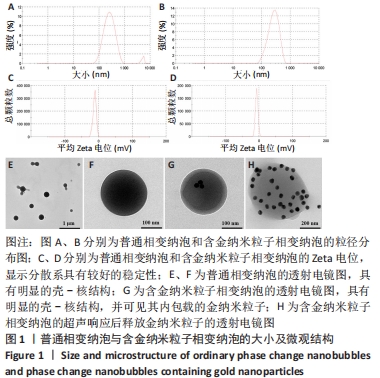
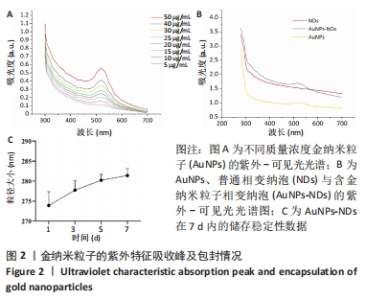
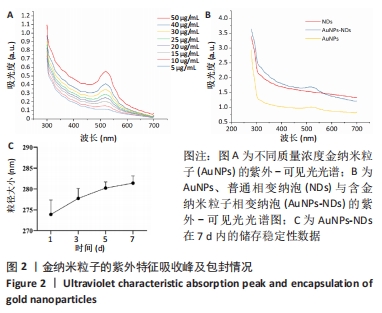
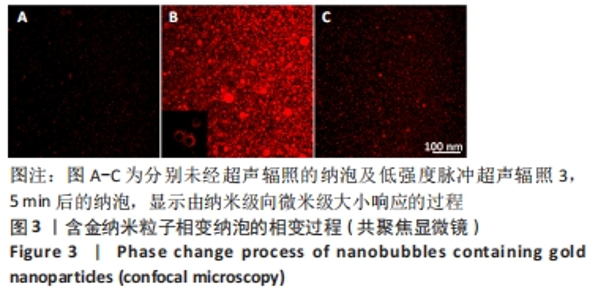
Chinese Journal of Tissue Engineering Research ›› 2024, Vol. 28 ›› Issue (15): 2330-2337.doi: 10.12307/2024.377
Previous Articles Next Articles
Ultrasound-optimized hydrogel scaffold used to promote transdermal delivery of gold nanoparticles
Guo Yuxin, Wang Hao, Li Mingqi, Chen Yueying, Pan Juhong, Huang Xin, Wang Zhiwen, Zhou Qing
- Renmin Hospital of Wuhan University, Wuhan 430060, Hubei Province, China
-
Received:2023-05-11Accepted:2023-06-25Online:2024-05-28Published:2023-09-19 -
Contact:Zhou Qing, PhD, Professor, Renmin Hospital of Wuhan University, Wuhan 430060, Hubei Province, China -
About author:Guo Yuxin, Master candidate, Renmin Hospital of Wuhan University, Wuhan 430060, Hubei Province, China -
Supported by:National Natural Science Foundation of China (General Project), No. 81971624 (to ZQ)
CLC Number:
Cite this article
Guo Yuxin, Wang Hao, Li Mingqi, Chen Yueying, Pan Juhong, Huang Xin, Wang Zhiwen, Zhou Qing. Ultrasound-optimized hydrogel scaffold used to promote transdermal delivery of gold nanoparticles[J]. Chinese Journal of Tissue Engineering Research, 2024, 28(15): 2330-2337.
share this article
Add to citation manager EndNote|Reference Manager|ProCite|BibTeX|RefWorks
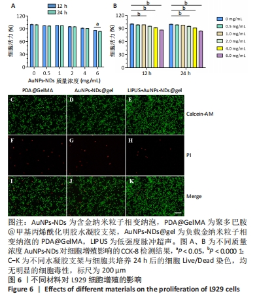
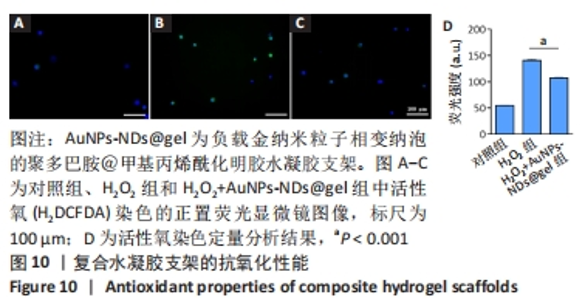
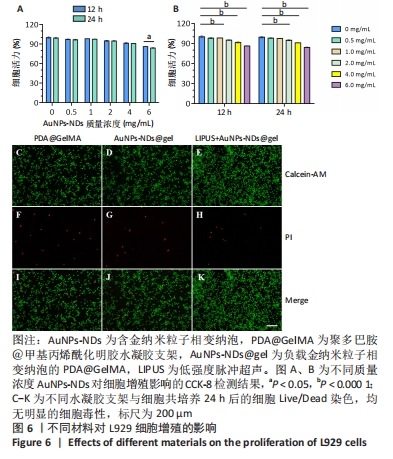
2.5 AuNPs-NDs和AuNPs-NDs@gel的生物相容性评价 将L929细胞分别与不同质量浓度的AuNPs-NDs孵育12 h和24 h,细胞存活率均> 90%。在相同AuNPs-NDs质量浓度条件下,孵育12 h与孵育24 h之后的细胞存活率比较差异无显著性意义(P > 0.05),当AuNPs-NDs质量浓度达6.0 mg/mL时,孵育24 h与孵育12 h相比细胞活力显著下降(P < 0.05);在相同时间条件下,AuNPs-NDs质量浓度低于1.0 mg/mL时细胞存活率与对照组比较差异无显著性意义(P > 0.05),大于此质量浓度后细胞活性开始下降,但总体存活率均> 85%,见图6A、B。 水凝胶支架的细胞相容性通过Live/Dead染色观察L929细胞的生长活性进行评价。培养24 h后,如图6C-K结果所示,水凝胶支架及其降解产物对L929细胞没有显示出明显的毒性,并且低强度脉冲超声+AuNPs-NDs@gel组还表现出更少的凋亡细胞。"

2.6 水凝胶支架的形貌观察 扫描电镜观察PDA@GelMA和AuNPs-NDs@gel的微观结构,见图7。两种材料截面均为多孔网状结构,孔隙呈类圆形,AuNPs-NDs@gel还可在较大孔内见到小孔分布。PDA@GelMA和AuNPs-NDs@gel的孔径大小分别为(73.24±16.26),(76.05±16.26) μm,这可能源于交联密度增大导致的孔径减小;定量分析测定PDA@GelMA的孔隙率为(57.6±5.0)%,AuNPs-NDs@gel的孔隙率为(65.7±3.6)%,二者比较差异有显著性意义(P < 0.01),说明低强度脉冲超声能够一定程度上提高水凝胶支架内部的孔隙率。"

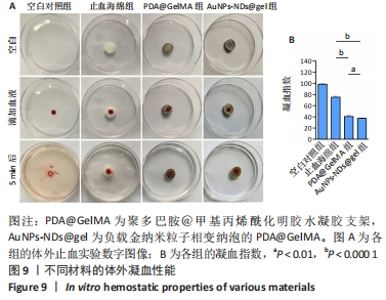
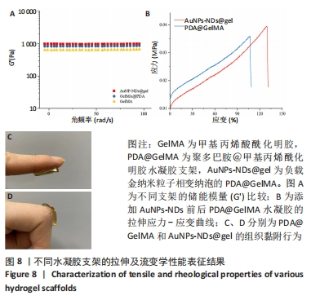
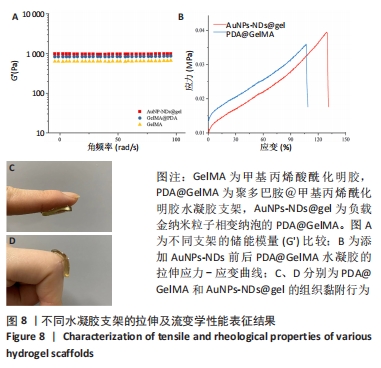
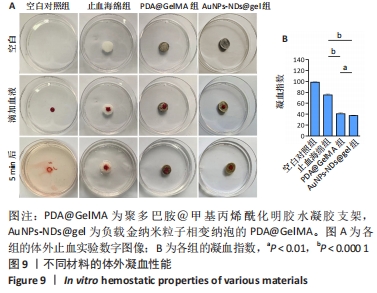
图8A显示,随振荡频率从0.1 rad/s增至100 rad/s时,PDA@GelMA和AuNPs-NDs@gel的储能模量(G’)数值都显示出小幅稳定增加,表明水凝胶支架稳定的力学性能。但15% GelMA的G’ 低于PDA@GelMA,这可能源于纯GelMA材料低交联密度所致其较差的力学性能,而PDA@GelMA则由于GelMA与PDA间形成的氢键及π-π键增加了交联密度,表现出更强的力学性能;AuNPs-NDs@gel的G’ 略高于PDA@GelMA,表现出更高的弹性模量,图8C、D展示了PDA@GelMA和AuNPs-NDs@gel良好的组织黏附性和延展性。 适配皮肤的支架材料需具备良好的延展性,因此有必要评估材料的拉伸性能。如图8B所示,AuNPs-NDs@gel相较PDA@GelMA显示出更好的延展性,二者拉伸断裂伸长率分别为122%,106%,优于人体皮肤的60%-70%[14]。AuNPs-NDs的添加使PDA@GelMA表现出更高的拉伸应力,这一结果与流变学相吻合,证明了其更优异的力学性能。 2.8 水凝胶支架的体外凝血性能检测结果 PDA@GelMA和AuNPs-NDs@gel组蒸馏水颜色均未发生明显变化,血液几乎均凝结于水凝胶上,而止血海绵组血块周边可见少量血液漫出,直观说明PDA@GelMA本身的凝血能力优于止血海绵,见图9A。凝血指数可反映材料的凝血性能,其值越小凝血性能越好。图9B是对材料凝血能力的定量分析,止血海绵的凝血指数为(75.3±1.81)%,PDA@GelMA的凝血指数为(40.83±2.06)%,AuNPs-NDs@gel组凝血指数为(37.64±1.16)%,3组间凝血指数两两比较差异有显著性意义(P < 0.01,P < 0.000 1)。"
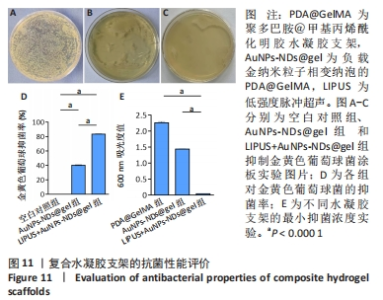
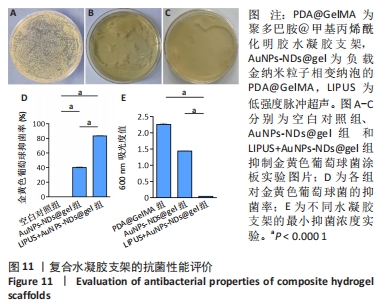
2.10 水凝胶支架的抗菌性能检测结果 AuNPs-NDs@gel发挥抗菌效能主要源自金纳米粒子的释放。实验组仅有少量菌落生长,对照组对菌落生长抑制不明显,见图11A-C;实验组经超声辐照后对金黄色葡萄球菌的抑菌率平均为83.17%,大于对照组(P < 0.001),见图11D,对照组表现出的抗菌作用可能源于少量未被包载的金纳米粒子。为了更好地评估AuNPs-NDs@gel的杀菌性能,可通过测量600 nm处细菌的吸光度值表征,见图11E。最小抑菌浓度测定结果显示,与PDA@GelMA相比,AuNPs-NDs@gel在超声辐照后的吸光度值显著降低,最小抑菌浓度为 6.5 μg/mL。结果表明AuNPs-NDs@gel具有良好的杀菌效能。"
| [1] GUNGOR S, KAHRAMAN E. Nanocarriers Mediated Cutaneous Drug Delivery. Eur J Pharm Sci. 2021;158:105638. [2] FENG X, CHEN Y. Drug delivery targets and systems for targeted treatment of rheumatoid arthritis. J Drug Target. 2018;26(10):845-857. [3] CHEN Y, FENG X. Gold nanoparticles for skin drug delivery. Int J Pharm. 2022;625:122122. [4] LABOUTA HI, LIU DC, LIN LL, et al. Gold nanoparticle penetration and reduced metabolism in human skin by toluene. Pharm Res. 2011; 28(11):2931-2944. [5] FABIILLI ML, WILSON CG, PADILLAF, et al. Acoustic droplet-hydrogel composites for spatial and temporal control of growth factor delivery and scaffold stiffness. Acta Biomater. 2013;9(7):7399-7409. [6] 陈丹丹,靳巧锋,谢明星,等.超声响应性纳米载体的研究进展[J].中华超声影像学杂志,2020,29(6):548-552. [7] CHOWDHURY SM, ABOU-ELKACEM L, LEE T, et al. Ultrasound and microbubble mediated therapeutic delivery: Underlying mechanisms and future outlook. J Control Release. 2020;326:75-90. [8] AKTURK O, KISMET K, YASTI AC, et al. Collagen/gold nanoparticle nanocomposites: A potential skin wound healing biomaterial. J Biomater Appl. 2016;31(2):283-301. [9] JIANG X, SAVCHENKO O, LI Y, et al. A Review of Low-Intensity Pulsed Ultrasound for Therapeutic Applications. IEEE Trans Biomed Eng. 2019; 66(10):2704-2718. [10] MA W, DONG W, ZHAO S, et al. An injectable adhesive antibacterial hydrogel wound dressing for infected skin wounds. Biomater Adv. 2022;134:112584. [11] ZHANG Z, KUANG G, ZONG S, et al. Sandwich-Like Fibers/Sponge Composite Combining Chemotherapy and Hemostasis for Efficient Postoperative Prevention of Tumor Recurrence and Metastasis. Adv Mater. 2018;30(49):e1803217. [12] SHI W, HASS B, KUSS MA, et al. Fabrication of versatile dynamic hyaluronic acid-based hydrogels. Carbohydr Polym. 2020;233:115803. [13] WIEGAND I, HILPERT K, HANCOCK RE. Agar and broth dilution methods to determine the minimal inhibitory concentration (MIC) of antimicrobial substances. Nat Protoc. 2008;3(2):163-175. [14] ANNABI N, RANA D, SHIRZAEI SANI E, et al. Engineering a sprayable and elastic hydrogel adhesive with antimicrobial properties for wound healing. Biomaterials. 2017;139:229-243. [15] 缪伟伟,王志雄,张俊利,等.聚羟基脂肪酸酯和聚酰胺-胺树状高分子构筑的经皮给药系统[J].中国组织工程研究,2014,18(52): 8433-8437. [16] KIM YJ, KIM B, KIM JW, et al. Combination of nanoparticles with photothermal effects and phase-change material enhances the non-invasive transdermal delivery of drugs. Colloids Surf B Biointerfaces. 2015;135:324-331. [17] BESSAR H, VENDITTI I, BENASSI L, et al. Functionalized gold nanoparticles for topical delivery of methotrexate for the possible treatment of psoriasis. Colloids Surf B Biointerfaces. 2016;141:141-147. [18] MAHMOUD NN, AL-QAOUD KM, AL-BAKRI AG, et al. Colloidal stability of gold nanorod solution upon exposure to excised human skin: Effect of surface chemistry and protein adsorption. Int J Biochem Cell Biol. 2016;75:223-231. [19] 任超宇,薛鹏程,焦雄,等.超声波作用下SiO2纳米颗粒的分离[J].高压物理学报,2018,32(4):60-66. [20] ANIRUDHAN TS, NAIR SS. Development of voltage gated transdermal drug delivery platform to impose synergistic enhancement in skin permeation using electroporation and gold nanoparticle. Mater Sci Eng C Mater Biol Appl. 2019;102:437-446. [21] ZHANG Y, YU J, KAHKOSKA AR, et al. Advances in transdermal insulin delivery. Adv Drug Deliv Rev. 2019;139:51-70. [22] CHENG CY, PHO QH, WU XY, et al. PLGA Microspheres Loaded with beta-Cyclodextrin Complexes of Epigallocatechin-3-Gallate for the Anti-Inflammatory Properties in Activated Microglial Cells. Polymers (Basel). 2018;10(5):519. [23] SHI NQ, ZHOU J, WALKER J, et al. Microencapsulation of luteinizing hormone-releasing hormone agonist in poly (lactic-co-glycolic acid) microspheres by spray-drying. J Control Release. 2020;321:756-772. [24] JOYCE P, PRESTIDGE CA. Synergistic effect of PLGA nanoparticles and submicron triglyceride droplets in enhancing the intestinal solubilisation of a lipophilic weak base. Eur J Pharm Sci. 2018;118:40-48. [25] LARESE FILON F, MAURO M, ADAMI G, et al. Nanoparticles skin absorption: New aspects for a safety profile evaluation. Regul Toxicol Pharmacol. 2015;72(2):310-322. [26] DUL M, NIKOLIC T, STEFANIDOU M, et al. Conjugation of a peptide autoantigen to gold nanoparticles for intradermally administered antigen specific immunotherapy. Int J Pharm. 2019;562:303-312. [27] CHANDAN R, MEHTA S, BANERJEE R. Ultrasound-Responsive Carriers for Therapeutic Applications. ACS Biomater Sci Eng. 2020;6(9):4731-4747. [28] RAHALI K, BEN MESSAOUD G, KAHN CJF, et al. Synthesis and Characterization of Nanofunctionalized Gelatin Methacrylate Hydrogels. Int J Mol Sci. 2017;18(12):2675. [29] GAN D, XU T, XING W, et al. Mussel-inspired dopamine oligomer intercalated tough and resilient gelatin methacryloyl (GelMA) hydrogels for cartilage regeneration. J Mater Chem B. 2019;7(10):1716-1725. [30] ALIABOUZAR M, ZHANG LG, SARKAR K. Lipid Coated Microbubbles and Low Intensity Pulsed Ultrasound Enhance Chondrogenesis of Human Mesenchymal Stem Cells in 3D Printed Scaffolds. Sci Rep. 2016;6: 37728. [31] HAMEED S, WANG Y, ZHAO L, et al. Shape-dependent significant physical mutilation and antibacterial mechanisms of gold nanoparticles against foodborne bacterial pathogens (Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus) at lower concentrations. Mater Sci Eng C Mater Biol Appl. 2020;108:110338. [32] LEUNG K. Poly(ethylene glycol)-coated gold nanocages bioconjugated with [Nle(4),d-Phe(7)]-alpha-melanotropin-stimulating hormone. Molecular Imaging and Contrast Agent Database (MICAD), Bethesda (MD), 2004. [33] CHI YF, QIN JJ, LI Z, et al. Enhanced anti-tumor efficacy of 5-aminolevulinic acid-gold nanoparticles-mediated photodynamic therapy in cutaneous squamous cell carcinoma cells. Braz J Med Biol Res. 2020;53(5):e8457. |
| [1] | Yang Yufang, Yang Zhishan, Duan Mianmian, Liu Yiheng, Tang Zhenglong, Wang Yu. Application and prospects of erythropoietin in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1443-1449. |
| [2] | Chen Kaijia, Liu Jingyun, Cao Ning, Sun Jianbo, Zhou Yan, Mei Jianguo, Ren Qiang. Application and prospect of tissue engineering in treatment of osteonecrosis of the femoral head [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(9): 1450-1456. |
| [3] | Mei Jingyi, Liu Jiang, Xiao Cong, Liu Peng, Zhou Haohao, Lin Zhanyi. Proliferation and metabolic patterns of smooth muscle cells during construction of tissue-engineered blood vessels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1043-1049. |
| [4] | Wang Shanshan, Shu Qing, Tian Jun. Physical factors promote osteogenic differentiation of stem cells [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(7): 1083-1090. |
| [5] | Shen Ziqing, Xia Tian, Shan Yibo, Zhu Ruijun, Wan Haoxin, Ding Hao, Pan Shu, Zhao Jun. Vascularized tracheal substitutes constructed by exosome-load hydrogel-modified 3D printed scaffolds [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 697-705. |
| [6] | Zhu Liwei, Wang Jiangyue, Bai Ding. Application value of nanocomposite gelatin methacryloyl hydrogels in different bone defect environments [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 753-758. |
| [7] | Chen Xiaofang, Zheng Guoshuang, Li Maoyuan, Yu Weiting. Preparation and application of injectable sodium alginate hydrogels [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 789-794. |
| [8] | Wang Jiani, Chen Junyu. Angiogenesis mechanism of metal ions and their application in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(5): 804-812. |
| [9] | Yang Yuqing, Chen Zhiyu. Role and application of early transient presence of M1 macrophages in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(4): 594-601. |
| [10] | Liu Xueli, Shen Li, Bi Wenguang, Mou Yang, Li Sen. Effect and mechanism of low intensity pulsed ultrasound on early angiogenesis in rats with acute tendon injury [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(32): 5097-5103. |
| [11] | Gu Mingxi, Wang Changcheng, Tian Fengde, An Ning, Hao Ruihu, Guo Lin. Preparation and in vitro evaluation of a three-dimensional porous cartilage scaffold made of silk fibroin/gelatin/chitosan [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 366-372. |
| [12] | Wang Xinmin, Yan Wenkai, Song Yahui, Liu Fei. Leukocyte- and platelet-rich fibrin with autologous hamstring tendon for traumatic patella dislocation [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 404-410. |
| [13] | Bi Yujie, Ma Dujun, Peng Liping, Zhou Ziqiong, Zhao Jing, Zhu Houjun, Zhong Qiuhui, Yang Yuxin. Strategy and significance of Chinese medicine combined with medical hydrogel for disease treatment [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 419-425. |
| [14] | Wang Xinyi, Xie Xianrui, Chen Yujie, Wang Xiaoyu, Xu Xiaoqing, Shen Yihong, Mo Xiumei. Electrospun nanofiber scaffolds for soft and hard tissue regeneration [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 426-432. |
| [15] | Gao Xueyu, Zhang Wentao, Sun Tianze, Zhang Jing, Li Zhonghai. Application of metal ions in bone tissue engineering [J]. Chinese Journal of Tissue Engineering Research, 2024, 28(3): 439-444. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||