jin Yuan, Zhou Jiabing. Applications and advances of hydrogels in bone tissue engineering repair related to sports injuries[J]. Chinese Journal of Tissue Engineering Research, 2026, 30(32): 8512-8520.
水凝胶自1960年被WICHTERLE和LíM[24]首次合成并用于医学材料以来,就为组织工程材料的研究奠定了基础。2000年,以壳聚糖、明胶、透明质酸为代表的天然水凝胶因优异的生物降解性和细胞相容性被广泛应用于骨缺损动物模型中,研究聚焦该水凝胶的成骨诱导分化及细胞黏附性能[14]。2010年后,功能化水凝胶策略迅速发展,研究者通过引入生物活性基团、可控交联网络及纳米材料实现了水凝胶在智能响应与生长因子控释方面的突破,极大地拓展了它在骨再生中的应用潜力[15]。2015年以后,随着3D生物打印技术的进步,具备结构支撑能力与细胞递送功能的可打印水凝胶复合支架逐渐成为研究热点,特别是在大体积骨缺损及异形组织修复方面展现出工程化优势[10]。2020年至今,新一代水凝胶进入智能化和临床前转化阶段,强调注射性、自修复性与免疫调节性,相关研究成果不断进入大动物实验,部分产品正处于临床前评价阶段,推动其由实验室向临床应用的转化[17-23]。
2.2 水凝胶的基本特点与功能化策略 水凝胶的基本特征与功能化手段是增强它在骨组织工程领域应用潜力的关键部分,合适的设计策略可优化水凝胶的生物相容性、机械性能、降解速率和功能化特点,进而更有效地满足骨再生的需求[25]。该部分将从水凝胶的基本组成、改性策略及常见的类型等方面展开讨论。
2.2.1 水凝胶的基本组成与结构特点 水凝胶属于一类由亲水性聚合物借助物理或化学交联形成的三维网络结构材料,能够吸收大量水分并保持自身结构稳定[26],主要结构特点有:①高含水量:给予细胞生存的湿润环境,类似于天然细胞外基质[27];②多孔结构:利于营养物质跟代谢废物相互交换,同时推动细胞迁移进而促进组织生长[28];③可实现调控的机械性能:采用交联密度、聚合物类别及复合增强举措改变水凝胶的弹性、强度以及降解速率[29];④智能化的响应属性:可依据外部刺激进行自适应的管控,符合个性化治疗渴望。水凝胶所拥有的这些特点使它成为骨组织工程里重要的支架材料,同时可作为生长因子与药物的承载物,为骨骼再生营造理想的微环境[30]。

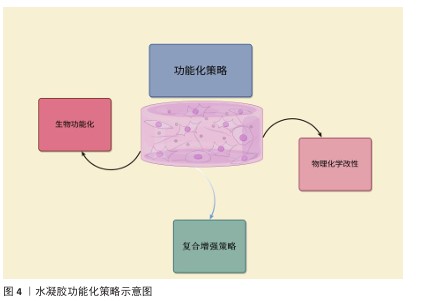
2.2.2 功能化策略概述 为了增强水凝胶在骨组织工程里的应用特性,研究者实施了多种功能化策略开发,主要包括物理化学改性、生物功能化及复合增强等,旨在提高水凝胶的生物相容性、机械强度、降解可控的特性以及药物递送功能(图4)。
物理化学改性:主要借助改变交联结构、进行表面修饰或掺杂纳米填料等途径来提升水凝胶的性能[31]。水凝胶的力学性能、降解速率以及细胞黏附能力受交联度影响[32],交联度升高,水凝胶的力学稳固性增强,但降解速度呈现降低态势。水凝胶的交联方式主要有:①物理交联(像氢键、静电相互作用):呈现可逆特性、响应性较好,然而力学性能欠佳;②化学交联(恰似共价键交联):具备较高稳定性,但材料降解速率需进行优化[33-35]。向水凝胶表面引入生物活性官能团能增强它的细胞相容性,加大成骨细胞的附着及增殖能力,例如在聚乙二醇水凝胶表面修饰三肽序列,可显著增成骨细胞的黏附能力[36],向水凝胶中掺入无机(纳米羟基磷灰石、二氧化硅、碳纳米管)或有机(纳米纤维素、聚乳酸-羟基乙酸共聚物)纳米填料可提升材料的力学强度、生物活性和矿化能力[37-39]。
生物功能化:主要借助模拟细胞外基质、输送生长因子和调节免疫反应等途径提高水凝胶的生物活性。水凝胶可借助整合天然细胞外基质成分或者仿生纳米纤维提升细胞黏附、增殖能力和成骨分化效率[40-41],例如,透明质酸基水凝胶可对天然软骨基质进行模仿,促进软骨-骨界面实现修复。功能化水凝胶可携带骨形态发生蛋白、血管内皮生长因子等生长因子,借助控释系统来推动骨再生[42]。经硫酸肝素修饰的水凝胶可稳定生长因子,强化材料的成骨效果,同时减少不良反应,通过调节巨噬细胞M1/M2的极化降低炎症反应程度,改善骨再生环境[43]。
复合增强策略:是将不同种类的材料组合起来,使水凝胶同时具备更强的机械强度、更好的生物活性以及可控制的降解性能[44]。将无机骨矿化成分和有机水凝胶结合起来,增进材料的机械强度,例如,壳聚糖-明胶复合水凝胶可营造更贴近天然骨组织的矿化环境,促进骨生成[45]。采用构建仿生梯度结构与层状结构的方式增进水凝胶的适应水平,使它在不同骨组织区域具有不同的功能,例如,软骨-骨界面修复材料可做成双层结构设计,其中软骨层含有大量透明质酸[46]。为适应不同组织的要求,基于温度、光、pH值这类刺激做到可控的降解及药物释放,温敏性水凝胶聚N-异丙基丙烯酰胺在体温状态下可形成凝胶,适合通过局部注射达成成骨应用[47]。
2.3 水凝胶促进骨再生的机制 功能化水凝胶在骨组织再生中起着至关重要的作用,作用机制主要包括细胞相互作用机制、生长因子与信号通路调控、机械信号与微环境调控以及药物与纳米递送系统(表2)。这些机制协同作用,优化水凝胶的成骨性能,使它成为理想的骨组织工程材料[48-50]。
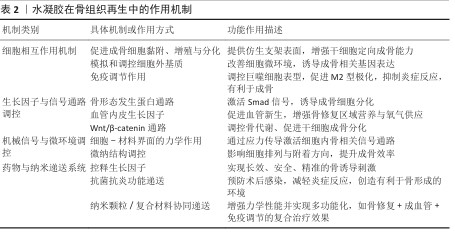
2.3.1 细胞相互作用机制 水凝胶通过调控细胞行为促进成骨细胞的黏附、生长和分化,同时模拟细胞外基质微环境,并调节免疫反应,优化骨再生效果[40-41,51]。
促进成骨细胞黏附、增殖与分化:水凝胶材料可凭借表面修饰RGD(精氨酸-甘氨酸-天冬氨酸)等整合素识别序列,提高成骨细胞(像骨髓间充质干细胞、成骨细胞MC3T3-E1等)的黏附能力,进而促进细胞的增殖与分化。例如,RGD修饰的壳聚糖-明胶水凝胶可提升骨细胞的存活和矿化能力[52-54]。
细胞外基质模拟及调控探索:将胶原蛋白、透明质酸、海藻酸盐等天然细胞外基质成分整合起来构建生物仿生水凝胶,使材料更贴合骨组织所处的天然微环境,可促进细胞黏附与骨基质沉积。例如,透明质酸修饰的水凝胶可增强成骨细胞的迁移能力,同时促进骨组织的重塑[55-56]。
免疫调节作用:在骨再生过程中,水凝胶可通过调控巨噬细胞的极化(M1/M2转化)降低炎症反应并促进组织修复。负载抗炎分子的水凝胶可促进M2型巨噬细胞的增殖,减少炎症环境对骨再生的抑制所用。
2.3.2 生长因子与信号通路调控 生长因子与信号通路在骨组织再生过程中发挥关键作用,功能化水凝胶可作为载体实现生长因子的可控释放,并通过特定信号通路促进成骨分化和血管生成[57]。骨形态发生蛋白家族是最重要的成骨生长因子,能够诱导间充质干细胞向成骨细胞分化,并促进骨基质沉积。骨形态发生蛋白2负载的明胶-壳聚糖水凝胶可实现缓释,提高成骨效率,并减少单纯骨形态发生蛋白2在体内快速降解的缺陷[58-60]。血管生成在骨修复过程中至关重要,血管内皮生长因子能够促进新生血管的形成,为成骨细胞提供充足的氧气和营养。血管内皮生长因子修饰的壳聚糖-明胶复合水凝胶可增强血管生成能力,从而加快骨修复速度[61]。Wnt/β-catenin 信号通路在成骨细胞分化过程中起核心作用,可通过激活Runx2、Osterix等成骨转录因子促进成骨分化,通过特定小分子药物(如Wnt激动剂)修饰的水凝胶可增强Wnt信号通路活性,提高骨组织再生能力[62]。
2.3.3 机械信号与微环境调控 机械信号在骨组织再生过程中同样起到关键作用,水凝胶可通过调控力学性能和微纳结构影响细胞行为优化骨修复效果。①细胞-材料界面力学作用:水凝胶的弹性模量对细胞行为有直接影响,软硬适中的水凝胶可促进成骨细胞的黏附和分化。模拟天然骨组织力学特性的壳聚糖-明胶-生物玻璃复合水凝胶,可显著提高成骨细胞的分化效率。②微纳结构对骨再生的影响:水凝胶的微纳结构可调控细胞的黏附方式,影响细胞骨架重塑和信号转导。采用3D打印或电纺技术制备的多孔水凝胶,可提供更优的细胞生长空间,促进骨基质沉积[63]。
2.3.4 药物与纳米递送系统 功能化水凝胶可以作为药物载体,实现成骨因子、抗炎药物及抗菌药物的可控释放,从而增强骨修复能力。采用可降解微球或纳米载体封装生长因子并负载于水凝胶中,可实现长时间可控释放。聚乳酸-羟基乙酸共聚物微球负载的骨形态发生蛋白2水凝胶,可在骨缺损部位持续释放生长因子,增强骨再生能力。植入材料的抗菌性能对于防止术后感染至关重要,水凝胶可通过负载抗菌药物提高抗菌能力,例如,壳聚糖-硝酸银复合水凝胶不仅具备优异的抗菌活性,还可促进骨组织的修复;此外,功能化水凝胶可负载抗炎药物有效抑制术后炎症反应,提高骨修复成功率。
在骨再生进程中,功能化水凝胶借助促进成骨细胞相互作用、调控生长因子信号通路、优化力学微环境以及药物递送等多种机制协同发力,以提升骨修复效果。将基因工程、智能响应材料及3D生物打印技术结合并应用于个性化骨组织工程领域,水凝胶将有更广阔的应用前景显现。
2.4 单水凝胶在骨组织工程中的应用进展
2.4.1 植入水凝胶支架 过去几十年里,依靠先进技术,多种水凝胶的合成与应用取得了迅速发展,作为生物医学组织再生重要分支的植入式水凝胶支架,已被开发及应用,采用合理设计并制造具有理想结构和性能的水凝胶,为克服传统骨修复材料的缺陷带来了新的机会。
(1) 3D打印水凝胶支架:为了有效治愈像颅骨缺损、颅面骨折和软骨下骨损伤之类的大范围骨缺损[16-17],研究者已开发了多种水凝胶支架的制备手段,当前研究趋势是构建既具备良好机械性能又兼具生物活性的功能化水凝胶支架,从而提高骨组织的愈合效率[18-20]。
3D生物打印技术引发了含材料科学与骨科医学多学科研究领域的普遍关注,研究人员以该技术为手段成功构建了机械强度较高的聚ε-己内酯基骨再生支架,但因缺乏生物功能以及结构诱导能力限制了细胞的黏附与增殖,最终影响了材料的长期稳定性和有效性[21]。为克服上述限制,自组装肽水凝胶因结构仿生性被广泛应用于组织再生研究。其中,基于β-折叠结构的自组装肽FEFEFKFK(F:苯丙氨酸,E:谷氨酸,K:赖氨酸)被证实是一种适用于细胞培养的理想支架。LI等[44]提出了一种新型复合支架设计,通过在3D打印聚ε-己内酯基支架表面涂覆β-sheet结构的FEFEFKFK肽,赋予材料优异的生物功能,实现了软骨及软骨下骨的协同修复。
针对创伤性脑损伤中普遍存在的颅内高压难题,研究者设计出一种含双交联网络的3D打印水凝胶支架,以达成颅骨修复在高力学强度和弹性适应性上的双重要求[23],该支架由亚甲基-双丙烯酰胺、海藻酸盐以及聚丙烯酰胺所组成,具有良好的剪切变稀特质,可开展3D打印,具有良好的细胞相容性与成骨能力。为解决天然骨组织结构复杂性跟血管化难题,CUI等[49]设计出一种双3D打印技术相结合的层状水凝胶支架,此系统采用熔融沉积式3D打印技术创建内部支撑的多孔聚乳酸结构,又结合立体光固化成型打印包裹细胞的甲基丙烯酰化明胶水凝胶外层,该支架在结构上与天然骨接近,而且可显著促进细胞生长和血管形成,在复杂骨组织再生方面展现出广阔得应用前景。
(2)纳米颗粒复合水凝胶支架:伴随着纳米技术的迅猛发展,纳米颗粒作为功能强化元件被普遍引入水凝胶支架当中,以构建更复杂、有多功能响应特性的骨组织工程材料。常见纳米颗粒类型主要有脂质体[29]、聚合物胶束[30]、介孔二氧化硅[31]、金纳米棒和纳米羟基磷灰石等[32-33],这些纳米材料来源广泛、结构呈现多样化,与水凝胶复合可提升材料的力学性能、生物活性、药物控释等。为降低纳米颗粒潜在的生物毒性隐患,研究者选取透明质酸、壳聚糖、聚乙二醇等高生物相容性的聚合物,也可借助仿生膜(像细胞膜、外泌体膜)对水凝胶表面开展功能化修饰,明显提升了水凝胶于体环境中的稳定性与安全性[34]。
聚合物胶束作为可编程的合成纳米单元,可与聚丙烯酰胺构建共价互穿/共交联网络,用以协同提升水凝胶的力学与缓释性能,该复合体系具有良好的参数可调性:在保持其他条件不变时,胶束含量加倍可使材料拉伸强度提升至约2倍、弹性模量可提升至约4倍[35]。由此可见,该复合材料在外载下表现出更显著的力学响应与承载稳定性,可为骨组织修复提供所需的支撑与缓释微环境。
鉴于两亲性结构、良好生物相容性与便捷的制备流程,纳米脂质体常被用作药物与生物活性分子的递送载体[36-40]。RAHALI等[39]构建了纳米脂质体-甲基丙烯酰化明胶复合水凝胶,研究发现随着材料中脂质体比例的提高,水凝胶的机械稳定性与流变学性能增强。
从无机纳米材料的视角来看,纳米羟基磷灰石因与天然骨矿物在成分和晶体结构上高度相似,已成为骨相关支架的核心组成部分。WANG等[41]构建的含黑磷修饰脂质体的胶原/羟基磷灰石复合支架,不仅实现了局部药物的持续释放,还通过脂质体与透明质酸之间的静电相互作用显著增强了支架的机械刚性。介孔二氧化硅纳米颗粒凭借极高的比表面积、优良的载药能力以及表面可调控性,被视为潜力巨大的药物递送平台。FILIPOWSKA等[42]研究显示,壳聚糖/胶原-介孔二氧化硅复合水凝胶对人骨髓间充质干细胞具有良好的细胞相容性,同时显著提升了人骨髓间充质干细胞碱性磷酸酶活性,有效促进成骨分化。四氧化三铁磁性纳米颗粒因优异的磁响应特性以及可与多巴胺基团形成稳定配位的能力,也被应用于自愈型水凝胶的构建。有研究制备了四氧化三铁与多巴胺修饰的星形聚乙二醇组装成动态共价网络,通过调控纳米颗粒含量实现了从流体态到固体态的可逆力学转变,为开发具备可调刚度与自修复性能的骨修复支架提供了新的策略[43-44]。
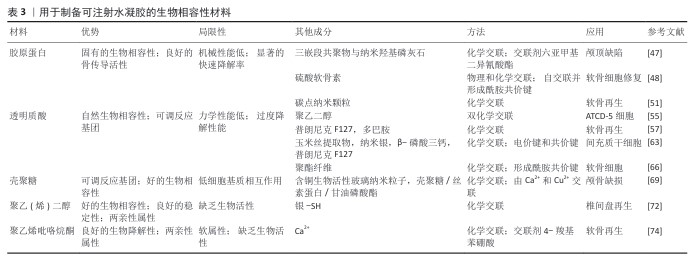
2.4.2 可注射水凝胶支架 可注射型生物材料因具备微创操作的特性,近年来在骨再生领域得到广泛关注与应用。与传统块状或成型支架相比,可注射水凝胶能够有效填充形态复杂或不规则的骨缺损区域,显著改善患者的个体顺应性,并在原位环境下促进组织修复与再生,因而展现出不可替代的优势。多类天然高分子物质,包括透明质酸、硫酸软骨素、胶原蛋白、壳聚糖及多肽等,均被视为制备可注射水凝胶的理想候选材料。依据现有的体内实验评价结果,这些材料具有良好的生物相容性与生物降解性,为它们的临床转化奠定了基础[45]。
(1)胶原蛋白基可注射水凝胶:胶原蛋白作为天然纤维蛋白,因独特的理化性质与分子构型在生物医学研究与临床应用中占据重要地位。作为细胞外基质的重要成分,胶原蛋白不仅具有优良的生物相容性,还能通过整合素介导的细胞-基质相互作用促进细胞在三维支架中的黏附、迁移与分化,从而在骨传导过程中发挥关键作用。尤其在软骨组织工程中,胶原蛋白凭借天然来源、可降解性以及诱导细胞向软骨表型分化的能力,被视为理想的组织工程支架材料[46]。然而,单一胶原蛋白水凝胶存在力学强度不足和降解速率过快的问题,限制了它在长期维持细胞功能和支持骨再生方面的应用潜力。
为解决这些局限,近年来大量研究集中于胶原蛋白基可注射水凝胶的结构优化与性能提升。FU等[47]开发了一种复合水凝胶体系,通过引入三嵌段共聚物与纳米羟基磷灰石,并利用共价交联方式将聚乙二醇-聚己内酯-聚乙二醇接枝于胶原蛋白主链,从而赋予水凝胶热响应特性,流变学与试管倒置实验结果表明,该材料在常温下呈流动状态,而在37 ℃时可迅速凝胶化;将该材料植入新西兰大白兔颅骨缺损模型中20周后,组织学与微型计算机断层成像分析证实了该材料良好的组织相容性与促成骨潜能。GAO等[48]构建了一种无需外源交联剂即可自交联形成的Ⅰ型胶原蛋白基可注射水凝胶,合成策略是将改性硫酸软骨素与Ⅰ型胶原蛋白混合于PBS中,在生理条件下即可形成网络,通过调节改性硫酸软骨素的取代度可有效控制水凝胶力学性能;该体系在体外表现出良好的细胞增殖与细胞外基质分泌功能,在SD大鼠体内注射4周后未见明显炎症反应,显示出优良的生物安全性。
为进一步提升胶原蛋白水凝胶的功能,研究者尝试引入纳米材料以增强其力学性能并实现药物的可控释放。碳点纳米颗粒因低毒性和在光照下产生活性氧的特性,被应用于构建具有光响应功能的胶原蛋白复合水凝胶,从而促进软骨细胞的增殖与分化[49-50]。LU等[51]将龙胆蓝交联剂与碳点纳米颗粒联合应用,形成胶原-龙胆蓝-碳点纳米颗粒柔性可注射水凝胶,该材料通过双重交联机制显著提高了材料的力学稳定性,压缩模量较传统胶原水凝胶提升约21倍,并且降解速率显著减缓;将该材料与骨髓间充质干细胞联合注射至雌性SD大鼠软骨缺损模型中,同时在治疗过程中周期性给予808 nm激光照射,诱导碳点纳米颗粒产生活性氧以促进细胞增殖和软骨再生,组织病理学与免疫组化结果显示骨髓间充质干细胞增殖率提高约50.3%,软骨修复效果显著优于对照组,该体系可通过调控转化生长因子β/SMAD与哺乳动物雷帕霉素靶蛋白信号通路有效促进软骨分化与修复。
(2)透明质酸基可注射水凝胶:透明质酸是一种由葡萄糖醛酸和乙酰葡萄糖胺交替聚合形成的天然多糖,是软骨基质和关节滑液中的主要糖胺聚糖之一[52]。作为机体自身的成分,透明质酸来源广泛、成本相对低廉且免疫原性极低,因此被广泛应用于关节疾病与骨组织修复的研究。功能上,透明质酸能够通过减少炎症反应、抑制蛋白聚糖降解、阻断前列腺素合成及下游信号转导等途径维持关节微环境的稳态,并在防止软骨退变方面发挥关键作用。
在结构层面,透明质酸属于高分子线性多糖,其分子链上含有羟基、羧基和乙酰基等可反应基团,这为透明质酸的进一步功能化改造与理化性能调控提供了可能。然而,由于透明质酸极易受到透明质酸酶、氧化还原酶以及自由基的降解作用,其降解速度往往较快,这一特性一方面使透明质酸基材料能够在组织中被快速吸收,但另一方面也限制了材料在承重骨组织修复中实现长期稳定支撑的能力[53]。
为提升透明质酸的力学性能与结构稳定性,研究者探索了多种改良策略。WANG等[54]通过光引发反应诱导透明质酸分子间形成共价键,随后利用狄尔斯-阿尔德反应进一步增强网络稳定性,使材料的压缩模量由5.9 kPa提高至21 kPa,尽管该方法显著增强了材料的力学性能,但反应耗时较长限制了其临床应用;为此,研究进一步引入巯基化学修饰,仅需2 s即可快速形成稳定水凝胶,材料压缩模量最高可达72 kPa。
在功能拓展方面,透明质酸常与热敏性聚合物复合,如聚异丙基丙烯酰胺、嵌段共聚物或明胶等,以赋予材料温度响应特性,这类复合体系在软骨修复中应用广泛,并通过负载骨形态发生蛋白2或曲安奈德等药物实现了对损伤软骨的显著修复效果[55-62]。与此同时,为避免在注射和植入过程中发生感染,研究者将银纳米颗粒、铜离子和季铵盐等抗菌组分引入水凝胶网络。例如,MAKVANDI等[63]利用植物提取物作为绿色还原剂合成银纳米颗粒,并与透明质酸和磷酸三钙复合,制备出具有热敏性与抗菌性的复合水凝胶,该材料在37 ℃下可实现溶胶-凝胶转变,既保持了材料良好的力学稳定性又显著提升了抗菌性能,在动物实验中,该复合材料能够有效促进间充质干细胞的成骨分化,表现出优良的促骨再生潜力。
(3)壳聚糖基可注射水凝胶:壳聚糖是由甲壳素脱乙酰化得到的天然多糖衍生物,是软骨组织中重要的糖胺聚糖之一[64]。壳聚糖分子链中富含氨基,不仅赋予了壳聚糖优良的生物相容性和可降解性,还为化学修饰提供了丰富的活性位点。常见的修饰方式包括酰化、烷基化和羧甲基化,这些改性方法能够有效改善壳聚糖的理化性能和生物功能,从而拓展它在骨组织工程中的应用前景[65-68]。壳聚糖基水凝胶常形成多孔结构,有助于细胞的黏附与迁移,同时利于营养和代谢物交换,为新生组织的生长提供有利条件。
目前,壳聚糖基水凝胶的制备方法主要包括氧化还原反应和光引发聚合,然而,传统光引发体系可能因引发剂毒性或长时间光照而对细胞造成损伤,限制了它在再生医学中的应用[64]。为解决这一问题,研究者逐渐转向温和条件下的交联方式,其中席夫碱反应因能够在氨基与醛基之间形成亚胺键而被广泛采用。CAO等[66]合成了一种醛基修饰的聚乙二醇,并与壳聚糖通过席夫碱反应形成共价交联网络,所得水凝胶在12周内保持良好的稳定性和降解性能,同时在体内外实验中支持软骨细胞的增殖和表型维持,显示出理想的软骨修复效果。
除了力学优化,壳聚糖基水凝胶的生物诱导功能同样重要。骨组织修复过程中血管化是促进新生组织与宿主骨整合的关键环节[67]。研究表明,铜离子能够促进血管生成,并诱导干细胞向成骨细胞分化[68]。WU等[69]设计了一种基于铜掺杂生物活性玻璃纳米颗粒的复合水凝胶体系,该材料由壳聚糖、丝素蛋白和甘油磷酸盐共同构建,具有明显的热响应特性,能通过缓释机制维持离子释放,从而在缺损部位保持持久的生物活性;动物颅骨缺损实验表明,该体系即使在无细胞和无外源生长因子的条件下也能有效促进骨组织再生,验证了它作为骨修复材料的应用前景。
(4)其他可注射水凝胶:除了透明质酸与壳聚糖外,合成高分子材料在可注射水凝胶的研究中同样受到关注。聚乙二醇是一种已获得美国食品药品监督管理局批准的高分子材料,因优良的生物相容性、化学稳定性及两亲性而被广泛应用于药物递送和组织修复[70]。在椎间盘退变研究中,聚乙二醇基水凝胶展现出较大潜力。研究发现,椎间盘退变通常伴随基质金属蛋白酶过度表达,导致髓核细胞外基质的降解与功能损失[71]。CHEN等[72]基于四臂巯基化聚乙二醇设计了一种多功能水凝胶,该水凝胶通过银-硫配位反应实现自发交联,并且具备自愈特性与优良注射性,植入动物模型中能显著改善椎间盘结构,表现出理想的修复效果。
聚乙烯醇是另一种广泛应用于组织工程和药物递送的材料,同样获得美国食品药品监督管理局的认可[73]。然而,传统聚乙烯醇基水凝胶往往力学性能不足,难以满足承重组织的需求。ZHAO等[74]提出了双动态交联策略,利用硼酸酯共价键与钙离子静电作用形成双重网络,从而显著提升聚乙烯醇基水凝胶的断裂能和稳定性,在兔软骨缺损实验中该材料不仅表现出良好的组织相容性,还在病理学和免疫组织学检测中展现出更完整的软骨结构层,进一步证明了它在软骨再生中的应用价值。
用于制备可注射水凝胶的生物相容性材料,见表3。
2.4.3 功能药物递送载体微/纳米凝胶 随着纳米技术的迅速发展,脂质体、纳米胶束、纳米颗粒以及纳米/微凝胶等一系列新型纳米体系已在多个生物医学领域得到应用[75-78]。这些材料凭借小尺寸优势能够跨越血管屏障并靶向特定组织,同时通过表面功能化修饰与生物分子产生特异性结合,从而实现主动靶向递送,在肿瘤治疗、骨组织工程、软骨修复及伤口愈合等领域,它们均显示出广阔的应用前景[79-82]。
与其他纳米材料相比,微/纳米凝胶的突出优势在于可调节粒径、内部结构高度多孔,有利于细胞附着和渗透;较大的比表面积不仅增强了与生物环境的相互作用,也提升了药物装载效率。更重要的是,这类凝胶表面分布有丰富的化学官能团,可通过修饰实现理化性质和生物行为的精准控制,对温度、酸碱度、离子强度等外界刺激的敏感性,使它具备环境响应性和仿生特性,因此被认为是极具潜力的功能性药物递送载体。
(1)微凝胶:微凝胶凭借良好的相容性、低成本与多功能调控性能,在组织工程领域受到广泛关注[83-87]。微凝胶可作为三维微支架,提供利于细胞黏附、增殖与迁移的微环境,并能封装蛋白质、多肽、金属或小分子药物,从而推动组织再生进程[84]。WU等[85]构建的复合微凝胶体系,通过聚异丙基丙烯酰胺赋予材料热敏特性,聚乙二醇增强材料体积稳定性,半乳糖配体促进细胞黏附,研究发现肝癌细胞在该体系中的显著提升,并形成致密的多细胞球体结构。
ARUN KUMAR等[88]采用溶剂再生法制备了天然多糖几丁质与聚己内酯、纳米羟基磷灰石的复合微凝胶,该材料具有良好的弹性模量和流变特性,适用于骨缺损微创治疗。LI 等[89]利用功能化明胶与交联剂制备了注射型微凝胶网络,在封装人骨髓间充质干细胞后不仅保持了细胞活性,还促进了软骨相关基因的表达。LI等[90-91]通过微流控液滴技术构建了能够促进血管化和软骨分化的自组装微凝胶体系,该材料在动物模型中亦显示出积极的修复效果。
综上所述,功能化微凝胶通过提供仿生三维环境与药物缓释功能,为构建高效骨软组织修复支架提供了有力支撑。
(2)纳米凝胶:纳米凝胶是一类尺寸在纳米范围内、由物理或化学交联构成的三维水网络,兼具生物可降解性与可调节力学性能[92]。与传统支架或注射水凝胶相比,纳米凝胶在药物递送中的表现更为突出,尤其在生物活性分子封装和靶向释放方面显示出更高的效率[93]。通过叶酸、肽链或抗体修饰,可实现纳米凝胶对肿瘤与关节炎病灶的特异性递送[94-95]。
近年来,刺激响应型纳米凝胶受到广泛关注。炎症部位常存在高浓度谷胱甘肽的氧化应激环境,研究者据此设计了谷胱甘肽响应型纳米凝胶,用于类风湿关节炎的智能给药[96]。此类材料在体外实验中可实现药物在高还原环境下的快速释放,在动物模型中显著缓解炎症[97]。
另一类研究聚焦于清除炎症环境中过量的一氧化氮[98-99]。YEO等[100]开发的响应性纳米凝胶在高一氧化氮条件下解聚并释放抑制因子有效降低炎症反应,在类风湿关节炎小鼠模型中恢复了骨密度。与此同时,热敏纳米凝胶因在生理温度下可实现相变,被广泛应用于骨组织修复。为解决传统聚异丙基丙烯酰胺纳米凝胶响应缓慢、力学性能差的问题,研究者提出利用活化纳米凝胶作为交联单元,制备响应速度快、弹性佳的复合水凝胶,能够在数分钟内完成体积变化并维持药物稳定释放[103]。
此外,骨形态发生蛋白作为临床常用的骨诱导因子,它在体内的半衰期短、脱靶风险高。为解决这一问题,有研究构建了基于热敏聚磷腈的纳米颗粒体系,能在体温下形成注射性支架,实现骨形态发生蛋白的持续释放,该材料在小鼠颅骨缺损实验中可诱导新骨形
成[104-105]。
2.4.4 水凝胶材料在骨组织工程中的关键因素 在骨组织修复中,水凝胶既要具备良好的生物相容性与无毒性,又要能够支持细胞增殖、分化与迁移。常用的水凝胶制备原料包括糖胺聚糖、天然蛋白质以及脱氧核苷酸等内源性物质,同时聚乙二醇、聚乙烯吡咯烷酮、羟丙基甲基纤维素等合成聚合物也被广泛用作替代性材料[106],尽管这些材料具备较好的相容性,但它们在促进细胞活性方面仍存在局限,因此,开发兼具生物活性和力学增强功能的新型水凝胶材料成为研究重点。
细胞可通过机械转导机制感知水凝胶的力学特性,并据此调节自身迁移与分泌行为,进而影响表型与再生潜能[107],因此,构建与天然骨组织力学性能接近的水凝胶有助于加速愈合并提高修复效果。目前,提升水凝胶力学性能的方法包括引入多重交联网络、掺杂纳米颗粒以调节物理化学特性以及利用3D打印构建具有结构支撑作用的聚合物内核等[108]。
水凝胶的孔隙结构也是影响修复效果的重要因素,小孔径(2-50 nm)有助于药物负载和活性保持,中等孔径促进磷灰石沉积与无机离子交换,大孔径(> 100 μm)更适合细胞黏附和迁移[109-111]。因此,依据不同治疗需求合理设计水凝胶孔径,是优化修复效果的关键。
功能化水凝胶在药物递送方面的应用也引起了广泛关注,功能化水凝胶的核心目标是实现药物或信号分子的可控释放,以满足骨修复过程中的时序和特异性要求。传统的直接包埋方式常导致药物或生物分子释放过快,难以满足长期治疗需求。研究显示,将功能性纳米颗粒引入水凝胶基体能够延长药物或生物分子的释放时间,并可通过光热、磁场等外部刺激实现按需释放[112-113],这一策略为骨缺损、类风湿关节炎及骨折等疾病的治疗提供了更精准和高效的手段。
| [1] HARADA S, RODAN GA. Control of osteoblast function and regulation of bone mass. Nature. 2003;423(6937):349-355. [2] STOLZING A, JONES E, MCGONAGLE D, et al. Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies. Mech Ageing Dev. 2008;129(3):163-173. [3] LEE KI, LEE JS, KANG KT, et al. In Vitro and In Vivo Performance of Tissue-Engineered Tendons for Anterior Cruciate Ligament Reconstruction. Am J Sports Med. 2018;46(7):1641-1649. [4] SEOW D, YASUI Y, HURLEY ET, et al. Extracellular Matrix Cartilage Allograft and Particulate Cartilage Allograft for Osteochondral Lesions of the Knee and Ankle Joints: A Systematic Review. Am J Sports Med. 2018;46(7):1758-1766. [5] WANG Z, WANG Y, WANG Z, et al. Engineered mesenchymal stem cells with enhanced tropism and paracrine secretion of cytokines and growth factors to treat traumatic brain injury. Stem Cells. 2015;33(2):456-467. [6] WILLEMS WF, LARSEN M, GIUSTI G, et al. Revascularization and bone remodeling of frozen allografts stimulated by intramedullary sustained delivery of FGF-2 and VEGF. J Orthop Res. 2011;29(9):1431-1436. [7] ARRINGTON ED, SMITH WJ, CHAMBERS HG, et al. Complications of iliac crest bone graft harvesting. Clin Orthop Relat Res. 1996;(329): 300-309. [8] KOFRON MD, LAURENCIN CT. Bone tissue engineering by gene delivery. Adv Drug Deliv Rev. 2006;58(4):555-576. [9] WANG SJ, JIANG D, ZHANG ZZ, et al. Biomimetic Nanosilica-Collagen Scaffolds for In Situ Bone Regeneration: Toward a Cell-Free, One-Step Surgery. Adv Mater. 2019;31(49):e1904341. [10] GRANDE DA, HALBERSTADT C, NAUGHTON G, et al. Evaluation of matrix scaffolds for tissue engineering of articular cartilage grafts. J Biomed Mater Res. 1997;34(2):211-220. [11] LI Y, RODRIGUES J, TOMÁS H. Injectable and biodegradable hydrogels: gelation, biodegradation and biomedical applications. Chem Soc Rev. 2012;41(6):2193-2221. [12] JEON YH, CHOI JH, SUNG JK,et al. Different effects of PLGA and chitosan scaffolds on human cartilage tissue engineering. J Craniofac Surg. 2007;18(6):1249-1258. [13] NISHIMOTO S, TAKAGI M, WAKITANI S, et al. Effect of chondroitin sulfate and hyaluronic acid on gene expression in a three-dimensional culture of chondrocytes. J Biosci Bioeng. 2005; 100(1):123-126. [14] XAVIER JR, THAKUR T, DESAI P, et al. Bioactive nanoengineered hydrogels for bone tissue engineering: a growth-factor-free approach. ACS Nano. 2015;9(3):3109-3118. [15] LI J, MOONEY DJ. Designing hydrogels for controlled drug delivery. Nat Rev Mater. 2016; 1(12):16071. [16] PAN P, CHEN X, XING HR, et al. A fast on-demand preparation of injectable self-healing nanocomposite hydrogels for efficient osteoinduction. Chin Chem Lett. 2021;32(7): 2159-2163. [17] ZHAI X, MA Y, HOU C, et al. 3D-Printed High Strength Bioactive Supramolecular Polymer/Clay Nanocomposite Hydrogel Scaffold for Bone Regeneration. ACS Biomater Sci Eng. 2017;3(6):1109-1118. [18] LEVENGOOD SL, ZHANG M. Chitosan-based scaffolds for bone tissue engineering. J Mater Chem B. 2014;2(21):3161-3184. [19] ZHAO C, QAZVINI NT, SADATI M, et al. A pH-Triggered, Self-Assembled, and Bioprintable Hybrid Hydrogel Scaffold for Mesenchymal Stem Cell Based Bone Tissue Engineering. ACS Appl Mater Interfaces. 2019;11(9):8749-8762. [20] KHURANA S, BEDI PM, JAIN NK. Preparation and evaluation of solid lipid nanoparticles based nanogel for dermal delivery of meloxicam. Chem Phys Lipids. 2013;175-176: 65-72. [21] SCHMITT F, LAGOPOULOS L, KÄUPER P, et al. Chitosan-based nanogels for selective delivery of photosensitizers to macrophages and improved retention in and therapy of articular joints. J Control Release. 2010; 144(2):242-250. [22] ABIOYE AO, ISSAH S, KOLA-MUSTAPHA AT. Ex vivo skin permeation and retention studies on chitosan-ibuprofen-gellan ternary nanogel prepared by in situ ionic gelation technique--a tool for controlled transdermal delivery of ibuprofen. Int J Pharm. 2015;490(1-2):112-130. [23] FUJIOKA-KOBAYASHI M, OTA MS, SHIMODA A, et al. Cholesteryl group- and acryloyl group-bearing pullulan nanogel to deliver BMP2 and FGF18 for bone tissue engineering. Biomaterials. 2012;33(30):7613-7620. [24] WICHTERLE O, LÍM D. Hydrophilic Gels for Biological Use. Nature. 1960;185(4706): 117-118. [25] KLIONSKY DJ, ABDELMOHSEN K, ABE A. Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy. 2016;12(1):1-222. [26] WANG J, YANG M, ZHU Y, et al. Phage nanofibers induce vascularized osteogenesis in 3D printed bone scaffolds. Adv Mater. 2014; 26(29):4961-4966. [27] BLACHE U, METZGER S, VALLMAJO-MARTIN Q, et al. Dual Role of Mesenchymal Stem Cells Allows for Microvascularized Bone Tissue-Like Environments in PEG Hydrogels. Adv Healthc Mater. 2016;5(4):489-498. [28] CUI H, ZHU W, NOWICKI M, et al. Hierarchical Fabrication of Engineered Vascularized Bone Biphasic Constructs via Dual 3D Bioprinting: Integrating Regional Bioactive Factors into Architectural Design. Adv Healthc Mater. 2016;5(17):2174-2181. [29] MARUYAMA K, ISHIDA O, TAKIZAWA T, et al. Possibility of active targeting to tumor tissues with liposomes. Adv Drug Deliv Rev. 1999; 40(1-2):89-102. [30] TANIGO T, TAKAOKA R, TABATA Y. Sustained release of water-insoluble simvastatin from biodegradable hydrogel augments bone regeneration. J Control Release. 2010;143(2): 201-206. [31] ÁLVAREZ E, ESTÉVEZ M, JIMÉNEZ-JIMÉNEZ C, et al. A versatile multicomponent mesoporous silica nanosystem with dual antimicrobial and osteogenic effects. Acta Biomater. 2021; 136:570-581. [32] LANKVELD DP, RAYAVARAPU RG, KRYSTEK P, et al. Blood clearance and tissue distribution of PEGylated and non-PEGylated gold nanorods after intravenous administration in rats. Nanomedicine (Lond). 2011;6(2):339-349. [33] THEIN-HAN WW, MISRA RD. Biomimetic chitosan-nanohydroxyapatite composite scaffolds for bone tissue engineering. Acta Biomater. 2009;5(4):1182-1197. [34] BATEMAN RJ, SMITH J, DONOHUE MC, et al. GRADUATE I and II Investigators and the Gantenerumab Study Group. Two Phase 3 Trials of Gantenerumab in Early Alzheimer’s Disease. N Engl J Med. 2023;389(20): 1862-1876. [35] XIAO L, LIU C, ZHU J, et al. Hybrid, elastomeric hydrogels crosslinked by multifunctional block copolymer micelles. Soft Matter. 2010; 6(21):5293-5297. [36] BANGHAM AD, HORNE RW. negative staining of phospholipids and their structural modification by surface-active agents as observed in the electron microscope. J Mol Biol. 1964;8:660-668. [37] KORSMEYER R. Critical questions in development of targeted nanoparticle therapeutics. Regen Biomater. 2016;3(2): 143-147. [38] DOLL TA, RAMAN S, DEY R, et al. Nanoscale assemblies and their biomedical applications. J R Soc Interface. 2013;10(80):20120740. [39] RAHALI K, BEN MESSAOUD G, KAHN CJF, et al. Synthesis and Characterization of Nanofunctionalized Gelatin Methacrylate Hydrogels. Int J Mol Sci. 2017;18(12):2675. [40] NAIN A, JOSHI A, DEBNATH S, et al. A 4D printed nanoengineered super bioactive hydrogel scaffold with programmable deformation for potential bifurcated vascular channel construction. J Mater Chem B. 2024; 12:7604. [41] WANG G, BABADAĞLI ME, ULUDAĞ H. Bisphosphonate-derivatized liposomes to control drug release from collagen/hydroxyapatite scaffolds. Mol Pharm. 2011; 8(4):1025-1034. [42] FILIPOWSKA J, LEWANDOWSKA-ŁAŃCUCKA J, GILARSKA A, et al. In vitro osteogenic potential of collagen/chitosan-based hydrogels-silica particles hybrids in human bone marrow-derived mesenchymal stromal cell cultures. Int J Biol Macromol. 2018;113:692-700. [43] WAITE JH, TANZER ML. Polyphenolic Substance of Mytilus edulis: Novel Adhesive Containing L-Dopa and Hydroxyproline. Science. 1981; 212(4498):1038-1040. [44] LI Q, BARRETT DG, MESSERSMITH PB, et al. Controlling Hydrogel Mechanics via Bio-Inspired Polymer-Nanoparticle Bond Dynamics. ACS Nano. 2016;10(1):1317-1324. [45] TEMENOFF JS, MIKOS AG. Injectable biodegradable materials for orthopedic tissue engineering. Biomaterials. 2000; 21(23):2405-2412. [46] SCHNEIDER U, RACKWITZ L, ANDEREYA S, et al. A prospective multicenter study on the outcome of type I collagen hydrogel-based autologous chondrocyte implantation (CaReS) for the repair of articular cartilage defects in the knee. Am J Sports Med. 2011;39(12):2558-2565. [47] FU S, NI P, WANG B, et al. Injectable and thermo-sensitive PEG-PCL-PEG copolymer/collagen/n-HA hydrogel composite for guided bone regeneration. Biomaterials. 2012;33(19): 4801-4809. [48] GAO Y, KONG W, LI B, et al. Fabrication and characterization of collagen-based injectable and self-crosslinkable hydrogels for cell encapsulation. Colloids Surf B Biointerfaces. 2018;167:448-456. [49] CUI Y, ZHU T, LI A, et al. Porous Particle-Reinforced Bioactive Gelatin Scaffold for Large Segmental Bone Defect Repairing. ACS Appl Mater Interfaces. 2018;10(8):6956-6964. [50] HINO K, HORIGOME K, NISHIO M, et al. Activin-A enhances mTOR signaling to promote aberrant chondrogenesis in fibrodysplasia ossificans progressiva. J Clin Invest. 2017; 127(9):3339-3352. [51] LU Z, LIU S, LE Y, et al. An injectable collagen-genipin-carbon dot hydrogel combined with photodynamic therapy to enhance chondrogenesis. Biomaterials. 2019;218: 119190. [52] KIM IL, MAUCK RL, BURDICK JA. Hydrogel design for cartilage tissue engineering: a case study with hyaluronic acid. Biomaterials. 2011;32(34):8771-8782. [53] BURDICK JA, PRESTWICH GD. Hyaluronic acid hydrogels for biomedical applications. Adv Mater. 2011;23(12):H41-56. [54] WANG G, ZHU J, CHEN X, et al. Alginate based antimicrobial hydrogels formed by integrating Diels-Alder “click chemistry” and the thiol-ene reaction. RSC Adv. 2018;8(20):11036-11042. [55] LI G, LIN Y, YANG J, et al. Intensive Ambulance-Delivered Blood-Pressure Reduction in Hyperacute Stroke. N Engl J Med. 2024; 390(20):1862-1872. [56] OHYA S, NAKAYAMA Y, MATSUDA T. Thermoresponsive artificial extracellular matrix for tissue engineering: hyaluronic acid bioconjugated with poly(N-isopropylacrylamide) grafts. Biomacromolecules. 2001;2(3):856-863. [57] GAO Z, SHENG T, ZHANG W, et al. Microneedle-Mediated Cell Therapy. Adv Sci (Weinh). 2024; 11(8):e2304124. [58] AKKARI ACS, PAPINI JZB, GARCIA GK, et al. Poloxamer 407/188 binary thermosensitive hydrogels as delivery systems for infiltrative local anesthesia: Physico-chemical characterization and pharmacological evaluation. Mater Sci Eng C Mater Biol Appl. 2016;68:299-307. [59] NAIR MB, KRETLOW JD, MIKOS AG, et al. Infection and tissue engineering in segmental bone defects--a mini review. Curr Opin Biotechnol. 2011;22(5):721-725. [60] MAKVANDI P, JAMALEDIN R, JABBARI M, et al. Antibacterial quaternary ammonium compounds in dental materials: A systematic review. Dent Mater. 2018;34(6):851-867. [61] LU Y, LI L, ZHU Y, et al. Multifunctional Copper-Containing Carboxymethyl Chitosan/Alginate Scaffolds for Eradicating Clinical Bacterial Infection and Promoting Bone Formation. ACS Appl Mater Interfaces. 2018;10(1):127-138. [62] ZHANG R, LEE P, LUI VC, et al. Silver nanoparticles promote osteogenesis of mesenchymal stem cells and improve bone fracture healing in osteogenesis mechanism mouse model. Nanomedicine. 2015;11(8):1949-1959. [63] MAKVANDI P, ALI GW, DELLA SALA F, et al. Hyaluronic acid/corn silk extract based injectable nanocomposite: A biomimetic antibacterial scaffold for bone tissue regeneration. Mater Sci Eng C Mater Biol Appl. 2020;107:110195. [64] FEDOROVICH NE, OUDSHOORN MH, VAN GEEMEN D, et al. The effect of photopolymerization on stem cells embedded in hydrogels. Biomaterials. 2009;30(3):344-353. [65] BALAKRISHNAN B, JOSHI N, BANERJEE R. Borate aided Schiff’s base formation yields in situ gelling hydrogels for cartilage regeneration. J Mater Chem B. 2013;1(41):5564-5577. [66] CAO L, CAO B, LU C, et al. An injectable hydrogel formed by in situ cross-linking of glycol chitosan and multi-benzaldehyde functionalized PEG analogues for cartilage tissue engineering. J Mater Chem B. 2015; 3(7):1268-1280. [67] QUINLAN E, PARTAP S, AZEVEDO MM, et al. Hypoxia-mimicking bioactive glass/collagen glycosaminoglycan composite scaffolds to enhance angiogenesis and bone repair. Biomaterials. 2015;52:358-366. [68] BURGHARDT I, LÜTHEN F, PRINZ C, et al.A dual function of copper in designing regenerative implants. Biomaterials. 2015;44:36-44. [69] WU J, ZHENG K, HUANG X, et al. Thermally triggered injectable chitosan/silk fibroin/bioactive glass nanoparticle hydrogels for in-situ bone formation in rat calvarial bone defects. Acta Biomater. 2019;91:60-71. [70] JEONG B, BAE YH, KIM SW. Drug release from biodegradable injectable thermosensitive hydrogel of PEG-PLGA-PEG triblock copolymers. J Control Release. 2000;63(1-2): 155-163. [71] HENRY N, CLOUET J, LE BIDEAU J, et al. Innovative strategies for intervertebral disc regenerative medicine: From cell therapies to multiscale delivery systems. Biotechnol Adv. 2018;36(1):281-294. [72] CHEN W, CHEN H, ZHENG D, et al. Gene-Hydrogel Microenvironment Regulates Extracellular Matrix Metabolism Balance in Nucleus Pulposus. Adv Sci (Weinh). 2019;7(1): 1902099. [73] MUSCHERT S, SIEPMANN F, LECLERCQ B, et al. Drug release mechanisms from ethylcellulose: PVA-PEG graft copolymer-coated pellets. Eur J Pharm Biopharm. 2009;72(1):130-137. [74] ZHAO Y, LI M, LIU B, et al. Ultra-tough injectable cytocompatible hydrogel for 3D cell culture and cartilage repair. J Mater Chem B. 2018;6(9):1351-1358. [75] ABBOTT BP, ABBOTT R, ABBOTT TD, et al. Observation of Gravitational Waves from a Binary Black Hole Merger. Phys Rev Lett. 2016;116(6):061102. [76] ZHANG Y, REN T, GOU J, et al. Strategies for improving the payload of small molecular drugs in polymeric micelles. J Control Release. 2017;261:352-366. [77] CHEN G, ROY I, YANG C, et al. Nanochemistry and Nanomedicine for Nanoparticle-based Diagnostics and Therapy. Chem Rev. 2016; 116(5):2826-2885. [78] GBD 2021 Nervous System Disorders Collaborators. Global, regional, and national burden of disorders affecting the nervous system, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Neurol. 2024;23(4):344-381. [79] DING J, XU W, ZHANG Y, et al. Self-reinforced endocytoses of smart polypeptide nanogels for “on-demand” drug delivery. J Control Release. 2013;172(2):444-455. [80] RANK LA, AGRAWAL A, LIU L,et al. Diverse Impacts on Prokaryotic and Eukaryotic Membrane Activities from Hydrophobic Subunit Variation Among Nylon-3 Copolymers. ACS Chem Biol. 2021;16(1): 176-184. [81] ESLAHI N, ABDORAHIM M, SIMCHI A. Smart Polymeric Hydrogels for Cartilage Tissue Engineering: A Review on the Chemistry and Biological Functions. Biomacromolecules. 2016;17(11):3441-3463. [82] ZHU J, LI F, WANG X, et al. Hyaluronic Acid and Polyethylene Glycol Hybrid Hydrogel Encapsulating Nanogel with Hemostasis and Sustainable Antibacterial Property for Wound Healing. ACS Appl Mater Interfaces. 2018;10(16):13304-13316. [83] HERIS HK, DAOUD J, SHEIBANI S, et al. Investigation of the Viability, Adhesion, and Migration of Human Fibroblasts in a Hyaluronic Acid/Gelatin Microgel-Reinforced Composite Hydrogel for Vocal Fold Tissue Regeneration. Adv Healthc Mater. 2016;5(2):255-265. [84] STEINHILBER D, ROSSOW T, WEDEPOHL S, et al. A microgel construction kit for bioorthogonal encapsulation and pH-controlled release of living cells. Angew Chem Int Ed Engl. 2013; 52(51):13538-13543. [85] WU Y, ZHAO Z, GUAN Y, et al. Galactosylated reversible hydrogels as scaffold for HepG2 spheroid generation. Acta Biomater. 2014;10(5):1965-1974. [86] WYSOKOWSKI M, BAZHENOV VV, TSURKAN MV, et al. Isolation and identification of chitin in three-dimensional skeleton of Aplysina fistularis marine sponge. Int J Biol Macromol. 2013;62:94-100. [87] WOODRUFF MA, HUTMACHER DW. The return of a forgotten polymer—Polycaprolactone in the 21st century. Prog Polym Sci. 2010; 35(10):1217-1256. [88] ARUN KUMAR R, SIVASHANMUGAM A, DEEPTHI S, et al. Injectable Chitin-Poly(ε-caprolactone)/Nanohydroxyapatite Composite Microgels Prepared by Simple Regeneration Technique for Bone Tissue Engineering. ACS Appl Mater Interfaces. 2015;7(18):9399-9409. [89] LI F, TRUONG VX, FISCH P, et al. Cartilage tissue formation through assembly of microgels containing mesenchymal stem cells. Acta Biomater. 2018;77:48-62. [90] LI G, LIN Y, YANG J, et al. Intensive Ambulance-Delivered Blood-Pressure Reduction in Hyperacute Stroke. N Engl J Med. 2024; 390(20):1862-1872. [91] GONG W, HUANG HB, WANG XC, et al. Construction of a sustained-release hydrogel using gallic acid and lysozyme with antimicrobial properties for wound treatment. Biomater Sci. 2022;10(23):6836-6849. [92] ELKHOURY K, RUSSELL CS, SANCHEZ-GONZALEZ L, et al. Soft-Nanoparticle Functionalization of Natural Hydrogels for Tissue Engineering Applications. Adv Healthc Mater. 2019;8(18):e1900506. [93] SASAKI Y, AKIYOSHI K. Nanogel engineering for new nanobiomaterials: from chaperoning engineering to biomedical applications. Chem Rec. 2010;10(6):366-376. [94] CULVER HR, STEICHEN SD, PEPPAS NA. A Closer Look at the Impact of Molecular Imprinting on Adsorption Capacity and Selectivity for Protein Templates. Biomacromolecules. 2016;17(12):4045-4053. [95] LI S, ZHANG T, XU W, et al. Sarcoma-Targeting Peptide-Decorated Polypeptide Nanogel Intracellularly Delivers Shikonin for Upregulated Osteosarcoma Necroptosis and Diminished Pulmonary Metastasis. Theranostics. 2018;8(5):1361-1375. [96] MATEEN S, MOIN S, ZAFAR A, et al. Redox signaling in rheumatoid arthritis and the preventive role of polyphenols. Clin Chim Acta. 2016;463:4-10. [97] FENG N, YANG M, FENG X, et al. Reduction-Responsive Polypeptide Nanogel for Intracellular Drug Delivery in Relieving Collagen-Induced Arthritis. ACS Biomater Sci Eng. 2018;4(12):4154-4162. [98] GEHRING J, TREPKA B, KLINKENBERG N, et al. Sunlight-Triggered Nanoparticle Synergy: Teamwork of Reactive Oxygen Species and Nitric Oxide Released from Mesoporous Organosilica with Advanced Antibacterial Activity. J Am Chem Soc. 2016;138(9):3076-3084. [99] HUANG Z, FU J, ZHANG Y. Nitric Oxide Donor-Based Cancer Therapy: Advances and Prospects. J Med Chem. 2017;60(18):7617-7635. [100] YEO J, LEE YM, LEE J, et al. Nitric Oxide-Scavenging Nanogel for Treating Rheumatoid Arthritis. Nano Lett. 2019;19(10):6716-6724. [101] ZHENG Y, LIANG Y, ZHANG D, et al. Gelatin-Based Hydrogels Blended with Gellan as an Injectable Wound Dressing. ACS Omega. 2018; 3(5):4766-4775. [102] HAN Y, CAO Y, LEI H. Dynamic Covalent Hydrogels: Strong yet Dynamic. Gels. 2022; 8(9):577. [103] ZHANG A, LIU Y, QIN D, et al. Research status of self-healing hydrogel for wound management: A review. Int J Biol Macromol. 2020;164:2108-2123. [104] HE X, ZHANG C, WANG M, et al. An electrically and mechanically autonomic self-healing hybrid hydrogel with tough and thermoplastic properties. ACS Appl Mater Interfaces. 2017; 9(12):11134-11143. [105] LÜ S, BAI X, LIU H, et al. An injectable and self-healing hydrogel with covalent cross-linking in vivo for cranial bone repair. J Mater Chem B. 2017;5(20):3739-3748. [106] GEORGE MN, LIU X, MILLER AL, et al. Phosphate functionalization and enzymatic calcium mineralization synergistically enhance oligo[poly(ethylene glycol) fumarate] hydrogel osteoconductivity for bone tissue engineering. J Biomed Mater Res A. 2020;108(3):515-527. [107] KUMAR R, PARASHAR A. Atomistic simulations of pristine and nanoparticle reinforced hydrogels: A review. WIREs Comput Mol Sci. 2023;13(4):e1655. [108] FU Y, CUI S, LUO D, et al. Novel Inorganic Nanomaterial-Based Therapy for Bone Tissue Regeneration. Nanomaterials (Basel). 2021;11(3):789. [109] WAN T, ZHANG M, JIANG HR, et al. Tissue-Engineered Nanomaterials Play Diverse Roles in Bone Injury Repair. Nanomaterials (Basel). 2023;13(9):1449. [110] GONG W, HUANG HB, WANG XC, et al. Construction of a sustained-release hydrogel using gallic acid and lysozyme with antimicrobial properties for wound treatment. Biomater Sci. 2022;10(23):6836-6849. [111] ZHENG Y, LIANG Y, ZHANG D, et al. Gelatin-Based Hydrogels Blended with Gellan as an Injectable Wound Dressing. ACS Omega. 2018;3(5):4766-4775. [112] MA Y, SUN L, ZHANG J, et al. Exosomal mRNAs for Angiogenic-Osteogenic Coupled Bone Repair. Adv Sci (Weinh). 2023;10(33): e2302622. [113] WANG Z, YAN HH, WANG M, et al. Enhancing Bone-Titanium integration through hydrogel coating mediated sequential M1/M2 polarization of interfacial macrophages. Chem Eng J. 2024;500:157088. |
| [1] | 范 筱, 李慧云, 窦馥国, 张 楠, 张新颜. 纳米磷酸铜水凝胶敷料促进小鼠皮肤创面愈合[J]. 中国组织工程研究, 2026, 30(32): 8413-8419. |
| [2] | 李轩泽, 方汉洪, 徐 哲. 海藻酸钠-羟基磷灰石-氧化石墨烯水凝胶的制备及细胞相容性[J]. 中国组织工程研究, 2026, 30(32): 8420-8426. |
| [3] | 刘邦定, 唐永亮, 李 妮, 任 波. 载槲皮素水凝胶材料治疗感染性骨缺损[J]. 中国组织工程研究, 2026, 30(32): 8427-8435. |
| [4] | 陈世超, 邓云艺, 赵任圣洁, 余 科, 李广文. 负载槲皮素-纳米银粒子光敏水凝胶应用于感染创口的抗菌性能[J]. 中国组织工程研究, 2026, 30(32): 8436-8442. |
| [5] | 丁 浩, 高 原, 李 彬, 于 瑞, 王建茹, 孙雨蝶, 王选阳, 张文评, 朱明军. 水凝胶心脏贴片和中药联合治疗急性心肌梗死的潜能与应用前景[J]. 中国组织工程研究, 2026, 30(32): 8479-8486. |
| [6] | 费孝渊, 许 姣, 史 惠. DNA水凝胶在组织修复中的应用策略[J]. 中国组织工程研究, 2026, 30(32): 8487-8495. |
| [7] | 李明慧, 郄浩宇, 潘 敏, 毕蕊洁, 吕晓萌, 张浩雅, 韩逸飞. 基于水凝胶载体的类风湿性关节炎治疗递送系统[J]. 中国组织工程研究, 2026, 30(32): 8496-8501. |
| [8] | 孟怡豪, 张 帅. 自愈水凝胶在运动损伤预防与康复中的应用[J]. 中国组织工程研究, 2026, 30(32): 8502-8511. |
| [9] | 邱佳静, 黄立渠. 组织工程尿道修复与重建中的水凝胶支架[J]. 中国组织工程研究, 2026, 30(32): 8521-8528. |
| [10] | 郑 颖, 李梦瑶, 郑帆帆, 何 昭, 张 宁, 邹嘉伦, 李优磊, 高 枫. 搭载生物材料细胞外泌体修复脊髓损伤的作用机制[J]. 中国组织工程研究, 2026, 30(32): 8544-8554. |
| [11] | 夏瑾燕, 苏梅芳, 杨文育, 胡琦兰, 龚 丽. 新型材料在糖尿病足创面组织修复中的应用[J]. 中国组织工程研究, 2026, 30(32): 8563-8568. |
| [12] | 胡丽群, 肖东琴, 马晨曦, 李卓韩, 闫吉元, 李 忠, 贺 葵, 段 可. 硫酸钙-氧化镁复合材料作为抗感染植骨材料的性能[J]. 中国组织工程研究, 2026, 30(32): 8309-8318. |
| [13] | 杨 光, 印治涛, 许 燕. 3D打印异烟肼脂质体光热支架及性能评价[J]. 中国组织工程研究, 2026, 30(26): 6701-6709. |
| [14] | 赵张红, 金东升, 阮世强, 黄文良, 万 喻, 田仁元, 邓 江. 淫羊藿苷缓释微球三维支架的体外促成骨与抗炎性能[J]. 中国组织工程研究, 2026, 30(26): 6710-6718. |
| [15] | 皮志龙, 李嘉源, 谭志超, 陆小梅, 张志强, 叶翔凌. 3D打印新补骨脂异黄酮涂层支架调节成骨/破骨细胞活性促进骨再生[J]. 中国组织工程研究, 2026, 30(26): 6736-6743. |
目前,骨缺损的治疗策略主要包括自体骨移植、同种异体骨移植和异种骨移植。其中,自体骨移植因良好的组织相容性和成骨能力被认为是骨缺损修复的“金标准”[7-8],然而,该方法仍存在诸多限制,如供体部位损伤、形态不匹配、骨源有限以及术后并发症等[9]。因此,基于生物材料的骨组织工程策略应运而生,并逐渐成为解决骨缺损问题的重要方向。近年来,以骨组织工程支架、纳米药物递送系统和3D打印技术为代表的新型治疗方法取得了显著进展,为骨修复提供了更具前景的方案[10]。
在骨组织工程领域,细胞外基质作为一种复杂的生物大分子网络,在细胞黏附、增殖、分化及组织重塑过程中发挥了至关重要的作
用[11]。水凝胶因高度仿生的三维网络结构,可模拟天然细胞外基质的微环境,为细胞提供适宜的生长支架,从而促进成骨细胞的分化和组织再生,因此,水凝胶被认为是骨组织工程中极具潜力的生物材料之一[12-13]。
水凝胶按照来源可分为天然聚合物水凝胶、合成聚合物水凝胶及复合水凝胶。天然聚合物水凝胶(如壳聚糖、海藻酸盐、胶原蛋白、明胶和透明质酸等)具有优异的生物相容性,然而,它的机械强度较低、降解速率难以调控,并且在某些情况下可能引发免疫原性反应,临床应用受限。相比之下,合成水凝胶(如聚乙二醇、聚乙烯醇、聚乳酸和聚己内酯)具有良好的力学性能和可调控的降解特性,但缺乏内源性生物活性[14]。因此,结合天然与合成材料优势的复合水凝胶成为当前研究的热点之一。此外,有机-无机纳米复合水凝胶的出现,不仅提高了材料的机械强度,还可通过负载生长因子(如骨形态发生蛋白、转化生长因子β等)实现局部控释,进一步促进骨再生[15-16]。
根据骨缺损的类型和治疗需求,水凝胶可被设计为不同形式的应用方式,例如:植入式水凝胶支架具有良好的机械强度和形态可调特性,可用于修复大范围骨缺损[17-18];注射型水凝胶具备可流动性和原位凝胶化特性,在治疗软骨缺损、小面积骨损伤时具有显著优势[19];此外,纳米凝胶或微凝胶也被探索为药物递送载体,通过局部或全身给药方式用于治疗骨相关疾病,如类风湿性关节炎[20-21]、骨关节炎[22]、骨缺损[23-24]。
综上所述,水凝胶作为一种高度仿生且可调控的生物材料,在骨组织工程领域展现出广阔的应用前景。该文重点分析水凝胶在骨组织修复中的最新研究进展,以期为骨组织工程的未来发展提供参考。
1.1.1 检索人及检索时间 由第一作者在 2025年1月进行检索。
1.1.2 文献检索时限 各数据库建库至2025年1月。
1.1.3 检索数据库 PubMed数据库和中国知网。
1.1.4 检索词 英文检索词为“Hydrogel,Bone Tissue Engineering,Nanomaterials,Bone Regeneration Mechanism,Bone Defect Repair”,中文检索词为“水凝胶,骨组织工程,纳米材料,骨再生机制,骨缺损修复”。
1.1.5 检索文献类型 研究原著及综述。
1.1.6 手工检索情况 手工查阅相关书籍。
1.1.7 检索策略 中国知网、PubMed数据库检索策略,见图1。
1.1.8 检索文献量 初步检索得到文献1 407篇,包括英文文献 1 254篇、中文文献153篇。
1.2 文章框架结构设计 见图2。
1.3 入组标准
纳入标准:①水凝胶在骨组织工程中的相关应用研究;②文献质量、相关性、可靠性高,或创新性较为突出的文献;③优先选择最近15年内发表的文献。
排除标准:①与研究主题无关的文献;②重复性文献;③观点陈旧或存在争议的文献。
1.4 文献质量评价和筛选 共检索到1 407篇文献,初筛剔除重复文献后,通过泛读对剩余文献的标题、摘要进行筛选,无法判别时精读全文,选取与主题更为相符的文献,最终纳入符合要求的英文文献113篇。文献筛选流程详见图3。
3.2 未来发展趋势 未来,水凝胶的研究将更加注重多功能一体化的系统构建,如通过引入温度、pH值、酶或磁场等智能响应模块,实现时空可控的生物因子或药物释放,增强水凝胶在骨再生全过程中的动态干预能力。同时,开发具有生物可降解性、力学性能可调和微环境适应性的高分子材料,将有效提升水凝胶在复杂生理环境中的稳定性与组织整合能力。借助高精度3D打印技术,未来可实现结构可控、功能分区、个性定制的水凝胶,以匹配患者个体化缺损的结构特征。
此外,针对不同类型骨组织缺损(如软骨、松质骨、皮质骨等)在力学需求与再生微环境上的差异,也将促使水凝胶材料向更精准应用导向演进。未来趋势还包括跨学科融合的多模态协同治疗策略,如结合骨免疫调节、血管化引导、电刺激与光热治疗等手段协同提升成骨与修复效率。同时,人工智能与多组学数据分析将被引入材料设计与修复方案优化过程,基于高通量筛选、生物信息学和深度学习预测算法,实现水凝胶组分选择、结构设计、响应模式与临床适配的系统性智能优化,推动水凝胶材料向智能化与临床可转化方向迈进。
3.3 挑战与建议 尽管当前在水凝胶支架领域已取得显著进展,但它在临床转化中仍面临诸多挑战:①力学性能与生物性能的兼容性问题:在提升支架力学强度的同时往往会牺牲其生物活性,建议未来研究应聚焦于构建兼具力学强度与生物功能性的复合材料体系;②药物释放的精准调控难度大:现有递送系统多存在释放过快或持续性差的问题,建议引入响应性材料(如温敏、磁敏、光敏材料)以实现按需释放,并通过系统建模优化释放参数;③个体化治疗的实现难度:个体骨损伤情况存在显著差异,标准化支架难以满足所有患者需求,建议结合临床影像数据,发展基于3D打印与计算机辅助设计的个性化支架构建平台;④长期安全性与生物降解性的评估不足:多数研究集中于短期修复效果,缺乏对材料在体内长期稳定性、降解产物毒性及免疫反应的系统评估,建议在动物实验基础上构建更为完善的生物安全性评价体系,为临床应用提供依据。
综上所述,水凝胶支架在骨组织修复中的应用潜力巨大,但它的临床应用仍需多学科协同推进,通过材料科学、生物学、医学及工程技术的融合创新,以实现高效、安全和可控的组织再生策略。
该文围绕水凝胶在骨组织工程中的应用展开系统综述,重点聚焦其设计与功能化改性策略、促进骨再生的作用机制以及近年来在骨缺损修复中的前沿研究成果。首先,文章深入探讨了通过交联方式调控物理化学性能、表面功能基团修饰以及纳米填料复合等方式提升水凝胶生物性能的研究进展。其次,系统梳理了水凝胶如何通过细胞黏附、免疫调控、生长因子递送和信号通路调节等机制促进骨形成。最后,总结了水凝胶在复杂骨组织修复、软骨-骨界面重建及智能递释系统等方面的创新性应用。该文的亮点在于整合多维度策略构建多功能复合水凝胶体系,并以功能化设计为核心视角,强调其在应对临床骨修复挑战中的潜力,为今后水凝胶材料在组织工程和个性化治疗领域的发展提供理论基础和技术指导。
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||