Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (7): 1115-1120.doi: 10.3969/j.issn.2095-4344.2014.07.022
Previous Articles Next Articles
Tissue-engineered tooth regeneration: hotspots and clinical application
Liu Peng-peng1, Shen Hui-juan2, Wang Zi-yi3, Wu Yin-fang1, Jin Guang-yong1, Qi Qian-xian1, Zeng Xian-zhi4
- 1School of Medicine, Jiaxing University, Jiaxing 314001, Zhejiang Province, China
2Electrical Engineering Center, School of Mechanical Engineering, Jiaxing University, Jiaxing 314001, Zhejiang Province, China
3Department of Stomatology, Zhejiang Provincial People’s Armed Police Corps Hospital, Jiaxing 314000, Zhejiang Province, China
4Department of Anatomy, School of Medicine, Jiaxing University, Jiaxing 314001, Zhejiang Province, China
-
Revised:2013-12-04Online:2014-02-12Published:2014-02-12 -
Contact:Zeng Xian-zhi, M.D., Associate professor, Department of Anatomy, School of Medicine, Jiaxing University, Jiaxing 314001, Zhejiang Province, China -
About author:Liu Peng-peng, School of Medicine, Jiaxing University, Jiaxing 314001, Zhejiang Province, China -
Supported by:the Natural Science Foundation of Zhejiang Province, No. Y2110158; the Science and Technology Program of Jiaxing City, No. 2011AY1053-1, 2012AY1075-11; the Science and Technology Innovation Program of Undergraduate of Zhejiang Province, No. 2012R417006; Key Program of Student Research Training of Jiaxing University, No. 851712004
CLC Number:
Cite this article
Liu Peng-peng, Shen Hui-juan, Wang Zi-yi, Wu Yin-fang, Jin Guang-yong, Qi Qian-xian, Zeng Xian-zhi . Tissue-engineered tooth regeneration: hotspots and clinical application[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(7): 1115-1120.
share this article
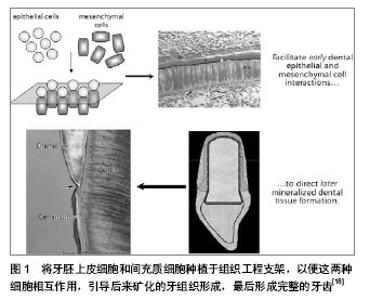
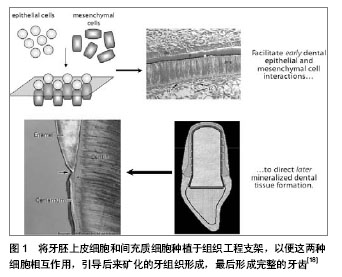
首先,组织工程牙齿再生常见的干细胞包括以下几种:基质干细胞是多潜能干细胞,能分化成各种细胞类型。可用于牙齿再生的潜在基质干细胞包括人类脱落的乳牙干细胞(stem cells from human exfoliated deciduous teeth, SHEDs)[9],成人牙髓干细胞(adult dental pulp stem cells, DPSCs)[10],来自牙乳头顶部的干细胞(stem cells from the apical part of the papilla, SCAPs)[11-13],来自牙泡的干细胞(stem cells from the dental follicle, DFSCs)[14],牙周韧带干细胞(periodontal ligament stem cells, PDLSCs)和骨髓来源的间充质干细胞 (mesenchymal stem cells,BMSCs)[15-17](图1)。"
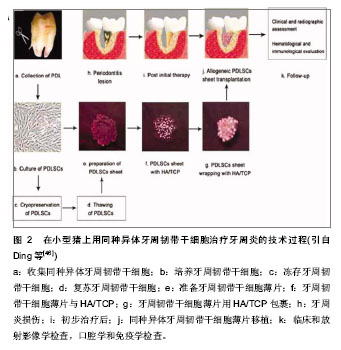
其次,用组织工程支架复合上述干细胞进行牙齿再生的实验有以下发展历程。 El-Backly等[19]尝试培养釉质器官,用来自6月龄猪的第三磨牙分离成单个的牙源性上皮细胞而次培养(无血清)在3T3-J2细胞滋养层。通过免疫染色和Western blotting实验发现次培养的牙源性上皮细胞表达釉质蛋白。次培养的牙源性上皮细胞和新鲜的牙间充质细胞被种植在胶原海绵支架中,然后移植到无胸腺大鼠。4周后釉质-牙质样复合物出现在移植结构上。结果说明这样的培养系统产生了能够分泌釉质蛋白且在体内形成釉质样组织的分化的生釉质样细胞。次培养技术的运用为进一步的牙组织工程和提高对釉质生物学的理解奠定了基础。 随着牙齿再生以及功能性牙齿组织修复机制知识的增多,当前的治疗策略开始让位给新进展的方法,比如组织工程和生物仿生学。 Peng等[20]培养出了牙本质结构。他们从兔牙分离得到牙泡干细胞,植入2种不同孔径的多孔性乳酸-共-羟基乙酸(lactic-co-glycolic acid)多聚体。这些细胞/支架结构被植入兔皮下。兔牙泡干细胞在扩增时呈现高的繁殖能力和克隆形成能力,植入支架12 d后产生矿化组织,组织学检查移植的样品可见形成了牙性骨质样结构和垂直平行排列的像管状牙质的管状双层结构。用组织工程的方法获得的组织非常像正常的牙质/牙泡样组织,今后可能用在牙齿再生或手术过程。 干细胞治疗牙齿缺陷或缺失是一个神奇而又先进的模式。然而,当牙齿来源的干细胞不是现成的背景下,这种策略又是一种挑战。Huo等[21]试图探索利用容易从皮肤组织得到的皮肤多潜能干细胞(dermal multipotent cells, DMCs)来诱导成牙齿源性的干细胞。通过有限的稀释技术,形成克隆的细胞群被游离且具有增生活动和多系分化潜能的特征。通过暴露在培养胚胎和新生的牙胚细胞的条件培养基培养,提高了皮肤多潜能干细胞的增生和矿物化活动,同时胚胎牙胚细胞条件培养基(embryonic tooth germ cell-conditioned medium, ETGC-CM)产生更多重要的作用。在体外用胚胎牙胚细胞条件培养基处理的皮肤多潜能干细胞,特异的细胞系标记物提示拟表型成牙质细胞。作为细胞小球移植到体内,胚胎牙胚细胞条件培养基处理的皮肤多潜能干细胞能够产生成块的矿物化组织,与牙泡干细胞移植到同样皮下口袋环境时相似。这些发现提示,尽管未分化的皮肤多潜能干细胞要成为成牙质细胞需要更充分和更连续的微环境,胚胎牙胚细胞条件培养基处理的皮肤多潜能干细胞实际上获得了牙泡干细胞的特性。他们的工作突显了利用皮肤多潜能干细胞作为一种可替代的细胞来源的潜能,希望发展成为牙齿再生研究更实用的策略和治疗方法。 Arany等[22]报道第一次尝试用一种自发永生化间充质细胞系制造生物工程牙齿。为了评估该细胞系的牙齿源性潜能,成牙质细胞系(odontoblast-lineage cells, OLC)与分离自具有相当分化能力的E14.5小鼠牙齿上皮复合。应用一种三维器官胚层培养方法来体外构建复合物。复合物被移植到宿主动物的肾囊下2周,三分之一的移植物发育成牙齿组织。成牙质细胞系源性的GFP阳性细胞用免疫组织化学方法证实能够矿化牙胚。它们最终分化成有功能的成牙质细胞。总之,牙齿再生实验中设法用成牙质细胞系作为牙齿间充质替代物。因此,该自发永生化间充质细胞系具有在未来应用中形成复合物、牙齿发育和生物工程研究的潜能。 Takahashi等[23]检测口腔上皮克隆的细胞系能否分化为成釉细胞然后与牙胚间充质细胞结合再生牙齿。带有明确特征形态的口腔上皮克隆细胞系来自p53缺乏的第18天小鼠胚胎。牙齿成型因子几乎完全而在E18时口腔上皮和牙齿上皮是分开的。用RT-PCR分析细胞系表明,可以观察到黏膜上皮标记物细胞角化素14,没有观察到成釉细胞标记物如成釉蛋白。当培养在滋养层或在胶原胶中1-3周时,形成分层上皮细胞和表达特定的分化标志物CK13,证明它们是口腔黏膜来源。接下来,生物工程牙齿用细胞系和胚胎磨牙间充质组织种植在肾囊培养两三周。6个细胞系中的5个细胞系再生如同自然牙齿的钙化结构。结果表示一些口腔上皮细胞在E18拥有分化成成釉细胞的能力。另外,在本研究建立的细胞系对研究牙齿口腔生成和牙齿再生的过程是有用的模型。 虽然已经准确地知道牙齿上皮-间充质细胞相互作用在牙齿发育中起关键作用,适宜的牙齿上皮-间充质细胞相互作用来再生牙齿的方法尚未建立。为接受这种挑战,形成预先设定好尺寸和形态的生物工程牙齿,Zhang等[24]描述了预先建立好的三维牙齿上皮-间充质细胞构筑的特征。人类牙髓细胞种植的胶原胶层与悬浮在低生长因子人工基质胶中的猪牙齿上皮细胞共培养。这种三维牙齿上皮-间充质细胞层在体外培养,或体内种植在裸鼠皮下。种植4周后组织学和免疫组织化学分析发现牙齿上皮-间充质细胞相互作用秩序良好,诱导表达牙组织特异标记物釉原蛋白和牙本质涎磷蛋白,以及基膜标记物Laminin 5和collagen Ⅳ,还有不规则的矿物质化组织形成。他们预期这些结果将为最终有利于建立可靠的详细描述牙组织和特定的形态和大小完全牙齿的方法。 Ohara等[25-26]选用胶原和纤维蛋白作为组织工程支架的材料,基于与牙乳头来源细胞的生物相容性,而结果与常用于牙齿、牙本质和骨组织工程的聚乙醇酸纤维和β-磷酸三钙多孔团块相比较。分离猪的牙胚来源细胞,植入其中一种支架,然后移植到裸鼠的背部。与聚乙醇酸或β-磷酸三钙比较,在胶原和纤维蛋白胶中更能经常观察到牙蕾样结构,但硬组织形成的数量更少。这些结果表明胶原和纤维蛋白胶支持早期牙蕾再生过程,可能与它们支持上皮和间充质细胞生长的能力有关。另外,在纤维蛋白和胶原胶中牙蕾的成熟是困难的,可能需要其他因子。 Kuo等[27-28]用另外一种支架来促进牙齿的再生。来自骨髓液的骨髓基质干细胞分泌的生长因子和形态发生素可能促进牙齿的再生。这个研究小组获取小型猪未萌出的第二磨牙胚基,并在体外培养以获得牙胚细胞。收集含有骨髓基质干细胞的下颌骨骨髓液。悬浮于骨髓液中的牙胚细胞被种植在由明胶、6-磷酸-软骨素和透明质酸形成的三聚体的支架(GCHT scaffold)中。牙胚细胞/骨髓液/GCHT支架被自体移植到原来的牙槽臼中。移植后40周放射影像学和组织学检查确认牙齿的再生。也取得相似的结果:1只动物长出了一个完整的牙,包括形态明确的牙冠、牙根、牙泡、牙釉质、牙本质、成牙质细胞、牙骨质、血管和牙周韧带。3只动物有一个未萌出的牙,表达牙本质基质蛋白1,血管内皮生长因子、骨桥蛋白;另2只动物有牙本质小管的牙状结构。结果提示了牙胚细胞加上含有骨髓基质干细胞的骨髓液和合适的支架能够促进自体移植的牙齿再生。 这个研究小组的的另外一个牙齿组织工程实验也得到了相似的结果[29-30]。富血小板纤维蛋白含有丰富的生长因子和细胞因子,可以促进组织再生。相应地,富血小板纤维蛋白被加入到纤维蛋白胶中使微环境富集生长因子。收集来自小型猪未萌出的第二磨牙牙蕾然后在体外培养3周来获得牙胚细胞(dental bud cells, DBCs)。在手术前取全血获得富血小板纤维蛋白和纤维蛋白胶。将牙胚细胞悬浮于纤维蛋白胶然后用富血小板纤维蛋白包裹,形成的牙胚细胞-纤维蛋白胶-富血小板纤维蛋白复合物自体移植到原位牙槽陷凹中。移植36周后放射影像学和组织学检查证明可以再生牙齿。免疫组织化学染色检测出与牙齿再生相关的蛋白表达,包括细胞角蛋白14、牙本质基质蛋白1、血管内皮生长因子和骨桥蛋白。形成的牙齿有牙冠、牙根、牙泡、釉质、牙本质、成牙细胞、牙骨质、血管和牙周韧带,并具有明确的形态。这个研究证明了在猪模型上用自体细胞移植,即牙胚细胞植入纤维蛋白胶-富血小板纤维蛋白能够再生一个完整的牙齿。 迄今为止,最为成功的牙齿再生研究当属Sujesh等[31-34]研究小组,在一个啮齿类动物模型上,用生物工程方法制造的单个牙齿单位(包含了成熟的牙齿、牙周韧带和牙槽骨),用受体骨塑形整合被成功地移植到一个合适大小的牙槽骨洞中。用生物工程方法制造的单个牙齿单位在垂直的方向上完整地修复了啮齿类动物下颌骨的缺陷。植入的生物工程牙齿展现了生理学牙齿功能比如咀嚼、牙周韧带功能。这个研究代表了一个实质性的进展,而且证明了作为下一代再生疗法的生物工程成熟器官替代的真正潜能。 另外,也有用组织工程方法修复牙槽骨缺损[35-39]。王荣等[40-43]探讨小型猪牙齿在牙槽骨缺损修复区的移动情况,选用6只实验用小型猪(8-10个月龄),在一侧前磨牙近中造成直径为15 mm,高为10 mm的圆柱状骨缺损区。获取小型猪的自体骨髓基质细胞,分离培养并诱导为成骨样细胞。将小型猪自体骨髓基质干细胞与生物复合体(60%羟基磷灰石+40%磷酸三钙)复合后,种植到骨缺损区。另一侧作为对照侧。术后14周用60 g力牵引双侧第一前磨牙近中移动。于12周后测量双侧前磨牙的移动距离。进行配对t检验比较。结果6只小型猪两侧第一前磨牙近中移动平均距离的差异无显著性意义(P > 0. 05)。结果表明应用细胞支架构建方式,可修复小型猪的上颌骨缺损,修复后不会对牙齿移动产生不良影响。 来自人类脱落牙齿的干细胞已经被证明是一种新的出生后干细胞群,具有多潜能分化能力,包括在体内产生矿化组织。Zheng等[44-45]为了检验利用这些细胞再生修复颌面骨骼缺损的有效性,分离自体小型猪脱落牙齿的干细胞,然后移植到下颌骨缺损处。结果表明来自小型猪脱落牙齿干细胞—一种自体的并且容易获得的干细胞来源,移植6个月后能够修复合适大小的下颌骨缺损。这种大型动物模型上特别是猪的临床前研究使得研究人员可以检验干细胞/支架复合物在恢复口腔面部骨骼缺损和提供快速应用在人类以干细胞为基础的口腔颌面重建治疗上的可行性。 牙周炎是人类最常见的感染性疾病。它的主要原因是牙齿缺失及其相关的许多系统性疾病。至今,仍然没有合适的方法来实现功能性牙周组织的再生。Ding等[46]建立了一种神奇的方法,在小型猪上用同种异体牙周韧带干细胞(allogeneic periodontal ligament stem cells,PDLSCs)治愈了牙周炎。在自体或同种异体的牙周韧带干细胞移植后12周取得了明显的牙周组织再生。基于临床评估,CT检查和组织学检查,在自体或同种异体移植组之间并没有什么不同。另外,也没有观察到免疫排斥反应。有趣的是,他们发现人类牙周韧带干细胞没有表达(HLA)-Ⅱ DR和协同刺激分子。牙周韧带干细胞不会诱导或抑制T细胞增生。他们的工作证明牙周韧带干细胞具有低免疫原性和明显的免疫抑制通过PGE2诱导的T细胞无力。他们还形成了一个标准的在小型猪上用同种异体牙周韧带干细胞治疗牙周炎的技术过程(图2)。"
| [1] Warreth A, McAleese E, McDonnell P,et al. Dental implants and single implant-supported restorations.J Ir Dent Assoc. 2013;59(1):32-43.[2] Cudney SM, Vieira AR.Molecular factors resulting in tooth agenesis and contemporary approaches for regeneration: a review.Eur Arch Paediatr Dent. 2012;13(6):297-304.[3] Lymperi S, Ligoudistianou C, Taraslia V,et al.Dental Stem Cells and their Applications in Dental Tissue Engineering. Open Dent J. 2013;7:76-81. [4] Sujesh M, Rangarajan V, Ravi Kumar C,et al. Stem cell mediated tooth regeneration: new vistas in dentistry. J Indian Prosthodont Soc. 2012;12(1):1-7. [5] Zheng Y, Wang XY, Wang YM, et al. Dentin regeneration using deciduous pulp stem/progenitor cells. J Dent Res. 2012;91(7):676-682.[6] Dadu SS.Tooth regeneration: current status.Indian J Dent Res. 2009;20(4):506-507. [7] Yang KC, Wang CH, Chang HH,et al. Fibrin glue mixed with platelet-rich fibrin as a scaffold seeded with dental bud cells for tooth regeneration.J Tissue Eng Regen Med. 2012; 6(10): 777-785. [8] Oshima M, Mizuno M, Imamura A,et al. Functional tooth regeneration using a bioengineered tooth unit as a mature organ replacement regenerative therapy. PLoS One. 2011; 6(7):e21531. [9] Sloan AJ, Smith AJ.Stem cells and the dental pulp: potential roles in dentine regeneration and repair. Oral Dis. 2007; 13(2):151-157.[10] Otaki S, Ueshima S, Shiraishi K,et al. Mesenchymal progenitor cells in adult human dental pulp and their ability to form bone when transplanted into immunocompromised mice.Cell Biol Int. 2007;31(10):1191-1197. [11] Yokoi T, Saito M, Kiyono T,et al. Establishment of immortalized dental follicle cells for generating periodontal ligament in vivo.Cell Tissue Res. 2007;327(2):301-311. [12] Kémoun P, Laurencin-Dalicieux S, Rue J,et al. Human dental follicle cells acquire cementoblast features under stimulation by BMP-2/-7 and enamel matrix derivatives (EMD) in vitro. Cell Tissue Res. 2007;329(2):283-294. [13] Wu J, Jin F, Tang L,et al. Dentin non-collagenous proteins (dNCPs) can stimulate dental follicle cells to differentiate into cementoblast lineages.Biol Cell. 2008;100(5):291-302.[14] Tsuchiya S, Honda MJ, Shinohara Y,et al. Collagen type I matrix affects molecular and cellular behavior of purified porcine dental follicle cells.Cell Tissue Res. 2008;331(2): 447-459.[15] Trubiani O, Orsini G, Zini N,et al. Regenerative potential of human periodontal ligament derived stem cells on three-dimensional biomaterials: a morphological report.J Biomed Mater Res A. 2008;87(4):986-993. [16] Yang ZH, Zhang XJ, Dang NN,et al. Apical tooth germ cell-conditioned medium enhances the differentiation of periodontal ligament stem cells into cementum/periodontal ligament-like tissues.J Periodontal Res. 2009;44(2):199-210. [17] Jing W, Wu L, Lin Y,et al. Odontogenic differentiation of adipose-derived stem cells for tooth regeneration: necessity, possibility, and strategy. Med Hypotheses. 2008;70(3): 540-542. [18] Yen AH, Yelick PC.Dental tissue regeneration - a mini-review. Gerontology. 2011;57(1):85-94.[19] El-Backly RM, Massoud AG, El-Badry AM,et al. Regeneration of dentine/pulp-like tissue using a dental pulp stem cell/poly(lactic-co-glycolic) acid scaffold construct in New Zealand white rabbits. Aust Endod J. 2008;34(2):52-67.[20] Peng L, Ye L, Zhou XD. Mesenchymal stem cells and tooth engineering.Int J Oral Sci. 2009;1(1):6-12. [21] Huo N, Tang L, Yang Z,et al. Differentiation of dermal multipotent cells into odontogenic lineage induced by embryonic and neonatal tooth germ cell-conditioned medium.Stem Cells Dev. 2010;19(1):93-104. [22] Arany S, Kawagoe M, Sugiyama T. Application of spontaneously immortalized odontoblast cells in tooth regeneration. Biochem Biophys Res Commun. 2009;381(1): 84-89.[23] Takahashi C, Yoshida H, Komine A,et al. Newly established cell lines from mouse oral epithelium regenerate teeth when combined with dental mesenchyme.In Vitro Cell Dev Biol Anim. 2010;46(5):457-468. [24] Zhang W, Ahluwalia IP, Yelick PC.Three dimensional dental epithelial-mesenchymal constructs of predetermined size and shape for tooth regeneration.Biomaterials. 2010;31(31):7995- 8003.[25] Ohara T, Itaya T, Usami K,et al. Evaluation of scaffold materials for tooth tissue engineering.J Biomed Mater Res A. 2010;94(3):800-805. [26] Liu L, Liu YF, Zhang J,et al. Ameloblasts serum-free conditioned medium: bone morphogenic protein 4-induced odontogenic differentiation of mouse induced pluripotent stem cells.J Tissue Eng Regen Med. 2013. doi: 10.1002/term.1742. [Epub ahead of print][27] Kuo TF, Lin HC, Yang KC,et al. Bone marrow combined with dental bud cells promotes tooth regeneration in miniature pig model.Artif Organs. 2011;35(2):113-121.[28] Wang Y, Preston B, Guan G.Tooth bioengineering leads the next generation of dentistry.Int J Paediatr Dent. 2012 ;22(6): 406-418.[29] Yang KC, Wang CH, Chang HH,et al. Fibrin glue mixed with platelet-rich fibrin as a scaffold seeded with dental bud cells for tooth regeneration.J Tissue Eng Regen Med. 2012;6(10): 777-785.[30] Yildirim S, Fu SY, Kim K,et al. Tooth regeneration: a revolution in stomatology and evolution in regenerative medicine.Int J Oral Sci. 2011;3(3):107-116. [31] Sujesh M, Rangarajan V, Ravi Kumar C,et al. Stem cell mediated tooth regeneration: new vistas in dentistry.J Indian Prosthodont Soc. 2012;12(1):1-7.[32] Liu Y, Jiang M, Hao W,et al.Skin epithelial cells as possible substitutes for ameloblasts during tooth regeneration.J Tissue Eng Regen Med. 2012. doi: 10.1002/term.1485. [Epub ahead of print][33] Zheng Y, Wang XY, Wang YM, et al. Dentin regeneration using deciduous pulp stem/progenitor cells.J Dent Res. 2012; 91(7):676-682. [34] Xiao L, Tsutsui T.Three-dimensional epithelial and mesenchymal cell co-cultures form early tooth epithelium invagination-like structures: expression patterns of relevant molecules.J Cell Biochem. 2012;113(6):1875-1885.[35] Zamiri B, Shahidi S, Eslaminejad MB,et al. Reconstruction of human mandibular continuity defects with allogenic scaffold and autologous marrow mesenchymal stem cells.J Craniofac Surg. 2013;24(4):1292-1297. [36] Su J, Xu H, Sun J,et al. Dual Delivery of BMP-2 and bFGF from a New Nano-Composite Scaffold, Loaded with Vascular Stents for Large-Size Mandibular Defect Regeneration.Int J Mol Sci. 2013;14(6):12714-12728. [37] Streckbein P, Jäckel S, Malik CY,et al. Reconstruction of critical-size mandibular defects in immunoincompetent rats with human adipose-derived stromal cells.J Craniomaxillofac Surg. 2013;41(6):496-503. [38] Sándor GK, Tuovinen VJ, Wolff J,et al. Adipose stem cell tissue-engineered construct used to treat large anterior mandibular defect: a case report and review of the clinical application of good manufacturing practice-level adipose stem cells for bone regeneration.J Oral Maxillofac Surg. 2013; 71(5):938-50. [39] Fricain JC, Schlaubitz S, Le Visage C,et al. A nano-hydroxyapatite--pullulan/dextran polysaccharide composite macroporous material for bone tissue engineering.Biomaterials. 2013;34(12):2947-2959. [40] 王荣,孙正,刘华松.BMSC-HA/CP修复牙槽骨缺损后牙齿移动的实验研究[J].中华老年口腔医学杂志, 2008, 6(1):35-37.[41] Bains R, Bains VK, Loomba K,et al. Management of pulpal floor perforation and grade II Furcation involvement using mineral trioxide aggregate and platelet rich fibrin: A clinical report.Contemp Clin Dent. 2012;3(Suppl 2):S223-227. [42] Xiao L, Tsutsui T.Three-dimensional epithelial and mesenchymal cell co-cultures form early tooth epithelium invagination-like structures: expression patterns of relevant molecules.J Cell Biochem. 2012;113(6):1875-1885. [43] Hayden RE, Mullin DP, Patel AK. Reconstruction of the segmental mandibular defect: current state of the art.Curr Opin Otolaryngol Head Neck Surg. 2012;20(4):231-236.[44] Zheng Y, Liu Y, Zhang CM,et al. Stem cells from deciduous tooth repair mandibular defect in swine.J Dent Res. 2009; 88(3):249-254.[45] Yu H, Yang X, Cheng J,et al. Distraction osteogenesis combined with tissue-engineered cartilage in the reconstruction of condylar osteochondral defect.J Oral Maxillofac Surg. 2011;69(12):e558-564. [46] Ding G, Liu Y, Wang W,et al. Allogeneic periodontal ligament stem cell therapy for periodontitis in swine.Stem Cells. 2010; 28(10):1829-1838. [47] Takahashi C, Yoshida H, Komine A,et al. Newly established cell lines from mouse oral epithelium regenerate teeth when combined with dental mesenchyme.In Vitro Cell Dev Biol Anim. 2010;46(5):457-468. [48] Snead ML.Whole-tooth regeneration: it takes a village of scientists, clinicians, and patients.J Dent Educ. 2008;72(8): 903-911. |
| [1] | Pu Rui, Chen Ziyang, Yuan Lingyan. Characteristics and effects of exosomes from different cell sources in cardioprotection [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 1-. |
| [2] | Lin Qingfan, Xie Yixin, Chen Wanqing, Ye Zhenzhong, Chen Youfang. Human placenta-derived mesenchymal stem cell conditioned medium can upregulate BeWo cell viability and zonula occludens expression under hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(在线): 4970-4975. |
| [3] | Zhang Tongtong, Wang Zhonghua, Wen Jie, Song Yuxin, Liu Lin. Application of three-dimensional printing model in surgical resection and reconstruction of cervical tumor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(9): 1335-1339. |
| [4] | Zhang Xiumei, Zhai Yunkai, Zhao Jie, Zhao Meng. Research hotspots of organoid models in recent 10 years: a search in domestic and foreign databases [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(8): 1249-1255. |
| [5] | Hou Jingying, Yu Menglei, Guo Tianzhu, Long Huibao, Wu Hao. Hypoxia preconditioning promotes bone marrow mesenchymal stem cells survival and vascularization through the activation of HIF-1α/MALAT1/VEGFA pathway [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 985-990. |
| [6] | Shi Yangyang, Qin Yingfei, Wu Fuling, He Xiao, Zhang Xuejing. Pretreatment of placental mesenchymal stem cells to prevent bronchiolitis in mice [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 991-995. |
| [7] | Liang Xueqi, Guo Lijiao, Chen Hejie, Wu Jie, Sun Yaqi, Xing Zhikun, Zou Hailiang, Chen Xueling, Wu Xiangwei. Alveolar echinococcosis protoscolices inhibits the differentiation of bone marrow mesenchymal stem cells into fibroblasts [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 996-1001. |
| [8] | Fan Quanbao, Luo Huina, Wang Bingyun, Chen Shengfeng, Cui Lianxu, Jiang Wenkang, Zhao Mingming, Wang Jingjing, Luo Dongzhang, Chen Zhisheng, Bai Yinshan, Liu Canying, Zhang Hui. Biological characteristics of canine adipose-derived mesenchymal stem cells cultured in hypoxia [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1002-1007. |
| [9] | Geng Yao, Yin Zhiliang, Li Xingping, Xiao Dongqin, Hou Weiguang. Role of hsa-miRNA-223-3p in regulating osteogenic differentiation of human bone marrow mesenchymal stem cells [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1008-1013. |
| [10] | Lun Zhigang, Jin Jing, Wang Tianyan, Li Aimin. Effect of peroxiredoxin 6 on proliferation and differentiation of bone marrow mesenchymal stem cells into neural lineage in vitro [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1014-1018. |
| [11] | Zhu Xuefen, Huang Cheng, Ding Jian, Dai Yongping, Liu Yuanbing, Le Lixiang, Wang Liangliang, Yang Jiandong. Mechanism of bone marrow mesenchymal stem cells differentiation into functional neurons induced by glial cell line derived neurotrophic factor [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1019-1025. |
| [12] | Duan Liyun, Cao Xiaocang. Human placenta mesenchymal stem cells-derived extracellular vesicles regulate collagen deposition in intestinal mucosa of mice with colitis [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1026-1031. |
| [13] | Pei Lili, Sun Guicai, Wang Di. Salvianolic acid B inhibits oxidative damage of bone marrow mesenchymal stem cells and promotes differentiation into cardiomyocytes [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1032-1036. |
| [14] | Guan Qian, Luan Zuo, Ye Dou, Yang Yinxiang, Wang Zhaoyan, Wang Qian, Yao Ruiqin. Morphological changes in human oligodendrocyte progenitor cells during passage [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1045-1049. |
| [15] | Wang Feng, Zhou Liyu, Saijilafu, Qi Shibin, Ma Yanxia, Wei Shanwen. CaMKII-Smad1 promotes axonal regeneration of peripheral nerves [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(7): 1064-1068. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||