中国组织工程研究 ›› 2026, Vol. 30 ›› Issue (32): 8364-8371.doi: 10.12307/2026.208
• 膜生物材料 membrane biomaterials • 上一篇 下一篇
新型胶原膜修复大鼠颅骨缺损
杨 平1,戚晓阳2,雷智杰3,陈一心1,2,邱旭升1,2,3
- 1南京中医药大学鼓楼临床医学院骨科,江苏省南京市 210008;2南京大学医学院附属鼓楼医院骨科,江苏省南京市 210008;3江苏大学鼓楼临床医学院骨科,江苏省南京市 210008
Novel collagen membrane in repairing skull bone defects in rats
Yang Ping1, Qi Xiaoyang2, Lei Zhijie3, Chen Yixin1, 2, Qiu Xusheng1, 2, 3
- 1Department of Orthopedics, Nanjing Drum Tower Hospital Clinical College of Nanjing University of Chinese Medicine, Nanjing 210008, Jiangsu Province, China; 2Department of Orthopedics, Nanjing Drum Tower Hospital Affiliated to Nanjing University Medical School, Nanjing 210008, Jiangsu Province, China; 3Department of Orthopedics, Nanjing Drum Tower Hospital Clinical College of Jiangsu University, Nanjing 210008, Jiangsu Province, China
摘要:
文题释义:
引导骨再生技术:是口腔颌面外科常用的一种骨缺损修复技术,该技术的基本原理:根据各类组织细胞迁移速度不同的特点,将屏障膜置于软组织和骨缺损之间建立生物屏障,创造一个相对封闭的组织环境,阻止结缔组织细胞和上皮细胞进入骨缺损区,允许有潜在生长能力、迁移速度较慢的前体成骨细胞优先进入骨缺损区优势生长,同时保护血凝块,减缓压力,实现缺损区的骨修复性再生。
屏障膜:是引导骨再生技术中所使用的一种膜,用于隔离骨缺损区与周围软组织。屏障膜分为不可吸收屏障膜(如钛膜、膨体聚四氟乙烯膜)和可吸收屏障膜(如胶原膜),不可吸收屏障膜有需要二次手术取出的缺点,而现有商用胶原膜存在机械强度差的缺点。
背景:引导骨再生技术常用的可吸收屏障膜——Bio-Gide胶原膜(猪皮来源)存在机械强度差、降解速度快的缺点,限制了它的临床应用。因此,研制理化性质更优的胶原膜具有重要意义。
目的:制备猪膀胱来源新型胶原膜,表征该膜的理化特性以及修复大鼠颅骨缺损的效果。
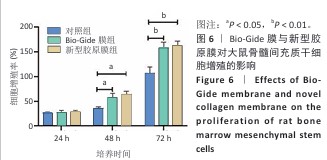
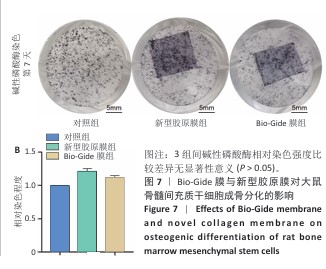
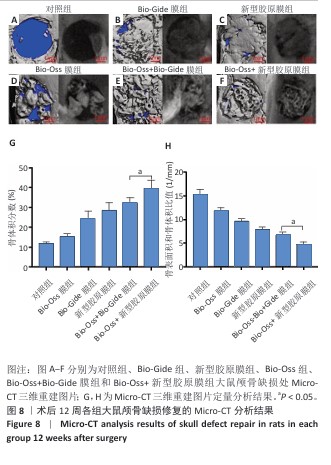
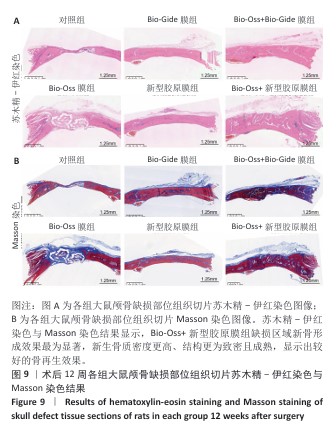
方法:①去除猪膀胱的浆膜层和部分肌肉层,制备新型胶原膜。表征新型胶原膜与Bio-Gide胶原膜的表面形貌、吸水率、孔隙率、降解率与拉伸弹性模量、极限载荷。②将大鼠骨髓间充质干细胞分别与新型胶原膜与Bio-Gide胶原膜共培养,以单独培养的细胞为对照,CCK-8法检测细胞增殖;成骨诱导后7 d,通过碱性磷酸酶染色评估成骨分化情况。③在18只SD大鼠颅骨矢状缝左右两侧各制作1个直径5 mm的圆形全层骨缺损,36个缺损部位随机分6组干预:对照组(n=6)缺损处不植入任何材料,Bio-Oss组(n=6)缺损处填充Bio-Oss骨粉,Bio-Gide组(n=6)缺损处覆盖Bio-Gide膜,新型胶原膜组(n=6)缺损处覆盖新型胶原膜,Bio-Oss+Bio-Gide组(n=6)缺损处填充Bio-Oss骨粉后覆盖Bio-Gide膜,Bio-Oss+新型胶原膜组(n=6)缺损处填入Bio-Oss骨粉后再覆盖新型胶原膜。术后12周取材,分别进行Micro-CT扫描与组织学观察。
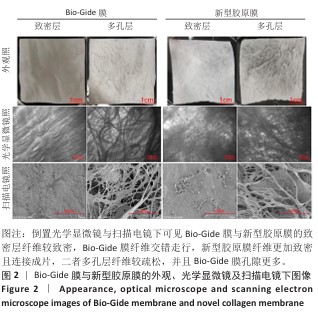
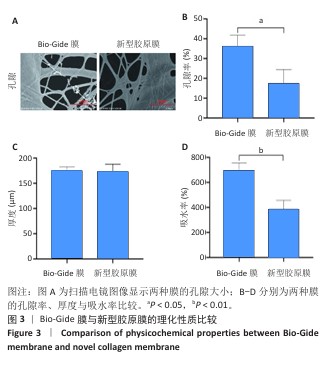
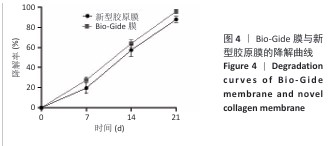
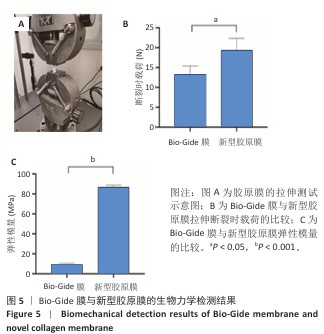
结果与结论:①扫描电镜下可见Bio-Gide膜与新型胶原膜的致密层纤维较致密,Bio-Gide膜纤维交错走行,新型胶原膜纤维更加致密且连接成片,二者多孔层纤维较疏松,并且Bio-Gide膜孔隙更多。新型胶原膜的孔隙率、吸水率均低于Bio-Gide膜(P < 0.05),拉伸弹性模量与极限载荷均高于Bio-Gide膜(P < 0.05)。②相较于Bio-Gide膜,新型胶原膜的降解更稳定。CCK-8检测显示,Bio-Gide膜与新型胶原膜均可促进大鼠骨髓间充质干细胞的增殖。碱性磷酸酶染色显示,Bio-Gide膜与新型胶原膜均不影响大鼠骨髓间充质干细胞的成骨分化。③Micro-CT扫描结果显示,对照组骨缺损处有少量新骨形成,其他5组骨缺损处有大量新骨形成,其中Bio-Oss+新型胶原膜组新骨形成量与骨结构成熟度最佳。苏木精-伊红染色与Masson染色显示,对照组缺损区新生骨形成较少,其余5组缺损区新生骨形成较多,其中Bio-Oss+新型胶原膜组新生骨质密度更高、结构更成熟。④结果表明,新型胶原膜的理化特性优于Bio-Gide膜,在配合使用Bio-Oss骨粉时,新型胶原膜表现出比Bio-Gide膜更优的骨修复效果。
https://orcid.org/0009-0004-6290-4213 (杨平)
中国组织工程研究杂志出版内容重点:生物材料;骨生物材料;口腔生物材料;纳米材料;缓释材料;材料相容性;组织工程
中图分类号: